Abstract
Keywords
Pervasive developmental disorder (PDD) consists of a group of childhood-onset developmental difficulties that include autism, Asperger's syndrome, PDD not otherwise specified (PDDNOS), Rett's disorder and childhood disintegrative disorder. PDD is a spectrum of disorders encompassing children with a wide range of abilities. There is thus gradation in the level of presentation. Children with Asperger's syndrome would not have significant communication difficulties and children with PDDNOS may not present with the full picture of the classical triad. Children and adolescents with autism will present with a triad of difficulties consisting of impairments in social interaction, impairments in communication and a restricted repetitive and stereotyped pattern of behaviours, interests and activities. Difficulties in social interaction manifest as impairments in non-verbal behaviours, failure to develop age-appropriate peer relationships, a lack of spontaneous sharing of enjoyment and a lack of social and emotional reciprocity. Communication difficulties present as a delay or lack in development of spoken language without compensatory modes of communication, impairment in initiating and sustaining conversation with others when language is adequately developed, stereotyped language and a lack of spontaneous make-belief or social imitative play. Restricted repetitive and stereotyped behaviours include encompassing preoccupations or interests that are abnormal in intensity or focus, inflexible adherence to specific, nonfunctional routines or rituals, stereotyped and repetitive motor mannerisms and persistent preoccupation with parts of objects.
The most well-replicated neurochemical finding is a 25–50% increase in blood serotonin levels in subjects with autism. Studies on the dopamine neurotransmitter system, glutamate, γ-aminobutyric acid (GABA) and neuropeptides have not produced significant findings [1–3]. Serotonin dysfunction may thus form a basis for the use of some pharmacological interventions of behavioural issues in PDD. Although pharmacological agents will not cure PDD, they may be efficacious in treating dysfunctional behavioural symptoms that interfere with rehabilitative efforts and cause impairment or distress [4]. Such behavioural symptoms may include aggression, irritability, stereotyped behaviours, anxiety, hyperactivity and sleep difficulties.
It should be noted that management of mental health and behavioural difficulties in people with learning disabilities should be based on multidisciplinary and multimodal approaches. Such treatment should follow a contextual and functional analysis of behaviour and pay careful attention to the language abilities and developmental level of the child or adult that possibly underlies their medical disorders and life events. Pharmacological intervention is only one component of the therapeutic package [5]. Children and adolescents on medications for behavioural and mental health issues should thus have their condition and need for medication periodically reviewed.
Pharmacological management in children and adolescents with PDD is an area of much interest. Many studies on various pharmacological agents have been done. Much of the current research, however, consists of case reports, open-label studies or small clinical trials. Very few well-conducted randomized controlled trials of good sample sizes are available. Studies were also conducted on different types of sample groups, such as differing age groups or on individuals in different ranges of the spectrum. This scenario limits understanding, interpretation and clinical application of the findings.
This article reviews the available research on pharmacological agents used in managing behavioural symptoms associated with PDD. The aim was to provide a better understanding of the efficacy and limitations in the currently available agents. Agents included were those commonly used in clinical practice in the authors’ setting and agents of specific interest for children and adolescents with PDD. Electronic literature searches were conducted from the following sources: MEDLINE, Cochrane Library, PSYARTICLES and PsycINFO. Search terms included, but were not limited to, ‘autism’, ‘PDD’, ‘autism spectrum disorder’ (‘ASD’), and ‘pharmacological management’. Searches were limited to English-language materials from 1975 to 2008 inclusive. Article abstracts obtained from the search strategy were perused and eligible articles were then retrieved as full text. Articles were then classified according to the Ministry of Health (Singapore) levels of evidence (Table 1). Studies included in this review used various terminologies, making standardization of terminology difficult. Hence, the ‘terms pervasive developmental disorder’ (PDD) and ‘autism spectrum disorder’ (ASD) are used in accordance with the terminologies used by the individual authors.
