Abstract
Evasion of the immune system is the tumor’s key strategy for its maintenance and progression. Thus, targeting the tumor microenvironment (TME) is considered one of the most promising approaches for fighting cancer, where immune cells within the TME play a vital role in immune surveillance and cancer elimination.
FasL is one of the most important death ligands expressed by tumor-infiltrating lymphocytes (TILs) and plays a vital role in eliminating Fas-expressing cancer cells via Fas/FasL pathway-induced apoptosis. However, tumor cells can express elevated levels of FasL inducing apoptosis to TILs. Fas/FasL expression is linked to the maintenance of cancer stem cells (CSCs) within the TME, contributing to tumor aggressiveness, metastasis, recurrence, and chemoresistance.
This study is considered the first study designed to block the overexpressed FasL on the tumor cells within TME mimicking tissue culture system using rFas molecules and supplementing the Fas enriched tissue culture system with blocked Fas - peripheral blood mononuclear cells PBMCs (using anti-Fas mAb) to protect them from tumor counterattack and augment their ability to induce tumor cell apoptosis and stemness inhibition.
A significantly increased level of apoptosis and decreased expression of CD 44 (CSCs marker) was observed within the east tumor tissue culture system enriched with Fas molecules and anti-Fas treated PBMCs and the one enriched with Fas molecules only compared to the breast tumor tissues cultured alone (p < 0.001). Accordingly, we can consider the current study as a promising proposed immunotherapeutic strategy for breast cancer.
Introduction
Breast cancer is the most frequently occurring cancer in women. There are several alterations that tumor cells possess to promote their growth and invasion including; evasion of apoptosis, evasion of immune surveillance, tissue invasion, and metastasis [1]. Several studies demonstrated that the normal development of breast tissues is controlled by a tight balance between apoptosis and cell proliferation [2]. That’s why evasion of apoptosis is considered one of the most important characteristics of tumor cells, which contributes to both tumorigenesis and treatment resistance [3]. Fas is a cell surface molecule that initiates an extrinsic apoptotic pathway upon binding to its specific ligand FasL on the target cell. Moreover, Fas/FasL is the main pathway for apoptosis induced by cytotoxic T lymphocytes (CTLs) during the tumor immunosurveillance mechanism [4]. On the other hand, this apoptotic pathway was found to have a significant role in tumorigenesis, and its impairment in cancer cells may lead to apoptosis resistance and contribute to tumor progression [5]. In addition, it has been demonstrated that the expression of FasL by apoptosis-resistant tumor cells enables a powerful ‘counterattack’ mechanism against the antitumor immune effector cells, such as CTLs, many of which are themselves sensitive to FasL-mediated apoptosis [6].
The concept of the tumor microenvironment (TME) had been extensively studied in the last few decades and a vast majority of studies demonstrated that the heterogeneous complex structure and the cell-cell interactions within the TME are the major factors responsible for tumorigenesis, metastasis, and resistance to therapy. Cancer stem cells are among the vital players within the TME. it was demonstrated that this special cell population has a vital role in tumor aggressiveness, therapy resistance, and metastasis [7]. Stem cell CD44+/CD24- phenotype is considered reliable for the isolation of breast cancer stem cells (BCSCs). Surface markers (CD44, CD24) used for the isolation and identification of BCSCs, not only contribute to the cell interactions but also endow them with unique properties [8].
According to the dynamic bidirectional model of CSCs evolution, non-stem cancer cells (NSCCs) can undergo a de-differentiation process and acquire CSC-like properties. This dynamic equilibrium between CSCs and NSCCs is strongly controlled by the epithelial-mesenchymal transition [9]. Fas and Fas signaling have been connected to normal stem cells [10] and are required for the survival of CSCs allowing new CSCs to emerge [11]. Stimulation of Fas signaling on multiple tumor cells induced a conversion from NSCCs to CSCs. This reprogramming activity of Fas signaling is thought to be independent of its apoptosis-inducing function [11]. Thus we aimed at the current study to investigate the apoptotic activity and stemness potential in our own designed tumor tissue culture system supplemented with recombinant Fas molecules and blocked Fas-peripheral blood mononuclear cells of breast cancer patients (Fig. 1).
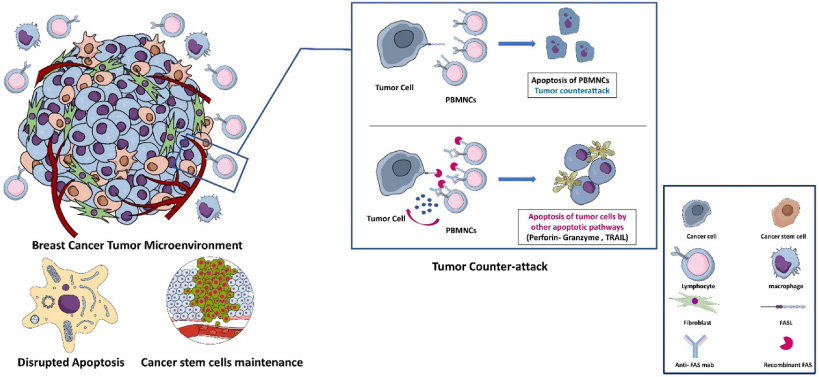
Schematic presentation for the TME, tumor counterattack and the proposed immunotherapeutic strategy for counterattack blocking.
