Abstract
Keywords
The prevalence of smoking has been reported to be as high as 65–90% in patients with schizophrenia [1–4], which is significantly higher than the 35–47% reported in the general population [5,6]. Previous studies have suggested that vulnerability to schizophrenia may be associated with increased proclivity to smoking initiation [7,8]. Schizophrenia patients are less likely to stop smoking than the general population [9], partly due to withdrawal symptoms including anger/irritability, poor concentration, impatience, and restlessness [1,10]. The genetic predisposition to smoking and the lower cessation rates could result in the high proportion of smoking in schizophrenia patients.
A meta-analysis found that schizophrenia patients have a 20% reduced life expectancy, with smoking being one of the major risk factors for early mortality [11]. Another study found that schizophrenia patients died on average 10 years earlier than the general population, and that smoking was an important cause of the increased mortality [12,13]. Therefore, understanding the patterns of smoking and its sociodemographic and clinical correlates in schizophrenia patients is vitally important for policymakers and health-care providers in the implementation of appropriate strategies and execution of effective measures to reduce its harmful consequences.
Numerous surveys on smoking in schizophrenia patients have been conducted in Western countries over recent decades. The correlates of smoking have included male sex, early onset of the illness, more hospitalizations, more frequent substance use, severity of psychopathology, cognitive deficits, high doses of antipsychotic drugs, antipsychotic drug-induced side-effects, and poor responses to treatment [12,14–20]. Rates of tobacco smoking in schizophrenia patients have ranged from 14% to 88%, with smoking being the most common form of substance abuse in this patient population [21,22]. Because smoking is influenced by a host of biological, psychological, and environmental factors [23,24], findings regarding Caucasian patients may not be generalizable to other ethnic groups.
In recent years, several studies on the frequency of smoking and its correlates in schizophrenia have been conducted in China, with conflicting results. The proportion of smoking varied between 23% [25] and 54% [26]. Sun and Zou surveyed male schizophrenia patients in a psychiatric hospital and found that 54% of the patients smoked [26]. Smokers had a lower quality of life. Only men, however, were involved in that study and assessment of smoking was not described. Chen et al. interviewed a convenience sample of 90 inpatients with first-episode schizophrenia [23]. The authors defined smokers as ‘a person who smokes a cigarette daily at least in five days per week’, and reported that the smoking rate was 30% in their sample. Smokers had more severe psychotic symptoms and impaired cognitive functioning than non-smokers. The methodological shortcomings of the Chen et al. study included the lack of a timeframe for smoking and the number of cigarettes smoked. Tang et al. reported a smoking rate of 23% for all inpatients with schizophrenia in a psychiatric hospital (male, 40.1%; female, 2.2%); smokers were more likely to be divorced, had lower education, a more episodic course, a great number of relapses, and were more likely to receive clozapine [25]. The items of the questionnaire to measure smoking developed by the authors, however, were not published. The common major limitations of these studies were that they recruited only schizophrenia inpatients and were restricted to one site. Given that more than 90% of schizophrenia patients in China live in the community, and that there are significant sociocultural and economic differences and varying clinical traditions across the country [27], the findings of these studies may not be applicable to the majority of Chinese schizophrenia patients.
This study set out to investigate the proportion of cigarette smoking and its sociodemographic and clinical characteristics in Chinese schizophrenia patients. The study involved 19 mental health centres nationwide that represented a range of clinical settings in which schizophrenia patients receive treatment in China.
Methods
Study participants and settings
This study was part of a nationwide project that explored the optimal length of risperidone maintenance treatment [Wang CY et al., unpublished data, 2010]. The recruitment period started on 1 December 2002 simultaneously at each site by screening schizophrenia patients who were in remission from an acute episode of their illness, and lasted until 31 January 2004.
