Abstract
BACKGROUND:
Agents targeting the PI3K pathway in triple negative breast cancer did not show any significant efficacy so far mostly because of the complex nature of these targeted inhibitors. Targeting the cancer cells with the combination of inhibitors may help in decelerating the regulatory pathways further achieving optimum clinical benefit. In this study, we investigated the effect of PIK3CA and mTOR inhibition in-vitro in triple-negative breast cancer (TNBC) cell lines.
OBJECTIVE AND METHODS:
Three TNBC cell lines; MDA MB231, MDA MB468, and MDA MB453 were subtyped using immunohistochemistry and were screened for hotspot mutations in PIK3CA and AKT1. All cell lines were treated with different concentrations of inhibitors; PI3K inhibitor (BKM 120), mTOR inhibitor (AZD 8055), and dual PI3K/mTOR inhibitor (BEZ 235), and cell viability was assessed by MTT (3-(4, 5-Dimethylthiazol-2-yl)-2, 5-Diphenyltetrazolium Bromide), Trypan blue and Annexin-V/PI Assays.
RESULTS:
Using immunohistochemistry, TNBC cell lines were subtyped as; mesenchymal subtype-specific cell line (MDA MB231), basal subtype-specific cell line (MDA MD468), and Luminal androgen receptor (LAR) subtype-specific cell line (MDA MB453). PIK3CA hot spot mutation (p.H1047R) in exon 20 was identified in the Luminal androgen receptor subtype (MDA MB453 cells) cell line. Cell viability assays showed that the Mesenchymal subtype-specific cell line (MDA MB231) was the most resistant to all inhibitors and the Luminal Androgen subtype (MDA MB453 cells) cell line was more sensitive to BKM120 (PI3K inhibitor) inhibition compared to other subtypes.
CONCLUSIONS:
This study identified that the Luminal androgen receptor subtype of triple-negative breast cancer with PIK3CA mutation may be targeted with PIK3CA inhibitors with a favorable outcome.
Introduction
Triple-negative breast cancer (TNBC) lacks expression of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) and accounts for 15–20% of all breast cancers [2]. Further molecular profiling of TNBC has shown genomic, transcriptomic, and histopathological heterogeneity in this subtype. Extensive research is going on to find a therapeutic target for TNBC, however, despite the best efforts, there has not been any effective targeted therapy against TNBC subtypes, and patients are treated with conventional chemotherapy. However, not all patients respond well to conventional chemotherapy, thus resulting in early relapse and distant metastasis [3,4]. Earlier studies have identified heterogeneity in triple-negative breast cancer based upon gene expression profiling. Lehmann et al. [12] reclassified TNBC into four molecular subtypes: Basal-like 1 (BL1), Basal-like 2 (BL2), Mesenchymal (M), and Luminal Androgen Receptor (LAR) subtypes. Based upon the information from gene expression studies, similar classification can now be provided by immunohistochemistry [5,7]. Exploiting the TNBC heterogeneity may help in deciphering new biomarkers and new targetable pathways.
PI3K/AKT/mTOR is one such pathway that is activated in different types of cancer including breast cancer. However, this pathway is less explored in the TNBC subtype [13]. Inhibitors targeting the PI3K pathway are already being tested in clinical trials for different malignancies. Currently, we do not have any biomarker which can predict the response to these inhibitors. Successful development of such inhibitors will be enhanced by the identification of the patients who are likely to respond to these targeted drugs through the use of predictive biomarkers. We aimed to identify putative biomarkers predictive of response to the selective PI3K/AKT inhibitors.
Materials and methods
Cell lines
Three human triple-negative breast cancer cell lines: MDA-MB231, MDA-MB468, and MDA-MB453 were used and were procured from the National Centre for Cell Sciences (NCCS), Pune, India.