Subjects
The current study was approved by the ethical committee of the medical research institute, Alexandria University, Egypt, and confines the provisions of the declaration of Helsinki. Written informed consent was provided by all subjects under study.
Thirty Egyptian females who were scheduled for modified radical mastectomy for histologically proven breast cancer were recruited from the department of experimental and clinical surgery, Medical Research Institute, Alexandria University. Patients with immunologically mediated diseases that may influence the normal behavior of the immune system and immuno-compromised individuals were excluded.
Methods
Preparation of anti-Fas treated Peripheral blood mononuclear cells (PBMCs)
Under strictly aseptic conditions Peripheral blood mononuclear cells (PBMCs) were isolated from heparinized blood, freshly drawn from all patients under study by density gradient centrifugation over Ficoll-Histopaque density gradient (1077) (Seromed, Germany) [12]. The viability of the isolated cells was then tested by “Trypan blue dye exclusion technique to assure that an acceptable number of viable isolated cells are obtained [13]. Isolated PBMCs count was adjusted and incubated with anti-Fas monoclonal antibodies (Thermo Scientific, USA) in final concentration (15 μg/106 cells) [14].
Breast normal and tumor tissue cultures
Under aseptic conditions, a fresh sterile tissue sample of the primary breast tumor was obtained from each patient immediately after surgical dissection, according to the approved medical ethics of Alexandria University. Each tumor sample was divided into two parts, one part for the routine histopathological studies, H&E staining, and hormonal receptors status detection, and the other part was maintained in an organ transportation medium on ice until use for the tissue culture. The breast tumor tissues were then embedded in complete RPMI media (Lonza, Belgium). Equal volumes of each tumor tissue sample were then cultured in a 96-well tissue culture plate according to the following design: Breast tumor tissues cultured in complete RPMI media alone. (T) Breast tumor tissues cultured in complete RPMI media supplemented with 2 μg/ml recombinant Fas molecules. (TS) Breast tumor tissues cultured with anti-Fas treated Peripheral blood mononuclear cells (PBMCs) in complete RPMI media supplemented with 2 μg/ml recombinant Fas molecules. (TSL)
The same designed tissue culture systems were performed using breast normal tissue samples excised from the same breast (N, NS & NSL). Tissue-cultured samples were then incubated at 37° C in a constant atmosphere of 5% CO2 for 24 hours [15]. After the incubation period, the tumor and normal cultured tissues were then fixed in 10% phosphate-buffered formalin PH 7.4 for 24 hours and then processed for preparation of-microscopic slides used for immunological investigations (Fig. 2).
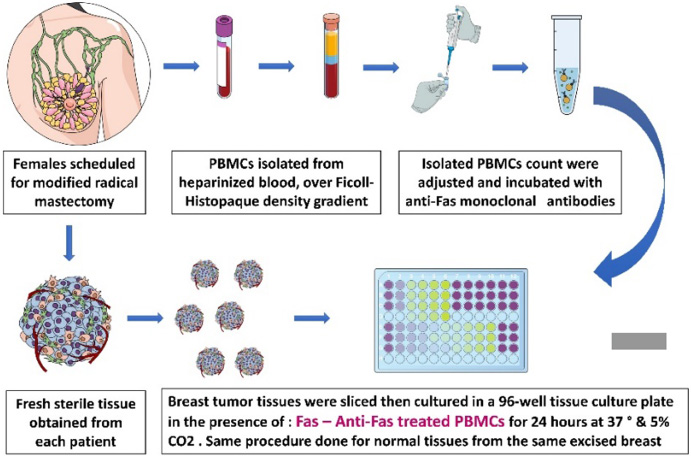
Schematic diagram of PBMNCs separation and tissue culture system designed.
Tissue sections were deparaffinized, rehydrated and slides were then incubated in 3% Hydrogen Peroxide Block H (Thermo Biochemicals, USA) and then placed in retrieval sodium citrate buffer 0.01M, pH: 6.0 and heated at 100° C then washed with PBS. Afterward, they were incubated with ultra V block (Thermo Biochemicals, USA) diluted 1:5 in PBS for 30 minutes to reduce the nonspecific binding of the conjugated secondary antibody.
The sections were then incubated with ”Rabbit polyclonal antibody for caspase-3 (NeoMarkers-Lab Vision Corporation, USA) (5 μg/ml) at 4° C in a humid chamber. Washed and incubated with Biotinylated goat anti-polyvalent secondary antibody (Thermo Biochemicals, USA) for 10 minutes at room temperature. Washed again and Streptavidin peroxidase (Thermo Biochemicals, USA) was applied for 10 minutes at room temperature.
