Abstract
Objective:
To review the recent academic literature surrounding the prevalence, aetiopathology, associations and management of depression in chronic kidney disease (CKD), in order to provide a practical and up-to-date resource for clinicians.
Methods:
We conducted electronic searches of the following databases: MEDLINE, EMBASE and PsycINFO. The main search terms were: depression, mood disorders, depressive disorder, mental illness, in combination with kidney disease, renal insufficiency, dialysis, kidney failure. Separate searches were conducted regarding antidepressant use in CKD.
Results:
A number of recent, large and well-conducted studies have confirmed markedly raised rates of depression amongst those with CKD, with meta-analysis suggesting the prevalence of interview-defined depression to be approximately 20%. The interactions between depression and CKD are complex, bidirectional and multifactorial. Depression in CKD has been shown to be associated with multiple poor outcomes, including increased mortality and hospitalisation rates, as well as poorer treatment compliance and quality of life. Clinical evaluation of depression in patients with CKD can be challenging; however, once a diagnosis is made, a range of treatment modalities can be considered.
Conclusions:
Depression is common in CKD and is associated with a significant risk of adverse outcomes. Given the importance of this issue, there is now an urgent need for well-conducted randomised trials of interventions for depression in CKD in order to provide information on the safety and efficacy of treatments.
Introduction
Until recently, the bulk of the academic literature around the overlap between chronic physical illness and mental health has tended to focus on cardiovascular disease and depression. However, there have been a number of large studies published which suggest that the association between depression and several other chronic somatic conditions, including renal disease, are equally important.
Chronic kidney disease (CKD) is a growing problem that affects approximately 10% of the global population (Palmer et al., 2013b). Most of the data on depression in CKD have focused on end-stage kidney disease (ESKD), which is defined when there is permanent kidney failure requiring regular renal replacement therapy (dialysis or transplant).
It is well established that depression is more common amongst those with chronic physical health problems (Harvey and Ismail, 2008; Olver and Hopwood, 2012; Rayner et al., 2010). However, depression amongst those with CKD has been estimated to be even greater than that reported for patients with other chronic diseases (Palmer et al., 2013b). Furthermore, depression in CKD has been shown to be associated with multiple poor outcomes (Chilcot et al., 2011; Hedayati et al., 2010; Kimmel et al., 2000; Koo et al., 2005; Palmer et al., 2013a). Unfortunately, despite these associations and the increasingly available evidence, clinicians remain cautious when managing depression in those with CKD, and rates of detection and treatment remain very low (Hedayati and Finkelstein, 2009).
This review will discuss the prevalence of depression in patients with CKD, propose a model for the complex interactions between depression and CKD, highlight the important consequences of comorbid depression, and, finally, synthesise the available evidence in order to provide guidance to clinicians for the appropriate management of depression in this population. As the bulk of the research to date has been in those with ESKD, this will be the primary focus of this review.
Review methodology
Electronic searches were conducted using title and subject headings for ‘depression’, ‘mood disorders’, ‘depressive disorder’, ‘mental health’, ‘mental illness’ and ‘psychiatric’, in combination with ‘kidney disease’, ‘renal dialysis’, renal insufficiency’ and ‘kidney failure’. The search was conducted up to and including 28 March 2013, and was limited to those papers written in English.
While not producing clinical management guidelines as such, this review aims to summarise the evidence regarding depression in CKD in order to aid clinical decisions. One issue which can be particularly challenging for clinicians is the use of antidepressant medications in the setting of CKD. A separate search was conducted to identify studies examining the efficacy and pharmacokinetics for antidepressants in the setting of impaired renal function. This, together with previous guidelines (Taylor et al., 2012) was used to provide recommendations on the safety of a range of commonly prescribed antidepressants. Each antidepressant was classified into one of three categories: ‘evidence available suggests agents are usually safe to use, but may require additional monitoring and dose alteration’, ‘evidence available suggests can be used in CKD, but greater caution needed’, or ‘not recommended for routine use in CKD’. In order to be classified in the first category, an antidepressant had to have published evidence of efficacy in CKD and not have any major or common adverse effects likely to be particularly problematic in renal disease.
Epidemiology
Attempts to quantify the prevalence of depression in CKD using self-report scales have often produced widely varying estimates. Palmer et al. (2013b) recently conducted a systematic review and meta-analysis of studies to summarise the point prevalence of depressive symptoms in adults with CKD. A total of 249 populations were included (55,982 participants) and analyses were limited to studies that used clinical interview and pre-specified criteria for diagnosis. The authors found the summary prevalence of interview-defined depression to be 20.3%. Using self or clinician-administered rating scales, the prevalence of depressive symptoms for CKD was higher, suggesting that self-report scales may overestimate the presence of depression, particularly in the renal setting. The frequency of somatic symptoms commonly experienced in these patients, such as fatigue, sleep disturbance and decreased appetite, may partly explain this overestimate.
Table 1 attempts to place these prevalence rates in the context of other chronic medical illnesses. This table shows the rates of interview-defined depression, using either structured or semi-structured interviews that have been observed in other clinical settings. Prevalence rates of depression were obtained from meta-analyses or systematic reviews, where available.
