Abstract
Interrelations between cytokines, produced by invasive ductal carcinoma (IDC) and fibroadenoma (FA) of the breast, and angiogenic growth factor VEGF-A, expressed in IDC and FA, were investigated. The analysis of the cytokine profiles of IDC and FA was performed by cultivation of tumor biopsy specimens in vitro. Testing of the cytokine-producing reserve of the tumors for production of VEGF-A was conducted by culturing samples of IDC and FA in a medium containing polyclonal activator (a complex of phytohemagglutinin, concanavalin A, and lipopolysaccharide). Levels of cytokines and growth factors (IL-2, IL-6, IL-8, IL-10, IL-17, IL-18, IL-1β, IL-1Ra, TNF-α, IFN-γ, G-CSF, GM-CSF, VEGF-A) and MCP-1 (monocyte chemoattractant protein-1) in tumor supernatants were determined by an ELISA. Expression of VEGF-A was analyzed in tumor biopsy specimens by immunohistochemical analysis. In the IDC supernatants, the concentrations of IL-17, IL-18, and IFN-γ were higher and the concentrations of IL-10 and MCP-1 were lower in comparison with the FA supernatants. We observed negative correlations between the macrophage infiltration and VEGF-A concentration in the IDC supernatants (r = −0.508;
Introduction
Many authors have highlighted the dual role of cytokines in the pathogenesis of malignant tumors.1–3 A growing tumor and cells in its microenvironment can produce a variety of cytokines, some of which inhibit its growth and metastasis, while others ensure tumor progression.4,5 Correlations have been found among increased concentrations of certain angiogenic cytokines, the degree of tumor vascularization, proliferative activity of tumor cells, and the depth of tumor invasion.3,5,6 The data obtained led to the following concept: in the course of tumor progression, a sufficiently autonomous cytokine network 4 can be formed in the tumor, and this network is one of the multi-component regulators of malignant tumor growth and metastasis. Nonetheless, the mechanisms of the “work” of this “network” have not been studied thoroughly. The relationship between tumor cytokine profile in general and production of angiogenic cytokines, particularly VEGF family cytokines, depending on the degree of malignancy of the tumor is not sufficiently studied. The aim of this study was to evaluate the cytokine profiles of invasive ductal carcinoma (IDC) and fibroadenoma (FA) of the breast and to analyze possible correlations with the expression of VEGF-A.
Materials and methods
Patients
The study included 34 women (age range, 45–60 years) with IDC and 11 women with FA of the breast, who were undergoing treatment at a Novosibirsk regional oncological center. The object of the study were biopsies of tumors, which were obtained under sterile conditions after removal of the tumors. Removal was performed at stage II of tumor development (T2) in patients not previously treated with radiation and chemotherapy. The diameter of removed tumors was in the range of 2–5 cm. In operated patients, the breast cancer had not spread to regional lymph nodes. After the surgical procedure, a histopathological analysis of the resected material was performed. Histological slides of FA and adenocarcinomas of the mammary gland were stained by hematoxylin and eosin (H&E). The degree of infiltration of tumors by immunocompetent cells (macrophages and others) were determined by cytomorphological criteria (the average number per field of view was evaluated, 10 fields of view, magnification ×1000). The assessment of tumor vascularization was performed on a 6-point scale (the number of vessels in the field of view was evaluated, 10 fields of view examined, magnification ×400).
All studies were performed according to the Declaration of Helsinki (Brasil, 2013) 7 . Recommendations of ICMJE were taken into account, each patient was informed about the upcoming research of obtained tumor biopsies, gave permission for the research, and signed the document in presence of the attending physician.
Measurement of cytokine secretion
Tumor samples, obtained by trepanobiopsy (8 mm3), were placed in DMEM-F12 and incubated at 37°С for 72 h, centrifugated at 900 g for 15 min to obtain the supernatant for further cytokine analysis. Concentrations of IL-2, IL-6, IL-8, IL-10, IL-17, IL-18, IL-1β, IL-1Ra, TNF-α, IFN-γ, G-CSF, GM-CSF, VEGF-A, and MCP-1 (monocyte chemotactic protein-1) in supernatants were determined by ELISA, using kits manufactured by AO Vector-best (Russia).