Grades of recommendation

Tricyclic antidepressants
Tricyclic antidepressants block presynaptic re-uptake of norepinephrine and serotonin to varying degrees. Although the tricyclic antidepressants were the drugs of choice for management of depression in the 1980s, their non-selective action on cholinergic, histaminergic, and presynaptic adrenergic receptors resulted in adverse effects, thus resulting in reduced use of these drugs in recent years.
Clomipramine
Clomipramine is a tricyclic antidepressant that has serotonin re-uptake inhibitor properties. In a double-blind, comparison study of 12 autistic subjects aged 6–18 years, Gordon et al. found that clomipramine was superior to desipramine and placebo in improving autistic symptoms, anger, compulsive and ritualized behaviours, with no differences between desipramine and placebo [6]. Clomipramine was equal to desipramine and both drugs superior to placebo in reducing hyperactivity.
Remington et al. conducted a double-blind, placebo-controlled cross-over trial comparing clomipramine (mean dose 128.4 mg), haloperidol (mean dose 1.3 mg) and placebo in 36 subjects (age 10–36 years) with autistic disorder [7]. They found that clomipramine was comparable to haloperidol in terms of improvement from baseline based on before–after measurements on the Childhood Autism Rating Scale and the Irritability subscale of the Aberrant Behaviour Checklist (ABC). Significantly fewer, however, receiving clomipramine (37.5%) versus haloperidol (69.7%) were able to complete the trial due to side-effects and behaviour problems (lethargy, tremors, tachycardia, insomnia, diaphoresis, nausea and decreased appetite).
In an open-label pilot study conducted by Sanchez et al., eight young autistic children (aged 3.5–8.7 years) were rated using the Children's Psychiatric Rating Scale, Clinical Global Impressions (CGI), Conner's Parent Teacher Questionnaire and the Clinical Global Consensus Ratings [8]. Clomipramine was found to be not therapeutic and was associated with serious, untoward effects, such as acute urinary retention, severe constipation, insomnia, behavioural problems and drowsiness. Young children seemed more prone to experience untoward effects.
While clomipramine may reduce irritability and stereotypical behaviour in autism, it may be associated with more frequent side-effects.
Selective serotonin re-uptake inhibitors
Selective serotonin re-uptake inhibitors (SSRIs) increase the extracellular level of the neurotransmitter serotonin by inhibiting its re-uptake into the presynaptic cell, increasing the level of serotonin available to bind to the postsynaptic receptor. Serotonin dysfunction has been postulated to be one of the underlying causes of autism. Persons with autism display repetitive thoughts and behaviours that are very similar to the presentation of obsessive– compulsive disorders. It is thus that SSRIs are considered as a pharmacological intervention for autism.
Fluvoxamine
McDougle et al. conducted a 12 week, double-blind, placebo-controlled trial of fluvoxamine in 30 adults with autistic disorder [9]. Eight (53%) of the 15 subjects who received fluvoxamine were rated as responders compared to 0 of the 15 placebo-treated subjects. Improvements were seen in repetitive thoughts and behaviour, maladaptive behaviours, repetitive language and aggression. Adverse effects included nausea and sedation, which were transient and of minor severity.
McDougle et al. conducted another 12 week, double-blind, placebo-controlled trial of fluvoxamine with 34 children and adolescents (5–18 years) with PDD [10]. The study found the medication to be poorly tolerated and ‘with limited efficacy at best’. Only one of the fluvoxamine-treated children demonstrated significant clinical improvement with the drug. Fourteen of the children demonstrated adverse effects, including insomnia, hyperactivity, agitation, aggression, increased rituals, anxiety, anorexia, increased appetite, irritability, decreased concentration and impulsivity.
In a 10 week, prospective open-label trial with fluvoxamine in 18 children and adolescents (7–18 years) with PDD, Martin et al. found that there was no response to fluvoxamine as a group [11]. Current research findings suggest that fluvoxamine is more efficacious in adults with autism and seems to be poorly tolerated with limited efficacy in children.