Prevalence of interview-defined depression in a variety of chronic medical illnesses.
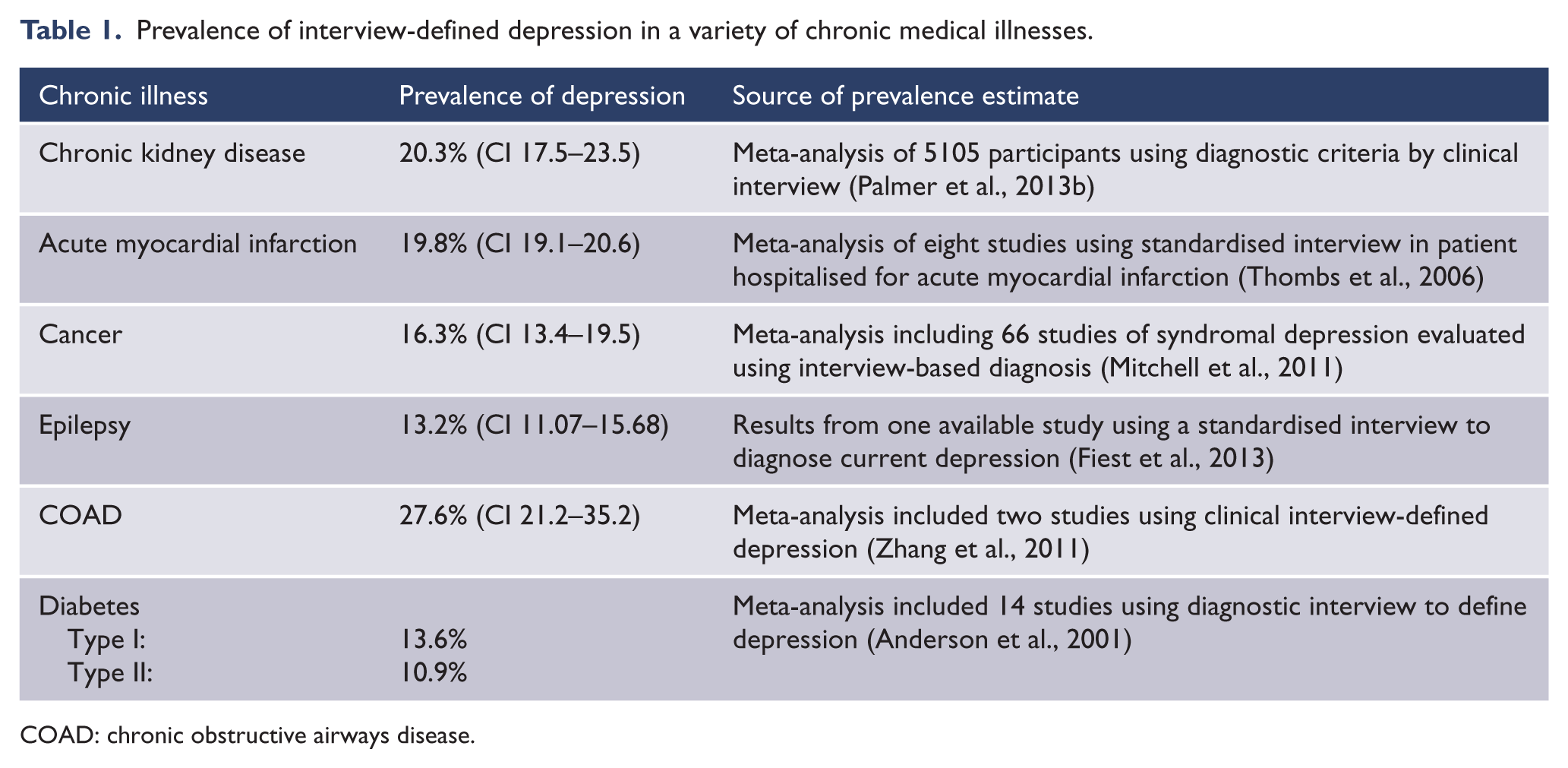
COAD: chronic obstructive airways disease.
Why is depression more common in CKD?
The complex interactions between depression and CKD are dynamic and multifactorial. In discussing the relationship between depression and CKD, it is helpful to consider shared ‘upstream’ risk factors, as well as the biopsychosocial or ‘downstream’ consequences of both illnesses. This is illustrated in Figure 1 using a model adapted from that originally proposed by Katon (2011), which can be applied to many chronic diseases.
