Abstract
Most patients with schizophrenia smoke cigarettes. A recent meta-analysis reported that smoking and schizophrenia are associated [1]. Patients with schizophrenia have considerable difficulty in giving up and suffer physical and social harm as a result of smoking [2–4]. For example, the prevalence of smoking in patients with schizophrenia is 88% compared with 33% in the general American community [4]. In addition, smokers with schizophrenia smoke more than patients diagnosed with other psychiatric disorders [5]. There are higher rates of smoking in male adolescents prior to the development of their first psychotic episode, compared with unaffected controls. This suggests that comorbid smoking may be related to the underlying disease process of schizophrenia [6].
Attention has been drawn to the neglect by psychiatrists of the physical health of their patients with serious mental illness, in particular, ischaemic heart disease [7]. The combination of both physical and psychiatric disability adds considerably to the disease burden experienced by those with schizophrenia [8, 9].
Smoking is the most common comorbid substancedependence condition in schizophrenia [10, 11] and this association is independent of substance abuse, treatment with antipsychotics and treatment setting [12]. This increased rate of smoking may be related to common aetiological factors, self-medication of symptoms (both positive and negative), medication adverse effects, mood symptoms and cognitive effects [5, 13].
Smoking is associated with significantly less Parkinsonism in patients with schizophrenia treated with typical neuroleptics [14]. A protective relationship between smoking and tardive dyskinesia has been reported [15] and smoking has also been hypothesized to protect against idiopathic Parkinson's disease [16]. Acute smoking of cigarettes results in a transient decrease in negative symptoms, but it is unclear whether this effect is due to nicotine, other components in cigarettes or the act of smoking [17]. The severity of nicotine dependence is also correlated with improvements in negative symptoms [18]. Collectively, these findings may have discouraged psychiatrists to assist patients to stop smoking.
Nicotine may have a role in improving cognitive impairments associated with schizophrenia. Nicotine normalizes deficits in auditory sensory gating found in many patients with schizophrenia and in their firstdegree relatives [19]. Smoking also improves the processing of auditory stimuli and lessens negative symptoms by increasing dopamine in the nucleus accumbens and the prefrontal and frontal cortex [20]. Harris et al. found that the effects of nicotine on cognition in schizophrenia were predominantly confined to attention [21].
Studies have suggested that clozapine, and perhaps other atypical neuroleptics, may decrease smoking in patients with schizophrenia, compared with typical antipsychotics [13, 20, 22, 23]. With the adoption of policies banning smoking in public places, patients who continue to smoke are likely to be further socially disadvantaged. Hospital policies are also changing and in-patient psychiatric units are now expected to provide smoke-free environments. The draft tobacco policy document of the Royal Australian and New Zealand College of Psychiatrists and the Royal Australasian College of Physicians details the high rates of smoking in schizophrenia and the importance of addressing this serious problem with appropriate access to treatment [24].
As smoking has been reported to produce possible beneficial effects in the areas of negative symptoms, cognitive impairment and extrapyramidal adverse effects, patients with varying degrees of nicotine usage were compared with non-smokers on these key clinical parameters. The nicotine consumption of patients treated with different classes of antipsychotic drugs was also compared.
Method
Subjects
One hundred and forty-six unrelated Caucasian patients attending two psychiatric units for the treatment of their schizophrenia were recruited for the study. All patients met Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) criteria for schizophrenia. They underwent a clinical history taking by either a psychiatrist (S.B., B.L., M.B.) or a clinical psychologist (R.Y.). Demographic details including ethnic background data were obtained. All subjects had received the same antipsychotic medication for at least 4 weeks at a stable dose. Patients on regular antidepressant, opiates, anxiolytic or mood stabilizing medication were excluded from the study. All patients were physically well and none had a history of epilepsy. The average age of those recruited was 36.26 years (SD = 12.0) and 125 (86%) were male and 21 (14%) were female. Sixty-two were inpatients and 84 were outpatients. The breakdown of medication was as follows: 49 patients were prescribed risperidone, 33 were taking olanzapine. Thirty-three patients were prescribed typical antipsychotics with 12 patients prescribed flupenthixol, two fluphenazine decanoate, 13 zuclopenthixol decanoate, three haloperidol decanoate, one thioridazine, one thiothixene, one trifluoperazine. Thirty-one patients were prescribed Clozapine. The dose was converted to chlorpromazine equivalents per kilogram (cpzek) [25]. The mean dose of risperidone was 4.8 (SD = 2.0) chlorpromazine equivalents per kilo (cpzek). The mean dose of olanzapine was 5.3 (SD = 2.6) cpzek, the mean dose of clozapine was 5.1 (SD = 2.9) cpzek and the mean dose of typicals was 5.6 (SD = 3.7) cpzek. There were no significant differences in dose between the different drug groups. Adherence in inpatients was sound as all subjects were administered medication by nursing staff. Outpatient adherence was estimated by self-report and assessment by the treating psychiatrist. Although it has limitations, self-report in patients with schizophrenia is accepted as the most cost-effective and time-efficient way of obtaining an indication of adherence [26]. Thirty of the 33 patients receiving typical medication were treated with nurse-administered depot preparations.
