Abstract
BACKGROUND:
The role of gluconeogenesis in cancer cells as the reverse pathway for glycolysis is not well known. Several studies of gluconeogenesis in cancer cells still show conflicting results. Expression of key enzymes such as FBP1 and LDHB in cancer tissues may explain the role of gluconeogenesis in tumor development.
OBJECTIVE:
This study aimed to analyze the expression of FBP1 and LDHB in fibroadenomas and invasive cancers of the breast.
METHODS:
The immunohistochemical staining technique was used to show the expression of FBP1 and LDHB in formalin-fixed, paraffin-embedded blocks of 32 fibroadenomas and 31 invasive breast cancer samples.
RESULTS:
FBP1 was expressed by the majority of fibroadenoma (68.7%) and invasive breast cancer (71%) samples. LDHB expression in fibroadenomas was significantly higher than in invasive breast cancers (P = 0.029). The expression of these two enzymes was found in invasive, lobular, and tubular breast carcinoma, and at well, moderately, and poorly differentiated breast malignancy.
CONCLUSIONS:
High expression of FBP1 and LDHB was found in fibroadenomas and invasive breast cancers. A higher level of LDHB expression was observed in fibroadenomas. These results may indicate the enzymes’ role in the pathogenesis of both breast diseases.
Introduction
Gluconeogenesis is de novo glucose synthesis from non-carbohydrate precursors such as glucogenic amino acids, pyruvate, lactate, and glycerol. This pathway is usually inhibited in cancer cells due to the activation of the glycolysis pathway [1,2]. However, many studies found that cancer cells are likely to adapt to the available nutrients from their microenvironment. Glucose supply fluctuates in the tumors. Therefore, gluconeogenesis should provide intratumoral glucose independently without the participation of gluconeogenic organs such as the liver and kidneys. Cancer cells can synthesize crucial metabolites using the abbreviated forms of gluconeogenesis [1–3]. Our study started by questioning the extent to which the activation of gluconeogenesis plays a role in the development of both benign and malignant breast tumors.
Fructose-1,6-bisphosphatase (FBP) is one of the main enzymes in the gluconeogenesis pathway. This enzyme catalyzes the hydrolysis of fructose 1,6 bisphosphate into fructose 6-phosphate and inorganic phosphate. This reaction is specific for FBP because different enzyme catalyzes the reversal reaction of the glycolysis pathway [4,5]. This enzyme has two isoenzymes, FBP1 and FBP2. Of these two enzymes, the FBP1 isoenzyme acts as a rate-limiting enzyme in gluconeogenesis, while FBP2 plays a role in glycogen synthesis. Several studies have shown that FBP1 may inhibit tumorigenesis [4,6–8]. Lactate dehydrogenase (LDH) is one of the primary metabolic enzymes that function of converting pyruvate to lactate and vice versa. There are two isoforms of this enzyme, firstly LDHA, expressed mainly in skeletal muscle with the preference of converting pyruvate to lactate. Secondly, LDHB, that is primarily expressed by the heart and brain, and preferentially converting lactate to pyruvate [9,10]. Several studies have shown an increase in LDHB expression associated with staging, therapeutic response, recurrence, metastasis, and prognosis with varying results [9,11–13]. The metabolic preference of LDHB converting lactate to pyruvate shows this enzyme’s role in the initial steps of the gluconeogenesis pathway.
Studies on the role of glycolysis (Warburg effect) in cancer development have been carried out quite a lot. Many results on this topic have been developed as new approaches for cancer treatments. Furthermore, studies on gluconeogenesis are growing recently, based on the evidence of a very high metabolic adaptability of cancer cells. Limited studies showed that cancer cells could utilize gluconeogenesis enzymes to grow and survive. This evidence is fascinating to be exploited further, considering the critical role of gluconeogenesis as a glucose-producing pathway, in which glucose is the primary energy substrate as well as the biosynthetic material for cancer cells. This pathway appears to have great potential as a new therapeutic target in efforts to increase the effectiveness of cancer treatment.
