Abstract
Part of three systematic reviews on the effects of psychotropic medication exposure in pregnancy, this paper critically reviews the literature on adverse effects of antidepressant use during pregnancy, and derives recommendations for clinical practice. Electronic databases were searched for original research studies examining the effects of gestational exposure to antidepressants on pregnancy, neonatal and longer-term developmental outcomes. Most results were derived from cohort (prospective and retrospective) and casecontrol studies. There were no randomized controlled trials. Congenital malformations: 35 studies identified, 12 demonstrated a significant association between antidepressant use in early pregnancy and congenital malformations. Pregnancy outcomes: 35 articles identified, outcomes measured rates of spontaneous abortion (4 out of 7 studies reporting elevated risk), preterm birth (15 out of 19 reporting elevated risk) and abnormal birth weight (8 out of 23 reporting elevated risk). Neonatal outcomes: 17 controlled studies including one meta-analysis were identified concerning neonatal adaptation. 15 studies showed an association between gestational exposure to antidepressants and neonatal adaptation difficulties. Three studies examined an association between selective serotonin reuptake inhibitor (SSRI) exposure and persistent pulmonary hypertension in the neonate with conflicting results. Longer-term developmental outcomes: 6 of 7 studies comparing developmental outcomes of children exposed to antidepressants in utero with non-exposed children reported no significant differences. Most of these medications remain relatively safe in pregnancy, but some significant areas of concern exist, particularly some evidence of higher risk of preterm birth, neonatal adaptation difficulties and congenital cardiac malformations (with paroxetine). The impact of these findings on the risk-benefit analysis when treating pregnant women with antidepressants is discussed.
Keywords
It is now widely recognized that women are at increased risk of depression in both pregnancy and the post-partum period. About 12% of women report symptoms of depression during pregnancy. Further, of women who experience significant depression in the post-partum period, 40% of these developed symptoms during pregnancy [1]. Studies have shown that in women with a pre-existing depression who are on antidepressants and discontinue medication during pregnancy the relapse rate is significantly higher than if they continue treatment [2].
There are considerable risks involved if depression is left untreated during pregnancy. Suicide has been identified as a major cause of maternal mortality in a number of studies [3,4]. Maternal depression is associated with poor attendance at antenatal clinics, poor nutrition, increased risk of smoking, alcohol and other drug abuse all of which have negative impacts on foetal well-being [5]. Some studies have suggested a relationship between obstetric complications such as preeclampsia and depression [6–8], whereas others suggest a relationship between ‘chronic stress’ and preterm delivery and low birth weight [8], perhaps due to the effect of chronic anxiety and depression on adrenocortical regulatory functions that influence growth, birth weight and gestation. Finally, maternal depression is associated with poor maternal–infant attachment and poorer long-term developmental outcomes for the child [9].
Clinicians have thus become aware of the importance of active treatment of perinatal depression. Several studies have established foetal exposure to antidepressants during pregnancy via both placental (measured in cord blood) [10–13] and amniotic fluid passage [14]. Variable cord blood-to-maternal serum ratios have been found for both tricyclic depressants (TCAs) [15] and selective serotonin reuptake inhibitors (SSRIs) [10,11,13]. Amniotic fluid levels of drugs have been documented in at least one study [14], which point to another potential avenue of foetal exposure that bypasses first-pass hepatic metabolism. It is therefore clear that exposure in utero must be a consideration for any pregnant woman using antidepressants.
Prescribers need to be aware of the current state of knowledge of the risks to the developing foetus and neonate of exposure to antidepressants. This article evaluates the most recent literature on the use and risks of antidepressants during pregnancy. This systematic review of antidepressants in pregnancy forms part of three separate systematic reviews covering antipsychotics, mood stabilizers and antidepressants in pregnancy, which was undertaken as a joint initiative across the perinatal mental health services in Melbourne.
Methodology
This study was designed with the objective of reviewing the literature on available antidepressants in Australia and outcomes of their use in pregnancy in terms of teratogenicity, pregnancy complications, neonatal complications and longer-term neurodevelopmental outcomes. A computerized search of the following databases was completed: MEDLINE, PsycINFO and EMBASE, for the period of 1950 to May 2009. The key terms used were: ‘pregnancy’, ‘depression’, ‘antidepressants’, ‘neonate’, ‘SSRI’, as well as individually named antidepressants. These were: fluoxetine, fluvoxamine, sertraline, paroxetine, citalopram, escitalopram, reboxetine, venlafaxine, mirtazapine, duloxetine, amitriptyline, imipramine, nortriptyline, clomipramine, doxepin, dothiepin, moclobemide, tranylcypromine and phenelzine. Papers were limited to English language and studies in humans. There was extensive cross-referencing and manual searching through journal articles, particularly review papers for relevant articles not identified through the initial searches. Where available, papers using a scientific, experimental methodology were preferentially reviewed. Only in their absence were case reports and case series considered for discussion.
Results
Congenital malformations
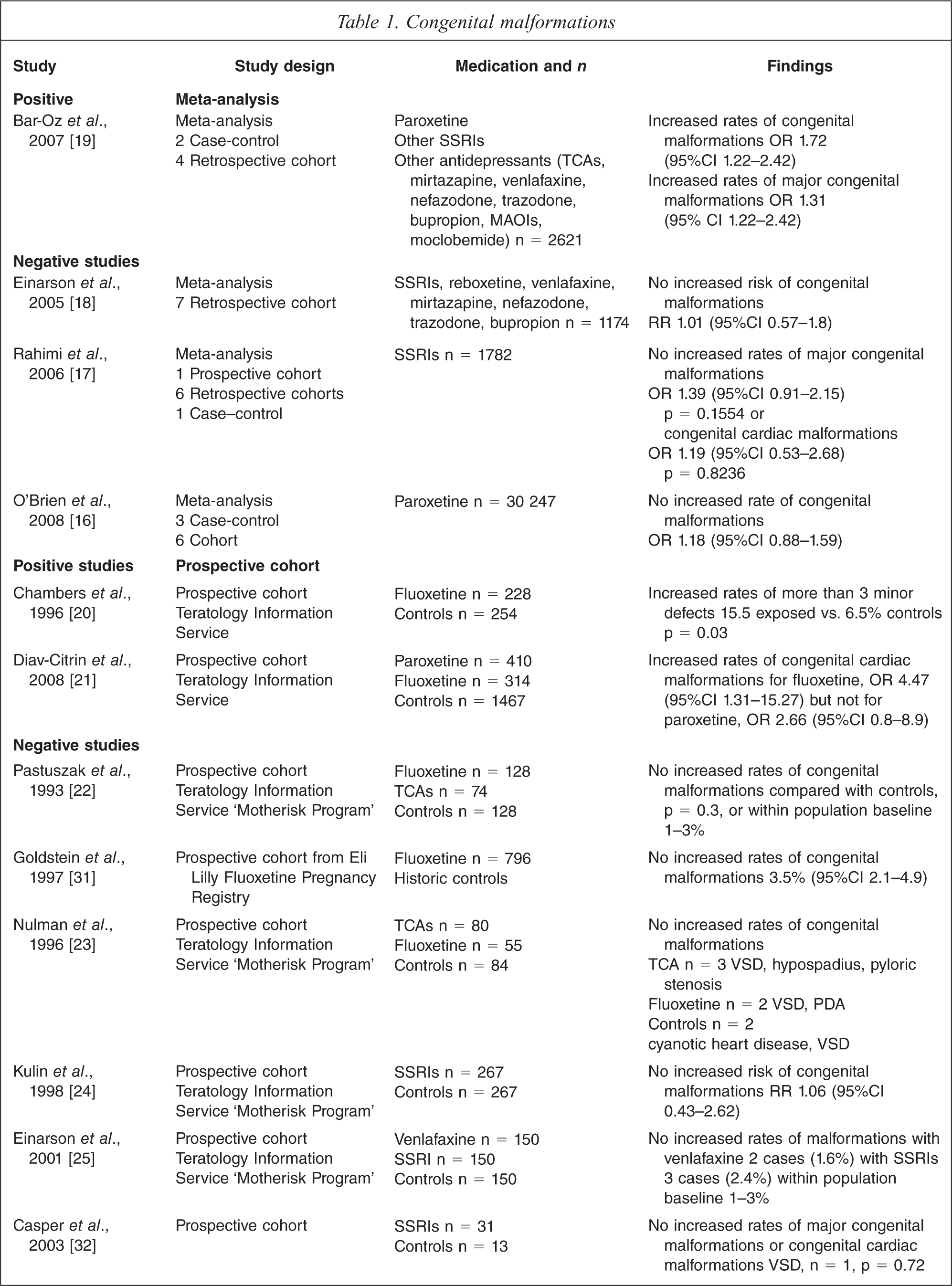
Thirty-five studies of three study types; meta-analyses, cohort and case–control studies were identified. See Table 1. Of these, only twelve (N = 18 451 out of 159 618) demonstrated a statistically significant association between antidepressant use in early pregnancy and congenital malformations.