Procedure
Patients were recruited from the Fortitude Valley Community Mental Health Centre, the Royal Brisbane Mental Health Unit and the Park Psychiatric Hospital. Inclusion criteria included being aged between 18 and 65 years and having a DSM-IV diagnosis of Schizophrenia. All subjects possessed an adequate comprehension of English. Potential participants were excluded if they had any other psychiatric disorder including schizoaffective disorder, bipolar disorder, dementia, organic brain syndrome and major depressive disorder with delusions.
A questionnaire was administered to determine the number of cigarettes smoked. Subjects were divided into three groups: non-smokers, those smoking less than 20 cigarettes day −1 (light smokers) and those smoking 20 or more cigarettes day −1 (heavy smokers). There were 30 non-smokers, 30 light smokers and 86 heavy smokers. Expired carbon monoxide was measured to validate self-report. There is good correspondence between self-report and biochemical measures of smoking in those with psychosis [27].
The Positive and Negative Symptom Scale (PANSS) [28], the General Health Questionnaire-28 (GHQ-28) [29], the Reitans Trail Making A and B [30], the Barnes Akathisia Rating Scale [31] and the Extrapyramidal Rating Scale [32] were administered. The interrater reliability of the PANSS was monitored throughout the data collection period through random and independent administration by two raters.
Results
The primary analyses undertaken were analysis of variance with post-hoc comparisons undertaken when significant effects or trends were evident. Data were analysed using SPSS software version 11.2. There was no significant difference in chlorpromazine equivalence between the medication groups (F (3,146) = 1.798, NS). Analysis of variance showed a significant difference in expired carbon monoxide (CO) between the heavy smoking group (Mean = 22.65 p.p.m., SD = 12.81), the light smoking group (Mean = 13.00 p.p.m., SD = 10.40) and the non-smoking group (Mean = 1.55 p.p.m., SD = 1.64) (F (2,145) = 39.64, p < 0.000001). This validates patient self-reported cigarette consumption. Post-hoc comparisons (Tukey HSD) showed the difference between non-smokers and the heavy smoking group to be highly significant (p < 0.001). The difference in expired CO between non-smokers and light smokers was also significant (p < 0.001) as was the difference between the light smoking and heavy smoking groups (p < 0.001).
Analysis of variance examining differences in consumption of nicotine in mg/day revealed a significant effect (F (3,128) = 6.25, p < 0.0.001) across the treatment groups. As a conservative test an analysis of covariance between typical and atypical medication groups was also conducted with baseline CPZEK as the covariate. This showed increased significance over the analysis of variance (F (2,128) = 9.41, p < 0.001). Post-hoc examination (Tukey HSD) revealed that there was a significant difference with patients treated with typical antipsychotic medications smoking more heavily than those treated with olanzapine (p < 0.013) and risperidone (p < 0.000). There was a trend for patients treated with clozapine to smoke less than those treated with typicals (p < 0.081). There was no significant difference in nicotine consumption between patients treated with atypical medications (olanzapine, risperidone and clozapine). Patients treated with typical medications smoked a mean of 430.92 mg day − 1 (SD = 394.52) while those on clozapine smoked 266.58 mg day −1 (SD = 183.30), on olanzapine smoked 218.84 mg day − 1 (SD = 235.32) and on risperidone smoked 158.50 mg day − 1 (SD = 198.10). There was no difference between typical and atypical antipsychotic medication groups in age, gender, inpatient or outpatient status, ethnicity, age of onset of schizophrenia, number of inpatient admissions, first degree family history of schizophrenia, past suicide attempts and non-compliance with medication. Furthermore, there was no difference between the two groups in symptom severity as measured by the PANSS total score. The means and standard deviations of all variables by medication group are available from the authors by request. Table 1 illustrates the number of patients within the three smoking groups treated according to medication.