In this study, we aimed to analyze the expression levels of FBP1 and LDHB in the tissue specimens of benign breast tumor (fibroadenoma) and invasive breast cancer patients. We need to know whether the expression of FBP1 and LDHB was specific to invasive breast cancers or whether the benign tumors also expressed these gluconeogenic enzymes. We expected this research could be our starting point to reveal the significant role of the gluconeogenesis pathway in both benign and malignant breast tumor pathogenesis.
Methods
Tissue samples
The samples used in this study were the formalin-fixed, paraffin-embedded blocks of fibroadenoma, and invasive breast cancer tissues. These were the sample collections of The Laboratory of Pathology Anatomy Hasanuddin University Teaching Hospital. The samples were collected based on the patient’s medical record for one year from January 2017–December 2018. During this period, 63 samples met the criteria and had complete clinical information, consisted of 32 samples of fibroadenoma and 31 samples of invasive breast cancer. The use of this sample has fulfilled the requirements of the ethics committee of the Faculty of Medicine, Hasanuddin University, with an ethical approval number 246/UN4.6.4.5.31/PP36/2019.
Immunohistochemistry staining
Immunohistochemistry (IHC) examination of FBP1 and LDHB was performed using a rabbit monoclonal antibody to FBP1 (Abcam Anti-FBP1 antibody [EPR4619]; ab109020; 1:100) and mouse monoclonal antibody to Lactate Dehydrogenase B (Abcam Anti-Lactate Dehydrogenase B/LDH-B antibody [60H11]; ab85319; 1:100). Tissue sections of the samples were cut to a thickness of 4 μm, affixed to the slide, then deparaffinized in xylene, and rehydrated in a list concentration of alcohol. The specimens were then incubated in the microwave oven for ten minutes, then cooled at room temperature for thirty minutes and rinsed with phosphate-buffered saline (PBS) solution. The specimens were dipped in 0.5% hydrogen peroxide solution for ten minutes to block endogenous peroxidase. After rinsing with PBS solution, the primary antibodies were added on the specimens and incubated for sixty minutes. The specimens were again rinsed using a PBS solution. For the addition of secondary antibodies, the staining procedure was continued by following the protocol of UltraTek HRP (Anti-Polyvalent) from ScyTek Laboratories. In the end, the counterstain of the specimens was made using hematoxylin staining.
Two investigators evaluated all the immunostained samples. Determination of FBP1 and LDHB expression was carried out by semiquantitative scoring by calculating the percentage of stained cells and the staining intensity. The percentage of stained cells is calculated by counting the number of cells stained divided by the number of all cells in one microscope field of view. This calculation is carried out in four fields of view in each sample, and then the average value is taken. The percentage of stained cells was then divided into 5 categories: 0 = < 5%; 1 = 5−25%; 2 = 26−50%; 3 = 51−75%; 4 = >75%. The staining intensity was divided into 4 categories: 0 all cytoplasm of the specimens colored blue; 1 brownish, 2 light brown; and 3 dark brown. The stained cell percentage category is then multiplied by the staining intensity to generate the immunoreactivity score (IS) ranging from 0 to 12. IS ≥ 4 indicates the high expression of the enzymes [4].
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics 24.0. A Chi-square test was used to assess the statistical significance of the association between the expression of FBP1 and LDHB and the state of tumors (fibroadenomas and invasive breast cancers). A P-value of 0.05 was considered to determine statistical significance.
Results
Characteristics of the samples
Table 1 summarized the clinical characteristics of this study samples. All tissue specimens of both breast fibroadenomas and invasive breast cancers were from female patients.
Clinical characteristics of the samples
Clinical characteristics of the samples
This study showed FBP1 was expressed by 22 of 32 (68.7%) breast fibroadenomas and 22 of 31 (71%) invasive breast cancers (Fig. 1). Statistical analysis with the Chi-Square test showed no significant difference (P = 1,000) between FBP1 expression and tumor status (Table 2), which meant that FBP1 could be expressed by most fibroadenomas and invasive breast cancers. The LDHB was expressed by 29 of 32 (91%) breast fibroadenomas and 20 of 31 (64.5%) invasive breast cancers. Chi-Square test showed a significant difference (P = 0.029) between LDHB expression and tumor status, which meant that although LDHB could be expressed by most fibroadenomas and invasive breast cancers, LDHB expression in invasive breast cancers was significantly lower than in breast fibroadenomas (Table 2).
