Abstract
Currently, a number of promising strategies and approaches to cancer treatment include differentiation therapy. However, theoretical and methodological foundations of this field are not yet well developed. The objective of this study was to determine the effects of a mixture of polyclonal activators (PAs; phytohaemagglutinin, concanavalin A and lipopolysaccharide) on cytokine production by biopsy samples of invasive breast carcinoma of no special type (IBC-NST) having various differentiation abilities and metastatic potentials as well as on differentiation status of the IBC-NST biopsy samples. We used ELISAs to investigate spontaneous and PA-stimulated cytokine production in the IBC-NST biopsy samples; from these data, we calculated a cytokine production stimulation index (SIPA). The effect of PAs on tumour cell differentiation was determined via a differentiation stimulation index (DSI). DSI was found to vary within the range 1.0–5.0. After treatment with PAs, in the IBC-NST biopsy samples of group I (DSI <1.25), the production of IL-2, IL-6, IL-8, IL-17, IL-18, IL-1β, IL-1Ra, TNF-α and GM-CSF increased; in the biopsy samples of group II (DSI >1.25), the production of IL-6, IL-1β, IL-1Ra, TNF-α, G-CSF and GM-CSF significantly increased, while the production of VEGF-A decreased. Receiver operating characteristic (ROC) analysis of SIPA revealed that increased production of IL-18 in the IBC-NST biopsy samples after exposure to PAs may block the PA-driven, cytokine-mediated differentiation of moderately differentiated into highly differentiated tumour cells. The ROC analysis also uncovered an association between the responses of tumour cells to PAs and lymph node metastasis observed in the patients. The findings suggest that there is a need for research aimed at finding new drugs for differentiating cancer therapy and at searching for targeted inducers of cytokine production or specific suppressors of their induction.
Keywords
Introduction
It is known that one of the principal approaches to the treatment of malignant tumours involves neoadjuvant therapy, which is the first step in the treatment of various cancers. This approach is used to reduce the number of cancer cells and prepare the patient for surgical treatment. Because the number of malignant cells in a tumour may be decreased by eliminating the most malignant poorly differentiated cells and by increasing the number of more differentiated cells, so-called differentiation therapy (as one of the promising areas of cancer treatment) may also be added to the aforementioned strategy for the treatment of cancers. 1 The differentiation therapy of malignant tumours is based on the idea that it is possible to initiate differentiation of tumour cells and revert them to the normal phenotype, which reduces tumour malignancy in the preoperative period.1,2 However, the known differentiation therapy drugs with anti-tumour activity include only two groups of agents that can efficiently increase differentiation of neoplastic cells: the retinoid group and compounds that affect gene methylation in transformed cells.3–6 There have been attempts to include retinoids in the treatment regimens for neuroblastomas, breast cancer and prostate cancer, 7 but these treatments did not yield the expected effect.8–10
Meanwhile, there are substances that are known to enhance differentiation of breast cancer cells and to partially return their phenotype to a relatively normal one, as described in the following experiments. Cultivation of breast cancer cells in a medium containing a β1-integrin inhibitor (AIIB2), a mitogen-activated protein kinase (MAPK) inhibitor and a phosphatidylinositol-3-kinase (PI3K) inhibitor increases the proportion of tumour cells with signs of reversion of some cytophysiological indicators to the normal phenotype. 11 Addition of a mixture of mycophenolic acid, phorbol 12-myristate 13-acetate and retinoic acid to the culture medium of tumour cells has been demonstrated to increase, within 4 days, the percentage of AU-565 cells with a more mature phenotype. 12 These findings suggest that stimulation of cell differentiation in the tumour tissue may occur rather quickly. All these data indicate the importance of the search for substances or compositions (made of chemical or biological substances) capable of stimulating cell differentiation in malignant tumours as well as the usefulness of investigation of such stimulation mechanisms.
Previously, we have demonstrated that in vitro cultivation of biopsy samples of invasive breast carcinoma of no special type (IBC-NST) in a medium containing a mixture of polyclonal activators (PAs), such as phytohaemagglutinin (PHA), concanavalin A (ConA) and lipopolysaccharide (LPS), stimulates in them the production of several cytokines. 13 We have also shown that the cytokine-producing potential of IBC-NST biopsy samples derived from primary tumours with metastases to regional lymph nodes is significantly different from that without metastases to lymph nodes. 14 In another study, we showed in a similar model that the cytokine profile of an IBC-NST biopsy sample supernatant with the prevalence of poorly differentiated cells differs from the cytokine profile of an IBC-NST biopsy sample with the prevalence of highly differentiated cells. 15 On the basis of these findings, we have proposed that cultivation of IBC-NST samples in a PA-containing medium may affect differentiation of the IBC-NST cells through stimulated production of certain cytokines, and that the magnitude of this effect may be determined by the initial status of IBC-NST, that is, its differentiation degree and its metastatic potential.