Smoking status according to medication group
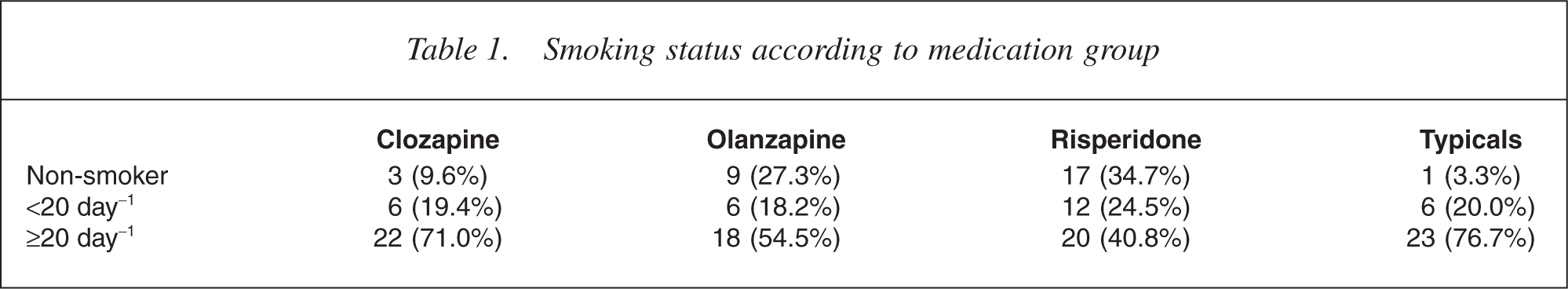
The differential distribution of smokers across the four medication groups was highly significant (χ(6) 2 17.08, p < 0.009). Further chisquare analysis revealed that patients prescribed typical medications were overrepresented in the heavy smoking group when compared with the non-smoking group (p < 0.004, Fishers exact test).
When patients on atypical medication were examined and those on typical medications excluded, there were no significant differences between the light, heavy and non-smoking groups in the PANSSnegative (F(2,110) = 0.399, NS), PANSS-positive (F(2,111) = 0.758, NS) and total scores (F 2,111 = 0.362, NS). Similarly, there were no significant differences in the extrapyramidal symptom rating scale (F(2,111) = 0.859, NS) and the Trail Making A (F(2,108) = 1.41, NS) and Trail Making Btests (F(2,98) = 0.561, NS). Furthermore, there were no significant differences in GHQ-28 scores between the groups (F(2,109) = 0.529, NS). However, analysis of variance revealed a significant difference between groups with regard to the Barnes Akathisia Clinical global impression Scale (F(2,110) = 6.18, p = 0.03). Post-hoc testing (Tukey HSD) revealed a significant increase in akathisia severity in the non-smoking compared with the 20 or more day − 1 group (p = 0.046).
Discussion
Treatment with typical medication was associated with a more than twofold greater consumption of cigarettes compared with treatment with atypical medication (risperidone, olanzapine and clozapine). There was no difference in cigarette consumption between patients prescribed risperidone, olanzapine and clozapine. Atypical medications are therefore associated with lower levels of smoking with no particular antipsychotic demonstrating an advantage. Previously clozapine [22, 23] has been reported to be superior compared with other atypicals; however, this was not reflected in our data.
The current study found no association between high levels of smoking and diminished negative symptoms. Although this observation is limited by the crosssectional nature of the study it does not support the notion that smoking improves negative symptoms in schizophrenia. Furthermore, cognitive impairment was not improved in either smoking group when compared with non-smokers. Although the Trail-making Test does not measure all domains of cognition, it is correlated with visual information processing, resistance to associative distracters and executive dysfunction [33, 34]. However, there was a trend towards significance when akathisia was compared in the non-smoking and heavy smoking groups treated with atypical agents at comparable doses.