One drop (40 μl) of DAB plus chromogen (Skytek Laboratories, USA) was added to 2 ml of DAB Plus substrate (Skytek Laboratories, USA), mixed by swirling, and applied to the tissue. The slides were then incubated for 5–15 minimum, depending on the desired stain intensity. The slides were finally counterstained in Mayer’shematoxylinn and coverslipped using a permanent mounting media.
The DAB chromogen yielded a brown color reaction end product at the site of the target antigen, the immunostain was expressed according to a semi-quantitative scale. The tumor samples were graded as negative when there was a complete absence of nuclear staining. The positive tumor samples were graded as negative (0) weak (1), moderate (2), and strong (3) according to the degree of nuclear and cytoplasmic staining [16].
Immunofluorescence detection of CD44 and CD24 expression
Tissue slides were deparaffinized, rehydrated, and incubated with retrieval sodium citrate buffer 0.01M, pH: 6.0 as previously described. The slides were then incubated with 1% bovine serum albumin (Euroclone, Italy) in phosphate buffer saline for 30 minimum to block the unspecific binding of the antibodies. The two primary antibodies were mixed at the right dilutions in PBS (CD24 at 1:200, CD44 at 1:50) in the same tube (eBioscience, UK), applied on the tissues and incubated overnight at 4° C.
The secondary antibodies, Alexaflour 488- conjugated anti-rat IgG (minimal cross-reaction to human, mouse, horse, bovine, and rabbit serum proteins) (Jackson Laboratories, USA) and Alexaflour 594-conjugated anti mouse IgG (minimal cross reaction to human, rat, horse, bovine and rabbit serum proteins) (Jackson Laboratories, USA) were mixed and diluted in PBS (1:300). The mixture was then added to the sections and the slides were incubated in the dark for 60 minutes and then washed twice in PBS for 5 minutes. Finally the slides were mounted by fluorescent mounting medium containing DAPI (eBioscience, UK) and examined by fluorescent microscope [17]. The slides were then photographed and the fluorescence intensity of the stain was calculated using imagej photos analyzing software as follows [18]:
Statistical analysis
Data were fed to the computer and analyzed using IBM SPSS software package version 20.0 (Armonk, NY: IBM Corp) [19]. Qualitative data were described using number and percent. The Kolmogorov-Smirnov test was used to verify the normality of distribution. Quantitative data were described using range (minimum and maximum), mean, standard deviation and median. The significance of the obtained results was judged at the 5% level. The used tests were: Friedman test: For abnormally distributed quantitative variables, to compare more than two studied groups. Mann Whitney test: For abnormally distributed quantitative variables, to compare between two studied groups. Spearman coefficient test: to correlate between two abnormally distributed variables.
Results
Clinicopathological Parameters
The clinicopathological parameters of the recruited subjects are summarized in Table 1.
The clinicopathological data of the patients enrolled in the study
The clinicopathological data of the patients enrolled in the study
According to the Friedman test, there is a significant increase in the apoptosis level detected within the (TSL) and (TS) tissue cultures compared to (T) tissue culture (mean ranks 2.9, 2 and 1.1 respectively, p < 0.0001) on the other hand, no significant difference was observed in the level of apoptosis within the breast normal tissue culture systems according to Friedman test (mean ranks 1.8, 2.1 and 2.2, p = 0.272) (Figs 3, 4).
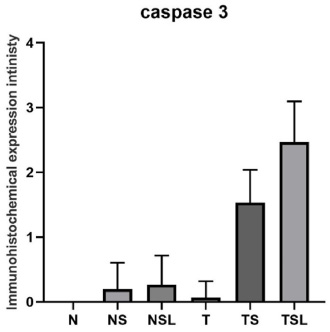
The level of caspase 3 staining within normal & tumor tissue culture systems. N: Breast normal tissue culture system alone. NS: Breast normal tissue culture system supplemented with recombinant FAS molecules. NSL: Breast normal tissue culture system supplemented with recombinant FAS molecules and anti-Fas treated PBMCs. T: Breast tumor tissue culture system alone. TS: Breast tumor tissue culture system supplemented with recombinant FAS molecules. TSL: Breast tumor tissue culture system supplemented with recombinant FAS molecules and anti Fas treated PBMCs.