Congenital malformations
Meta-analyses
Of four meta-analyses that collectively examined 102 835 cases, three meta-analyses did not find that first trimester paroxetine, SSRI or other antidepressant exposure increased the risk of major or cardiac malformations [16–18]. In contrast, one meta-analysis involving 2621 cases found that first trimester paroxetine use was associated with a significantly increased risk of major congenital malformations, OR 1.31 (95%CI 1.03–1.67) and cardiac congenital malformations, OR 1.72 (95%CI 1.22–2.42) [9]. In this study, however, matching to controls was incomplete with exposed women and exposed infants having statistically significantly more obstetric diagnostic tests.
Cohort studies
There were twenty-six cohort studies with matched controls. The studies were divided into the following types:
Prospective cohort studies including teratology information services and observational studies, and
Retrospective cohort studies which include birth registries, database studies and ‘other’ retrospective cohort studies
Prospective cohort studies
Fifteen prospective cohort studies were identified. Eleven studies [20–30] were conducted by Teratology Information Services (TIS), nine of which were conducted by the Motherisk Program in Toronto, Canada and [22–30] and four from unrelated centres [31–34].
Only two of the 15 studies (N = 952 out of 6663 patients) found a significant association between fluoxetine or paroxetine exposure and congenital malformations [20,21]. Diav-Citrin identified 410 women who used paroxetine and 314 who used fluoxetine during the first trimester and matched them to 1467 controls [21]. After exclusion of genetic and cytogenetic anomalies and controlling for cigarette smoking, they found fluoxetine, but not paroxetine was significantly associated with an increased risk of cardiac malformations. The authors state that the strength of the findings is open to question because of the broad definition of caseness and the wide range of the 95% confidence intervals. In the other positive study, Chambers found a significant association between fluoxetine exposure and at least three minor congenital malformations [20]. Neither of these findings has been replicated in other studies.
The best study design would take into account the effect of maternal depression on the foetus and include a control group of depressed but not medicated women in their design. Of the two studies that did this, both had negative results but were underpowered and had multiple confounders [32,34].
Retrospective cohort studies
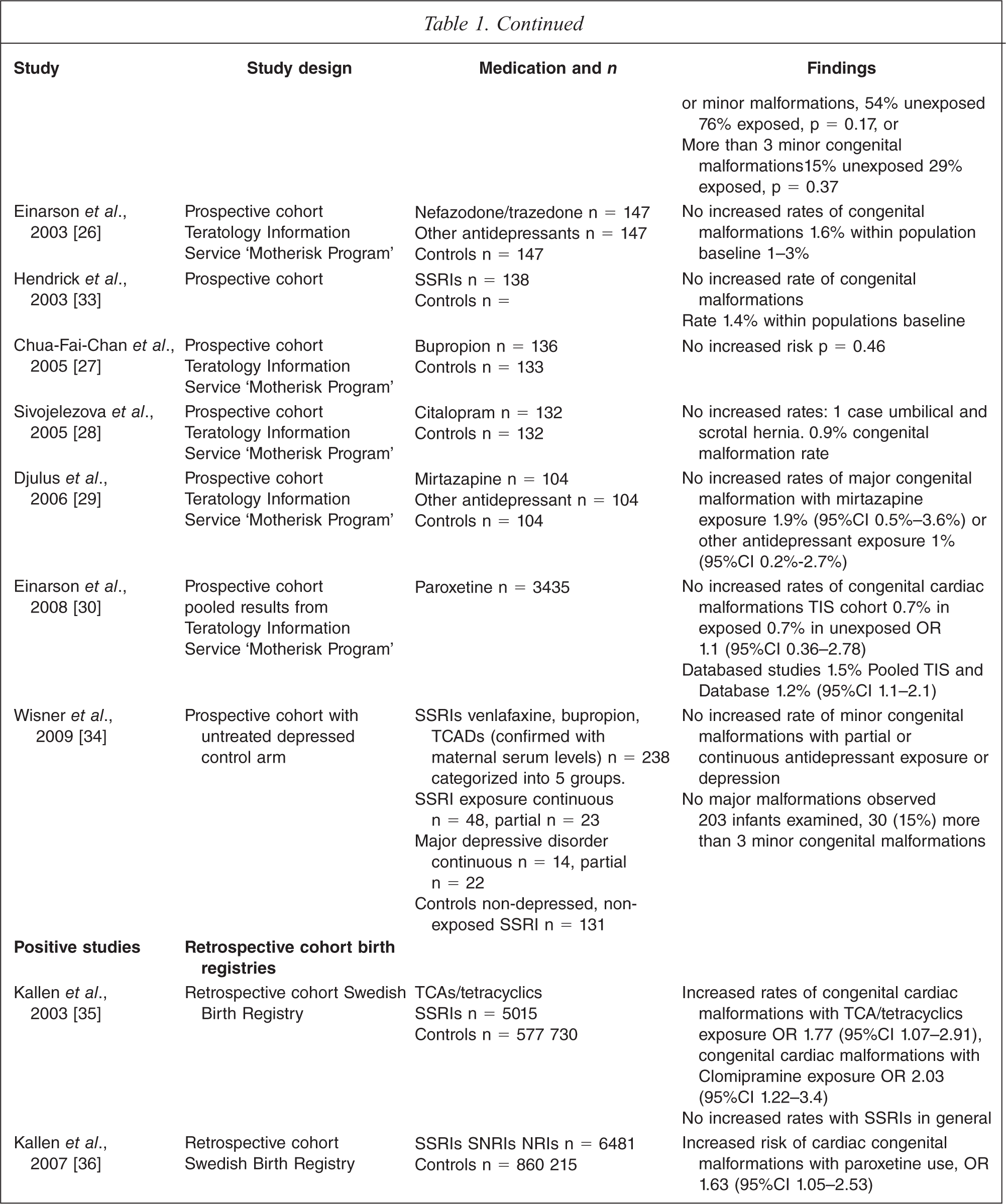
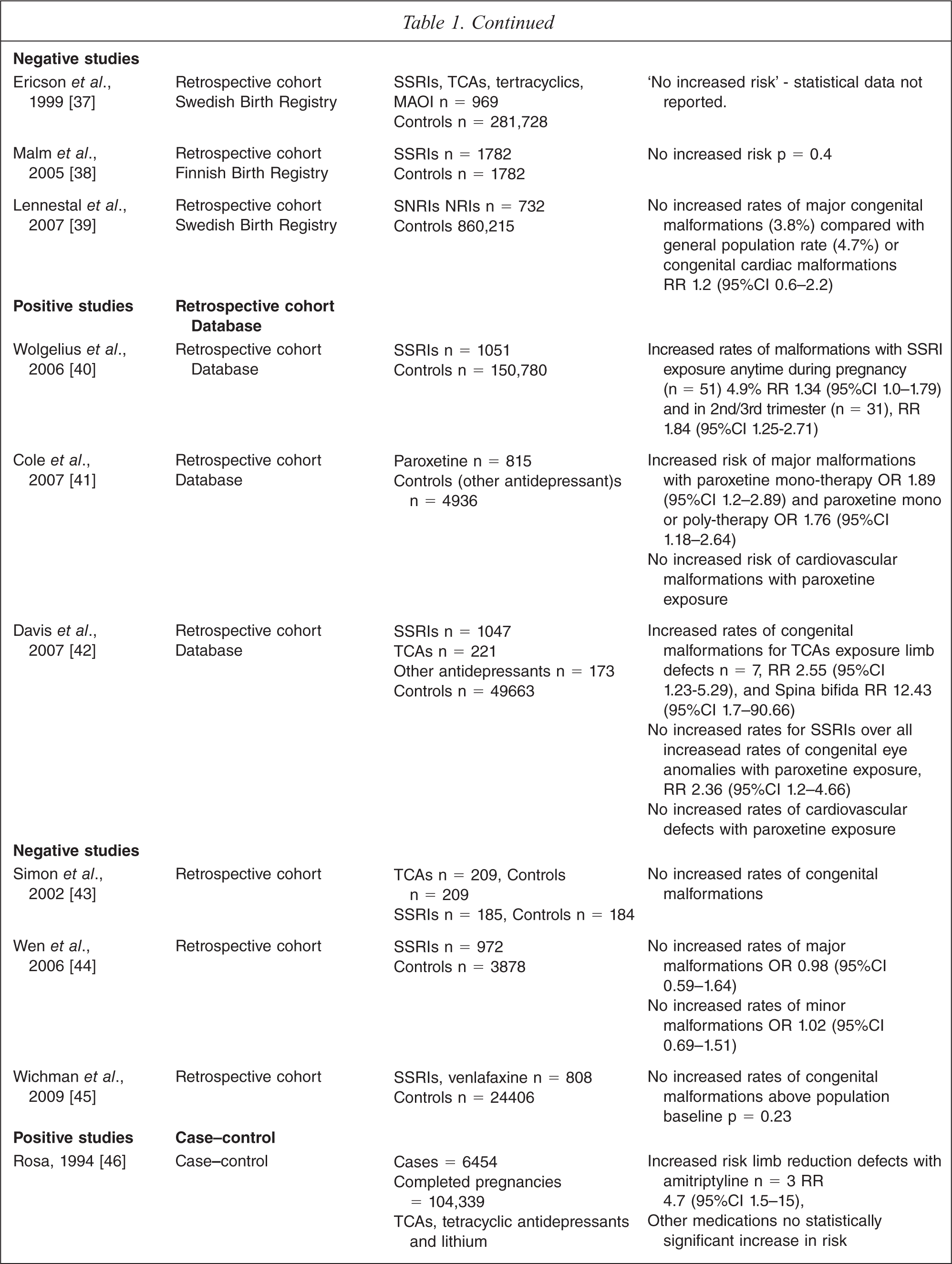
Eleven retrospective cohort studies are included in this review with five studies yielding positive results (N = 14 803 out of 20 460 patients) Five studies utilized data from birth registries, and of these, there were two positive studies both by Kallen utilizing data from the Swedish Birth Registry [35,36]. In 2003, Kallen found TCA or tetracyclic antidepressant use in early pregnancy was significantly associated with cardiac malformations. The association was strongest with clomipramine, OR 2.03 (95%CI 1.22–3.4) [35]. In 2007, Kallen reported a significant and positive association between the use of paroxetine in early pregnancy and cardiac malformations, OR 1.63 (95%CI 1.05–2.53) but not between SSRIs in general and congenital malformations [36]. In contrast, two other registry studies also using the Swedish Birth Registry and one using data from the Finnish Birth Registry did not find an increased risk of major or cardiac malformations in infants exposed to antidepressants [37–39].