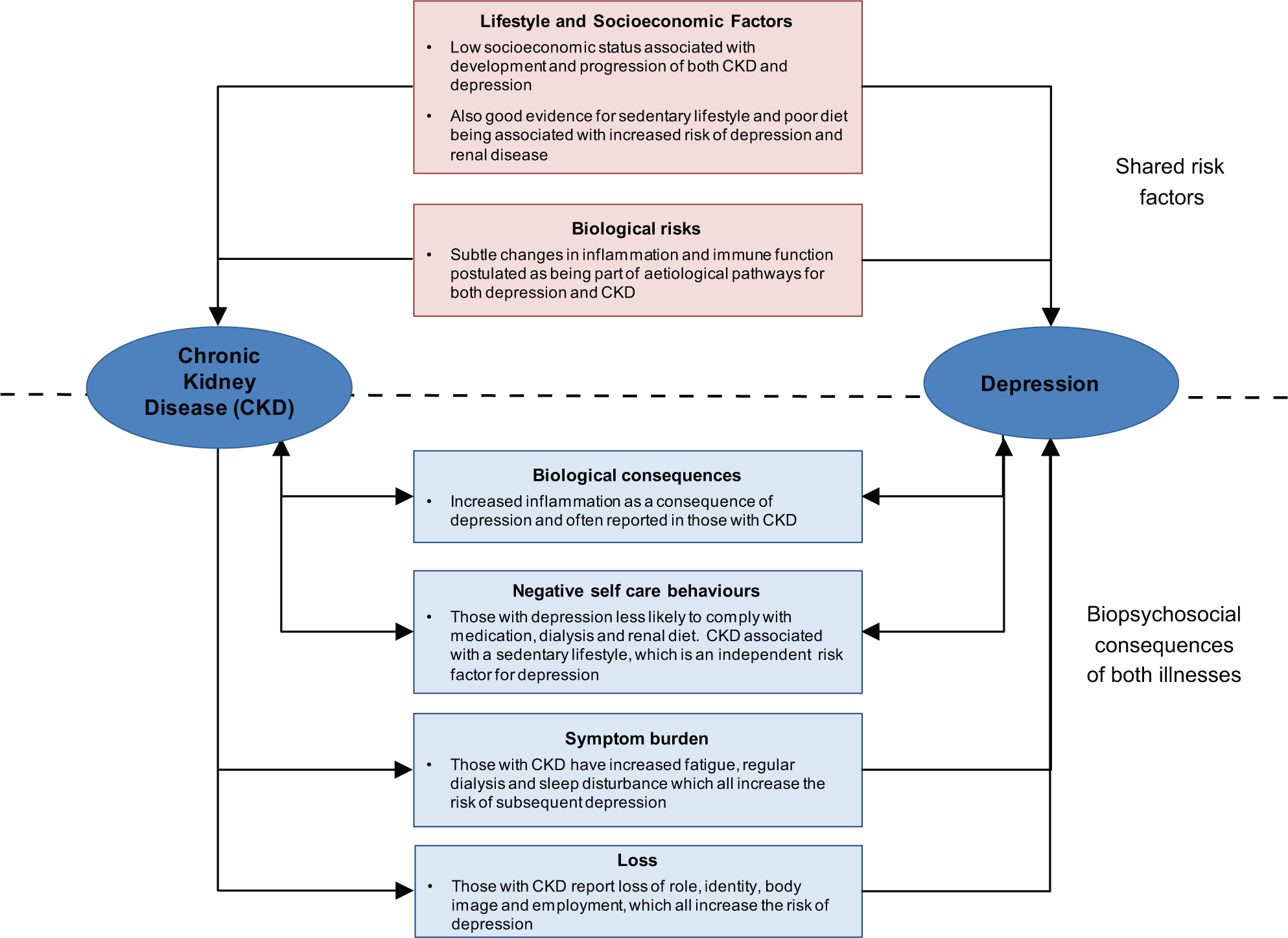
Complex relationship between chronic kidney disease and depression.
Both childhood adversity and socioeconomic factors are some of the most important risk factors for depression (Hatch et al., 2010; Lorant et al., 2003). Socioeconomic adversity is also associated with adverse health behaviours such as smoking, poor diet and sedentary lifestyle, as well as delayed help-seeking for established health problems. These factors increase the risk of, and impair the management of, diabetes and cardiovascular disease, as well as increasing the risk of depression (Harvey et al., 2010; Rivenes et al., 2009). As diabetes and cardiovascular disease are common causes of renal impairment, the importance of these socioeconomic and lifestyle factors cannot be overlooked.
There are other biological elements that can be considered to be both part of the shared ‘upstream’ risk factors, as well as consequences of CKD and depression. These include the immune system, inflammatory pathways, disturbances of the hypothalamic pituitary axis and changes in the parasympathetic and sympathetic nervous systems (Ahrens et al., 2008; Raison et al., 2006; Zunszain et al., 2013). One biological element that has been suggested as being particularly relevant for understanding the bidirectional link between CKD and depression is inflammation. Higher concentrations of pro-inflammatory cytokines have been identified among clinically depressed individuals and those with symptoms of depression, although there is considerable heterogeneity between studies (Dowlati et al., 2010; Howren et al., 2009). Furthermore, there is evidence to suggest that these pro-inflammatory cytokines interact with many of the pathophysiological mechanisms of depression, including neurotransmitter metabolism, neuroendocrine function, synaptic plasticity and behaviour (Raison et al., 2006). CKD is often considered to be a pro-inflammatory state with increased inflammatory markers and oxidative stress (Himmelfarb, 2008; Stenvinkel and Alvestrand, 2002), although attempts to demonstrate this have been somewhat mixed. This chronic pro-inflammatory state is thought to be related to the higher than expected rates of cardiovascular disease and other causes of increased mortality in this population (Kimmel et al., 1998b; Stenvinkel et al., 2005). It is possible that these factors could also partly explain the high rates of depression seen in this population.