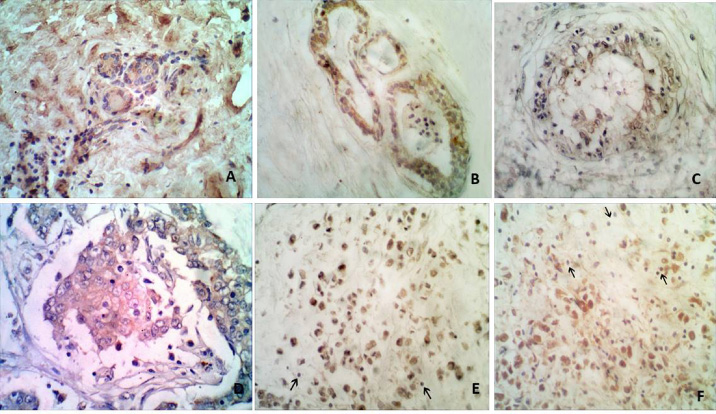
Immunohistochemical analysis of tumor and normal breast tissue culture systems for Caspase 3 examined and imaged by Olympus Light Microscope, A: Breast normal tissue cultured alone (N) showing negative staining for caspase-3. (IHC-caspase × 200) B: Breast normal tissue culture enriched with recombinant Fas molecules (NS) showing negative nuclear staining for caspase 3. (IHC × 200) C: Breast normal tissue culture enriched with recombinant Fas molecules and anti-Fas treated PBMNCs (NSL), showing mild cytoplasmic staining and negative nuclear staining with negative lymphocytic staining for caspase 3. (IHC × 400) D: Breast tumor tissue cultured alone (T), a case of invasive ductal carcinoma showing negative staining for caspase 3. (IHC × 400) E: Breast tumor tissue culture enriched with recombinant Fas molecules (TS). A case of invasive ductal carcinoma section showing moderate positive staining for caspase-3 among tumor cells with negative staining within the infiltrating lymphocytes. (IHC × 400) F: Breast tumor tissue culture enriched with recombinant Fas molecules and anti-Fas treated PBMCs (TSL). High power view showing scattered mixed tumor cells showing strong staining and infiltrated lymphocytes showing negative staining for caspase-3 (IHC × 400).
There is no significant difference detected between the apoptosis level within the breast tumor tissues cultured alone (T) compared to the breast normal ones (N) (p = 0.77) (Table 2). While a significant increase in the apoptosis level detected within (TS) compared to (NS) (p < 0.0001), and a significantly increased apoptosis level was detected in (TSL) compared to (NSL) (p < 0.0001) (Table 2).
Comparison between apoptosis levels in breast tumor tissue culture systems and breast normal tissue ones
Comparison between apoptosis levels in breast tumor tissue culture systems and breast normal tissue ones
∗: p value for Mann Whitney -U test∗∗: Statistically significant at p ≤ 0.05.
Concerning the level of CD 44 expression in breast tumor tissue culture systems, a significant difference was observed between the fluorescence intensity within the three breast tumor tissue culture systems, where the mean ranks of Friedman test between T, TS & TSL are 3,1.7&1.3 respectively (p < 0.001) while the difference in the level of expression of CD24 within the three breast tumor tissue culture systems T, TS and TSL was shown to be insignificant according to Friedman test with mean ranks 1.8, 2.3 & 1.9 respectively (p = 0.282), reflecting a significant decrease in the number of breast cancer stem cells (BCSCs) within the TME (Figs 5, 6, 7, 8).
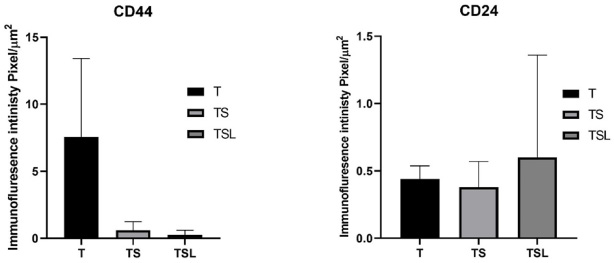
The fluorescence intensity of CD44 & CD24 staining within the breast tumor tissue culture systems.
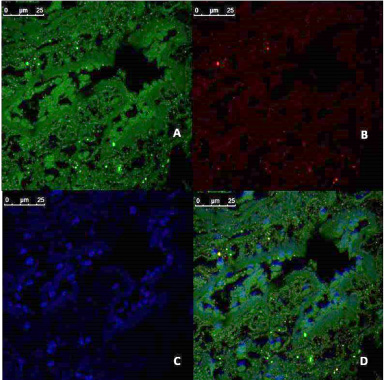
Immunofluorescences analysis of CD44/CD24 expression in tumor tissue culture system alone (T), examined and imaged by Leica Confocal microscope. A: CD44 expression, showing fluorescence intensity ≥10 pixel/μm2 for Alexaflour 488 staining (Green) indicating high CD44 expression. (Confocal microscopy, 600 ×) B: CD24 expression, showing fluorescence intensity <1 pixel/μm2 for Alexaflour 594 staining (Red), indicating the absence of CD24 marker expression. (Confocal microscopy, 600 ×) C: DAPI nuclear staining (blue) (Confocal microscopy, 600 ×) D: merged image showing the abundance of CD 44+/CD 24- BCSCs. (Confocal microscopy 600 ×).