Four retrospective cohort studies utilised databases, three with positive [40–42] and one with negative results [43]. Cole and colleagues found an increased risk of major congenital malformations with both first trimester paroxetine monotherapy OR 1.89 (95%CI 1.2–2.89) and paroxetine polytherapy OR 1.76 (95%CI 1.18–2.64) but not cardiac malformations [41]. Davis found that first trimester tricyclic antidepressant exposure was associated with an increased rate of limb abnormalities RR 2.55 (95%CI 1.23–5.29) and spina bifida RR 12.43 (95%CI 1.7–90.66) [42]. The validity of this last finding is questionable given the extremely wide range of the 95% confidence interval. Davis also found that paroxetine exposure was associated with an increased risk of congenital eye abnormalities, RR 2.36 (95%CI 1.2–4.66) [42]. The final positive study found that first trimester SSRI exposure was associated with an increased risk of major malformations, RR 1.34 (95%CI 1.0–1.79) as were second and third trimester exposure, RR 1.84 (95%CI 1.25–2.71) [40].
The two final non-database retrospective cohort studies by Wen-Shi Wu [44] and Wichman [45] failed to find a significant association between SSRIs or other antidepressant exposure and major, cardiac or minor congenital malformations.
Case–control studies
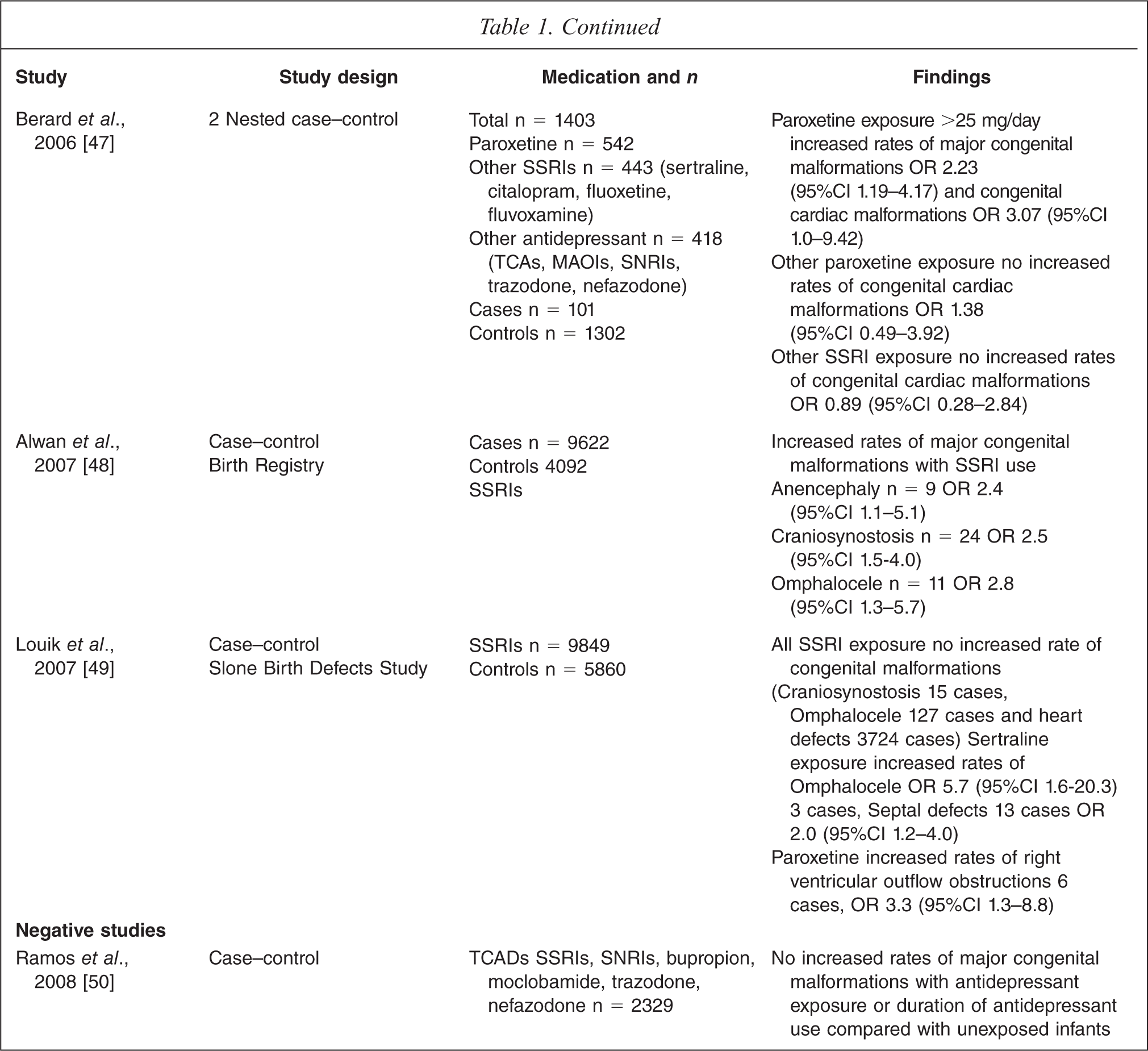
Five case–control studies were included in this review. Four studies found an association between antidepressant use and congenital malformations [46–49] and one did not [50]. Alwan and colleagues using data from the National Birth Defects Prevention Study identified 9622 cases of congenital malformations and 4092 controls. They found maternal SSRI use in early pregnancy was associated with anencephaly, OR 2.4 (95%CI 1.1–5.1), craniosynostosis, OR 2.5 (95%CI 1.5–4.0) and omphalocele, OR 2.8 (95%CI 1.3–5.7) [48]. It is important to note that the authors made 265 comparisons without adjustment to the level of significance hence increasing the likelihood that some of the positive outcomes could be due to chance. Louik and others conducted a case–control study using data from Sloane Epidemiology Centre Birth Defects Study [49]. They identified 9849 infants with congenital malformations and matched them to 5860 control infants. They found that SSRI exposure during pregnancy was not associated with congenital malformations but was associated with right ventricular outflow tract obstruction defects, OR 2.0 (95%CI 1.1–3.6). Despite the large study size, the authors warned against viewing their findings as strong evidence given the small number of exposed infants with rare outcomes. Berard and colleagues studied the association between exposure to a range of antidepressant types in a nested case–control study of 1403 cases and found a statistically significant association between paroxetine and major congenital, OR 2.23 (95%CI 1.19–5.17) and cardiac malformations, OR 3.07 (95%CI 1.0–9.42), but only with doses of paroxetine greater than 25 mg/day [47]. The final case–controlled study was reported in a letter by Rosa in 1994, it is included in this review because of the large sample size [46]. The author reported on an analysis performed by the Food and Drug Administration using the Medicaid database of 6454 infants identified with congenital malformations linked to 104 339 completed pregnancies. They found that amitriptyline use was associated with an increased risk of limb reduction malformations, RR 4.4 (95%CI 1.5–5.1).