Finally, loss is an important psychological theme to consider in examining depression in those with CKD. In a study of 151 ESKD patients, perceptions of loss were the strongest predictors of depression, which in turn predicted the quality of life (QOL) (Chan et al., 2009). In the early stages of CKD, people may feel a loss of wellness, as well as the more practical losses relating to lifestyle and independence. Loss of identity and primary role function are also important areas to consider. Those on dialysis often dislike, or have ambivalent feelings towards the treatment; being dependent on such treatment for life, while having to endure a loss of autonomy and control. Additionally, these factors may contribute to patients with CKD having feelings of guilt for the burden they perceive they cause for family members and carers. Not surprisingly, self-concept and self-esteem may be challenged.
Why does depression in CKD matter?
Accumulating evidence has shown that clinical depression and subthreshold depressive symptoms are associated with an increased risk for adverse clinical outcomes in patients with CKD. These negative outcomes include increased mortality and rates of hospitalisation, poor adherence to treatment and decreased QOL.
Mortality
The relationship between ESKD and survival has been somewhat controversial. Studies from the 1980s suggested that depression did significantly predict mortality in ESKD patients. However, as Halen et al. (2012) highlight, these studies used relatively small sample sizes and had some methodological limitations. Some later studies (Koo et al., 2003; Watnick et al., 2003), using more standardised measures for depression and advanced statistical methods, have failed to demonstrate an effect of depression on all-cause mortality in ESKD patients.
The Cochrane Renal Group have recently completed a systematic review and meta-analysis that helps to clarify this issue of depression and mortality in CKD (Palmer et al., 2013a). This paper included 22 cohort studies (83,381 participants) of depressed adults with CKD. Overall, a strong and statistically significant association was found between depression status and risk of all-cause mortality (RR 1.59; 95% CI: 1.35–1.87). These results remained consistent across stages of CKD, regardless of sex or age. Furthermore, the excess mortality risk attributable to depression in CKD is higher than that seen in other chronically diseased populations, such as cancer, diabetes and heart disease (Palmer et al., 2013b).
Hospitalisation
Multiple studies have shown that depression is associated with increased healthcare costs, including primary, pharmacy, inpatient medical, inpatient psychiatric and outpatient mental health care (Katon, 2011). DOPPS (The Dialysis Outcomes and Practice Patterns Study) was a large, international, prospective, observational study of 5256 haemodialysis patients which found that, after adjusting for time on dialysis, age, race, socioeconomic status, comorbid indicators and country, depression was associated with an increased risk for first hospitalisation (Lopes et al., 2002). Hedayati et al. have also conducted several studies attempting to address the association between depression and a variety of negative health outcomes in renal disease, including dialysis initiation, hospitalisation rates and length of stay, and death. In each of these studies an association between depression and hospitalisation has been shown (Hedayati et al., 2005, 2008, 2010).
Adherence
Treatment non-adherence represents one of the potential pathways through which depression may affect mortality and morbidity in patients with CKD. Treatment compliance in CKD patients is quite complex, with patients having to comply not only with the dialysis schedule, but also prescribed medication and dietary regimens.
Measuring adherence in ESKD patients presents particular logistical difficulties (Chilcot et al., 2010); however, most studies have indicated a relationship between depressive affect and both laboratory and behavioural markers of poor compliance in the dialysis patient (Kaveh and Kimmel, 2001; Koo et al., 2003; Leggat, 2005; Rosenthal Asher et al., 2012). Depressive symptoms of low motivation, impaired concentration and memory, and apathy can significantly interfere with patients’ adherence to complex and challenging treatment plans. This is important, as decreased behavioural adherence (skipping dialysis sessions, shortening dialysis time) has been shown to be associated with decreased survival (Kimmel et al., 1998a; Rosenthal Asher et al., 2012).
Adherence to a dietary prescription is a particular area of importance as nutritional status has been shown to significantly impact the course and outcome of ESKD. Depression is associated with impaired nutrition in dialysis patients (Koo et al., 2003, 2005), with non-adherence rates to prescribed diet and fluid restriction recommendations of approximately 50% (Kugler et al., 2005). Importantly, antidepressant therapy in conjunction with supportive psychotherapy appears to improve nutritional status in depressed individuals with CKD (Koo et al., 2003, 2005).
Finally, depressed individuals on dialysis therapy are also significantly more likely to withdraw from treatment (Lacson et al., 2012; McDade-Montez et al., 2006) than non-depressed dialysis patients.