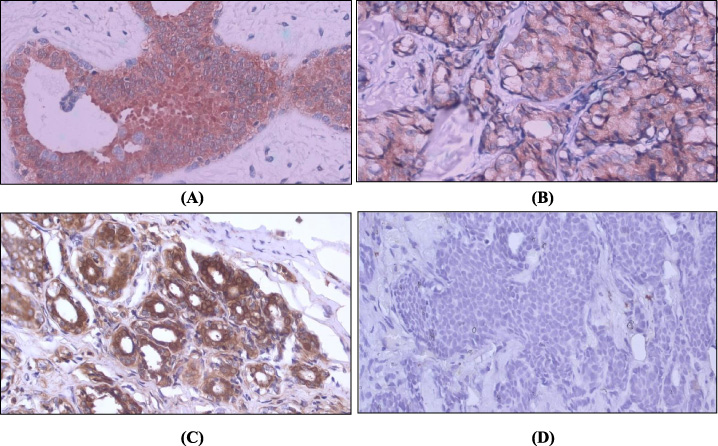
Representative pictures of FBP1 and LDHB expression in breast tumors. FBP1 and LDHB were localized in the cytoplasm of the tumor cells. (A) (B) Represent high FBP1 expression in breast fibroadenoma and invasive breast cancer, respectively. (C) Represent high LDHB expression in breast adenoma. (D) Represent low LDHB expression in invasive breast cancer. All images shown at 400× magnification.
FBP1 and LDHB expression in breast fibroadenomas and invasive breast cancer samples
Based on the FBP1 and LDHB expression data, we next calculated the conformity of the expression of the two enzymes in breast fibroadenomas and invasive breast cancers. Table 3 showed that although the percentage of positive conformity in fibroadenomas (65.6%) was higher than that of invasive breast cancers (41.9%), this has not been statistically confirmed. These results indicated that the co-expression of FBP1 and LDHB in breast fibroadenomas was more common than in invasive breast cancers. Whereas, the low negative conformity showed that these two enzymes were almost always expressed by fibroadenomas and invasive breast cancers, either co-expressed or only one of the two.
FBP1 and LDHB expression conformity for determining the co-expression of the two enzymes
FBP1 and LDHB expression conformity for determining the co-expression of the two enzymes
The majority of the breast carcinoma samples (93.6%) in this study had the morphology of invasive breast carcinoma (8500/3). Sixty-seven percent of this morphology expressed FBP1 and LDHB and showed 44.8% coexpression of the two proteins. There is one morphology, respectively, of lobular carcinoma (8520/3) and tubular carcinoma (8211/3), both of which express FBP1 but do not express LDHB. There was no significant difference in the expression of FBP1 and LDHB and their coexpression in well, moderately, or poorly differentiated invasive breast carcinoma.
Discussion
Studies of FBP1 expression in breast cancers and other cancers still show variable results. Several previous studies have shown FBP1 to be down-regulated in multiple human cancers, including breast cancers [6,7,14–16]. Different data were obtained from Oncomine, Cancer Cell Line Encyclopedia database (CCLE), and The Cancer Genome Atlas database (TCGA), which showed a pattern of increased expression of FBP1 in breast cancers. The high expression of FBP1 in our study was in line with the data and Li et al. that found high FBP1 expression in breast cancers [17]. Our study also showed that high expression of FBP1 might be found in all types of the observed specimens, including invasive, lobular, and tubular carcinoma. Likewise, with the degree of malignancy, FBP1 expression might be found in invasive breast cancers with well, moderately, or poorly differentiated breast carcinoma. It certainly requires further research because previous studies have shown that FBP1 expression could be a predictor for a good prognosis in breast cancers [17,18].