Summary
It is reassuring that of the 35 studies reviewed, only twelve demonstrated statistically significant associations between antidepressant use in early pregnancy and congenital malformations. The methodological limitations of the study designs are notable and only some findings have been replicated. Bar-Oz's meta-analysis [19] finding of an association between paroxetine and cardiac malformations is replicated by Kallen [36], and Berard [47] but is refuted by others [21,41,42,50]. Bar-Oz's other finding of an association between paroxetine exposure and a major congenital malformation is replicated by Cole and Berard [40,41]. Though Berard found an increased risk with paroxetine only for doses greater than 25 mg [47]. Kallen's findings of an association between cardiac malformations and TCAs and tetracyclics, in particular clomipramine and Diav-Citrin's finding with fluoxetine failed to be replicated by many others [22,23,34,37,40,42,46,50]. Rosa's report of an association between amitriptyline and limb reduction malformations [46] is replicated by Davis in 2007 [42], while Alwan's finding of an association between SSRI use and omphalocele [48] was partially replicated by Louik's finding of an association between sertraline and omphalocele [49].
It is clear that the majority of studies examining the full range of antidepressants (SSRIs, TCAs, SNRIs, MAOIs, tetracyclics, moclobemide) yield negative results, but interpreting the various results from studies of different methodology is difficult. On balance, with the studies included in this review, it would appear that antidepressants as a group and individual antidepressants apart from paroxetine, are unlikely to be teratogenic.
Pregnancy outcomes
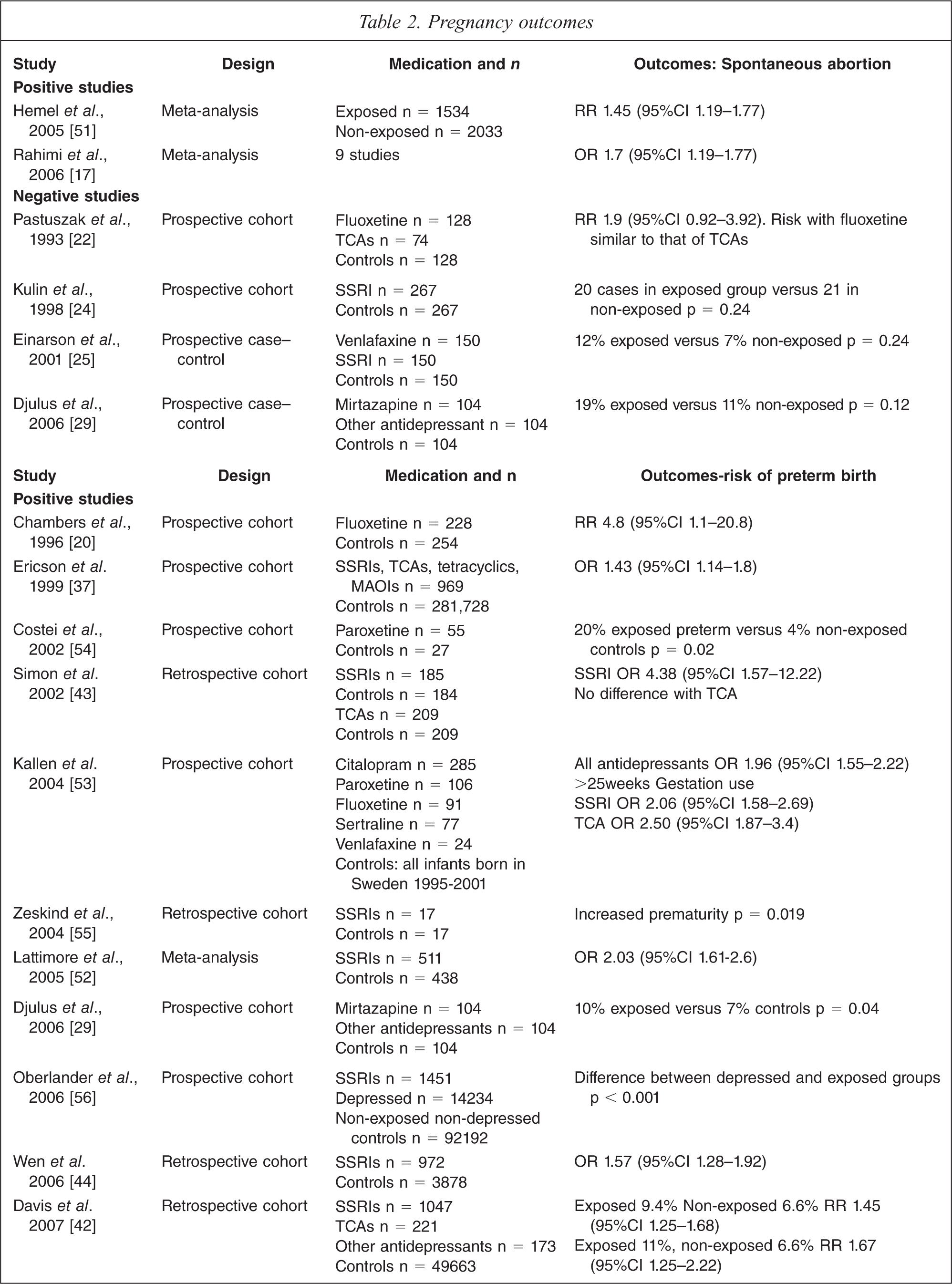
Thirty-five articles were analysed that reported pregnancy outcomes in infants exposed to antidepressants (Table 2). The outcomes measured were rates of spontaneous abortion (two out of six yielding positive results N = 3318 out of 3422), preterm birth defined as birth earlier than 37 weeks of gestation (15 out of 19 with positive results N = 8286 out of 8735), abnormal birth weight (8 out of 23 with positive results N = 6907 out of 10 890), gestational age (4 out of 17 with positive results N = 2271 out of 6853) and stillbirth (one study).
Pregnancy outcomes
Spontaneous abortion
No specific studies focused on the impact of antidepressants on spontaneous abortion. However, it was a secondary outcome in two meta-analyses. Hemel reported that in 1534 treated patients there was a statistically significant increase in spontaneous abortions of 3.9%, RR of 1.45 (95%CI 1.19–1.77) [51]. Rahimi reviewed nine studies and also found an increase risk OR 1.7 (95%CI 1.28–2.24) [17]. Unfortunately, none of these studies adequately controlled for previous miscarriages, age, smoking, other drug use or depression.
Preterm birth
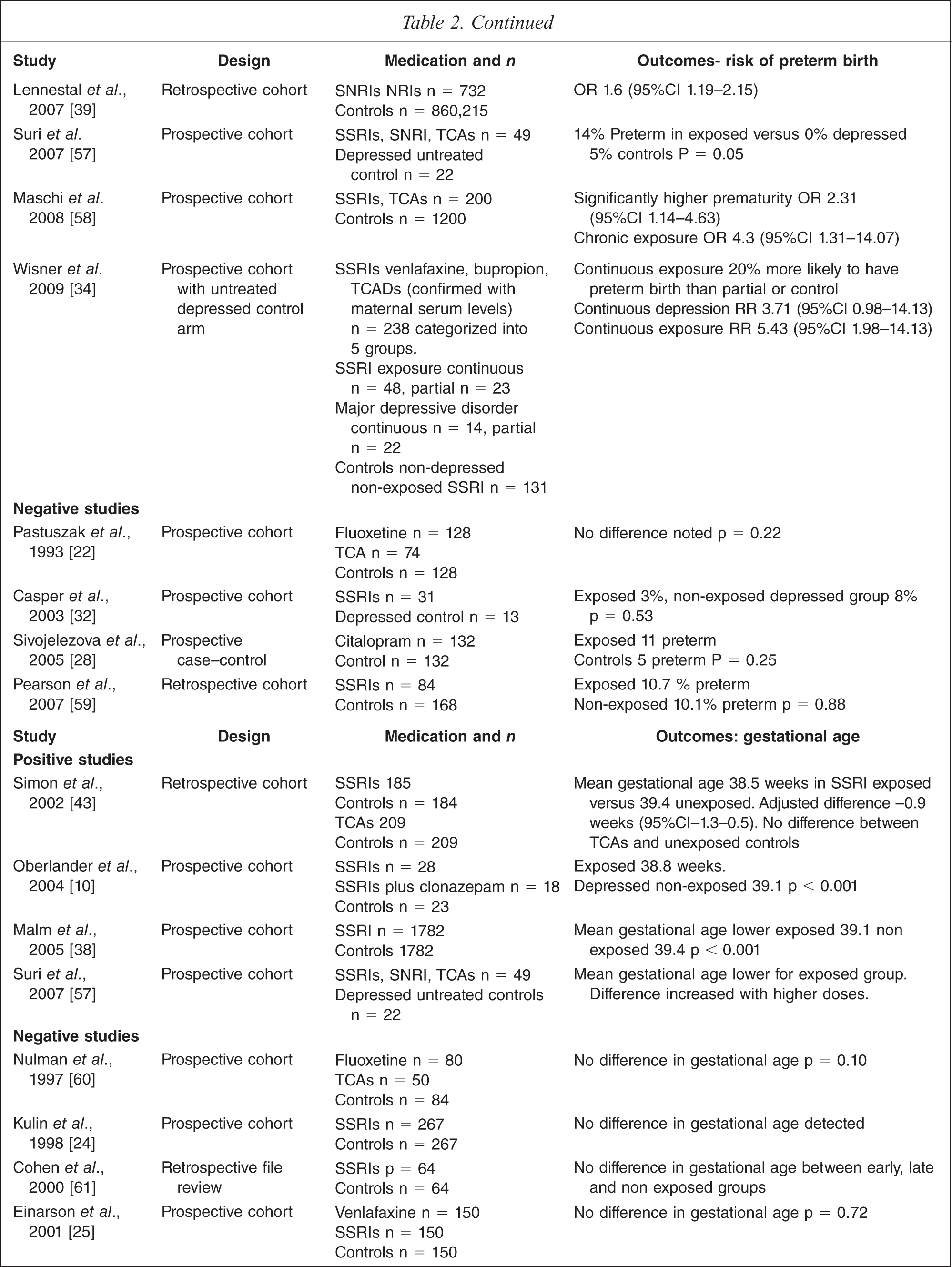
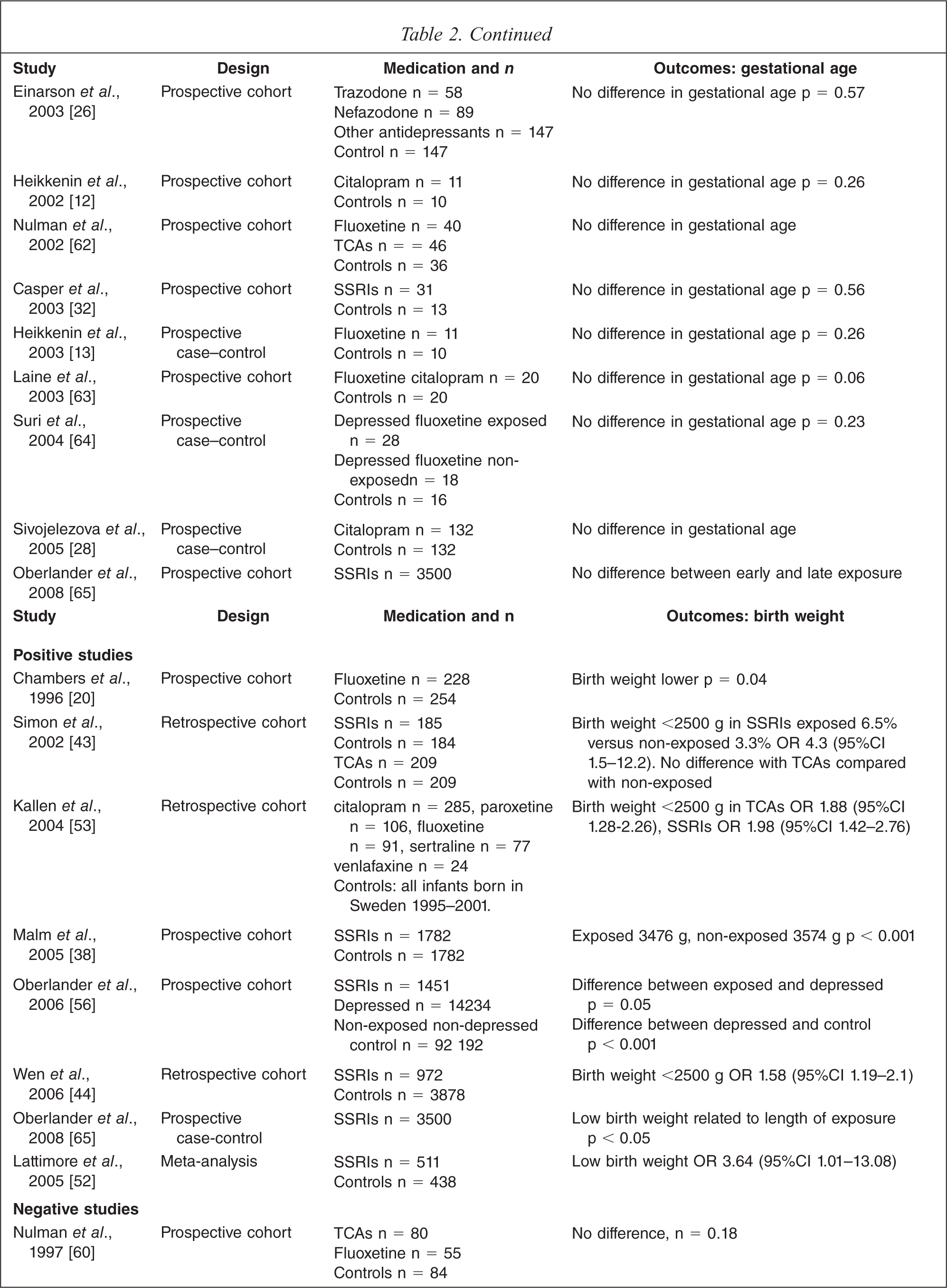