Quality of life
It is now widely accepted that health-related quality of life (HRQOL) is significantly compromised in patients with ESKD (Soni et al., 2010). Furthermore, HRQOL has been associated with increased morbidity and mortality. Depression has been found to have a profound negative impact on HRQOL in CKD patients (Kimmel, 2000; Weisbord et al., 2005). Additionally, it has been proposed that depression and anxiety may be more strongly associated with HRQOL in CKD than clinical and socio-demographic variables taken together (Vazquez et al., 2005). Depression can impact on HRQOL in a number of ways. Patients with depression have been found to have two- to threefold more medical symptoms compared with controls without depression (Katon, 2011). In addition to this higher symptom burden, ESKD patients with depression also experience more fatigue, cognitive difficulties, pain, sleep disturbances, sexual dysfunction and relationship difficulties (Soni et al., 2010). Aside from increased levels of symptoms, depression is also linked with reduced functional performance and increased rates of occupational disability (Harvey et al., 2011; Knudsen et al., 2013), often leading to financial strain and reduced well-being.
HRQOL is important in and of itself; however, as both HRQOL and depression are related to survival in dialysis patients (Hedayati et al., 2008; Kimmel, 2000; Mapes et al., 2003), this negative association has a clear clinical importance.
Clinical aspects of depression in CKD
Depression is often underdiagnosed in patients with serious physical illness and CKD is no exception. Dialysis physicians and nurses often fail to recognise symptoms of depression (Feroze et al., 2010). There may be many reasons for this, including a lack of knowledge or confidence when asking about mental health symptoms, as well as a focus, both from the medical staff and the patients, on physical symptoms alone. Although it has been shown that somatic symptoms are frequently the presenting complaint of those with a chronic medical illness who are eventually diagnosed with depression, physicians often pay less attention to somatic complaints of psychiatric illness (Farrokhi, 2012). An additional problem is the overlap between the physical symptoms of depression and those of the underlying medical illness. This is critical to the clinical evaluation of a patient with ESKD. In particular, the depressive symptoms of psychomotor agitation or retardation, appetite and weight changes, sleep disturbance, and aches and pains are often difficult to distinguish from anorexia, sleep disturbance and neuropathy secondary to underlying ESKD, particularly when uraemia is present (Halen et al., 2012).
Other factors to consider in evaluating the potential diagnosis of depression in a patient with ESKD include anaemia, electrolyte disturbances, side effects from medication and symptoms from other systemic illnesses. It is generally recommended that the diagnosis of depression in a patient with comorbid CKD should rely more heavily on psychological features, such as anhedonia, guilt, loss of self-esteem, hopelessness and suicidal ideation (Hedayati et al., 2012). Structured clinical interviews allow for a more thorough examination of the patient’s cognitive set, and as such are considered the gold standard method for distinguishing depressive symptoms from somatic complaints.
Screening
The issue of screening for depression in CKD has been somewhat controversial. Some, such as Palmer et al. (2013b), assert that there is insufficient high-quality data available to show that screening for, or treatment of, depression in CKD populations improves clinical symptoms and QOL. Farrokhi (2012) also suggests that before screening programs for depression in CKD are recommended, the benefits and harms of psychiatric intervention in this population need to be evaluated.
However, others, such as Hedayati and Finkelstein (2009), argue for routine screening based on the high prevalence of depression and associated poor outcomes in this population. Zalai asserts that, although routine screening for depression per se might not lead to better outcomes, it may help identify people whose psychosocial distress would otherwise remain undetected (Zalai et al., 2012). Hedayati has suggested that screening for depression should take place at key points of transition, such as on the initial evaluation of the CKD patient, at dialysis initiation, and then at regular intervals thereafter (Hedayati et al., 2012).
Several scales have been validated against Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV)-structured interviews among patients with CKD and ESKD, including the Beck Depression Inventory (BDI), the Hospital Anxiety and Depression Scale (HADS) and the Center for Epidemiologic Studies Depression Scale (CES-D). However, because of the overlap between the symptoms of depression and CKD, the cut-off scores suggested when screening depressive symptoms in the CKD population are generally the same or higher than those used in the general population. For example, when using the BDI, the recommended cut-off score to define clinically significant depression in the general population is 11 or greater (Beck et al., 1988); however, a cut-off of 11 or higher (Hedayati and Finkelstein, 2009) and between 14 and 17 (Hedayati et al., 2006; Preljevic et al., 2012) is suggested for the CKD and ESKD populations, respectively.
Management
Despite the high prevalence of depression and its association with poor outcomes, only a minority of patients with ESKD are given a diagnosis of, or receive treatment for, depression (Hedayati and Finkelstein, 2009). This may in part be due to the limited systematic study in this area, with only six small randomised controlled trials (RCTs) available to guide treatment safety and efficacy (Ancarani, 1993; Baines et al., 2004; Blumenfield et al., 1997; Duarte et al., 2009; Koo et al., 2005; Lii et al., 2007). The following section reviews what pharmacological and non-pharmacological approaches are available for the management of depression in CKD.