To determine the possible ability of the tumor to express VEGF-A, the tumor samples (8 mm3), obtained by trepanobiopsy, were placed in two vials: the first with DMEM-F12 (spontaneous production) and the second with a polyclonal activator (PA) solution in the same volume of DMEM-F12 (production induced by PA) and incubated at 37°С for 72 h. PA is a complex, consisting of phytohemagglutinin (4 µg/mL), concanavalin A (4 µg/mL), and lipopolysacharide (2 µg/mL) (“Cytokine-Stimulus-Best” kit, manufactured by AO Vector-Best, Novosibirsk, Russia). Tumor biopsies were fixed in 12% formalin, followed by dehydration in ethanol with increasing concentration and embedded in paraffin. One tissue slice was subjected to the conventional H&E staining.
Immunohistochemistry
This analysis was based on the indirect avidin-biotin peroxidase ABC method7,8 according to the manufacturer’s instructions. Deparaffinized tissue slices were placed in a solution of Triton X-100 (0.1%) for antigen demasking procedure for 5 min. After incubation in 1% H2O2 solution for 5 min, slices were incubated with blocking serum (VECTASTAIN Elite kit ABC Kit, PK-7200, Vector Laboratories, USA) for 10 min, then with primary antibodies: VEGF-A (polyclonal rabbit, cat. no. RB-9031-P, Thermo Scientific, USA) in a solution for dilution of antibodies (559148, BD Pharmigen) at the final antibody concentration of 2.5 µg/mL for 60 min at room temperature. Then the slices were incubated with the second biotinylated antibodies (VECTASTAIN Elite ABC Kit, PK-7200, Vector Laboratories, USA) for 30 min. The incubation with avidin-peroxidase complex (VECTASTAIN Elite ABC Kit, PK-7200, Vector Laboratories, USA) lasted for 30 min. After staining with a chromogenic substrate containing diaminobenzidine (ImmPACT DAB Substrate, SK-4105, Vector Laboratories, USA) for 5 min, the tissue slices were stained with H&E.
Image analysis
Tissue slices were viewed by light microscopy at magnification ×400 and ×1000. Histopathological analysis of histological slides stained by standard methods with H&E was conducted by a pathologist. Histological analysis was performed by means of Micros MC 300A microscope and digital camera CX 13c (“Baumer,” Germany). Computer-assisted quantification of the immunostaining in the standard area (digital photos with the size 133,9000 square pixels) was carried out in the ImageJ software 1.5 (National Institute of Health, USA). Eight slices from each tumor sample were examined.
Data analysis
VEGF-А expression index (relative units) represented the product of a percentage of the stained area and digital density of the staining. The index of polyclonal activators influence (IPAI) on VEGF-А expression by tumor biopsies was estimated using the formula IPAI = А/B, where A is immunostaining intensity during PA-induced production of VEGF-A and B is the level of spontaneous production of VEGF-A. The results were expressed as the median (Me) and quartiles (Q1; Q3). Statistical analyses involved the Mann–Whitney
Results
Significant differences in concentrations between IDC and FA were shown for only 5/14 cytokines studied in the tumor supernatants (Table 1). The concentrations of IL-17, IL-18, and IFN-γ were higher whereas the concentrations of IL-10 and MCP-1 were lower in the IDC supernatants compared to the FA supernatants. Negative correlations between the macrophage infiltration and VEGF-A concentration in IDC supernatant (r = −0.508;
Concentrations of cytokines in supernatants of tumors from patients with IDC or FA of the breast.

Data are presented as the median (Me) and quartiles (Q1; Q3).