Immunohistochemistry and subtyping
Immunohistochemistry was done on cell-block prepared from cell pellets. Immunostaining was performed on an automated Ventana, Biotek system. Cell lines were tested for estrogen receptor (ER), progesterone (PR), and human epidermal receptor2 (HER2). For the classification of cell lines of TNBC subtypes, the following antibodies were used: CK5/6 (1:100 dilution; Clone D5/16B4, Cell Marque, Rocklin, California, United States), CK4/14 (1:250 dilution; clone LL002, Cell Marque, Rocklin, California, United States), EGFR (1:100 dilution; clone H11, Dako), Vimentin (1:100 dilution; clone V9, Dako), E-cadherin (1:100 dilution; clone NCH-38, Dako), claudin3 and claudin7 (ready to use; Cell Marque, Rocklin, California, United States), Androgen Receptor (1:100 dilution; clone SP107, Cell Marque, Rocklin, California, United States). Based upon the expression of these proteins, TNBC cell lines were categorized as [7]: Basal-like (BL): Positive expression for Cytokeratin 5/6 and/or 4/14 and EGFR Mesenchymal (MES): Decreased expression of E-cadherin, Claudin 3, and 7 and positive expression for vimentin Luminal Androgen receptor (LAR) subtype: Positive nuclear expression for the androgen receptor
PIK3CA hotspot mutations
PIK3CA and AKT1 hotspot mutations were evaluated in all three TNBC cell lines by the Sanger sequencing method. Genomic DNA was extracted from TNBC cell lines using a commercial DNA extraction kit (QIAamp DNA Mini Kit, Qiagen, Hilden, Germany). The concentration of DNA was measured using a spectrophotometer (NanoDrop TM 2000, Thermo Scientific™, USA). Isolated DNA was subjected to PCR amplifications targeting PIK3CA (exon 4, 9, and 20) and AKT1 (exon2) using the primer pairs (Primers details given in Supplementary Table S1). The amplified genomic DNA was subjected to purification using a commercial kit (QIAquick, Qiagen, USA) and checked on 2% agarose gel electrophoresis. Sequencing was done using the Big Dye terminator sequencing kit version3.1 (Life Technology) on ABI 3500 series genetic analyzers (Life Technologies). The sequencing profiles were analyzed on Finch TV (PerkinElmer, Geospiza, USA).
PIK3CA/mTOR inhibitors
All inhibitors were purchased from Selleck chemical (Houston, TX 77014, USA). Stock solutions of drugs NVP BKM120 (Selective PI3K inhibitor class1, PI3K𝛼, 𝛽, 𝛾, 𝛿), AZD 8055 (mTOR inhibitor) and BEZ 235 (Dual ATP competitive PI3K and mTOR inhibitor) were prepared in sterile dimethyl sulfoxide (DMSO, Sigma). Treatment of the cells was done by adding the desired amount of agent into the culture medium to gain the desired concentration.
Cell Viability assays
MTT assay
Cytotoxicity of the inhibitors was assessed by MTT based reduction of 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT), a tetrazolium dye [6]. Briefly, cells were seeded at a density of 5 × 103 cells per well in 96 well plates for 24 h. Upon reaching 60–80% confluency, cells were treated with different concentrations of inhibitors (BKM-120, AZD 8055, BEZ 235) for 24 h. At the end of the incubation period with inhibitors, MTT (Cat. M5655; Sigma) solution (5 mg/ml) was added into each well and incubated for 4 h at 37 °C in dark. DMSO was added to wells and placed in a shaker for 10 min. OD was taken at 570 nm on a spectrophotometer (SPECTROstar Nano , BMG Labtech, India). Cell viability was calculated by using the following formula:
Percentage cell viability of the control = OD of the control/OD of the control × 100 and Percentage cell viability of the treated = OD of the treated/OD of the control × 100. After calculating percentage cell viability, the dose at which 50% cells were found to be dead was considered as the inhibitory concentration-50, (IC-50) dose of the inhibitors.