The purpose of the study was to investigate the effect of a mixture of PAs (PHA, ConA and LPS) on cytokine production by IBC-NST biopsy samples showing various parameters of tumour cell differentiation and metastatic potential as well as on the differentiation status of the IBC-NST samples under study.
Materials and methods
Patients
We analysed tumour biopsy samples from 38 female patients (aged 42–67 years) with grade II or III IBC-NST. The sample size of tumour biopsies needed for this study was determined but was limited by the number of patients who signed the agreement to participate in this study and by the terms of this agreement itself. This agreement stipulated that at least seven tumour bioptate samples from each patient would be studied, where one sample would be examined by a histopathologist when making a final diagnosis, and four samples would be used to analyse the expression of HER2/neu, PR (progesterone receptor), ER (oestrogen receptor) and Ki-67 (proliferation marker) to determine the tumour’s molecular genetic status, whose assessment is necessary for treatment planning; two samples would be used directly to evaluate the effect of a mixture of PAs on cytokine production and to evaluate changes in the quantitative ratios of tumour cells with different differentiation status after exposure to the PAs. Preliminary testing showed that the proportions (%) of highly differentiated tumour cells (HDTCs), poorly differentiated (PDTCs) and moderately differentiated tumour cells (MDTCs) did not change in the tumour biopsies when they were cultivated in a culture medium without PAs and the possible deviations of these parameters do not exceed 5% of the initial values (before the cultivation of the biopsies). If inflammatory infiltrates were detected in tumour biopsy specimens (according to preliminary histopathological analysis), such biopsy specimens were excluded from the study. During the study, the tumour biopsy samples were subdivided into two groups (each biopsy sample corresponded to one patient). Group I included 21 biopsy IBC-NST samples (i.e. 21 patients) whose in vitro tissue culture showed no changes in the proportions (%) of various categories of differentiated cells in response to PAs (Figure 1(a)). Group II included 17 biopsy IBC-NST samples (i.e. 17 patients) whose in vitro tissue culture manifested changes in the proportions of various categories of differentiated cells in response to PAs (Figure 1(b)). The whole study was conducted in accordance with the Helsinki Declaration (Brazil, Fortaleza, 2013). All recommendations of ICMJE were followed. Each patient was informed about the study being conducted, its objectives, and methods. A written informed consent form for participation in the study and for tumour biopsy procedures to be performed was signed by each patient and verified by a physician. The study and all study protocols were approved (approval No. 2016-3) by the Ethics Committee of the Institute of Molecular Biology and Biophysics, a subdivision of the Federal Research Center of Fundamental and Translational Medicine (Novosibirsk, Russia).

The proportions (%) of poorly differentiated tumour cells (PDTCs), moderately differentiated tumour cells (MDTCs) and highly differentiated tumour cells (HDTCs) in IBC-NST samples: (a) Group I tumour samples (n = 21) did not show cell differentiation after exposure to PAs; (b) Group II tumour samples (n = 17) showed cell differentiation after treatment with PAs. PA: after exposure to polyclonal activators (PAs); Control: without exposure to PAs. Data were the means ± SEM.
Evaluation of the cytokine-producing potential of the tumours
To assess the cytokine-producing potential of each tumour and its microenvironment, we employed a mixture (PAs; as mentioned above) of 4 μg/ml PHA, 4 μg/ml ConA and 2 μg/ml LPS. The CYTOKINE-STIMUL-BEST standardised assay kit (AO Vector-Best, Novosibirsk, Russia) was utilised for cytokine assays. Tumour biopsy samples (volume 8 mm3) obtained by trepanobiopsy were placed into two glass vials each containing 1 ml of a medium; one vial contained only the DMEM-F12 medium (for evaluation of spontaneous cytokine production, control experiment), while the second vial contained the above PAs (at the specified concentrations) in the same amount of the medium (for evaluation PA-stimulated cytokine production).13–15 Tumour biopsy samples were cultivated in vitro for 72 h. To obtain the pure supernatant, tumour cells remaining in the vials were precipitated by centrifugation at 900 g for 15 min. Enzyme-linked immunosorbent assays (ELISAs) were performed to determine the concentrations of IL-2, IL-6, IL-8, IL-10, IL-17, IL-18, IL-1β, IL-1Ra, TNF-α, IFN-γ, G-CSF, GM-CSF, VEGF-A and MCP-1 (monocyte chemotactic protein 1) in the supernatant via assay kits from AO Vector-Best. The stimulation index of polyclonal activators (SIPA, determined individually for each patient), which measures production of cytokines by a tumour and its microenvironment was calculated using the following formula: SIPA = A/B, where A is the concentration of a cytokine after stimulation of the tumour by PAs, and B is the concentration of this cytokine in the supernatant without stimulation (spontaneous production). The SIPA for each cytokine was expressed in arbitrary units (a.u.).