Fluoxetine
In 2005 Hollander et al. conducted an 8 week placebo-controlled cross-over trial of liquid fluoxetine in 45 child and adolescent patients with ASD [12]. Liquid fluoxetine in low doses was found to be more effective than placebo in the treatment of repetitive behaviours, and did not differ from placebo on treatment-emergent side-effects. In a retrospective chart review, Fatemi et al. observed the effects of fluoxetine on seven adolescents and young adults with autistic disorder [13]. There was a reduction in the subscales of irritability, lethargy, stereotypy and inappropriate speech as measured on the ABC. Side-effects reported included transient appetite suppression, vivid dreams and increase in hyperactivity.
DeLong et al. followed up 129 children (aged 2–8 years) with ASD on fluoxetine between 5 and 76 months [14]. They reported that 22 (17%) had an excellent response, 67 (52%) demonstrated good response, 10 (8%) had fair response and 30 (23%) had a poor response. In an open trial, Cook et al. examined the effect of fluoxetine treatment on children and adults (aged 7–28 years) with autism and mental retardation [15]. Fifteen (65%) of the 23 autistic subjects and 10 (63%) of the 16 mentally retarded subjects had improvement in overall clinical severity. Side-effects consisted primarily of restlessness, hyperactivity, agitation, decreased appetite or insomnia. Fluoxetine may be considered as an option in management of ASD.
Citalopram
Namerow et al. studied the effects of citalopram in a retrospective review of 15 children and adolescents (aged 6–16 years) with PDD [16]. They reported that 11 (73%) exhibited significant improvement in PDD symptoms, or anxiety and mood scores on the Clinical Global Improvement Scale. A total of 66% of patients improved significantly on anxiety symptoms associated with PDD while 47% improved on mood symptoms. Mild side-effects were reported by five patients.
Escitalopram
Owley et al. reported a 10 week, open-label, prospective study of escitalopram on 28 subjects with PDD [17]. A total of 17 (61%) were rated as responders based on the irritability subscale on the ABC. Dose-related side-effects included hyperactivity and aggression.
Sertraline
Three open-label trials have been conducted on the effects of sertraline on PDD. McDougle et al. conducted a 12 week, prospective, open-label study in 42 adults with PDD [18]. Twenty-four (57%) of the subjects were treatment responders with improvements primarily in the aggressive and repetitive behaviour symptoms. Sertraline was generally well-tolerated with no adverse cardiovascular effects, extra-pyramidal symptoms or seizures identified.
Hellings et al. conducted an open-label trial of sertraline in nine adults with mental retardation and autistic disorder presenting with target symptoms of self-injury and/ or aggression [19]. Eight subjects (89%) demonstrated significant reduction in aggression and self-injurious behaviour. In another open-label study of nine children (aged 6–12 years), Steingard et al. administered sertraline to children with autism and transition-associated anxiety and agitation [20]. Eight of the nine children (89%) demonstrated ‘some degree of response to sertraline treatment’.
Typical antipsychotics
The therapeutic actions of typical antipsychotics lie in their antagonism of dopamine receptors, particularly the D2 receptors. These antipsychotics produce extra-pyramidal symptoms, such as parkinsonism, dystonia, and tardive dyskinesia. In addition, typical antipsychotics also increase serum prolactin concentration.
Haloperidol
Campbell et al. conducted a double-blind, placebo-controlled, cross-over trial of haloperidol in 33 children (aged 2.3–7.9 years) with autism and mental retardation [21]. They reported a significant reduction in maladaptive behaviours and that it facilitated learning. Above the optimal range of dosage, however, increased irritability and dystonic reactions emerged. Campbell et al. also conducted a 15 year, prospective, longitudinal evaluation of haloperidol in 118 children (aged 2.3–8.2 years) with autism [22]. A significant proportion of the children developed dyskinesias.
Anderson et al. demonstrated that haloperidol decreased maladaptive behaviours such as temper tantrums, hyper-activity and stereotypies in 45 autistic children aged 2.02–7.58 years (double-blind, placebo-controlled) [23]. With careful regulation of dosage, none of the children had adverse effects at therapeutic dosages.
Haloperidol reduces maladaptive behaviours in children with PDD. At therapeutic doses with careful monitoring, side-effects may be minimized. Above therapeutic doses, the probability of developing extrapyramidal side-effects increases. There is a risk of developing tardive dyskinesia with the use of haloperidol.