Trypan blue assay
To analyze the effect of inhibitors, cells were seeded (5 × 104 cells/well) in 12 well plates and treated with selected IC50 of inhibitors of all cell lines for 24 h. The cell suspension was then centrifuged and the pellet was suspended in serum-free media. Suspension of cells was suspended in Trypan blue (0.4%) solution (Cat. 15250061, Gibco) in a ratio of 1:1. The mixture was incubated at room temperature for 2 min and loaded into a hemocytometer. Cells (viable: unstained, non-viable: stained) were counted manually. Cell viability was measured using the following formula:
Assessment of apoptosis using flow cytometry
For induction of apoptosis 50,000 Cells/well were seeded in 24 well plates for 24 h. When the cells achieved about 80% confluency, they were treated with a selected concentration of inhibitors. After treatment for 24 h, an Annexin-V/PI assay was done to detect apoptosis. Briefly, cells were collected by centrifugation after 24 h of treatment and transferred into FACS tubes. Cell pellets were suspended in a total volume of 50 μl of the binding buffer (1X), Annexin V (2.5 μl/sample) was added to the suspension of cells and incubated for 30 min in dark. The final volume of each tube was made up to 300 μl by adding 250 μl 1X binding buffer and 0.75 μl Propidium Iodide. Fluorescence was analyzed by Flow cytometer (BD FACS Aria) and interpreted as: Annexin V (+)∕PI (−) = Early Apoptotic cells Annexin V (+)∕PI (+) = Late apoptotic cells PI (+)∕ Annexin V (−) = Necrotic cells PI (−)∕ Annexin V (−) = Live cells.
Statistical analysis
All data are expressed as mean ± S.D. of three independent experiments. Experiments were performed in triplicates. Student’s t-test and one-way variance analysis were applied. Dunnett’s multiple comparison tests were applied to compare the control group with the test. P values of less than 0.05 (typically ≤ 0.05) were considered to be statistically significant.
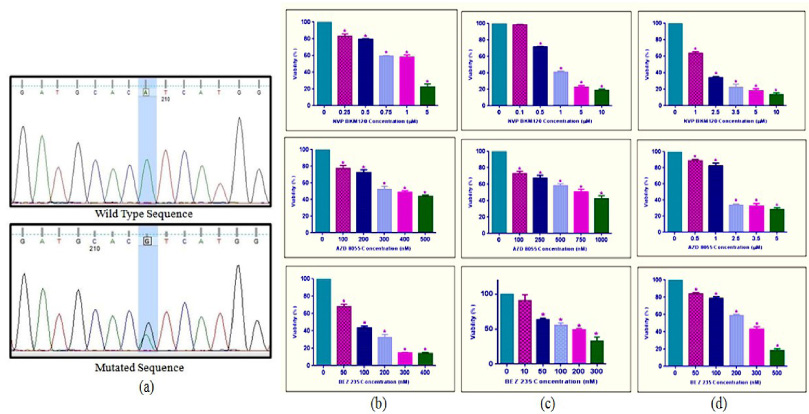
(a) Sequencing chromatogram showing the wild type and mutated sequence of c.3140A>G (p.H1047R) in exon 20 of PIK3CA gene in LAR subtype-specific cell line (MDA-MB453). The inhibitory results of PI3K and mTOR inhibitors, (b) Basal subtype (MDA MB468), (c) Luminal Androgen Receptor subtype (MDA MB453), and Figure (d) Mesenchymal subtype (MDA MB231) showed that treatment with increased concentration of BKM120, AZD 8055 and BEZ 235 reduced viability and growth kinetics in a concentration-dependent manner. Values are shown as mean ±S.D. of three independent experiments. ∗P ≤ 0.05 represents significant changes compared to untreated control.