Histopathological analysis
After incubation at 37°C for 72 h, the IBC-NST biopsy samples were removed from the medium and fixed in a formalin solution for subsequent histopathological analyses. IBC-NST tissue sections were stained with haematoxylin and eosin according to the standard procedure. Evaluation of the differentiation degree of the tumour cells and their classification as HDTCs, MDTCs, or PDTCs were based on the cellular polymorphism degree, nuclear/cytoplasmic ratio, presence of mitoses (including pathological ones) and capacity for tissue structure formation.16,17 HDTCs had cytomorphology similar to that of cells with a normal phenotype and showed the predominance of the cytoplasm over nucleus and capacity for gland formation. PDTCs had an irregular shape and featured pronounced polymorphism, predominance of the nucleus over cytoplasm, inability to form tissue structures, diffuse growth and numerous mitoses (including pathological ones). MDTCs were somewhere between HDTCs and PDTCs in terms of these parameters.
Molecular genetic subtypes of breast cancer were identified according to the international classification by immunohistochemical analysis of the IBC-NST samples (Gallen International Expert Consensus, 2011). The HER2/neu status of IBC-NST and expression of oestrogen receptor (ER, SP1), progesterone receptor (PR, 1E2) and cell proliferation marker Ki-67 in the IBC-NST biopsy samples were evaluated by an immunohistochemical method. 18
The effect of PAs on differentiation of IBC-NST tumour cells
This effect of PAs on differentiation of IBC-NST tumour cells was evaluated based on the differentiation stimulation index (DSI, determined individually for each patient), which was calculated via the formula DSI = HDTCPA/HDTCC, where HDTCPA is the proportion (%) of highly differentiated cells in a tumour sample after stimulation by PAs, and HDTCC is the proportion (%) of highly differentiated cells in this tumour sample without stimulation by the PAs (control). The DSI was expressed in a.u.
Statistical analysis
Histograms were generated in Microsoft Excel and IBM SPSS Statistics v.22.0 software. Mean values, standard error of the mean (SEM), medians and 25th–75th percentiles were calculated using the Statistica v.7 software suite. The significance of differences between groups was determined in Statistica v.7 by the following methods: the nonparametric Mann–Whitney U test (comparison of independent variables), Fisher’s exact test (comparison of values expressed in %) and the paired Wilcoxon t test (comparison of dependent variables). The receiver operating characteristic (ROC) analysis was performed using the IBM SPSS Statistics v.22.0 software suite.
Results
The influence of PAs on the proportions of cells with varying degrees of differentiation in the IBC-NST biopsy samples
The DSI among the IBC-NST samples was found to vary within a range of 1.0–5.0, that is, from no increase in the proportion of HDTCs (after exposure to PAs) to a pronounced increase in the proportion of HDTCs in the IBC-NST biopsy samples. The PAs stimulated formation of HDTCs in the IBC-NST biopsy samples of 44.7% patients. The number of HDTCs in the IBC-NST biopsy samples after exposure to PAs increased by 25% (among all the samples) on average. According to the paired Wilcoxon t test, the hypothesis of PA-stimulated HDTC formation was correct (P < 0.008). For further analysis of the effects of the PAs on the IBC-NST samples, these tumour samples (n = 38, each corresponded to a patient) were conditionally subdivided into two groups: group I showed no differentiation response to the PAs, with the SIPA <1.25 (n = 21), and group II showed a differentiation response to the PAs, with the SIPA >1.25 (n = 17). In group I, the numbers of PDTCs, MDTCs and HDTCs in the IBC-NST samples after exposure to PAs did not change significantly (Figure 1(a)). In group II, the proportion (%) of MDTCs in the tumour samples after exposure to the PAs significantly diminished, and the number of HDTCs increased accordingly. At the same time, the proportion of PDTCs remained almost unchanged (Figure 1(b)).