Atypical antipsychotics
The therapeutic action of atypical antipsychotics is characterized by a broader spectrum of pharmacological properties because they work on serotonin receptors as well as dopamine receptors. Thus, the atypical antipsychotics have a reduced propensity to produce extrapyramidal symptoms and may have less prolactin elevation. Atypical antipsychotic medications, however, have recently been associated with potential adverse metabolic effects, such as weight gain, insulin resistance, dyslipidemia and hyperglycemia [24]. Hence, taking of a good medical history, and regular weight, blood pressure, fasting blood glucose and lipid profile monitoring are recommended.
Risperidone
Risperidone is an antagonist of both dopamine (D2) and serotonin (5HT2 and others) receptors. Because conventional antipsychotics have been shown to improve behaviour in children with autism, and because serotonin dysfunction has been postulated to be a possible underlying mechanism of autism, interest has developed in investigating the efficacy of risperidone in ASD. Risperidone is currently the only medication that the US Food and Drug Administration (US FDA) has approved for the treatment of irritability and concomitant aggressive behaviours in autism.
In 2002 McCracken et al. at the Research Units on Pediatric Psychopharmacology (RUPP) Autism Network conducted a multisite, randomized, double-blind trial of risperidone compared with placebo for the treatment of autistic disorder accompanied by severe tantrums, aggression, or self-injurious behaviour in 101 children aged 5–17 years old [25]. The rate of positive response, as defined by >25% decrease in irritability score and a rating of much improved or very much improved on the Clinical Global Improvement Scale, was 69% in the risperidone group and 12% in the placebo group. They concluded that risperidone was effective and well-tolerated for the treatment of tantrums, aggression or self-injurious behaviour in children with autistic disorder.
In the same cohort of children, McDougle et al. investigated whether risperidone improved the core symptoms of autism [26]. They reported that risperidone led to significant improvements in restricted, repetitive and stereotyped patterns of behaviour, interests, and activities in autistic children but did not significantly change their deficit in social interaction and communication.
To investigate longer-term benefits of risperidone in the group of children who showed a positive effect in the RUPP [27] trial, the children were followed up in a two-part study. Part 1 consisted of a 4 month open-label treatment with risperidone. Part 2 was an 8 week randomized, double-blind, placebo-substitution study of risperidone withdrawal. The authors concluded that risperidone showed persistent efficacy and good tolerability for intermediate-length treatment of children with autism characterized by tantrums, aggression, and/or self-injurious behaviour. Discontinuation after 6 months was associated with rapid return of disruptive and aggressive behaviour in most subjects.
A second multi-centre, placebo-controlled study of risperidone in 79 children (aged 5–12 years) with PDD was conducted by Shea et al. [28]. Risperidone was associated with 64% reduction in ABC Irritability subscale scores as compared to 31% in placebo. More risperidone-treated subjects (87%) had global improvement in their condition compared to the placebo group (40%). Adverse events included somnolence, increases in weight, pulse rate and systemic blood pressure. Side-effects were manageable.
Olanzapine
Two case series and two randomized control trials have been done with olanzapine. Potenza et al. reported a case series of response to olanzapine in seven children, adolescents and adults with PDD [29]. Six of the seven subjects were responders, with significant improvement in overall symptoms of autism, motor restlessness, social relatedness, affectual reactions, sensory responses, language usage, self-injurious behaviour, aggression, irritability or anger, anxiety and depressive symptoms. Adverse events were increased appetite and weight gain.
Kemner et al. conducted an open-label trial of olanzapine in 25 children with PDD (aged 6–16 years) [30]. In that study olanzapine was found to be effective in only three subjects (12%) in terms of improvement on CGI ratings. This low response may have been related to the young age of the children and the level of cognitive functioning. There were positive changes found on the sub-scales Hyperactivity, Excessive Speech and Irritability of the ABC. In a parallel group design comparing olanzapine and haloperidol in 12 children with autism, Malone et al. reported that five of six subjects in the olanzapine group and three of six in the haloperidol group were rated as responders [31]. Weight gain was significantly higher in the olanzapine group.