Patients were recruited if they (i) were inpatients or outpatients of either sex diagnosed with DSM-IV schizophrenia; (ii) were clinically stable for ≥ 4 but < 8 weeks following an acute episode, with ‘clinical stability’ defined as < 36 points on the Brief Psychiatric Rating Scale (BPRS); (iii) were between 18 and 65 years of age; (iv) were on risperidone monotherapy titrated to optimal level in the acute episode; (v) were local residents, living with at least one family member; (vi) had satisfactory treatment adherence defined by a pill count that yielded >80% adherence to risperidone prescription over the past 4 weeks; and (vii) understood the aims of the study and signed the consent form. Patients were excluded if they (i) were taking antidepressants, mood stabilizers, and Chinese herbal remedies concomitantly with risperidone, or had received electroconvulsive therapy, or were participating in any other drug trials or interventional studies over the 4 weeks before study entry; (ii) had a history of, or ongoing major medical or neurological condition(s); (iii) had past or current drug/alcohol abuse other than nicotine; and (iv) were pregnant or had plans to become pregnant, were lactating, or were not practising an effective method of birth control.
During the recruitment period, all patients who met the entry criteria were randomly assigned to one of three groups with a different duration of risperidone therapeutic dose. The study continued until the last recruited patient completed his/her 12 month follow up, meaning that all patients were followed up for 12–26 months.
The study protocol was approved by the clinical research ethics committees of the respective study centres. Each participant signed a consent form.
Outcome measures and assessment
Patient sociodemographic data were collected by the raters using a questionnaire designed for the study. Current cigarette smoking (in the following referred to as ‘smoking’) was confirmed if patients answered affirmatively to the question ‘Have you ever smoked daily in the past month’. This definition of smoking has also been used in other recent studies [6,28]. The average number of cigarettes smoked was also recorded. Smoking cigarettes is the most common way of consuming tobacco worldwide [21] and in China [23]; hence, only cigarette smoking was investigated. A current alcohol user (in the following referred to as ‘alcohol consumption’) was defined as a person who drank at least one alcoholic beverage per month during the past year [29,30]. Patient responses on smoking and on alcohol use were confirmed by their family members.
BPRS [31] was used as a screening tool at entry. Psychopathology was evaluated on the Chinese version of the Positive and Negative Syndrome Scale (PANSS) [32], using the positive, negative, anxiety, and depressive symptom scores and the hostility–excitement and disorganized thoughts scores of the PANSS [33]. The Treatment Emergent Symptoms Scale [34] was used to assess the safety of the treatment. Outcome measures were assessed monthly during the first 6 months, and then every 2 months until the last enrolled patient completed the study. Before the study, all raters were trained in the use of the instruments and in judging relapse on 20 symptomatic schizophrenia patients, for which the interrater reliability yielded a satisfactory–good agreement.
Statistical analysis
The data were analysed using SPSS 13.0 for Windows (SPSS, Chicago, IL, USA). The comparison between smokers and non-smokers with respect to sociodemographic and clinical characteristics was analysed on independent sample t-test, Mann–Whitney U test, and χ2 test as appropriate. Multiple logistic regression analysis involving the ‘Enter’ method was used to adjust for relevant covariates and to determine the independent relationship of each variable with smoking. Smoking was the dependent variable while the independent variables included age, sex, education level, marital and employment status, age at onset, duration of illness, number of admissions, severity of psychopathology, baseline risperidone dose, medication adherence, side-effects at baseline, relapse, drinking and mean change in psychopathology and side-effects from entry to the end of follow up (mean change = scores at the end–scores at entry). The one-sample Kolmogorov–Smirnov test was used to check the normality of distribution of the continuous variables. The level of significance was set at 0.05 (two-tailed).
Results
Altogether 512 patients with schizophrenia were screened, of whom 108 patients did not meet the study criteria, leaving 404 patients who participated in the study. By the end of the study 30 patients were excluded for not following prescription rules, thus the statistical analyses were run on the 374 patients who completed the study.
Because the smoking status of the participants did not change during the study period, only the rate of smoking at baseline is given here. Smokers accounted for 13.9% of the whole sample (n = 52); 26.2% of male (n = 45) and 3.5% of female patients (n = 7). Among the smokers, the mean number of cigarettes consumed per day was 13.2 ± 8.2, while 44.2% of them smoked ≤ 10 cigarettes daily, 40.4% smoked 11–20 cigarettes, and 15.4% smoked >20 cigarettes.
Table 1 lists the sociodemographic and clinical characteristics of the whole sample and the comparison between smokers and non-smokers with regard to sociodemographic and clinical data. Male sex, unemployment, alcohol consumption, older age, older age at onset, longer duration of illness, more frequent admissions, more severe initial hostility–excitement score and less deterioration in the hostility–excitement scores over the study period were significantly associated with smoking.