The effect of PAs on cytokine production in the IBC-NST biopsy samples
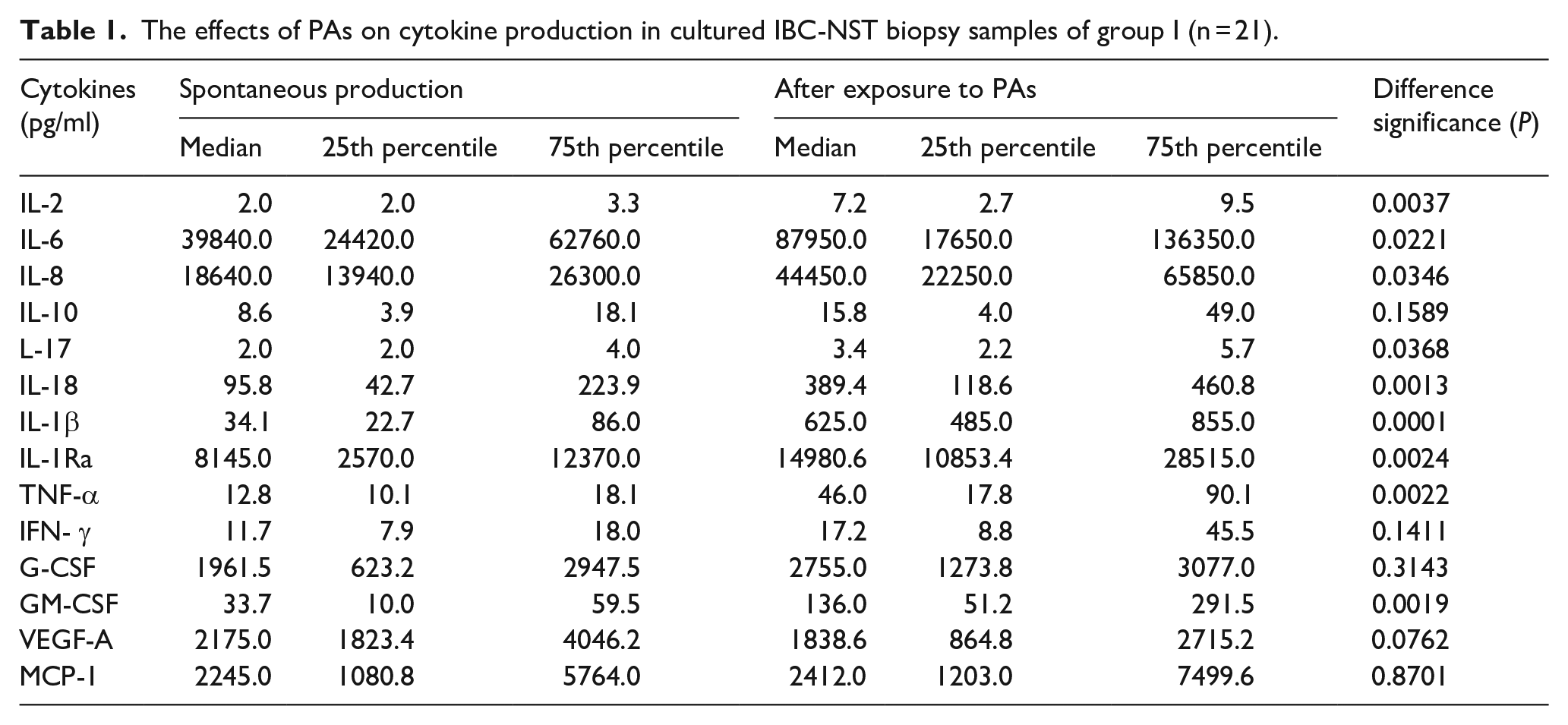
In group I, PA-treated IBC-NST biopsy samples significantly up-regulated the production of nine out of the 14 studied cytokines: IL-2, IL-6, IL-8, IL-17, IL-18, IL-1β, IL-1Ra, TNF-α and GM-CSF (Table 1). In group II, PA-treated IBC-NST biopsy samples significantly up-regulated IL-6, IL-1β, IL-1Ra, TNF-α, G-CSF and GM-CSF production and decreased VEGF-A production (Table 2). As presented in Tables 1 and 2, stimulation of HDTC formation in group II after the exposure to the PAs was accompanied by the absence of PA-dependent stimulation of IL-2, IL-8, IL-17 and IL-18 production in group I as well as by apparent PA-driven stimulation of G-CSF production and ‘inhibition’ of VEGF-A production.
The effects of PAs on cytokine production in cultured IBC-NST biopsy samples of group I (n = 21).
The influence of the PAs on cytokine production in cultured IBC-NST biopsy samples of group II (n = 17).

According to these data, we supposed that stimulation of MDTC differentiation and accordingly an increase in the number of HDTCs in the IBC-NST biopsy samples in group II may be mediated by PA-stimulated production of IL-6, IL-1β, IL-1Ra, TNF-α, GM-CSF and G-CSF. We also hypothesised that cell differentiation in group I may somehow be ‘blocked’ by the up-regulated cytokines IL-2, IL-8, IL-17 and IL-18.
Table 3 shows data on molecular genetic IBC-NST subtypes of all the patients whose biopsy samples were investigated herein. As readers can see in this table, groups I and II were heterogeneous and included various molecular genetic subtypes. The differences between the groups in the number of tumour samples of the Luminal B HER2-positive subtype were not significant. Therefore, we did not find a relation between the capacity of cells for PA-enhanced differentiation and molecular genetic IBC-NST subtypes.
Molecular genetic subtypes of the IBC-NST biopsy samples (the number of patients with metastasis to regional lymph nodes is indicated in parentheses).

Testing the hypothesis of cytokine-mediated inhibition of PA-induced cellular differentiation in the IBC-NST biopsy samples
To test the hypothesis of a cytokine-mediated inhibitory mechanism behind the PA-induced differentiation of tumour cells in the IBC-NST biopsy samples, we carried out ROC analysis of SIPA regarding the production of cytokines IL-2, IL-8, IL-17 and IL-18 to evaluate the quality of several statistical models for the allocation of all the studied IBC-NST samples into two groups: with and without stimulation of tumour cell differentiation.