A small double-blind placebo-controlled study of olanzapine in the treatment of 11 children and adolescents with PDD was conducted by Hollander et al. in 2005 [32]. A total of 50% on olanzapine versus 20% on placebo were responders. Olanzapine was associated with significant weight gain.
Current research suggests that olanzapine leads to improvement in motor restlessness, self-injurious behaviour, aggression and irritability. The risk of significant weight gain is a concern. More randomized controlled trials are indicated.
Quetiapine
Two reports on open-label trials and two case series on quetiapine have been published. Findings have been inconsistent. Martin et al. conducted an open-label study involving six children and adolescents (aged 6–15 years) with autism and mental retardation [33]. Overall, there was no significant improvement. Two of the six subjects were reported to be responders. Adverse effects reported included sedation, possible seizures, increased appetite and weight gain. The investigators concluded that quetiapine was poorly tolerated and generally ineffective.
Findling et al. reported a 12 week open-label trial of quetiapine in nine autistic adolescents (aged 10–17 years) [34]. Two of the nine were judged as responders. Adverse events reported were sedation, weight gain, agitation and aggression. They concluded that quetiapine may not be a particularly effective agent in the treatment of adolescent patients with autistic disorder.
A retrospective case series reported by Corson et al. found that eight of the 20 subjects (aged 5–28 years) were responders to quetiapine [35]. Adverse events were reported in 50% of the subjects and led to drug discontinuation. The authors concluded that quetiapine was modestly effective for maladaptive behaviour in patients with a PDD. In a second case series, Hardan et al. reported clinically significant improvement in symptoms of hyper-activity and inattention in six of 10 patients treated with quetiapine [36]. Adverse events were mild.
Ziprasidone
McDougle et al. published a report of an open-label trial of ziprasidone in 12 autistic subjects (aged 8–20 years) [37]. Six of the 12 were judged as treatment responders with improvements in symptoms of aggression, agitation and irritability. Adverse effects were minimal. They concluded that ziprasidone appeared to have potential for improving symptoms of aggression, agitation and irritability in autism. Cohen et al., in their retrospective chart review of 10 adults, noted that seven of the 10 patients had an improvement or no change in their maladaptive behaviour [38]. There were no significant adverse events associated with ziprasidone.
The US FDA has raised concerns about the potential for QTc interval prolongation with ziprasidone on electrocardiogram [39], so the drug should not be given to individuals with cardiac disease or those who are taking other medications that can prolong QTc interval without careful monitoring.
Aripiprazole
Stigler et al. conducted a prospective, open-label case series of five children with autism treated with aripiprazole [40]. All five children were reported to be responders with clinically significant improvements in aggression, self-injurious behaviour and irritability. Stigler et al. further reported a 14 week prospective, open-label study of aripiprazole in 25 children and adolescents with PDD [41]. A total of 22 of the 25 children were considered responders, with improvements in aggression, self-injurious behaviour and severe tantrums.
In a retrospective chart review, Valicenti-McDermott and Demb treated 32 children and adolescents with developmental disabilities with aripiprazole [42]. Aripiprazole was effective in 56% of the subjects and in 37% of the autistic subjects. Side-effects were reported in 50% of the subjects.
Clozapine
Clozapine blocks 5-HT2A, 5-HT2C, 5-HT3 and DA D1-D4 receptors. There is a paucity of reports on the use of clozapine in persons with autism, with only three case reports to date. Zuddas et al. described three children with hyperactivity, fidgetiness and aggression treated with clozapine [43]. Two of the three showed sustained improvement. Chen et al. reported on a 17-year-old boy with autism and severe mental retardation who was treated with clozapine [44]. He showed marked reduction of signs of ‘overt tension’, hyperactivity and repetitive movements. Gobbi and Pulvirenti described long-term treatment with clozapine in an adult with autistic disorder and aggression [45]. Clinical improvement was evident in aggressiveness and social responsiveness. Although clozapine could possibly be useful in the management of ASD, it is rarely considered because it has the potential to cause life-threatening agranulocytosis and requires regular blood count monitoring.