Nineteen studies examine the incidence of preterm birth in women exposed to antidepressants in late pregnancy (see Table 2). These include one meta-analysis, 12 prospective cohort studies, and six retrospective cohort studies. In contrast, 17 studies looked at mean gestational age (rather than preterm birth) and of these only four showed a lowering of gestational age in exposed babies (see Table 2).
Lattimore's meta-analysis of nine studies showed a significant increase in preterm births with an OR 2.23 (95%CI 1.61–2.6) [52]. Of the 12 prospective cohort studies, nine (involving antidepressants (SSRIs, Mirtazapine, TCA and SNRI)) demonstrated a statistically significant increase in preterm delivery rates. Two of these studies showed a relationship between an increase in preterm delivery rates and late exposure [20,53]. Maschi and colleagues revealed a correlation between preterm birth and chronic exposure to antidepressants but not with short term exposure [58]. Five out of six retrospective cohort studies also demonstrated a positive finding of exposure and preterm birth (see Table 2). All of these studies have similar weaknesses, with no measures of actual drug exposure or controls for confounders such as smoking and in particular the effects of underlying depression.
Some of the more rigorous studies have tried to tease out the role of untreated maternal depression. Oberlander [56] examined 119 547 prescription records matched with hospital separation records and found that SSRI-exposed babies had a higher rate of preterm birth than babies exposed to depression alone (p < 0.01). A further study, by the same group, of 3500 cases found that increased length of drug exposure was related to higher risk of low gestational age and low birth weight [65]. These studies are weakened by the fact that hospital records may be unreliable and that the authors did not correct for multiple comparisons. A small prospective study compared women with depression alone and treatment with SSRIs and found a statistically significant higher preterm birth rate in the exposed infants (14% exposed group, 0% depressed group) [62]. In contrast, another small study did not detect any difference between treated and depressed groups [32].
Finally, a recent carefully designed study looked at five overlapping groups using a prospective design [34]. Patients were classified into those with no SSRI or depression exposure, those with continuous or partial SSRI exposure and those with continuous or partial depression exposure. Unlike most other studies, exposure was confirmed with serum SSRI levels. The group with continuous SSRI exposure had a significantly higher preterm delivery rate with a RR of 5.43 (95%CI 1.98–14.84). The group with continuous depression also had a higher rate but this was no longer statistically significant when controlled for maternal age and race, RR 3.7 (95%CI 0.98–14.13). The partial SSRI and partial depression groups were no different from controls.