Figure 2(a) illustrates the ROC curves generated using SIPA for the IL-2, IL-8, IL-17 and IL-18 production; these curves characterise the capacity of the models for detection of biopsy samples of group I (i.e. no PA-induced differentiation of tumour cells in the IBC-NST). As depicted in Figure 2(a), the SIPA of PA-stimulated IL-18 production yields a good-quality model (area under the curve (AUC = 0.715)). If SIPAs of the IL-18 production were more than 1.50, then the quality of the model increased: AUC = 0.75 (Figure 2(b)). In contrast, if the SIPA cut-off for the IL-18 production was set to <1.50, then the accuracy of this statistical model for the identification of group I samples became unsatisfactory (Figure 2(c)). SIPAs of IL-2, IL-8 and IL-17 production yielded unsuitable models. As presented in Figure 2(d), the use of SIPAs for the G-CSF and VEGF-A production cannot predict PA-induced stimulation of cellular differentiation in the IBC-NST biopsy samples. Thus, we demonstrated that the PA-stimulated increase in IL-18 production in the IBC-NST biopsy samples may inhibit or block MDTC-to-HDTC differentiation after exposure to PAs; this differentiation may be mediated by other cytokines (IL-6 and/or IL-1β, IL-1Ra, TNF-α and GM-CSF).
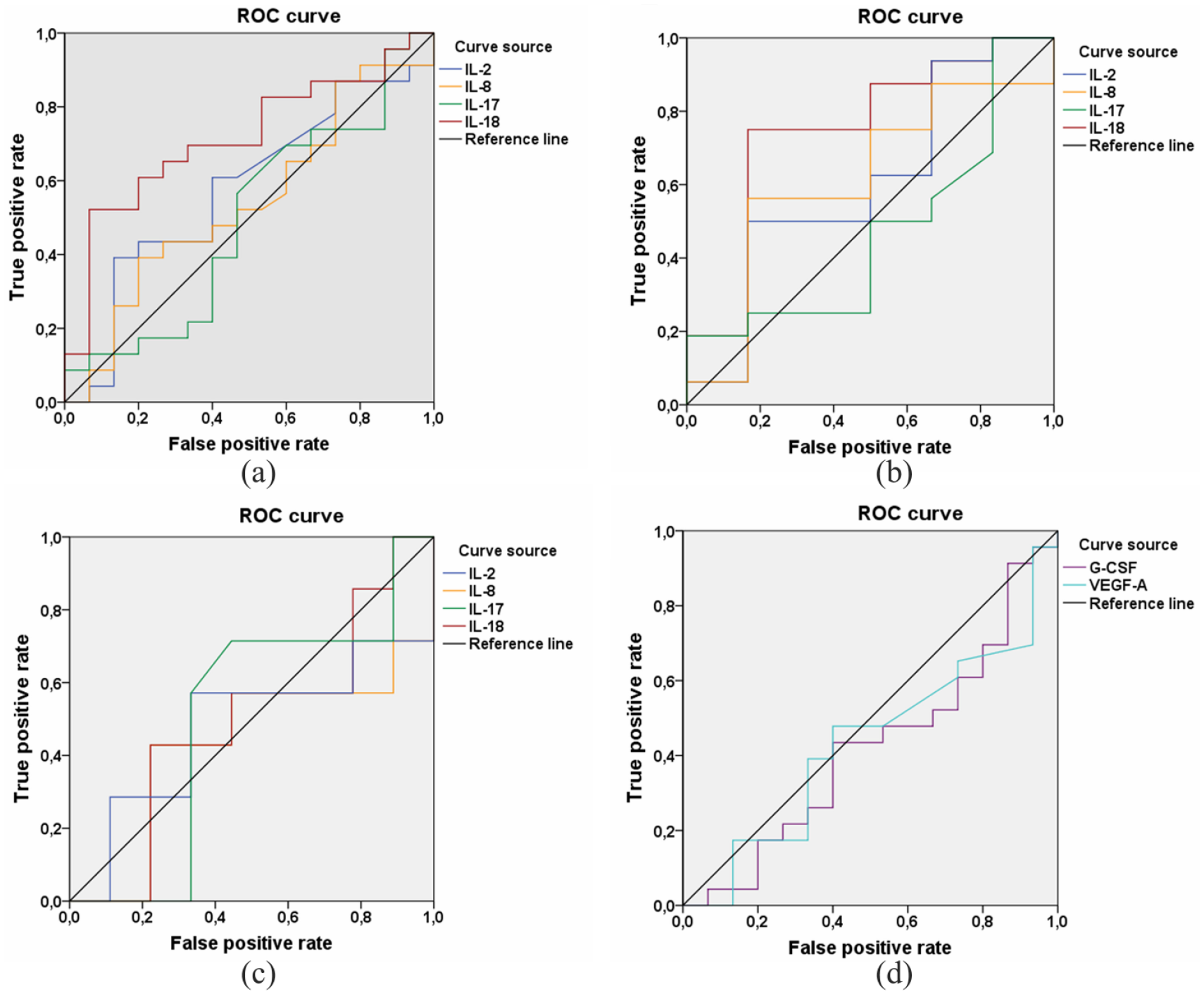
ROC curves generated using SIPA for the IL-2, IL-8, IL-17 and IL-18 production, which characterise quality of models for identification of the IBC-NST biopsy samples without an increase in the amount of HDTCs after exposure to PAs: (a) for all values of SIPA on IL-18 production, that is, for all patients (IL-2 AUC = 0.577, IL-8 AUC = 0.487, IL-17 AUC = 0.496, IL-18 AUC = 0.716); (b) for SIPA on IL-18 production >1.50 (IL-2 AUC = 0.604, IL-8 AUC = 0.417, IL-17 AUC = 0.469, IL-18 AUC = 0.750); (c) for SIPA on IL-18 production <1.50 (IL-2 AUC = 0.476, IL-8 AUC = 0.429, IL-17 AUC = 0.500, IL-18 AUC = 0.492); (d) ROC curves generated using SIPA for the G-CSF (AUC = 0.583) and VEGF-A production (AUC = 0.568) with allowance for SIPA data of all patients, which characterise quality of models for identification of the IBC-NST biopsy samples without an increase in the amount of HDTCs after exposure to PAs (identification of the IBC-NST biopsy of group I). True positive rate – Sensitivity. False positive rate – Specicity.