Subtyping of Cell lines
All cell lines were confirmed to be triple-negative by assessing expression for ER (estrogen), PR (progesterone), and HER2 (human epidermal growth factor receptor 2) by immunohistochemistry (Supplementary Fig. S1). Cell lines were classified into TNBC subtypes with the help of immunohistochemistry markers mentioned in the methodology part. MDA MB231 cell line showed positive expression for panCK and Vimentin and was negative for E-cadherin, hence classified as Mesenchymal subtype. MDA MB468 cell line showed positive expression for CK5/6 and EGFR and negative for CK4/14 hence classified as Basal-like subtype. MDA MB453 cell line showed positive nuclear expression for Androgen receptor, hence classified as LAR (luminal androgen receptor) subtype (Supplementary Fig. S2).
PIK3CA Mutation analysis
Only one non-synonymous pathogenic mutation c.3140 A > G (p.H1047R, COSM775) in exon 20 of PIK3CA was found in the Luminal Androgen Receptor specific cell line (MDA MB453) (Fig. 1a).
Cytotoxicity
To investigate the inhibitory concentration (IC50) of three drugs (BKM 120, AZD 8055, and BEZ 235) on MDA MB468, MDA MB453, and MDA MB231 cell lines, the cells were treated with increasing concentrations of inhibitors for 24 h. Results of inhibition are shown in (Fig. 1b–d). IC50values of the drugs for respective cell lines were found to be: BKM 120 (1.2 μM, 750 nM, 1.8 μM), AZD8055 (300 nM, 740 nM, 2 μM) and BEZ235 (75 nM, 140 nM, 250 nM) for MDA MB468, MDA MB453 and MDA MB231.
MTT assay showed that the number of cells in the inhibitor-treated group was significantly decreased as compared to non-treated cells. But when IC50 of drugs was compared among all subtype-specific cell lines, the Androgen subtype-specific cell line (MDA MB453) was most sensitive to PI3K inhibitors (BKM120) as compared to other cell lines. Mesenchymal subtype-specific cell line (MDA MB231) was the most resistant to all inhibitors compared to others.
The inhibitory effect of all three drugs on cell growth and viability in all TNBC subtype-specific cell lines was also assessed through trypan blue assay (Supplementary Figs S3 and S4). For this, cell lines were treated with selected concentrations of drugs i.e BKM 120 (750 nM), AZD 8055 (300 nM), and BEZ 235 (75 nM). Results showed viability percentage with inhibitors BKM 120 (55%, 52%, and 74%), AZD 8055 (66%, 56%, and 69%), BEZ235 (50%, 54%, and 55%) for Basal-like (MDA MB468), Androgen (MDA MB453) and Mesenchymal (MDA MB231) TNBC subtype-specific cell lines respectively. This showed Mesenchymal subtype-specific cell line (MDA MB231) was less sensitive to all inhibitors as compared to other subtype-specific cell lines and these results are similar to MTT assay results. We also found that the dual PI3K/mTOR inhibitor (BEZ 235) was more effective in less concentration i.e. 75 nM for MDA MB468, 140 nM for MDA MB453, and 250 nM for MDA MB231 than single inhibitors in all cell lines.
Based on MTT and Trypan blue assay’s results, it was assessed whether the cytotoxicity of these inhibitors in three cell lines is likely because of induction of apoptosis or not. Cells were treated with a selected concentration of the inhibitors same as the trypan blue assay. FACS analysis of Annexin-V/PI staining showed that inhibition of PI3K and mTOR significantly increased the proportion of apoptotic cells in all cell lines. NVP BKM120 which is PI3K inhibitor (750 nM) significantly increased annexin-V positive MDA MB468 cells to 3.4% in test compared to 2.2% in un-treated (control), 23.2% MDA MB453 cells in test compared to 0.6% in control and 13.5% MDA MB231 cells in test compared to 3.3% in control (Fig. 2A). AZD 8055, which is a potent mTOR inhibitor (300 nM) showed 5.3% annexin-V positive cells in MDA MB468, 14.4% in MDA MB453, and 12.5% in MDA MB231 (Fig. 2B). BEZ 235, which is a dual inhibitor (75 nM) showed increased apoptotic cells, 8.9% in MDA MB468, 17.6% in MDA MB453, and 12.7% in MDA MB231 compared to controls (Fig. 2C). All values are given as mean ±S.D of two independent experiments∗, P ≤ 0.05 showed significant changes from untreated control. Comparison of Annexin+ cell populations (%) of three TNBC subtype-specific cell lines is given in supplementary Table .