Pharmacological treatment
Depression in CKD is often undertreated. In their 2006 study using a DSM-IV-validated interview, Hedayati et al. (2006) found that less than half of the depressed patients with ESKD were being treated with antidepressants and about half of those on drug treatment were receiving subtherapeutic doses. Undertreatment of depression and underdosing of antidepressant agents may be in part caused by physicians’ concerns regarding efficacy and adverse effects. Little research has been performed regarding the safety of antidepressant medication use in patients with CKD, with such patients often being excluded from trials because of concerns for safety. In fact, to date, there has been only one completed RCT of antidepressant medication in CKD (Blumenfield et al., 1997). This was a small trial of 14 chronic haemodialysis patients with major depression. A statistically significant improvement in depression was found after 4 weeks amongst those given fluoxetine; however, this was not sustained at 8 weeks. Non-randomised, observational studies (Ancarani, 1993; Baines et al., 2004; Blumenfield et al., 1997; Duarte et al., 2009; Koo et al., 2005; Lii et al., 2007) suggest that antidepressant medication may be useful in CKD; however, further studies are needed.
Generally, antidepressant medications are hepatically metabolised, highly protein bound and not removed significantly by dialysis (Hedayati and Finkelstein, 2009). Thus, the relative activity and mode of excretion of metabolites of these drugs in patients with CKD is often uncertain. Further concerns include an increased risk of drug interactions (monoamine oxidase inhibitors, tricyclics and selective serotonin reuptake inhibitors (SSRIs)) (Cohen et al., 2004; Hedayati and Finkelstein, 2009), anticholinergic effects, including urinary retention (tricyclics) (Cohen et al., 2004; Hedayati and Finkelstein, 2009), QTc prolongation (tricyclics) (Cohen et al., 2004; Hedayati and Finkelstein, 2009), accumulation of toxic metabolites (venlafaxine and bupropion) (Cohen et al., 2004) and increased risk of bleeding (SSRIs) (Yuan et al., 2006). Given the limited evidence base, the decision regarding which antidepressant to use in those with CKD is based on its pharmacokinetics, drug-to-drug interactions and adverse event profile. Information regarding the pharmacokinetics and recommended dosage alterations for a range of commonly used antidepressants is provided in Table 2.
Evidence relating to the use of some common antidepressant medications in patients with CKD.
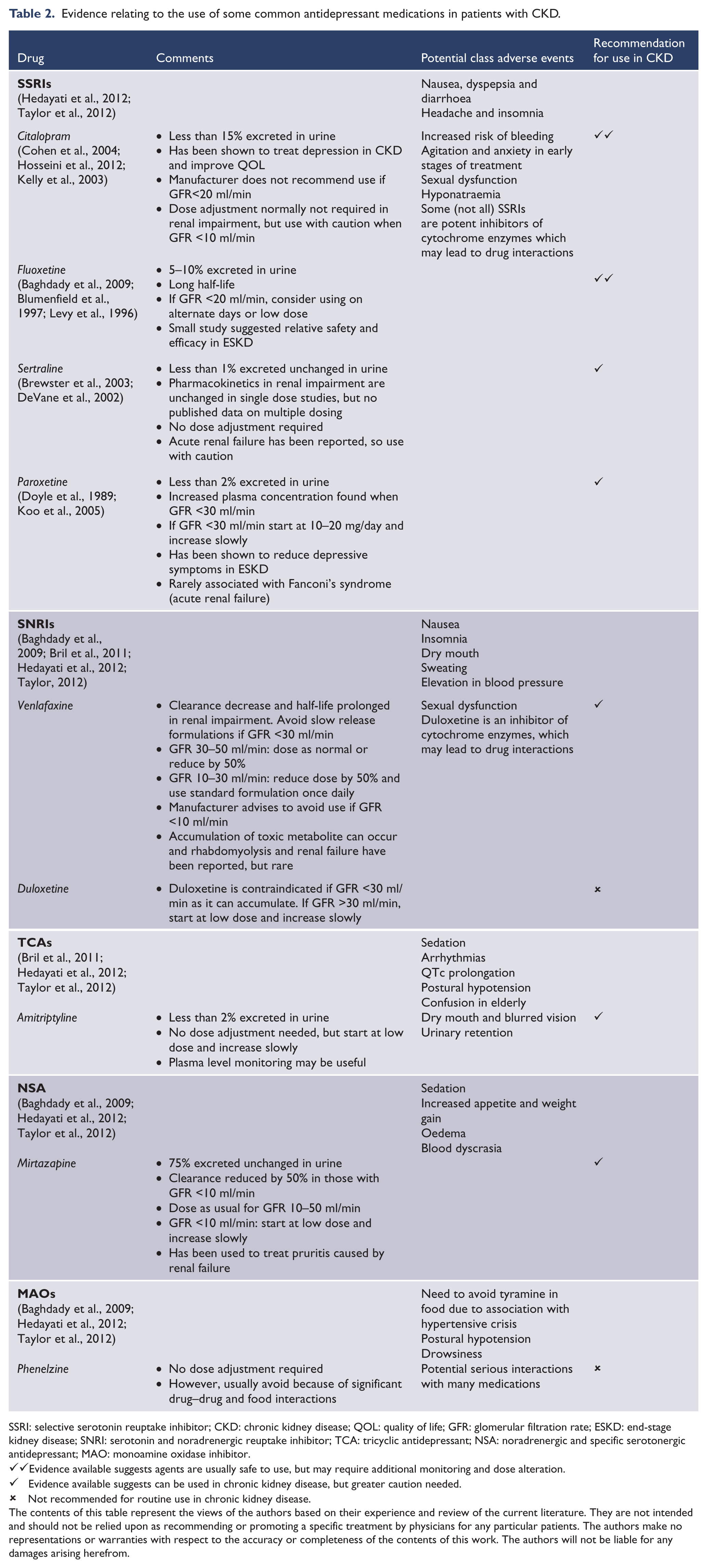
SSRI: selective serotonin reuptake inhibitor; CKD: chronic kidney disease; QOL: quality of life; GFR: glomerular filtration rate; ESKD: end-stage kidney disease; SNRI: serotonin and noradrenergic reuptake inhibitor; TCA: tricyclic antidepressant; NSA: noradrenergic and specific serotonergic antidepressant; MAO: monoamine oxidase inhibitor.