Correlation between lymph node metastasis and the ability of PAs to increase the proportion of HDTCs in the IBC-NST biopsy samples
In the course of this study, we made the following interesting observation. Among the patients corresponding to group I IBC-NST biopsy samples (no PA-induced tumour cell differentiation), metastases to regional lymph nodes were more common than in group II (where the PAs induced tumour cell differentiation). In group I, 42.9% of the patients had metastases to regional lymph nodes; in group II, only 11.8% of the patients had them. According to Fisher’s exact test, these differences were significant (P < 0.0385). Therefore, a question came up: to what extent may the presence of metastases be associated with the initial proportions of PDTCs, MDTCs and HDTCs in the IBC-NST samples? The second question was whether the differentiation response of the tumour samples to the PAs correlates with the metastases in the patients. ROC analysis revealed that the proportion of HDTCs in the IBC-NST biopsy samples, both before and after exposure to PAs, yielded good-quality models for the identification of patients with IBC-NST metastasising to regional lymph nodes: AUC of 0.87 and 0.714, respectively (Figure 3(a)). At SIPA values of <1.5, the quality of these models was almost unchanged: AUC of 0.83 and 0.69, respectively (Figure 3(b)). At SIPA values of <1.25, the quality of these models (for HDTCs) increased to the ‘excellent model’ and ‘very good model’ level: AUC of 0.93 and 0.84, respectively (Figure 3(c)). At SIPA values of <1.15, the quality of these models corresponded to the ‘excellent model’ level: AUC of 0.96 and 0.92, respectively (Figure 3(d)). The analysis suggested that the risk of metastasis to regional lymph nodes is associated with two indicators of the IBC-NST differentiation potencies: the initial amount of HDTCs in the IBC-NST biopsy samples and the amount of HDTCs after the stimulation of the biopsy samples by the PAs. Therefore, the ROC analysis revealed a relation between PA-induced stimulation of tumour cells and metastasis, that is, the larger the initial amount of HDTC and the greater the ability of tumour cells for PA-induced differentiation, the lower is the prevalence rate of lymph node metastasis among the patients.