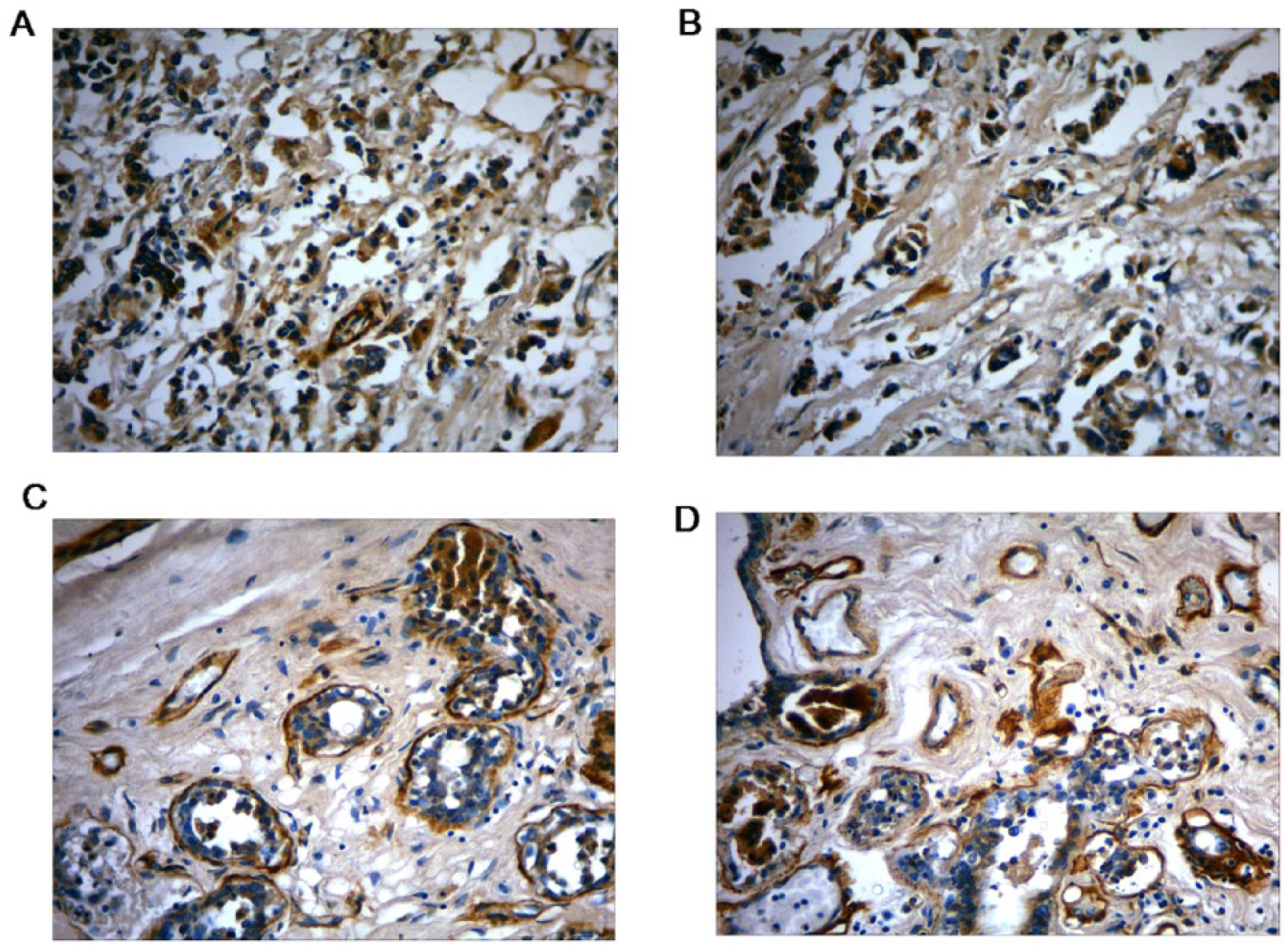
Expression of VEGF-A in breast IDC and breast FA. Expression of VEGF-A in IDC samples before (a) and after (b) incubation with polyclonal activator (PA). Expression of VEGF-A in FA samples before (c) and after (d) incubation with PA. Immunohistochemical staining with 3,3′-diaminobenzidine (DAB; brown) as a substrate and additional staining with H&E (400× magnification).