Evidence available suggests agents are usually safe to use, but may require additional monitoring and dose alteration.
Evidence available suggests can be used in chronic kidney disease, but greater caution needed.
Not recommended for routine use in chronic kidney disease.
The contents of this table represent the views of the authors based on their experience and review of the current literature. They are not intended and should not be relied upon as recommending or promoting a specific treatment by physicians for any particular patients. The authors make no representations or warranties with respect to the accuracy or completeness of the contents of this work. The authors will not be liable for any damages arising herefrom.
Non-pharmacological management
The challenges associated with utilising pharmacological strategies for the treatment of depression in CKD suggest effective non-pharmacological treatments may be of particular use in this population. Examples of non-pharmacological treatments include electroconvulsive therapy, cognitive behavioural therapy (CBT), exercise therapy and changes to the dialysis regimen.
Electroconvulsive therapy (ECT)
ECT is a highly effective treatment for severe depression, including medication-resistant depression (Carney, 2003; Rasmussen et al., 2002). The use of ECT has not been studied in RCTs in CKD, but there are case reports of excellent response to ECT in patients with CKD and severe depression refractory to antidepressant medication (Varghese et al., 2006; Williams and Ostroff, 2005). Special precautions are required in the CKD patient, including control of abrupt increases in blood pressure, adequate muscle relaxation to prevent strong contractions and the subsequent risk of fractures in an osteopenic patient (Williams and Ostroff, 2005), careful management of potassium levels which are further increased by succinylcholine – a muscle relaxant commonly administered during ECT (Horton and Fergusson, 1988), and attention to ECT dose levels, ideally established by individual seizure threshold titration, as the latter may be altered by acidosis and hypocalcaemia (Varghese et al., 2006).
Cognitive behavioural therapy (CBT)
CBT is a well-documented, evidence-based therapy for the treatment of depression. Importantly, for those in the general hospital setting, CBT may also have benefits beyond the treatment of depression, including improving medication adherence and reducing the severity of pain and other symptoms (Lloyd and Guthrie, 2007). The ability of CBT to change underlying attitudes to illness is also very relevant, given that negative beliefs regarding underlying illness schemata have been shown to be associated with higher scores on the BDI (Ghuzman and Nicassio, 2003).
Although there have been many RCTs of CBT for the treatment of depression in the physically ill (Berkman et al., 2003; de Godoy and de Godoy, 2003; Safren et al., 2014), there is a distinct lack of such trials in the CKD population. However, the observational studies and the few small RCTs that are available show mostly promising results. In a recent study of group CBT (Duarte et al., 2009), 85 dialysis patients with interview-diagnosed depression were randomly assigned to standard care versus group CBT with a psychologist weekly for 12 weeks. After 3 months, a significant difference in BDI scores was found between treatment and control groups. Additionally, improvement was found in several domains on a QOL measure. An interesting study conducted in Louisiana after hurricanes Katrina and Rita shows that CBT may not have to be delivered by psychologists (Weiner et al., 2010). In this study, 22 social workers implemented a cognitive behavioural intervention to 69 ESKD patients. Patients who participated in these sessions showed a significant reduction in depressive symptoms compared to patients who did not discuss the material.
Exercise therapy
There is increasing evidence of a link between levels of physical activity and an individuals’ mood (Harvey et al., 2010). In keeping with this emerging research, it appears that exercise programs, in addition to their primary aims of optimising physical function, cardiovascular risk and efficacy of dialysis, may also have a beneficial effect on depressive symptoms and various measures of HRQOL in patients with CKD (Greenwood et al., 2012).
In a study (Ouzouni et al., 2009) of 35 haemodialysis (HD) patients randomly assigned to a 10-month intradialytic exercise training program, a 39% reduction in depressive symptoms (as measured by BDI scores) was found in the exercise group compared to the control group. Additionally, another study (Kouidi et al., 2010) of 24 haemodialysis patients randomised to a 1-year intradialytic exercise training program showed a BDI score decrease of 34.5% (p < 0.001). Greenwood et al. (2012) conducted a larger, albeit uncontrolled, study of 131 participants at various stages of CKD, and showed that there was a significant improvement in symptoms of depression (29%) following a 12-week renal rehabilitation exercise program. However, this study also illustrates one of the main problems with exercise therapy, with only 55% of participants regularly completing the exercise program.