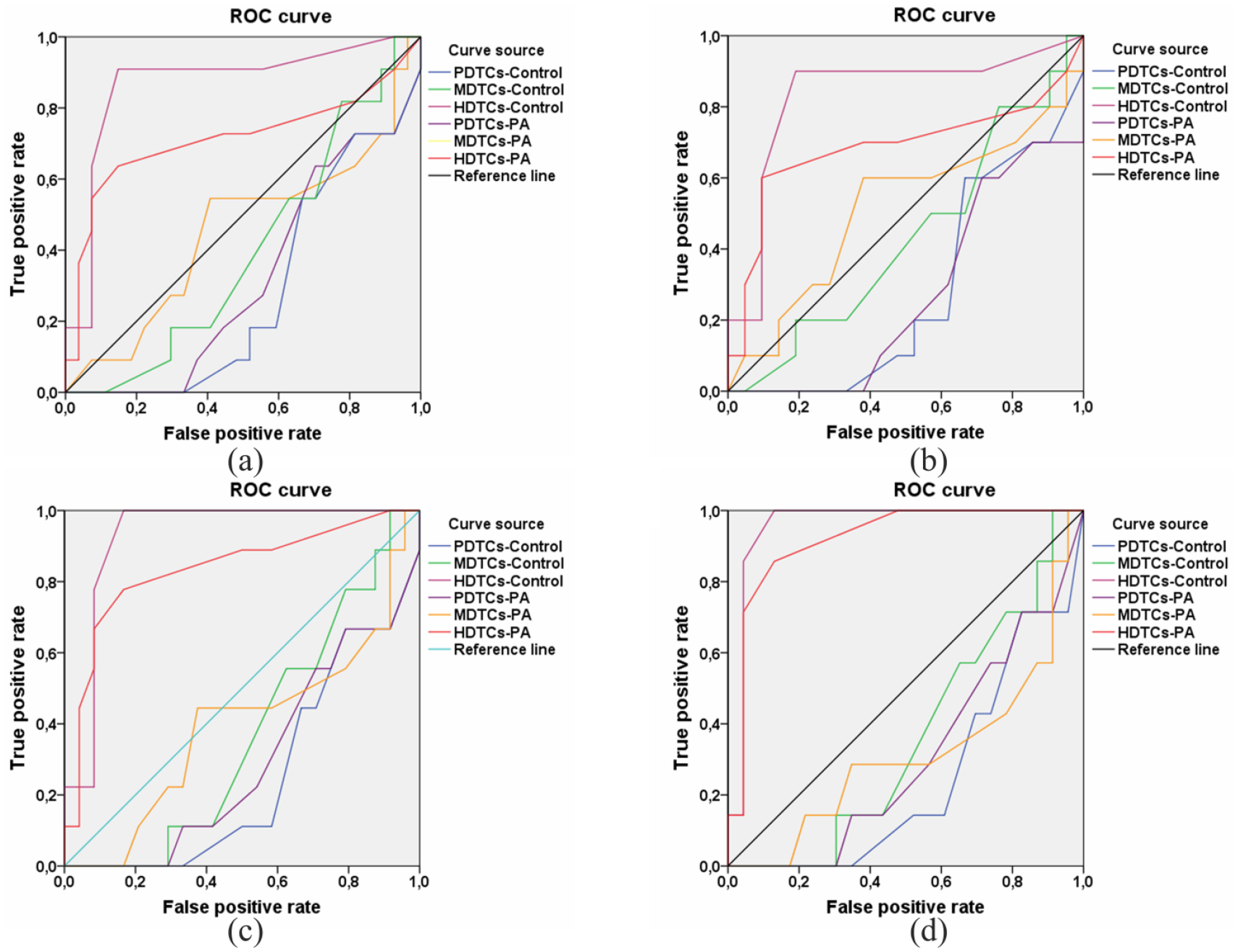
ROC curves generated using PDTCs-Control, MDTCs-Control, HDTCs-Control, PDTCs-PA, MDTCs-PA and HDTCs-PA data (see Figure 1), which characterise quality of models for identification of the IBC-NST biopsy samples of patients with metastases to regional lymph nodes: (a) for all values of DSI, that is, for all patients (PDTCs-Control AUC = 0.283, MDTCs-Control AUC = 0.399, HDTCs-Control AUC = 0.869, PDTCs-PA AUC = 0.320, MDTCs-PA AUC = 0.455, HDTCs-PA AUC = 0.714); (b) for patients with DSI <1.50 (PDTCs-Control AUC = 0.281, MDTCs-Control AUC = 0.433, HDTCs-Control AUC = 0.833, PDTCs-PA AUC = 0.274, MDTCs-PA AUC = 0.514, HDTCs-PA AUC = 0.686); (c) for patients with DSI <1.25 (PDTCs-Control AUC = 0.267, MDTCs-Control AUC = 0.356, HDTCs-Control AUC = 0.929, PDTCs-PA AUC = 0.307, MDTCs-PA AUC = 0.378, HDTCs-PA AUC = 0.842); (d) for patients with DSI <1.15 (PDTCs-Control AUC = 0.248, MDTCs-Control AUC = 0.363, HDTCs-Control AUC = 0.957, PDTCs-PA AUC = 0.307, MDTCs-PA AUC = 0.314, HDTCs-PA AUC = 0.919). True positive rate – Sensitivity. False positive rate – Specicity.