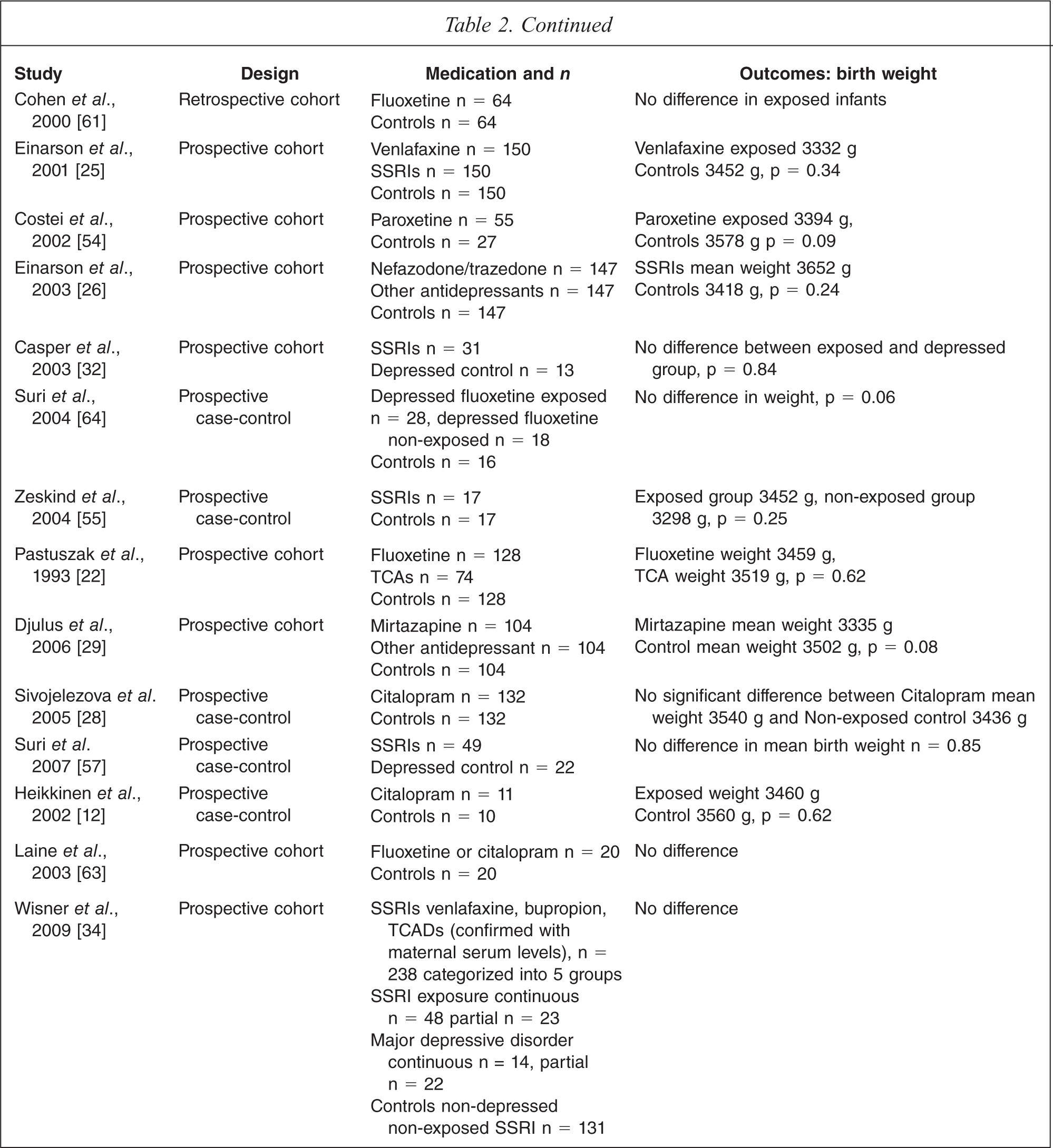
Birth weight
Twenty-three studies were identified that examined birth weight, with eight yielding positive findings. Lattimore's meta-analysis revealed a statistically significant risk of low birth weight for gestational age [52]. Seven other studies showed similar findings [20,38,43,44,53,56,65), but fifteen did not [12,22,25,26,28,29,32,34,54,57,60,61,64,66]. Oberlander attempted to increase the sensitivity of the study by using propensity scoring to control for confounders and still found an effect on birth weight related to exposure [56]. However, only two studies controlled for confounders such as depression, smoking, maternal age or maternal weight, and these showed no differences in birth weight in babies born to exposed or non-exposed mothers [34,64].
An increased possible risk of large birth weights following TCA treatment was reported in one study [37]. Although partially related to higher body mass index in mothers taking antidepressants, the effect did not completely disappear when this was controlled for.
Stillbirth
No studies specifically designed to look at the risk of stillbirth (i.e. late trimester obstetric loss) upon exposure to antidepressants were identified. Very large numbers would be needed to show any statistically significant differences. Of note, none of the large birth registry studies has shown any increased incidence of stillbirth in exposed infants.
Pregnancy complications
One study has shown an increased risk of hypertension and possibly pre-eclampsia in women exposed to SSRIs beyond the first trimester with a RR 4.9 (95%CI 2.7–8.8) [66]. However, this retrospective study was flawed, as it relied on patient recall of the diagnosis rather than any objective medical measures of blood pressure or other criteria for pre-eclampsia.
Summary
It is evidence that incidence of preterm birth is elevated in the majority of the studies that specifically examined this outcome, including some that attempt to control for the impact of the underlying depressive illness. The relationship is strengthened by one study finding that longer drug exposure is more likely to decrease gestational age. Other pregnancy outcomes such as reduced birth weight are weaker in their association. The data on rates of spontaneous abortions is limited and contradictory.
Antidepressants and neonatal outcomes
Neonatal adaptation
Seventeen controlled studies, including one meta-analysis, were analysed for information on neonatal outcomes after antidepressant exposure (Table 3). Fifteen of the studies show some association between gestational exposure to antidepressants and neonatal adaptation difficulties (N = 7869 out of 7900). Neonatal adaptation is measured in various ways, from gross markers such as NICU admission to more subtle evaluations such as behavioural observations, in different studies making it difficult to draw general conclusions.
Neonatal outcomes
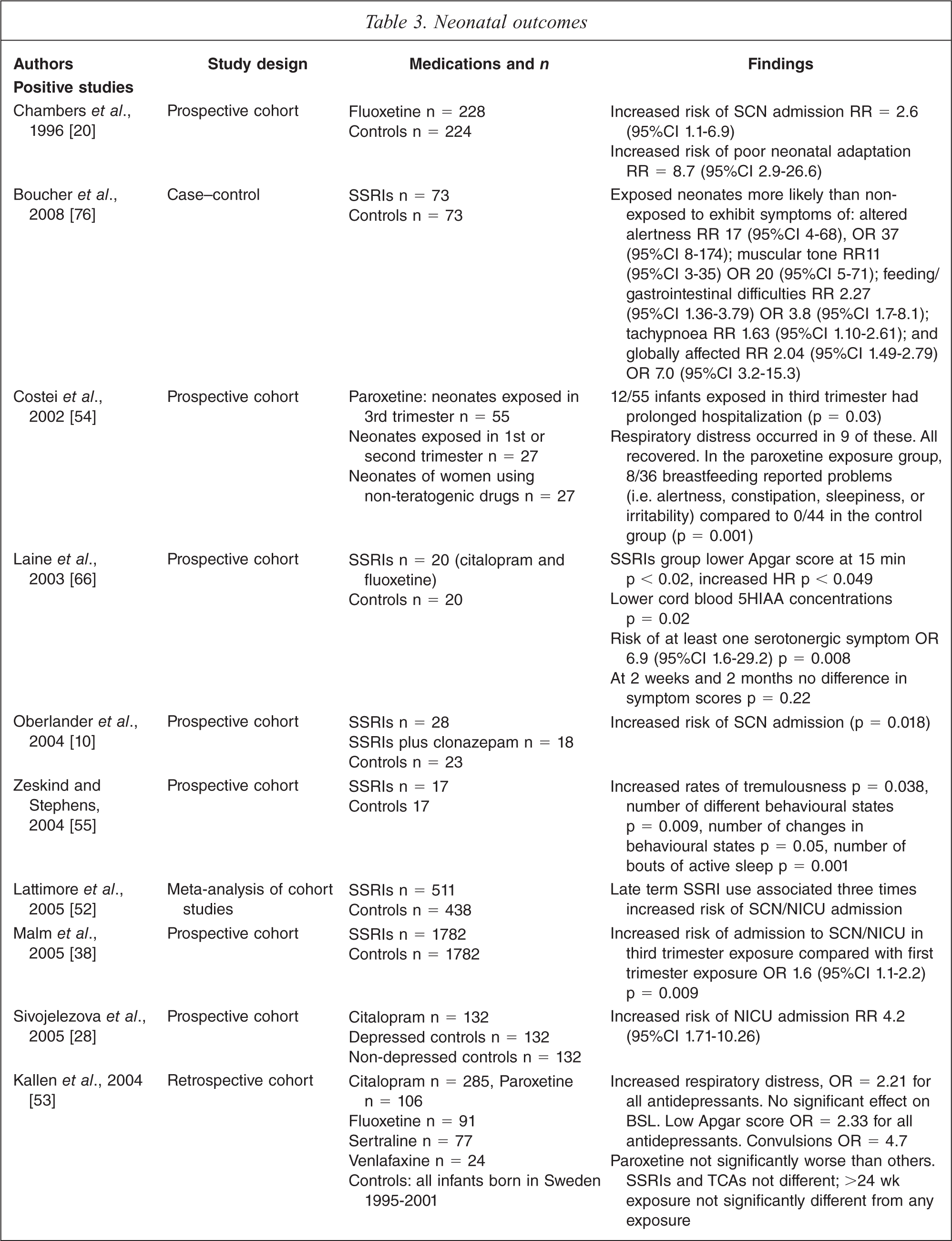
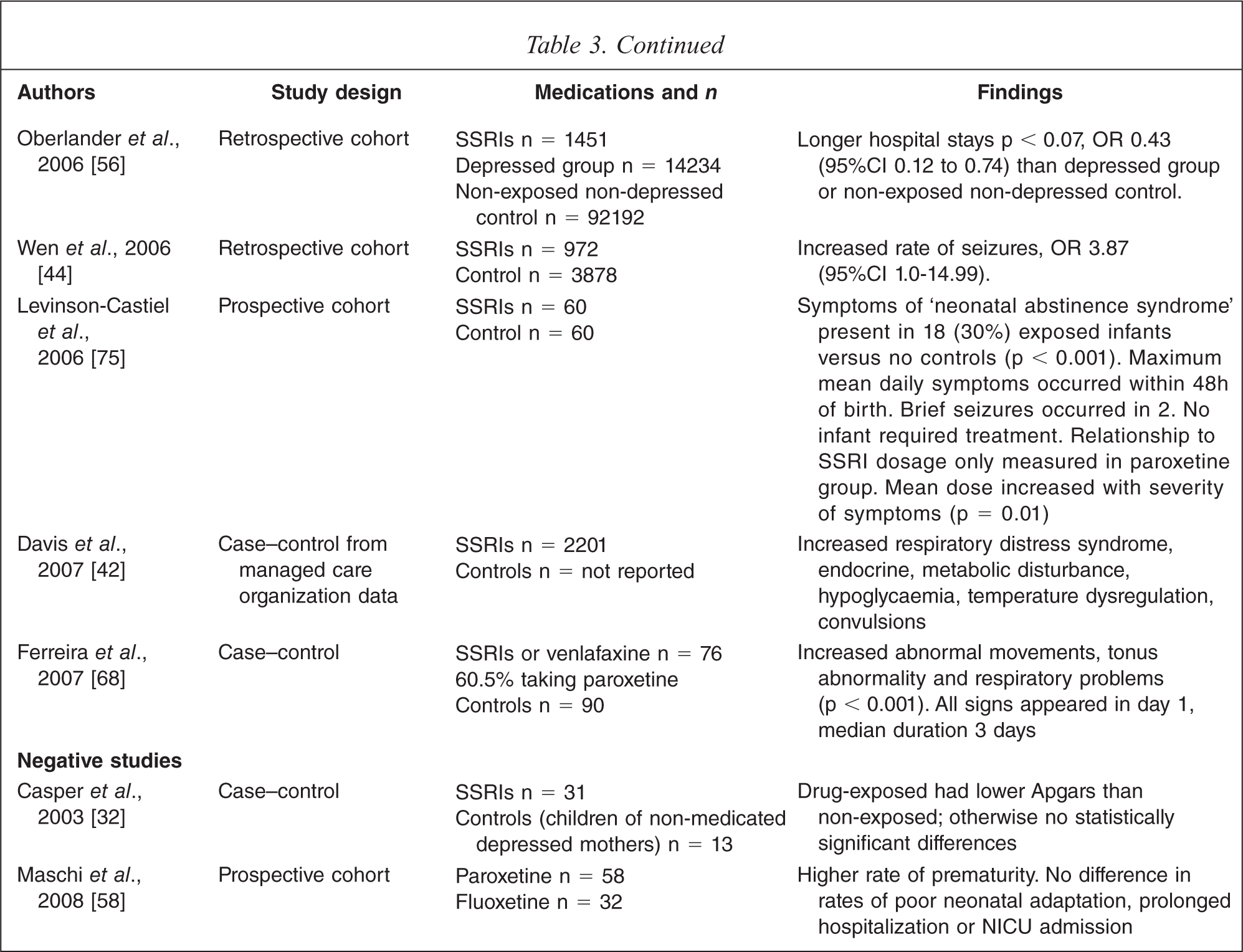
Large database or registry studies have variously suggested an increased risk of neonatal seizures [41–44,53,56], 1.5 times increased risk of NICU admission with third trimester exposure compared to first trimester exposure [38], and increased risks of respiratory distress and low APGAR scores [53].
The 2005 meta-analysis [52] of prospective controlled trials included consideration of 1066 mother–infant pairs and found a three-fold increased risk of SCN/NICU admission. None of the trials in the meta-analysis included a depressed, non-drug-treated group. Two subsequent studies do include such a group. Of these, Ferreira [68] found a significant (p < 0.001) increase in abnormal movements, tone and respiratory symptoms in 76 infants exposed to SSRIs or venlafaxine in the third trimester. Sivojelezova [28] found that 132 infants exposed to citalopram in pregnancy had a four-fold increased incidence of NICU admission compared to matched infants exposed to untreated maternal depression or controls.
Of the two controlled studies that did not find an association between antidepressant exposure and neonatal adaptation difficulties Casper [32] was possibly underpowered, whereas Maschi [58] was a larger trial including 200 women treated with antidepressants and 1200 controls.
Studies examining the neonatal effects of antenatal exposure to TCAs are limited to case reports and case series. Several case reports describe an association between gestational clomipramine [69–71] and imipramine [71–73] exposure with signs of neonatal adaptation difficulties. In a prospective case series [74] of 18 pregnant women on TCAs (predominantly imipramine), all nine with third trimester exposure had infants with adaptational difficulties.