A surprising result from this study was the expression of FBP1 in fibroadenoma, which was not significantly different from the expression of FBP1 in invasive breast cancer. The data regarding energy metabolism in benign tumors, especially fibroadenomas, were still very limited. We assumed that the glucose requirements in fibroadenomas are not as high as in invasive breast cancers. Tumor cells in benign lesions such as fibroadenomas have a lower glycolytic capacity and mitotic index than malignant lesions [19,20]; hence, gluconeogenesis activity in fibroadenomas is probably low. We suggest that high expression of FBP1 in breast fibroadenomas can also be the basis for understanding the role of FBP1 in suppressing the development of benign tumors toward malignancies. The pathogenesis of breast fibroadenomas is still unclear and is very important to be traced through the metabolic role approach, especially the enzymes involved in glycolysis and gluconeogenesis, which are critical pathways in providing biosynthetic substrates. Further research is also needed to assess whether the high expression of FBP1 indicated the high activity of gluconeogenesis in invasive breast cancers and how it is associated with the pathogenesis of the diseases. Previous studies have shown that in addition to its gluconeogenic function (canonical function), FBP1 inhibits glycolytic gene expression in several signaling pathways (non-canonical function) [3,8]. It is still unclear which of the two functions involved in the pathogenesis of breast diseases, including benign and malignant tumors.
Like FBP1 expression, this study showed LDHB could also be expressed by invasive breast cancers and breast fibroadenomas. The increase in LDHB expression in invasive breast cancers was in line with several studies that showed a high LDHB expression in several types of cancer [21]. This elevated expression is related to the role of LDHB as a regulator of cell proliferation [22]. Clinically, increased LDHB expression is related to tumor progression and can be used as a metabolic marker in response to neoadjuvant chemotherapy. As a marker of prognosis, LDHB expression showed inconsistent results, but in breast cancers, an increase in LDHB expression was associated with poor clinical outcome [11,23–25]. The increase in LDHB expression in our study occurred in all types and grades of malignancy. This result was in line with the expression of FBP1. It raises questions of whether the expression of these two enzymes is sustainable at every stage of cancer development in all types. Is this a canonical function related to carbohydrate metabolic pathways, especially gluconeogenesis or LDHB involved in cancer cell signaling pathways?
Not much is known about the role of LDHB in benign breast tumors, especially fibroadenoma. Our study showed LDHB expression appeared to be significantly higher in breast fibroadenomas compared to invasive breast cancers. This result is interesting for further studies to elucidate whether LDHB also acts as a regulator of cell proliferation in breast fibroadenomas. Is there a role for this enzyme in the development of breast fibroadenomas toward malignant phyllodes tumors in the breast [26]? It is not yet known whether the Warburg effect also occurs in breast fibroadenomas. LDHB has a catalytic preference to promote the conversion of lactate to pyruvate. This reaction is the first step of gluconeogenesis with lactate as its substrate. However, this LDHB preference is strongly influenced by cell types and microenvironment conditions. The specific role of LDHB in both benign and malignant tumor pathogenesis, mainly the canonical function of this enzyme in carbohydrate metabolism, still needs to be explored further.
Another interesting thing to be explored further from this study is the pattern of FBP1 and LDHB co-expression. This pattern is important, considering the different types of reactions of these two enzymes in the gluconeogenesis pathway. FBP1 is a rate-limiting enzyme of gluconeogenesis whose reaction is irreversible [2]. Meanwhile, LDHB plays a role in providing substrate for gluconeogenesis when the lactate influx into cells increases. LDHB then converts lactate to pyruvate. This enzyme can catalyze the reverse reaction by converting pyruvate to lactate. The conformity of these two enzymes’ expression patterns may indicate the active gluconeogenesis pathway. This pathway significantly suppresses glycolysis and other metabolic pathways, including tricarboxylic acid cycle, oxidative phosphorylation, pentose phosphate pathway, glutaminolysis, and serine and nucleotides biosynthesis in cancer cells [2]. Our research data have not elucidated this conformity, where the percentage of FBP1 and LDHB co-expression is low even though it is not statistically significant. If these results lead to the asynchronous expression of the two enzymes, it may indicate a metabolic shift in cancer development.
In conclusion, FBP1 and LDHB may be expressed both by benign fibroadenoma breast tumors and invasive breast cancers. LDHB expression levels were significantly higher in fibroadenomas compared to invasive breast cancers. Further studies are needed to clarify the role of these two enzymes in the development of both benign and malignant tumors.
Footnotes
Acknowledgements
The present study has been supported by Higher Education Basic Research Grant from Directorate General of Research Strengthening and Development, Ministry of Research, Technology, and Higher Education, Indonesia
Conflict of interest
None.