Stimulants
Stimulant medication has been found to be an effective treatment strategy in attention-deficit–hyperactivity disorder (ADHD). Methylphenidate is a potent stimulant derived from amphetamine, and it exerts its effect by enhancing dopaminergic transmission in the brain.
Conventional diagnostic guidelines (such as the DSM IV-TR and the ICD-10) exclude the diagnosis of ADHD in PDD. Symptoms of hyperactivity and inattention, however, are frequently found in children with PDD, and practical management strategies useful in ADHD, including pharmacological intervention, may be considered.
Methylphenidate
Quintana et al. conducted a double-blind cross-over study of methylphenidate in 10 autistic children, aged 7–11 years [46]. They reported improvement in irritability and hyperactivity. No significant side-effects, including worsening of stereotypic movements, were observed. In another double-blind, placebo-controlled cross-over trial, Handen et al. observed 13 autistic children with symptoms of ADHD (aged 5.6–11.2 years) on methylphenidate [47]. Eight subjects responded positively, based on a minimum 50% decrease on the Conner's Hyperactivity Index. Significant adverse effects observed included social withdrawal and irritability.
In a multisite, double-blind, placebo-controlled, crossover trial conducted by the RUPP Autism Network, 72 children with PDD (aged 5–14 years) were given a 1 week test-dose phase of low, medium and high doses of methylphenidate, followed by a randomized, double-blind, cross-over phase [48]. Methylphenidate was found to be superior to placebo on improvement of the hyper-activity subscale of the ABC, with 49% of subjects classified as methylphenidate responders. Adverse effects (primarily irritability) led to discontinuation of the study medication in 18% of the patients. The authors concluded that methylphenidate was often efficacious in treating hyperactivity associated with PDD, but the magnitude of response was less than that seen in typically developing children with ADHD. Adverse effects were more frequent.
Selective norepinephrine re-uptake inhibitors
Atomoxetine
Atomoxetine is non-stimulant drug used as a second-line treatment in the treatment of children with ADHD. Jou et al. conducted a retrospective assessment of atomoxetine in 20 children and adolescents with PDD [49]. Improvements were observed in the Conners Parent Rating Scale subscales of conduct, hyperactivity, inattention and learning.
Posey et al. described an open-label study of the use of atomoxetine for ADHD symptoms associated with high-functioning PDD [50]. Twelve of the 16 participants (75%), were rated as ‘much’ or ‘very much improved’ on the CGI–Improvement scale. The most significant improvements were in the area of ADHD symptoms.
In another open-label trial, Troost et al. examined the effects of atomoxetine on ADHD symptoms and autistic features in 12 children with PDD [51]. They found a reduction of 21% for changes in the subscale Hyperactivity of the ABC. Gastrointestinal symptoms, irritability, sleep problems and fatigue were the most frequent side-effects.
In a small placebo-controlled cross-over trial with 16 children and adolescents with ASD, Arnold et al. reported that atomoxetine was superior to placebo on the primary outcomes of the Hyperactivity subscale of the ABC [52], but it did not reach statistical significance on inattentive symptoms. Adverse events were tolerable. They concluded that the effects appeared as large as with methylphenidate with fewer side-effects.
Atomoxetine may be a promising agent in the treatment of ADHD symptoms in children with ASD.
Anti-convulsants
Anti-convulsants are usually indicated for the treatment of epilepsy. They may also be used as mood stabilizers and in the management of impulsivity and aggression.
Lithium carbonate
Limited studies have been done on lithium carbonate. Kebeshian et al. described two patients with infantile autism and atypical bipolar symptomatology treated with lithium carbonate [53]. Both children demonstrated a significant response at levels >1.0 mEq L−1. Steingard et al. reported on the treatment of two patients with infantile autism and mental retardation who developed acute manic-like symptoms [54].
Oxcarbazepine
Oxcarbazepine is a structural derivative of carbamazepine. It has a reduced impact on the liver, and reduced frequency of agranulocytosis occasionally associated with carbamazepine. It generally has the same mechanism as carbamazepine and is used to treat the same conditions.