Statistical analysis was done to reveal the correlation between the immunofluorescence intensity of CD44 expression and CD24 expression within the different breast tumor tissue culture systems. It showed that there is an insignificant negative correlation between the immunofluorescence intensity of CD44 and the CD24 expression within the (T) and (TS) tissue culture systems. (r s = −0.08844, p = 0.754 and r s = −0.152, p = 0.587 respectively) (Table 3) While within the TSL tissue culture system, there was an insignificant positive correlation between the immunofluorescence intensity of CD44 and the CD24 expression. (r s = −0.048, p = 0.864)
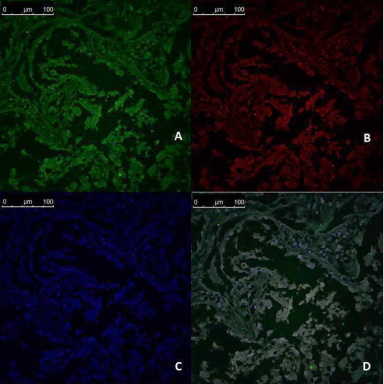
Immunofluorescences analysis of CD44/CD24 expression in breast tumor tissue culture system enriched with recombinant FAS molecules (TS), examined and imaged by Leica Confocal microscope A: CD44 expression, showing fluorescence intensity <1 pixel/μm2 for Alexaflour 488 staining (Green) indicating low CD44 expression. (Confocal microscopy, 200 ×) B: CD24 expression showing fluorescence intensity <1 pixel/μm2 for Alexaflour 594 staining (Red), indicating the absence of CD24 marker expression. (Confocal microscopy, 200 ×) C: DAPI nuclear staining (blue) (Confocal microscopy, 200 ×) D: Merged image showing the absence of CD 44+/CD 24- BCSCs. (Confocal microscopy 200 ×).
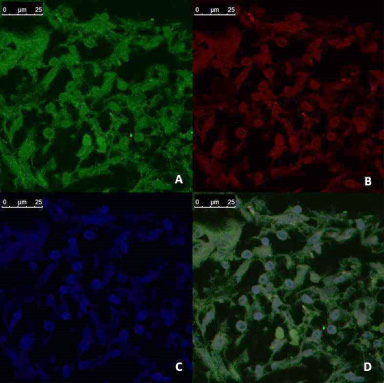
Immunofluorescences analysis of CD44/CD24 expression in breast tumor tissue culture system enriched with recombinant FAS molecules and anti-Fas treated PBMNCs (TS), examined and imaged by Leica Confocal microscope A: CD44 expression showing fluorescence intensity <1 pixel/μm2 for Alexaflour 488 staining (Green) indicating low CD44 expression. (Confocal microscopy, 600 ×) B: CD24 expression, showing fluorescence intensity 1–5 pixel/μm2 for Alexaflour 594 staining (Red) indicating moderate CD24 marker expression. (Confocal microscopy, 600 ×) C: DAPI nuclear staining (blue) (Confocal microscopy, 600 ×) D: Merged image showing the absence of CD 44+/CD 24- BCSC (Confocal microscopy 600 ×).
Correlation between immunofluorescence intensity of CD44 and CD24 expression
∗: P value for spearman rho test. ∗∗: Statistically significant at p ≤ 0.05.
Correlation between the measured immunological parameters within the breast tumor culture systems and the patients’ clinicopathological parameters.
Correlation between the measured immunological parameters within the breast tumor culture systems and the patients’ clinicopathological parameters.
∗: P value for spearman rho test. ∗∗: Statistically significant at p ≤ 0.05.
Statistical analysis was done to reveal the correlation between the immunofluorescence intensity of CD44 expression and CD24 expression and the level of apoptosis within the different breast tumor tissue culture systems. It showed that there is an insignificant negative correlation between the level of apoptosis and the immunofluorescence intensity of CD44 expression within the three breast tumor tissue culture systems T, TS and TSL. While an insignificant positive correlation was found between the level of apoptosis and the immunofluorescence intensity of CD24 expression within the breast tumor tissue culture systems (Table 5).
Correlation between immunofluorescence intensity of CD44 and CD24 expression and apoptosis levels within the breast tumor culture systems
Correlation between immunofluorescence intensity of CD44 and CD24 expression and apoptosis levels within the breast tumor culture systems
∗: p value for spearman rho test. ∗∗: Statistically significant at p ≤ 0.05.
Breast cancer tumor microenvironment (TME) consists of a variety of cellular and molecular elements interacting with each other and deciding the fate of the tumor. It is well established that TME tends to be immunosuppressive rather than immune-supportive via different overlapping mechanisms [20]. Among these mechanisms, tumor cells overexpress FasL that induce apoptosis to Fas expressing tumor-infiltrating peripheral blood mononuclear cells (PBMCs) through Fas/FasL apoptotic pathway instead of being killed themselves through the same pathway that is known as tumor counterattack [6]. In addition, PBMCs are more or less disabling cells that can’t achieve their anti-tumor function properly [21]. Moreover, Fas/FasL apoptotic pathway has been demonstrated to have a role in the progression of cancer stem cells (CSCs) which are responsible for tumor recurrence as well as chemo- and immune-therapy resistance [22].