Spontaneous VEGF-A expression and the expression induced by polyclonal activator (PA) in samples of IDC and breast FA according to immunohistochemical analysis (8 slices from each tumor sample were examined). (a) A notable increase in VEGF-A expression in FA samples after incubation with PA. (b) A markedly higher index of PA influence (IPAI) on VEGF-A expression in FA samples compared to IDC samples. Data are presented as a median and interquartile range (25–75%). Significance of differences in the indicator: **
Discussion
The study revealed that the cytokine profiles of IDC and FA differed in only five cytokines. We can assume that the maintenance of IDC growth is attributable mostly to such regulators as IL-10, IL-17, IL-18, IFN-γ, and MCP-1. It is known that IL-17 may induce the synthesis of pro-angiogenic and pro-oncogenic factors by fibroblasts (which are part of the stromal microenvironment of the tumor), for example, VЕGF, TGF-β, IL-6, PGE2 and IFN-γ.9–12 IL-18 is a cytokine that is involved in angiogenesis by enhancing endothelial cell migration and by increasing VEGF production by the tumor itself. 13 Thus, in our study, cytokines IL-18, IL-17, and IFN-γ could be assigned to one of the “cytokine blocks” of positive regulation of VEGF in tumors, where IFN-γ plays the role of a “deterrent factor.” Concentrations of IL-10 and MCP-1 were lower in IDC supernatants than in FA supernatants. IL-10 is related to “proinflammatory” cytokines and inhibits angiogenesis because of downregulation of the M1 macrophage population, which expresses VEGF.10,14 These phenomena may explain the observed negative correlation between the intensity of macrophage infiltration and the concentration of VEGF-A in the IDC supernatants.
The role of MCP-1 in tumor development is controversial. On the one hand, CC chemokines, which include MCP-1, stimulate the production of proteases by tumor cells; these enzymes can facilitate the migration and proliferation of tumor cells. On the other hand, strong expression of this chemokine inhibits tumor growth; this phenomenon is associated with activation of dendritic cells. 15 Thus, according to our study, cytokines IL-10 and MCP-1 may be assigned to one of the blocks of negative regulation of angiogenesis in a tumor, wherein downregulation of IL-10 and MCP-1 in tumors ensures conditions accelerating tumor angiogenesis accompanied by increased production of IL-18 and IL-17.
According to our data, VEGF-A is expressed in IDC samples both in tumor cells and in the cells of the tumor microenvironment. The proportion of VEGF-A expressed by macrophages is negligible due to the relatively small number of macrophages infiltrating the tumor. This phenomenon is probably due to the observed negative correlation between the VEGF-A expression in IDC supernatants and the degree of macrophage infiltration. We can hypothesize that the increased production of VEGF-A by IDC tumor cells reduces infiltration of the tumor by macrophages. Our findings are consistent with the results of other authors, 6 who showed that with increasing malignancy of tumors, a paradoxical effect can take place: weakening of tumor angiogenesis with the increasing production of VEGF-A by tumor cells and by cells of the tumor microenvironment. The absence of differences in VEGF-A concentrations between IDC and FA supernatants but noticeable differences in VEGF-A expression between IDC and FA samples may be indicative of different secretory activity of FA and IDC cells. It should be noted that IPAI on the production of VEGF-A was higher in FA biopsy specimens than in IDC biopsy specimens. This result suggests that stronger suppression of tumor growth by VEGF-A inhibitors may be expected in patients with FA than in patients with IDC.
In conclusion, our findings are suggestive of complex mechanisms of positive and negative regulation of the processes of tumor angiogenesis mediated by cytokines that are produced by the tumor and by the cells in its microenvironment, whose cytokine profiles may change at different stages of tumor progression.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