Subject characteristics
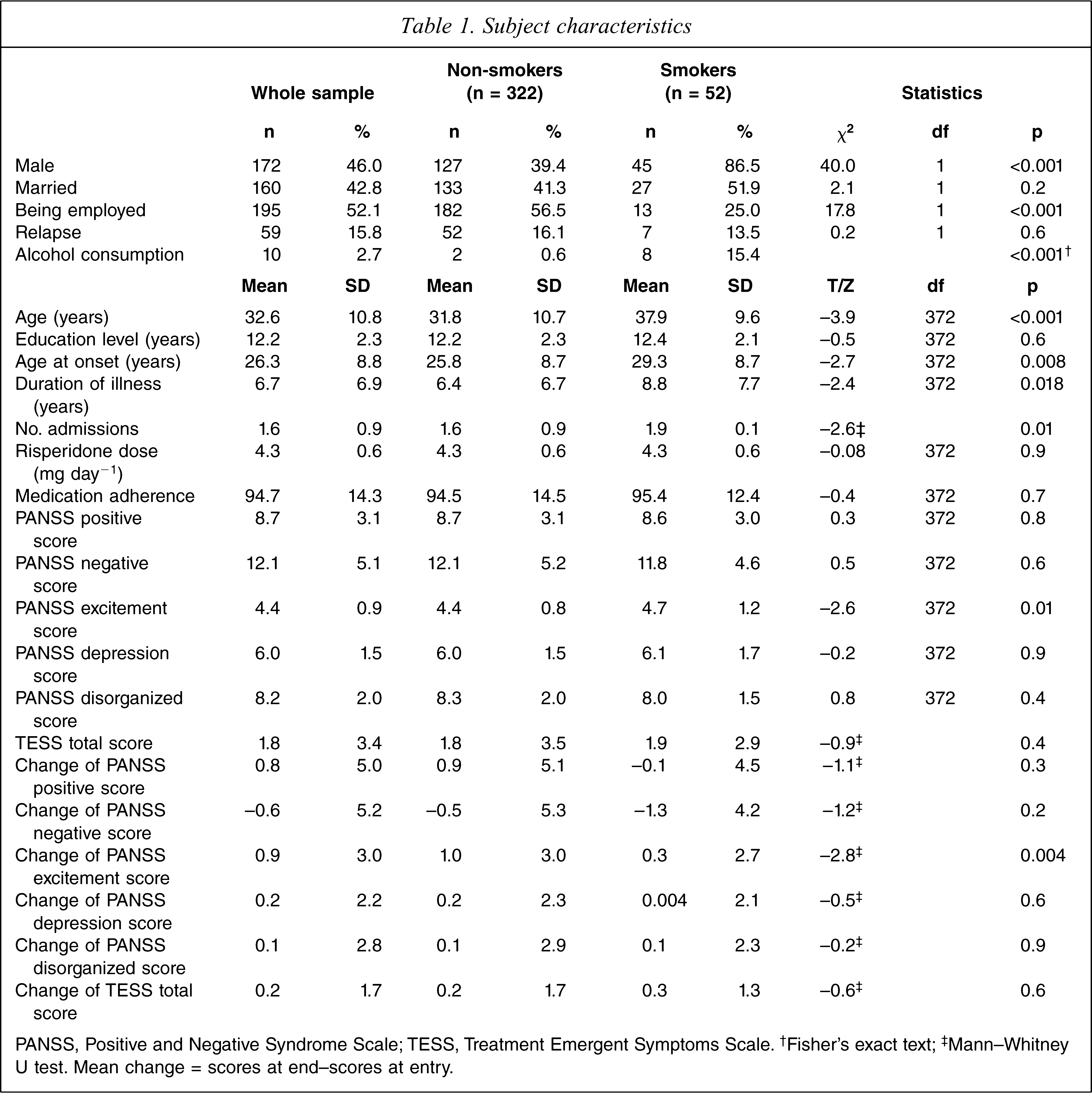
PANSS, Positive and Negative Syndrome Scale; TESS, Treatment Emergent Symptoms Scale. †Fisher's exact text; ‡Mann–Whitney U test. Mean change = scores at end–scores at entry.