So, we aimed in the current study to investigate the apoptotic activity and stemness potential in our own designed tumor tissue culture system supplemented with recombinant Fas molecules and blocked Fas- peripheral blood mononuclear cells of breast cancer patients.
To achieve this goal, firstly; our designed tissue culture system was enriched with recombinant Fas molecules to block the overexpressed FasL on the tumor cells preventing them from counterattacking TILs within the TME. This step allows the TILs to perform their vital role in immune surveillance and tumor elimination more effectively (TS tissue culture system). Secondly; isolated peripheral blood mononuclear cells (PBMCs) from peripheral blood samples withdrawn from each were treated with anti-Fas monoclonal antibodies (mAbs) to block their Fas receptors and protect them against killing by tumor cell-inducing Fas /FasL apoptosis. These protected PBMCs were infused into the Fas- enriched TME (TSL tissue culture system). Levels of apoptosis and markers of CSCs (CD44, CD24) were detected in all the tumor tissue culture systems of the study.
The tumor tissue culture system designed in our study gave us the ability to study the interaction between different cell populations and cytokine networks within the TME. It is now widely accepted that interaction between different elements is a must for both tumor growth and progression on one hand and designing an effective treatment strategy on the other hand [23]. So in contrast to the conventional cell lines used in the most of published studies, our designed tissue culture system enables us to manipulate the actual TME.
Results of the current study revealed a significant increase in the apoptosis level detected within the breast cancer tissue culture system supplemented with recombinant FAS molecules and anti-Fas treated peripheral blood mononuclear cells (PBMCs) (TSL) and breast cancer tissue culture system supplemented with recombinant FAS molecules (TS) compared to breast cancer tissue culture system alone (T) and insignificant difference in the level of apoptosis within the breast normal tissue culture systems that could be explained by two hypotheses. The first hypothesis is that by blocking the FasL within the TME, the tumor-promoting activities of FasL within the TME were successfully inhibited and abolished the tumor-promoting Fas signaling on tumor cells.
That tumorigenic non-apoptotic signaling of FasL was shown to promote tumor growth and favor the establishment of tumor metastases [24], via the involvement of multiple potentially tumorigenic signaling pathways such as the activation of NF-𝜅𝛽 and the three major mitogen-activated protein kinases (MAPK), Erk1/2, JNK1/2, and p38, as well as the Src/PI3K/GSK3𝛽/MMP (matrix metalloproteinase) pathway [25,26] as well as abolishing the anti-tumor activity of various cells such as NK cells [27], where it is now widely accepted that once cancer cells acquire resistance to Fas-mediated apoptosis, any further stimulation of Fas is tumorigenic [28].
Furthermore, FasL is expressed in two forms, membrane-bound and soluble form that is generated through the cleavage of membrane-bound FasL by metalloproteinases. It was reported that the membrane-bound FasL is the one responsible for the apoptotic activities while the soluble form promotes tumor invasion [29]. On the other hand, soluble FasL was found to be upregulated in breast cancer patients after the administration of the first chemotherapeutic cycle indicating a possible role in the recurrence and metastasis following the chemotherapy cycles [30].
In addition, tumor progression and invasiveness are increased by the expression of FasL by tumor cells and also as a result of over-expression by different cells within the TME such as CTLs. These cells were demonstrated to be expressing a high level of FasL within the TME, and not only have diminished ability to induce tumor cells’ apoptosis but also induced tumor cells to secrete PGE2 and increase tumor cell chemoattraction of MDSCs via Fas signaling to form an immunosuppressive TME, suggesting that CTLs infiltrated to the TME may participate in the process of tumor immune evasion [31].
The second hypothesis explaining the significantly increased apoptosis within the tumor tissue culture system enriched with recombinant Fas molecules compared to the untreated ones is that this strategy successfully prevented the tumor counterattack mechanism within the TME, where blocking FasL prevented the ability of the tumor cells to induce apoptosis in the TILs through the Fas/FasL pathway giving them the chance to induce tumor cells’ apoptosis through other pathways such as tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)/Death receptor (DR) pathway or the perforin granzyme pathway.
It was reported that FasL expression by apoptosis-resistant tumor cells enables a powerful ‘counterattack’ against antitumor immune effector cells, such as CTLs, many of which are themselves sensitive to FasL-mediated apoptosis, which support the immunosuppressive role of FasL in the TME [6,32,33]. In addition to the membrane-bound FasL, the soluble form of FasL besides its tumor-promoting activities was reported to have a role in the tumor counterattack within the TME as well [5]. Tumor counterattack mediated by FasL expression may not be only restricted to the cancer cell, many types of non-tumorigenic cells may be involved as well such as regulatory T-cells (CD4+ CD25+Treg) [34], tumor endothelial cells [35] and even tumor secreted pro-apoptotic exosomes expressing FasL on their surface, that possess the ability to eliminate activated T-cells by a simple ligand–receptor interaction [36]. Thus we can suggest that blocking the Fas/FasL pathway within the TME is more beneficial than its induction, which will result in immune modulation to enhance the immune response within the TME that in turn will support tumor regression and decrease recurrence and metastasis.