Discussion
In this study, we found that in vitro cultivation of IBC-NST biopsy samples in a medium containing a mixture of PHA, ConA and LPS stimulated both tumour cell differentiation in some IBC-NST biopsy samples and production of several cytokines. We supposed that the transition of IBC-NST tumour cells to the HDTC phenotype may be mediated by PA-dependent stimulation of IL-6, IL-1β, IL-1Ra, TNF-α, GM-CSF and G-CSF production. All these cytokines can be produced by tumour microenvironment cells (dendritic cells, monocytes/macrophages and others).19–22 All cytokines are known to act directly only on the target cells expressing appropriate receptors.23,24 This mechanism is also valid when cytokines act on tumour cells with complementary receptors for appropriate cytokines. According to the literature, IBC-NST tumour cells have receptors only for three out of the six cytokines being analysed here: IL-1β, IL-1Ra (IL2RA), 25 and TNF-α (TNF-R). 26 Because the PA-treated IBC-NST samples had no obvious signs of apoptotic changes, and because increased TNF-α production was also detected in group I, we ruled out TNF-α as a factor mediating the PA-induced differentiation in the IBC-NST biopsy samples. In group I, the PA-treated IBC-NST biopsy samples significantly increased production of the six above-mentioned cytokines as well as IL-2, IL-8, IL-17 and IL-18. All these cytokines can be produced by tumour microenvironment cells (e.g. dendritic cells and monocytes/macrophages),19–22 and IL-18 can be produced by IBC-NST cells. 27 IL-2 and IL-8 receptors are not expressed on the IBC-NST cells. Low expression of IL-17 receptors on IBC-NST cells is detected very rarely. 28 On the contrary, there is evidence of high expression of IL-18 receptors on IBC-NST cells (IL18R1). 29 Thus, according to the analysis of well-known international scientific databases, analysis of the literature, and a comparison with our findings, we propose that the PA-induced differentiation of these tumour cells may be controlled by IL-1β. In this case, IL-1Ra, being relatively overproduced, can inhibit the PA-induced differentiation of the tumour cells by binding to receptor IL2RA. IL-18, by binding to IL18R1 located on some IBC-NST tumour cells, probably suppresses the PA-induced differentiation of tumour cells in the IBC-NST biopsy samples whose supernatants are characterised by significantly increased production of IL-18 as compared to the biopsy samples whose incubation with the PAs leads to an increase in the relative count of HDTCs. Our ROC analysis of the SIPA revealed that increased production of IL-18 in the IBC-NST biopsy samples after the exposure to the PAs may block the cytokine-mediated induction of MDTC-to-HDTC differentiation by the PAs. The ROC analysis also uncovered an association between the responses of the tumour cells to the PAs and lymph node metastasis observed in the patients. Our assumptions about possible cytokine-dependent mechanisms underlying the regulation of PA-induced differentiation of tumour cells in IBC-NST biopsy samples – and associations of the effect of PA-induced differentiation of IBC-NST cells with metastasis – are largely based on the results of statistical analysis of our data obtained by the tissue culture method. Accordingly, we will refine the proposed mechanisms in further studies on in vitro cell models.
Probably, the ability of IBC-NST cells to enhance cellular differentiation after exposure to PAs may be associated with a direct influence of some components of the PA mixture on tumour cells through so-called Toll-like receptors (TLRs). To prove this assumption, it is necessary to conduct a separate in vitro study on the relationship of TLR expression on IBC-NST cells and their differential response to PA. However, we do not yet have data on the profile and levels of the TLRs expressed on the IBC-NST tumour cells to discuss these mechanisms.
It is important to emphasise that there are numerous studies showing that cytokines should be considered an important factor affecting the mechanisms of formation, progression and metastasis of malignant tumours.30,31 During tumour growth, cytokines are produced not only by the immune organs and immunocompetent cells infiltrating the tumour but also directly by tumour cells together with connective tissue cells that form the tumour microenvironment.32,33 Some research indicates that depending on the concentration and balance of cytokines and their antagonists, they can either enhance or inhibit breast cancer growth and metastasis.34,35 There is evidence that changes in the concentrations of some cytokines (IL-1, IL-6, IL-8, IL-18 and IL-19) and growth factors (G-CSF, GM-CSF and TGF-β) in the blood may be associated with breast cancer progression.36,37 However, the mechanisms behind this relation are not well understood because most data on the role of cytokines in tumour progression are usually based only on a comparison of blood concentrations of certain cytokines with tumour growth rates and metastasis. We hope that our study can fill this gap to some extent.
Conclusion
By means of a mixture of PAs (PHA, ConA and LPS), we found that the differentiation potential of IBC-NST tumour cells differs significantly among the patients. The ability of tumour cells to differentiate is likely associated with a range of cytokines produced by the tumour and its microenvironment cells. The ability of the IBC-NST tumour cells to differentiate, as detected by means of the mixture of PAs, may be related to the metastatic potential of the tumour. Our findings also suggest that the research aimed at finding new drugs for differentiating cancer therapy and at searching for targeted inducers of cytokine production or specific suppressors of their induction deserves further effort.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the Ethics Committee of the Institute of Molecular Biology and Biophysics, the Subdivision of Federal Research Centre of Fundamental and Translational Medicine, Novosibirsk, Russia (APPROVAL NUMBER No. 2016-3).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Each patient was informed about the study being conducted, its objectives, and methods. A written informed consent form for participation in the study and for tumour biopsy procedures to be performed was signed by each patient and verified by a physician.
Trial registration
Not applicable.