On multiple logistic regression analysis, male sex, unemployment, alcohol consumption, more frequent admissions, less severe positive and negative symptoms at study entry, less deterioration in negative symptoms, and a worsening trend in disorganized thoughts at study end were independently associated with smoking (Table 2).
Multiple regression for correlates of smoking, with non-smoking as reference
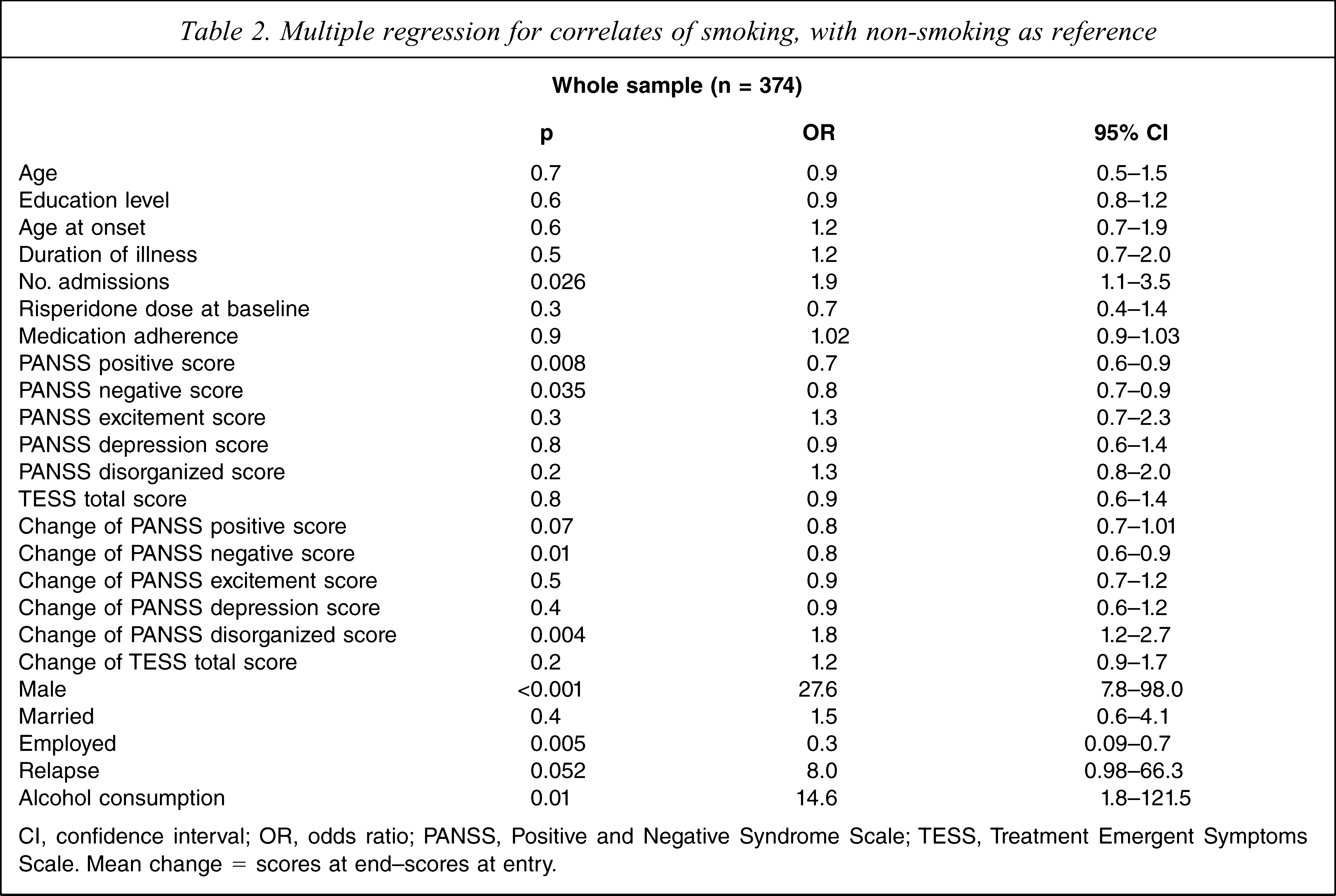
CI, confidence interval; OR, odds ratio; PANSS, Positive and Negative Syndrome Scale; TESS, Treatment Emergent Symptoms Scale. Mean change = scores at end–scores at entry.