On comparing Annexin V/PI populations (%) in all cell lines after treatment of same concentration of all inhibitors, Androgen receptor subtype (MDA MB453 cells) showed 23.2% apoptotic cells in response to PI3K inhibitor (BKM 120) as compared to 3.4% in Basal (MDA MB468) and 13.5% in Mesenchymal (MDA MB231) cell line. This indicates that the androgen receptor subtype was more sensitive to PI3K inhibitors compared to other subtypes.
Discussion
Triple-negative breast cancer has been characterized as a heterogeneous disease that behaves differently in terms of gene expression, pathological aspects, and biological behavior. Due to this heterogeneity, TNBC poses major challenges in exploring molecular targets and restricted the benefits of various therapies in clinical trials. Major focuses of recent studies have been shifted to molecular subtyping of TNBC using gene expression profiling and to identify the druggable targets for this breast cancer subtype [10]. Subtyping of TNBC by immunohistochemistry may be an alternative method to explore this heterogeneity in TNBC. Our previous study on molecular subtyping of TNBC by IHC showed classification of TNBC into subgroups with type-specific clinicopathological characteristics: LAR (16.7%), Basal-like 1 (13.1%), Basal-like 2 (1.6%), mixed type (Basal + Mesenchymal, 15.1%), Mesenchymal (28.6%) and unclassified (24.9%) [7]. We used the same immunohistochemical markers and classification system for deciphering heterogeneity in TNBC cell lines. In our previous study, we also screened hot spot mutations in PIK3CA and AKT1 in TNBC subtypes and found 16.2% PIK3CA and 2.5% AKT1 mutations [8]. PIK3CA mutations were most frequent in exon 20 and LAR subtype (33.3%). Similarly, we also looked for PIK3CA and AKT1 hot spot mutations in these TNBC cell lines and found pathogenic variant c.3140A > G (p.H1047R) in exon 20 of PIK3CA in LAR subtype-specific cell line (MDA-MB453).
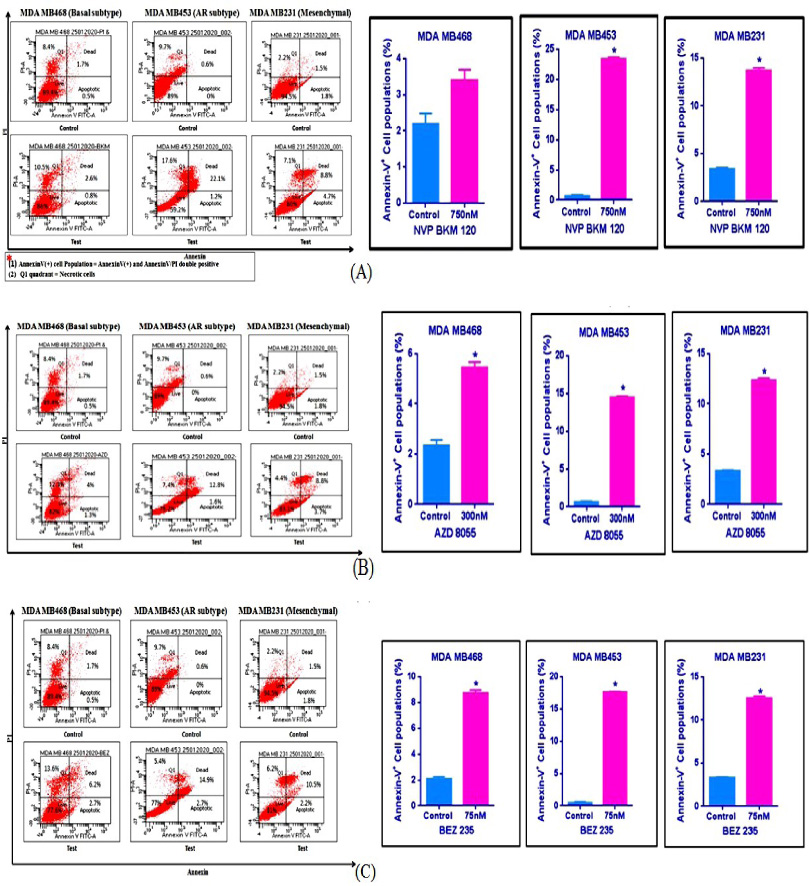
Apoptotic effect of (A) NVP BKM 120 (B) AZD 8055 (C) BEZ 235 inhibitors on TNBC cell lines. Values are shown as mean ± S.D. of three independent experiments. ∗P ≤ 0.05 represents significant changes compared to untreated control.