The significantly increased apoptosis level in breast tumor tissue culture enriched with recombinant FAS molecules and anti-Fas treated PBMCs (TSL) culture than that in both breast tumor tissue culture enriched with recombinant Fas molecules (TS) and breast tumor tissue culture alone (T) tissue cultures is expected as in this tumor tissue culture system (TSL) not only we blocked the FasL within the TME to prevent the tumor counterattack mechanism, but also we infused the patients’ own peripheral blood mononuclear cells (PBMCs) after blocking their Fas receptor to the TME. These PBMCs can effectively induce apoptosis to the cancer cells without being at risk to be killed themselves by apoptosis through Fas/FasL mechanism.
This tissue culture system designed in the current study can be considered a first step in developing an effective adoptive immunotherapeutic strategy against breast cancer. Immunotherapy holds much promise for the treatment of various types of cancer. Adoptive immunotherapy, involving the transfer of ex vivo activated autologous T-cells, has shown promise for the treatment of various types of cancers [37]. Putting into consideration that T cells are capable of homing to tumor sites throughout the body, provides its use an advantage over antibodies [38]. It is a highly personalized cancer therapy that had mediated durable and complete regressions in patients with melanoma. In addition, the ability to genetically engineer lymphocytes to express conventional T-cell receptors or chimeric antigen receptors has further extended the successful application of adoptive cell therapy for cancer treatment [39].
But on the other hand, multiple inhibitory mechanisms were reported to restrain endogenous TILs and modified T-cells used for adoptive therapy from effectively eradicating tumor, such as the upregulation of regulatory Tregs and myeloid-derived suppressor cells as well as the upregulation T cells inhibitory receptors CTLA-4 and PD-1 [40]. Overexpression of those checkpoints within the TME has been demonstrated to hinder endogenous T-cells from performing their vital duties [41].
So, blocking Fas expressed on the administered PBMCs in the current study can be also considered as a type of checkpoints inhibitors, where blocking Fas receptor unleash the breaks that limit the CTLs activities within the TME which in turn enhances its ability to perform its antitumor response.
In addition, the selectivity of the breast tissue culture systems designed in the current study in the induction of tumor cells’ apoptosis sparing the normal cells unharmed could encourage the proceeding to the next phase trails of the proposed immunotherapeutic strategy. This selectivity could be explained by the fact that the strategies proposed within the different breast tissue culture systems are dealing with FasL-expressing cells and it is well documented that tumor cells have higher expression of FasL compared to normal ones [42]. Moreover, putting into consideration that the designed tissue culture systems don’t induce apoptosis within the TME but only they pave the road for the immune system to perform its natural immunosurveillance mechanisms, could clarify the observed selectivity.
The decreased expression of CD44 in the designed breast tumor tissue culture systems can be explained also by two hypotheses. The first hypothesis is that blocking FasL on the tumor cells leads to shifting the cells’ dynamic equilibrium towards the differentiation side, where this blocking leads to the increase in the differentiation of BCSCs to NSCCs through mesenchymal-epithelial transition (MET) and losing their stemness traits. What makes this immunotherapeutic strategy a promising one, is that targeting cancer stem cells is a very tempting goal, where aiming at this cell population within the TME, will affect in turn two important hallmarks of cancer proposed by Hanahan et al. (2000) [43]; limitless replicative potential, invasion, and metastasis. In addition, targeting the CSCs will eradicate the cells responsible for tumor recurrence and chemo-, radio-, and immunotherapy resistance which will enhance the chance of complete tumor remission [43].
Although different studies showed success to some extent in apoptosis induction within the CSCs population [44,45], other studies demonstrated that some apoptotic pathways such as the Fas/FasL pathway may have a role in CSCs progression [11], and its stimulation induced a conversion from NSCCs to CSCs [11]. It also prevented the differentiation of CSCs back to NSCCs, which makes the downregulation of the death ligands and death receptors a more profitable anti-tumor immunotherapeutic strategy. That CSCs maintenance role of FasL was correlated to several signaling pathways such as AKT, ERK, and STAT1 [46], and to several known EMT marker genes such as VIM, FN1, and SNAI2 [47].