Spearman correlation analysis showed that smoking was not associated with the severity of positive (r = 0.2, p = 0.3), negative (r = −0.03, p = 0.3) or depressive symptoms (r = −0.1, p = 0.6), hostility–excitement (r = −0.1, p = 0.6), disorganized thoughts (r = 0.02, p = 0.9) and severity of adverse events (r = 0.04, p = 0.8) at entry, or with the change of positive (r = −0.2, p = 0.2), negative (r = 0.1, p = 0.6) and depressive symptoms (r = 0.1, p = 0.4), hostility–excitement (r = 0.1, p = 0.4), disorganized thoughts (r = 0.003, p = 0.98) and extrapyramical side effects (EPS) (r = 0.05, p = 0.7) over the study period.
Discussion
To the best of our knowledge, this was the first prospective, nationwide study to determine the association of smoking with sociodemographic and clinical characteristics in Chinese schizophrenia patients.
Over the past two decades, two national surveys on smoking were carried out in China: in 1996 and 2002. In these surveys current smoking was confirmed if participants answered affirmatively to the question ‘Have you ever smoked daily in the past month’ [5,35]. The rate of smoking was 35.3% in general, and 63.0% in men and 3.8% in women in 1996; the corresponding figures were 31.4%, 57.4% and 2.6%, respectively, in 2002 [5,35]. The rate of smoking cessation increased from 9.4% in 1996 to 11.5% in 2002, probably due to the effects of public education on the prevention and cession of smoking, increase in the price of cigarettes and wide advertising of the harmful effects of smoking. In 2002 the smoking rates in men were highest in Yunnan, Qinghai, Guangdong and Jiangxi provinces and the Inner Mongolia Autonomous Region (68.8–80.2%); more women smoked in Heilongjiang, Jinlin and Hebei provinces, the Mongolia Autonomous Region and Tianjin Municipality (4.7–17.3%). The mean cost of smoking per smoker was USD 0.34 day-1 in 2002 [5]. Previous studies indicated that the smoking rate was lower among younger Chinese; for example, the regular smoking rate in male secondary school students in Zhouzhou city was of 7.6% [36]. The current smoking rate was 17.5% in the 15–24 year age group in Beijing [28], which could be attributed to the strict prohibitive measures and intensive anti-smoking education on smoking in primary and secondary schools and colleges in China. In recent years, only one controlled study explored the measures on smoking cessation involving a convenience sample of 69 Chinese schizophrenia patients [37]. Patients were randomly assigned to two groups, with 36 receiving bupropion 150–300 mg day−1, and 33 receiving placebo. At the 8 week follow up, 28% and 6% in the bupropion and placebo groups, respectively, had stopped smoking [37]. The assessment instruments were described in sufficient detail only in two of the aforementioned studies [5,28].
The frequency of smoking ranged from 4.2% to 40% across different centres in the present study. The pooled rate (13.9%) for the whole sample was at the low end of the range reported for schizophrenia patients in a meta-analysis surveying 20 countries (14–88%) [21]. It was also considerably lower than the earlier reported figures in both the Chinese general population (31.4% in 1996 and 35.3% in 2002) [5,38] and schizophrenia patients (23–31%) [23,25,39]. The discrepancy in the smoking rates between the present and previous studies could be due to the following reasons. First, differences in public health policies and regulations. The 19 mental health centres in this study were all in urban areas of China, and prohibition on smoking in public places was implemented in these areas, which could lower the smoking rate to some extent. Second, different sampling methods, such as consecutive versus convenience sampling, were used. Third, potential selection bias: in the present study patients who had past or current drug or alcohol abuse, and who lived independently were excluded, while they might be more likely to be at higher risk of smoking. Fourth, differences in the definition of smoking may also contribute to the diverse findings. Finally, given the fact that many smokers were fully aware of the harmful effects of smoking, it may well be that they denied smoking, knowing that this was the ‘right’ answer (i.e. what the investigators wanted to hear). In China the contents and quality of anti-smoking campaigns vary across different areas and periods, so their influence on the patients involved in the present study is uncertain.
The smoking rates reported in Chinese schizophrenia populations are consistently lower than the pooled worldwide rate of 62% [21]; the sociocultural and/or biological reasons for this need to be further explored.