The development of PI3K inhibitors and several drugs that target PI3K and mTOR are already in clinical trials. In this study, PI3K inhibitor (BKM120), mTOR inhibitor (AZD8055), and dual PI3K/mTOR inhibitor (BEZ235) were used against basal-like (MDA MB468), Androgen (MDA MB453), and Mesenchymal (MDA MB231) subtype-specific TNBC cell lines. On targeting all subtype-specific cell lines with inhibitors, we found differential effects on cell viability. The androgen subtype-specific cell line (MDA MB453) was most sensitive to PI3K inhibitors (BKM 120) as compared to other cell lines measured by IC50values. Sensitivity of PI3K inhibitor in androgen subtype correlated with PIK3CA mutation status. Lehmann et al. [11] reported PIK3CA kinase mutation as a frequent event in AR+ TNBC cell line and showed that growth and viability of AR+ cell line is significantly reduced after treatment with PI3K inhibitors. In addition, we also found that the Mesenchymal subtype-specific cell line (MDA MB231) was the least sensitive to all inhibitors compared to other subtypes.
A combination of two or more therapies that target the cancer cells and their related pathway is the mainstay of cancer treatment [1,15]. Monotherapy is still a very common treatment option in many cancer types; however, this conventional approach is less effective than combination therapy. Conventional single-agent therapies target actively proliferating cells, which includes both cancerous cells and healthy cells. On the contrary, combination therapies act on normal cells with less toxicity and produce cytotoxic effects on tumor cells [9,14]. In the present study, we found that the dual PI3K/mTOR inhibitor (BEZ 235) was more effective in lower concentrations than single inhibitors in all cell lines.
An active apoptotic process conciliated the cell killing induced by inhibitors at the lowest concentration in all TNBC cell lines. The results showed androgen subtype-specific cell line (MDA MB453) was more sensitive to selective PI3K inhibitor (BKM 120) with 23.2% apoptotic cells in response to inhibitor. Similar results were found with AZD 8055 and NVP BEZ235 in androgen specific cell line in which the apoptotic cells were more as compared to other cell lines after treatment with equal concentrations. Here, in the present study with the same low doses, we have shown that cell killing induced by all inhibitors in TNBC cell lines was mediated by apoptosis.
The findings of this study should be elucidated in context of its limitations. In order to validate the results for clinical use, the effects of drugs should be investigated in multiple cell lines and patient samples of each TNBC subtype.
In conclusion, based on the in-vitro experiments, this study identified subtype-specific TNBC cell lines and demonstrated that the LAR subtype of triple-negative breast cancer with PIK3CA mutation may be targeted with PIK3CA inhibitors, especially with the dual inhibitors with a favorable outcome.
Footnotes
Acknowledgements
None.
Conflict of interest
No conflicts of interest to disclose.
Availability of data and material
The datasets are available from the corresponding author on reasonable request.
Consent for Publication
All authors declare their consent to the publication of this work.
Funding
Not applicable.
Code availability
Not applicable.