Change in dialysis regimen
It is not clear whether increasing the frequency of dialysis improves depression rates. The Following Rehabilitation, Economics and Everyday-Dialysis Outcome Measurements (FREEDOM) study (Jaber et al., 2010) was an observational cohort study with 128 completers. Dialysis frequency was increased from the standard three times weekly to six times weekly, with a targeted standardised weekly KT/V – a measure of dialysis adequacy – of greater than 2.1. At the end of the trial, a significant decrease in BDI values was found, and this was sustained at 12 months. However, the results of the intention-to-treat analysis were less clear. Additionally, it is important to note that this study did not have a control group, and participants were found to have had an increase in prescribed antidepressant use during the study. A recent, large, randomised trial (Group et al., 2010) did not find a statistically significant difference in BDI scores after a 12-month period of six times per week HD versus the conventional three times per week, although scores were lower in the six times per week group. It is difficult to determine if these lower scores are due to expected improvements in somatic symptoms secondary to more frequent dialysis, or are true improvements in depressive symptoms.
Others
Given the magnitude of the problem of depression in patients with CKD, additional therapeutic approaches need to be examined. Marriage and family tensions in CKD patients are well documented (Daneker et al., 2001). Interventions that address these relationship issues need to be explored, using, for example, family and marital counselling. Additionally, building up meaningful social supports, through the involvement of community, leisure or religious organisations, may help (Hedayati et al., 2012), both for the patient and caregivers. This is important as caregiver burden and burnout can be a major problem in long-term debilitating conditions like CKD. A Cochrane analysis also suggested that music therapy can have a beneficial impact on depressive symptoms (Maratos et al., 2008). Importantly, patient acceptance of this therapy was high, with dropout rates being low in all five studies included in the analysis.
Future directions could also include further exploration of inflammation in CKD. Data suggest that the reduction in cytokine activation associated with inflammatory conditions alone, without the use of antidepressant medications, can result in amelioration of depressive symptoms (Tyring et al., 2006). Additionally, novel brain stimulation treatments, such as repetitive transcranial magnetic stimulation (Slotema et al., 2010) and transcranial direct current stimulation (tDCS) (Berlim et al., 2013; Loo et al., 2010) also show promise in the treatment of depression. Such techniques may be of particular use in patients in whom pharmacological treatment of depression is a particular challenge.
Conclusions
Depression is a very common but complex problem amongst those individuals with CKD. Recent research reports provide strong evidence that around one in five patients with ESKD will be suffering from depression. Given that comorbid depression has been associated with higher rates of mortality, increased hospitalisation, reduced treatment adherence and lower QOL, the identification and appropriate treatment of depression in those with CKD is extremely important and needs to be given greater priority. However, the overlap of symptomatology makes diagnosing depression in the setting of CKD challenging, and the evidence base for effective treatments is lacking. Given the size and importance of this issue, there is now an urgent need for well-conducted randomised trials for interventions focused on the treatment of depression in CKD in order to provide information on their safety and efficacy. However, despite the lack of available data to date, a range of pharmacological and non-pharmacological treatment modalities are available and should be considered in this population.
Footnotes
Acknowledgements
Funding
Dr Bautovich was funded by a grant from the New South Wales Institute of Psychiatry. The research was also assisted by a grant from the Royal Australian and New Zealand College of Psychiatrists.
Declaration of interest
All authors have completed the Unified Competing Interest form at ![]() and declare that: (1) AB received support from the New South Wales Institute of Psychiatry and the Royal Australian and New Zealand College of Psychiatrists for the submitted work; (2) CL has received financial support from the Stanley Medical Research Foundation to fund research into tDCS treatment in depression and honoraria from Astra-Zeneca for speaking at an ECT conference; IK has received payments as a consultant for National Renal Care and has received grants from, and sits on, the Chronic Kidney Disease Advisory Panel for Amgen; and (3) CL has had tDCS equipment donated from Soterix as part of other ongoing research; IK is a member of the Kidney Health Australia (KHA) Advisory Committee and Chronic Kidney Disease Surveillance Group.
and declare that: (1) AB received support from the New South Wales Institute of Psychiatry and the Royal Australian and New Zealand College of Psychiatrists for the submitted work; (2) CL has received financial support from the Stanley Medical Research Foundation to fund research into tDCS treatment in depression and honoraria from Astra-Zeneca for speaking at an ECT conference; IK has received payments as a consultant for National Renal Care and has received grants from, and sits on, the Chronic Kidney Disease Advisory Panel for Amgen; and (3) CL has had tDCS equipment donated from Soterix as part of other ongoing research; IK is a member of the Kidney Health Australia (KHA) Advisory Committee and Chronic Kidney Disease Surveillance Group.