With regard to the newer antidepressants, evidence is scant. Some of the controlled trials include venlafaxine exposure [53,68,75,76] and suggest similar neonatal adaptation difficulties to SSRIs. Mirtazapine features only in case reports [77,78] and in one controlled study [21,29], with a suggestion of both respiratory and thermoregulatory problems. One case report exists for duloxetine [79] documenting neonatal adaptation symptoms. No data were found for desvenlafaxine or reboxetine.
Summay
Taken together, the evidence strongly suggests that antenatal antidepressant exposure is associated with a higher risk of neonatal adaptation difficulties, though it is not possible at present to quantify that risk or to distinguish between individual medications on the basis of risk. Less credible evidence implicates TCAs, SNRIs and mirtazapine. While some of the syndromes have included such serious problems as seizures, or warranted NICU admission, the vast majority of cases appear to have been mild-to-moderate manifestations that resolved in a matter of days.
Persistent pulmonary hypertension of the newborn
Chambers et al., in a retrospective case–control design nested within the larger Slone Epidemiology Centre Birth Defects Study, noted an association between SSRI exposure beyond 20 weeks and persistent pulmonary hypertension of the newborn (PPHN) [80]. PPHN is a condition occurring in infants, leading to respiratory distress, and hypoxaemia and is a rare but potentially fatal condition with the possibility of adverse longer term developmental outcomes. There were 377 women whose infants had PPHN and 836 matched controls. The risk of PPHN associated with SSRI use at any time during pregnancy was not elevated. However, SSRI use after the 20th week of gestation was found to be significantly associated with PPHN OR 5.1 (95%CI 1.9 to 13.3). There was no increased risk with TCAs and venlafaxine. Extrapolating from population prevalence data for PPHN of 1–2 infants per 1000 live births, Chambers suggests that the absolute risk with SSRI use in late pregnancy would be 6–12 per 1000.
Subsequently, Kallen and Olausson [81], using the Swedish Medical Birth Register, conducted a prospective study that supports Chamber's findings. There were 504 infants with PPHN of the 831 324 born, with a mortality rate of 8.9%. Particularly for gestational ages greater than 34 weeks, PPHN was associated with early exposure RR 2.40 (95%CI 1.2–4.3) and with early and late SSRI exposure RR 3.6 (95%CI 1.2–8.3). In contrast, Andrade [82] conducted a retrospective study of SSRIs in the third trimester with 1104 exposed infants and 1104 matched unexposed infants. There was no increased risk of PPHN in the exposed group.
In all three studies to date addressing PPHN, there have been significant methodological weaknesses. Chambers’ study was retrospective and used self-report data, with the potential for recall bias [80]. Furthermore, it is unclear as to why 20 weeks gestation was chosen as a demarcation point. Kallen's group used prospective data, however, most of the information was collected at interview in the first trimester, thus missing women who ceased or started medication later [81]. Furthermore, multiple comparisons were made to demonstrate a statistically significant finding and there were large confidence intervals. One negative study [82], had relatively small numbers and it was only adequately powered to find a 6-fold increase in risk and may in fact have missed a smaller, increase.
Summay
The evidence concerning the association between late pregnancy SSRI exposure and this rare but serious condition could be summarized as suggestive of a real effect that is nonetheless small in absolute terms. It cannot be construed as sufficient to contraindicate the use of these agents in pregnancy. Complicating the risk–benefit analysis is the list of other putative maternal risk factors for PPHN, these include higher pre-pregnancy BMI, diabetes, Caesarean section delivery, and the use of non-steroid anti-inflammatory drugs.
Longer term developmental outcomes
Seven studies examined longer-term development as a primary outcome (Table 4). Six of these studies reported no significant difference in developmental outcomes in children exposed to antidepressant medication compared with the variously used control groups (N = 375 out of 402) [23,60,62,83–85]. The measures of developmental outcomes have ranged from the use of formal scales (Bayley Scales of Infant Development, Carey Temperament Scales, Achenbach Behaviour Checklist) to standard hospital physical examination.