The second hypothesis explaining the significantly decreased fluorescence intensity of BCSC marker CD44 in TSL and TS compared to T is the ability of those immunotherapeutic strategies to eliminate BCSCS either by apoptosis or DICE (death induced by CD95R/L elimination) where it is demonstrated that when either Fas or FasL are eliminated, cancer cells die through a process called DICE (death induced by CD95R/L elimination). DICE is a necrotic form of mitotic catastrophe programmed cell death characterized by cell swelling and ROS production followed by DNA damage, activation of caspase-2, and loss of mitochondrial outer membrane potential (MOMP) [48]. Interestingly it was illustrated that CSCs and NSCSCs have differential sensitivities to Fas-mediated apoptosis and DICE. Where NSCCs are more sensitive to Fas-mediated apoptosis than CSC, and in contrast CSCs are more sensitive to be killed by DICE compared to NSCCs. Suggesting that the killing of cancer cells can be maximized by concomitant induction of both cell death mechanisms [11].
These findings may explain the significantly increased apoptosis levels within the TSL tumor tissue system in our study on one hand (targeting NSCC apoptosis by anti-Fas treated PBMCs) and significantly decreased BCSCs’ marker CD44 fluorescence intensity (targeting CSCs by DICE through FasL blockage) on the other hand. Supporting the proposed hypotheses, the insignificant negative correlation observed between the immunofluorescence intensity of CD44 and the CD24 expression within the (T) and (TS) tissue culture systems FasL blockage leads to the shift in the dynamic equilibrium between CSCs and NSCCs to the differentiated side. While within TSL tissue culture system, there was an insignificant positive correlation between the immunofluorescence intensity of CD44 and the CD24 expression indicating that in this tumor tissue culture system, the decreased level of CD44 expression is associated with a decrease in the CD24 expression as well supporting our second hypothesis that this tissue culture system targets CSCs within the TME either by apoptosis or DICE.
Several studies had identified and isolated BCSCs with the aid of CD44 and CD24 markers, where the BCSCs population was found to be CD44+/CD24−∕low [49,50 ]. Although it was reported that the shift in the dynamic equilibrium between CSCs and NSCSs is accompanied by a decrease in CD44 expression on one hand and an increase in CD24 expression on the other hand, suggesting that CD24 could be a NSCC marker in breast cancer [51,52 ], other studies failed to correlate CD24 expression with NSCCs abundance within the TME [53 ]. Putting into consideration that the tumor tissue culture systems designed in the current study not only did they have the ability to shift the dynamic equilibrium toward the differentiated side but also they could induce cancer cells elimination within the TME either by apoptosis or DICE, it will be inaccurate to judge the expression of CD24 marker.
Regarding the correlation between the immunofluorescence intensity of CD44 expression and the patients’ clinicopathological parameters, a significant negative correlation was observed within TSL breast tumor tissue culture in contrast to the insignificant positive correlation obtained within the control T tissue culture, suggesting that in the breast tumor tissues cultured alone, the level of abundance of BCSCs is directly proportional to its ability to reach the vasculature, and upon its culture with recombinant Fas molecules alone or in the presence of anti-Fas treated PBMCs this correlation is inversed, this could be owed to the efficacy of the two designed tissue culture systems in eliminating BCSCs population.
Furthermore, the statistical analysis done to reveal the correlation between the immunofluorescence intensity of CD44, CD24 expression and the level of apoptosis within the different breast tumor tissue culture systems, indicates that the level of abundance of BCSCs reflected by the level of CD44 expression is inversely proportional to the level of apoptosis within the TME, which supports previous studies that linked the population of CSCs to apoptosis resistance. It also refers to the successful targeting of the CSCs population within the two tumor tissue culture systems designed in our study TS and TSL that seemed to suppress this resistance. In addition, decreased CD44 expression is associated with increased CD24 expression thus losing stemness and increased CD24 expression is directly proportional with the increased level of apoptosis within the breast tumor tissue culture systems.
Accordingly, from the above findings of our study, we suggest that the breast tumor culture systems designed and examined in the current study (TS & TSL) can effectively and selectively induce apoptosis in breast tumor cells while sparing the breast normal cells unharmed, and have the ability as well to eliminate BCSCs population within the TME with the superiority of the breast tumor tissue culture system enriched with both recombinant Fas molecules and anti-Fas treated PBMCs. Further studies are recommended for the characterization of the PBMNCs used, in addition to studies to reveal the mechanistic signal transduction pathways involved in the immunotherapeutic strategies proposed.
Footnotes
Ethical statement:
The current study was approved by the ethical committee of the medical research institute, Alexandria University, Egypt, and confines the provisions of the declaration of Helsinki. Written informed consent was provided by all subjects under study.
Conflict of interest:
All of the authors declared no conflict of interest.
Funding information:
This study is not funded by any institution or a funding body.
Author contributions
Abou Shousha S designed the study, developed the methodology, performed the experiments, interpreted the experiments data, wrote and revised the manuscript. Ghoneim H interpreted the immunofluorescence results, Zohir M interpreted the histology and IHC data. Hemida M provided the surgical tissue samples; Baheeg S participated in the practical and tissue culture part of the study. Shahine Y performed the experiments and the statistical analysis of data and wrote the manuscript.