Patients with schizophrenia may smoke in order to improve symptoms [14, 17, 18, 35]. However, we report that smoking is not generally associated with amelioration of symptoms as measured by the GHQ and the PANSS. The GHQ is a well-accepted measure of general psychiatric symptomatology in schizophrenia [35]. Our data support an association of smoking with reduced akathisia in patients prescribed atypical agents. This association has been previously found in patients treated with typical medications [14] and implementation of a smoking ban in a locked psychiatric unit has also been reported to significantly increase agitated behaviour [36]. This could be a result of akathisia or other factors such as nicotine withdrawal. Nicotine patches significantly reduce neuroleptic induced akathisia [37]. This may be due to either the direct action of nicotine on enhancing nigrostriatial dopamine release, or the effects of nicotine on antipsychotic serum levels. Heavy smokers have reduced serum antipsychotic levels [38, 39], and cessation of smoking can result in these levels increasing. Akathisia may emerge in patients treated with atypical agents who quit smoking. A combination of nicotine replacement therapy and a possible reduction of antipsychotic dose could avert this problem.
Patients with schizophrenia are more prone to nicotine addiction as increased nicotine reward is associated with this disorder [40]. This enhanced reward may be related to abnormal expression and function of the neuronal nicotinic receptor gene family in schizophrenia [41]. Additionally, the personal and social disadvantage associated with schizophrenia further increase the vulnerability of this clinical group [42].
Nicotine induces the release of dopamine in the nucleus accumbens [43]. Drug reward is mediated by binding to postsynaptic dopamine receptors (DRD2) and activation of a reward cascade [44]. Atypical medications stimulate the release of dopamine via 5HT-1 A agonism and 5HT-2 A receptor antagonism [45]. Stimulation of dopamine release may result in greatly reduced postsynaptic DRD2 upregulation produced by the DRD2 antagonistic properties of these agents. Animal studies reveal that the upregulation of DRD2 produced by haloperidol alone is attenuated by coadministration of ritanserin, a 5HT-2 A receptor antagonist [46]. Conversely, typical medications which antagonize DRD2 in the absence of serotonergic effects result in markedly upregulated DRD2 [47]. Therefore, typical agents may amplify the postsynaptic DRD2 reward cascade via upregulation of these receptors. Patients taking typical medications may consequently be primed to experience greater reward from cigarette smoking. The differential effects of atypical and typical agents on dopamine may explain the differences in smoking found between these treatment groups.
These results should encourage clinicians to assist patients with schizophrenia in ceasing smoking. Other studies have found that no-smoking policies in psychiatric hospitals do not negatively affect the patient's mental state, even if withdrawal symptoms are precipitated [2, 48]. Withdrawal symptoms, however, should be alleviated by nicotine replacement therapy as case reports suggest that nicotine withdrawal may lead to an exacerbation of psychotic symptoms [49]. Smoking cessation can be achieved in patients with schizophrenia by using the usual methods to help people stop smoking, albeit with more difficulty [13, 49, 50]. For example, motivational interviewing techniques are of assistance in helping these patients cease smoking [51] and more generally in the treatment of other comorbid addictions [52].
Limitations of the current study include its crosssectional design. Further prospective studies are indicated to examine the effects of antipsychotic medication on smoking behaviour over time in both those with schizophrenia and smokers resistant to other interventions. In addition, future research could examine the impact of nicotine withdrawal on psychotic symptoms and to determine which smoking cessation interventions are more useful in patients with schizophrenia.
Our study provides strong support for the consideration of atypical agents as part of a strategy to assist with cigarette cessation in patients with schizophrenia. Furthermore, nicotine replacement therapy is indicated for treatment of nicotine withdrawal symptoms and the prevention of possible emergent akathisia upon smoking cessation. With the use of nicotine replacement therapy, clinicians can provide therapeutic interventions for smoking cessation in schizophrenia without fear of worsening symptoms and disability. Ultimately, this may lead to a reduction in the number of patients with schizophrenia who continue to die from this ‘habit’.
Footnotes
Acknowledgements
This research was supported in part by a grant from Janssen-Cilag Pharmaceuticals Pty Ltd. and AstraZeneca Pharmaceuticals Pty Ltd. The authors express their gratitude for the assistance given by patients and staff in the services involved. The authors declare that they have no financial interest.