Developmental outcomes
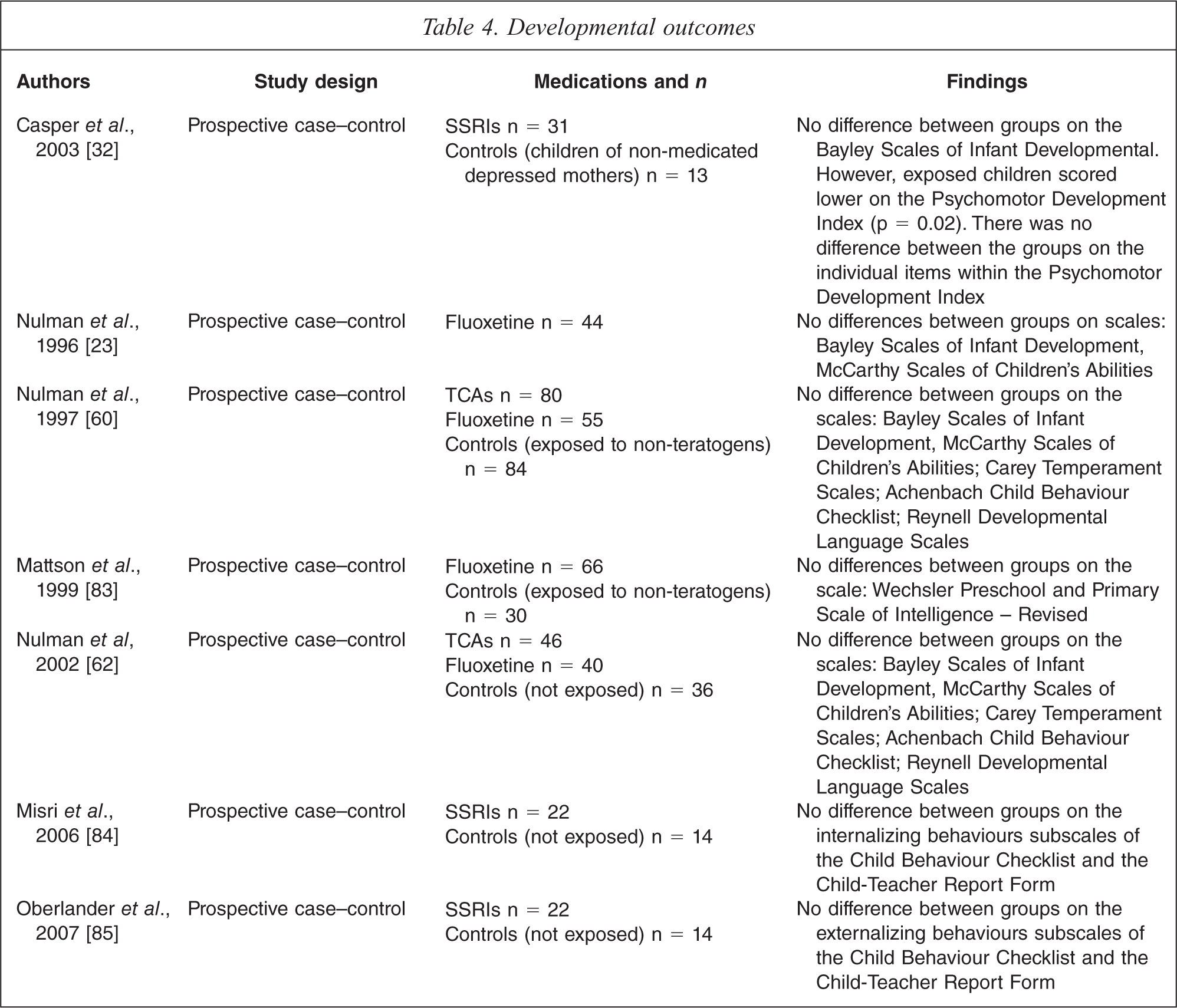
The only positive study was by Casper [32], which looked at 31 children of depressed mothers treated with SSRIs and a control group of 13 children of depressed mothers, who had elected not to take medication. At follow-up, the children ranged from 6 months to 40 months of age. There was no difference between the two groups on the Bayley Scales of Infant Development – Mental Developmental Index. There was a significant difference in the overall score for the Psychomotor Development Index with exposed children scoring lower. However, there was no difference in scores on individual items.
There are a numbers of studies comparing anti-depressants in pregnancy in terms of obstetric and neonatal outcomes [10,12,13,43]. Some of these studies also report on infant development. None of these studies reported a significant difference in developmental outcomes.
Summay
Of the small number of studies available, it is reassuring that the majority of these report no developmental differences between children exposed to antidepressant medication during pregnancy and those who are unexposed. Most single agent data is available for fluoxetine. There is little to no information on the newer antidepressants: venlafaxine, mirtazapine, escitalopram, reboxetine and duloxetine. Further, methodological issues include the wide range in age at which the children are assessed, and this heterogeneity lowers the possibility of positive findings. Whilst the majority of the studies were prospective and aimed to use a structured approach to developmental assessment, each of the scales has limitations, especially in terms of the clinical significance of statistical findings.
Discussion
The results outlined above reveal the complexity of studies in this area. Although a large number of pregnant women whose infants have been exposed to antidepressants have been investigated, a clear interpretation of the results is difficult. Studies vary in quality and many of the studies had insufficient numbers for adequate power to detect rare events. The larger register studies were limited by lack of proof of drug exposure and difficulty with follow-up. Study design is a further issue of concern. All designs had multiple confounders, which in many lacked controls. Few of the studies had controls for the underlying illness – a significant weakness. Retrospective designs are weakened by recall bias and often a high rate of non-responders. Teratology information studies (which were the prominent source of malformation studies) also had difficulties such as susceptibility to selection bias due to self recruiting. Finally, many studies conducted statistical tests on multiple parameters without the necessary correction.
With these limitations in mind, the overall impression is that these medications remain relatively safe in pregnancy but some notable areas of concern exist. In particular, a higher risk of preterm birth, neonatal adaptation difficulties, congenital cardiac malformations (with paroxetine) and possibly PPHN are outcomes for which there is more credible evidence.
Recommendations
As for all therapy, the clinician faced with the decision of how to treat depression and/or anxiety in a pregnant woman has to balance overall risks and benefits to both the mother and the foetus.
The decision in cases of mild illness is straightforward as evidence is available for the efficacy of non-pharmacological treatments such as psychotherapy. Similarly in women with severe illness, the risk of not treating far outweighs the risk of treating for both mother and infant.
The more complex decision making involves women with moderate illness where the risk–benefit balance is far more subtle. In these situations, thorough clinical assessment and detailed discussion with the patient and partner, providing clear information about options, is essential. Close liaison with other clinicians involved is prudent clinical practice. It is also vital that if the decision is made to withdraw medication, the patient is closely supervised so that early intervention is available should the woman relapse. In women presenting with their first episode of major depressive or anxiety disorder, who are medication naïve, we would recommend first line treatment with a SSRI (other than paroxetine).
In women with a clear history of recurrent illness and relapse off medication a case can be made for continuation of the most effective previous medication, with the exception of paroxetine where a trial of another SSRI could be considered. In these women, a history of premature labour or recurrent spontaneous abortions may raise the threshold for consideration of antidepressant use.
There is insufficient information available at the moment to give recommendations to women taking venlafaxine, mirtazapine, and the newer antidepressants such as duloxetine. In these cases, the clinician should discuss the limitations of knowledge and guide the patient through the process of evaluating their history, and the risks of altering beneficial treatment versus the risks of continuing or changing to other options.
Given the evidence for neonatal adaptation difficulties with antidepressant use, the availability of paediatric support is recommended.
Some guidelines such as the Therapeutic Guidelines [86] and NICE [87] recommend the use of TCAs over SSRIs. Our review does not support this, as TCAs are represented in positive studies outlined above, and there have been no specific studies comparing the efficacy of TCAs and SSRIs in pregnancy. Our findings and recommendations broadly concur with those of a recent report published jointly by the American Psychiatric Association and the American College of Obstetricians and Gynaecologists [88].
Withdrawal or tapering of antidepressants before delivery is recommended by some clinicians, with the rationale of preventing neonatal adaptation difficulties. However, one study [89] that specifically addressed this found no differences in neonatal outcomes after cessation of SSRIs fourteen days prior to delivery. In view of the absence of evidence for positive outcomes and the high risk of leaving mothers without antidepressant cover at the time of peak vulnerability, this practice is not recommended.
Future directions
Further research is essential to increase the information available, to clarify areas of concern and to widen the range of medications studied. It is also important that some of the methodological difficulties in study design are addressed so that clarity can be provided. Future studies should aim to ensure adequate sample sizes to detect real differences, have clear stated prior outcomes to minimize multiple comparisons and should control for confounders including the women's underlying illness so that drug effects and illness effects can be separated.
Footnotes
Acknowledgements
The authors are grateful to the other members of the Melbourne Perinatal Psychotropic Review Group: Anne Buist, Megan Galbally, Matthew Roberts, Fiona Judd, Lia Laios, Justin Dwyer; and would also like to thank Ross Coppell and Malcolm Hopwood.