It is reported that smoking is essentially self-medication for schizophrenia patients because nicotine could reduce negative symptoms and ameliorate EPS and cognitive deficits by increasing dopaminergic and glutamatergic neurotransmission in the prefrontal cortex [40–42]. Nicotine products are potent inducers of the cytochrome P450 1A2 enzyme (CYP1A2), which is involved in the metabolism of several antipsychotic medications [43]. The induction of CYP1A2 lowers the serum concentrations of antipsychotic drugs in schizophrenia patients who smoke [1]. Based on this we hypothesized that smoking would have an impact on the severity of psychopathology, EPS, or even short-term outcomes. The results partly supported this hypothesis because smoking was significantly associated with more admissions, less severe positive and negative symptoms at entry, a smaller decline in negative symptoms, and more deterioration in disorganized thoughts over the study period, but had no relationship with relapse rate at the end of the study. More frequent admissions are an indirect measure of illness severity, which was associated with smoking, thus replicating an earlier finding [44]. In contrast, the association between psychopathology and smoking was not in line with data from the literature. Some studies found that smoking was associated with more severe psychotic symptoms [23,45], while others reported no such association [25,46,47]. The inconsistent results concerning smoking and psychopathology could be attributable to the differences in patient characteristics [48] and pharmacotherapy [25].
The predominance of smoking among men in the present study is consistent with earlier reports for both the general population [28,49,50] and schizophrenia patients [25,48]. Strict prohibitions on smoking in the workplace may account for the lower rates of smoking among patients who were employed [28]. In agreement with the results of earlier studies [28,51], smoking was more prevalent in patients with alcohol consumption in this sample.
The results should be interpreted with caution because of certain methodological shortcomings. First, more smoking variables, such as ‘former smoker’ and ‘average age at onset of smoking’, should have been examined. Second, the definition of smoking used in the present study (‘ever smoking in the past month’) is relatively generous, and may not have captured minor changes in smoking rates. Third, as a spin-off of a nationwide risperidone trial on relapse prevention, the study criteria were restrictive. This was a convenience sample that could not represent the whole schizophrenia patient population in China. Further, the number of patents excluded in relation to individual study criteria was not recorded, and this aspect of the study could not be analysed. Previous studies in China described smoking only in schizophrenia inpatients in a cross-sectional design, and were restricted to only one site. This is the first study that surveyed smoking in Chinese schizophrenia inpatients and outpatients with a prospective, multicentre design, thus the findings could approximate the real situation regarding smoking. Finally, although it is well established that the use of illicit drugs is very common in Western psychiatric patients [9], no information on the use of illicit substances was collected in the present study. Except for alcohol, however, other, mostly illicit substances are rarely used in schizophrenia patients in China [52].
In conclusion, smoking is prevalent in Chinese schizophrenia patients, but significantly lower than the pooled worldwide figures. Smoking had no relationship with sociodemographic characteristics except for sex and employment status; illness-related factors appeared to be more important correlates of smoking in this patient population.
Given the harmful consequence of smoking, the Chinese government is considering implementing further preventive measures such as increasing the price of and taxes on cigarettes, restricting their availability and intensifying education on the harmful effects of smoking for the public and for psychiatric patients [5]. Chinese psychiatrists rarely ask patients about smoking, probably due to the inadequate emphasis on smoking in the medical curriculum, hence the importance of smoking prevention in clinical practice should be given more emphasis in the undergraduate and postgraduate curriculum.
Previous studies (e.g. de Leon and Diaz [21]) found that major anti-smoking campaigns in the general population hardly affected severely ill psychiatric patients. Therefore, specific interventions targeting this patient population should be developed.
Footnotes
Acknowledgements
The present study was supported by grants from the 10th National Five-Year Plan Foundation of the Ministry of Science and Technology Program, People's Republic of China (No. 2004BA720A22), the Research Foundation of Xian-Janssen Pharmaceutical (No. RIS-CN-MCCT-0201), the National Natural Science Foundation of China (No. 30800367), and the Beijing Nova Program of the Beijing Municipal Science and Technology Commission (No. 2008B59). Xian-Janssen Pharmaceutical donated drug supplies and offered advice on the dosing strategies, but was otherwise not involved in the design, execution, data analysis, interpretation, or writing of the study. No investigator benefited from participating in the study.