Kapetanovic reported the use of oxcarbazepine in managing disruptive behaviours in three youths with autistic disorder [55]. Improvements with oxcarbazepine included decreased aggression and improved compliance. The author suggested that oxcarbazepine has a favourable side-effect profile and may be more convenient for administration in view of an available liquid formulation.
Divalproex sodium
Divalproex sodium consists of a compound of sodium valproate and valproic acid in a 1:1 molar relationship in an enteric coated form. Hollander et al. reported an open trial of divalproex sodium in 14 children with ASD [56]. A total of 71% of the subjects were rated as having sustained response to treatment. It was generally well tolerated. The authors concluded that divalproex sodium may be beneficial to patients with ASD, particularly those with associated features of affective instability, impulsivity, and aggression as well as those with a history of electroencephalography abnormalities or seizures.
Hollander et al. then conducted an 8 week, double-blind, placebo-controlled trial of divalproex sodium versus placebo in 13 children with ASD [57]. There was a significant group difference on improvement of repetitive behaviours as measured on the Children's Yale–Brown Obsessive–Compulsive Scale. Hellings et al., however, also conducted another randomized, double-blind, placebo-controlled study of valproate in 30 subjects with PDD [58]. They found that no treatment difference was observed statistically between valproate and placebo groups in the ABC–Community scale (ABC-C) Irritability Subscale. In view of the inconsistent findings, more studies on divalproex sodium are indicated.
Opiate antagonist
Naltrexone
Naltrexone is an opiate antagonist. In the 1980s it was postulated that abnormalities of the endogenous opioid system led to maladaptive behaviours, cognitive deviances and other problems associated with autism. As such, naltrexone was considered a promising pharmacological treatment for autism. In 1989 an open-dose trial by Campbell et al. was conducted on 10 autistic children [59]. They reported promising results, with increased verbal production and reduced stereotypies, but a subsequent parallel group study by Campbell et al. suggested that although naltrexone significantly reduced hyperactivity, no effect on discrimination learning was observed [60].
Subsequent studies have failed to show any improvement in the core symptoms of autism with naltrexone. Kolmen et al. conducted a double-blind, placebo-controlled cross-over study of naltrexone in 13 young autistic children [61, 62]. They found significant improvement in impulsivity, hyperactivity and restlessness. Feldman et al. reported a randomized, double-blind, placebo-controlled, cross-over trial of naltrexone on 24 children with autism [63]. They found that the medication did not lead to improvement in communication. WillemsenSwinkels et al. conducted a double-blind, placebo crossover study of naltrexone treatment in 23 young children [64]. They reported a decrease in hyperactivity and irritability but no effects on social and stereotypic behaviour. As such, naltrexone has limited effect on reducing irritability and hyperactivity. It does not lead to improvements in the core symptoms of autism.
α2-Adrenergic agonists
α2-Adrenergic agonists have been used as anti-hypertensive agents. They have also been used in the management of ADHD.
Clonidine
Clonidine is an
Fankhauser et al. investigated the efficacy of transdermal clonidine in autism in a double-blind, placebo-controlled study involving nine autistic male subjects (aged 5–33 years) [66]. Significant improvement was seen on the CGI scale and in abnormal sensory responses, affectual reactions and social relationships. The most common adverse effects were sedation and fatigue.
Clonidine has demonstrated modest improvements in hyperactivity, aggression and irritability, but side-effects such as drowsiness may occur. There is a risk of hypertensive crises on withdrawal.
Guanfacine
Guanfacine is a newer
Scahill et al. conducted an open-label trial of guanfacine involving 25 children with PDD accompanied by hyperactivity [68]. They concluded that guanfacine may be useful in the treatment of hyperactivity in children with PDDs. Common adverse effects included irritability, sedation, sleep disturbances and constipation. Preliminary studies suggest that guanfacine may be useful in treating hyperactivity in children with PDD.
Glutamatergic antagonist
Amantadine
Amantadine hydrochloride is a non-competitive N-methyl-
Memantine
Memantine works as a glutamatergic antagonist and is used in the treatment of Alzheimer's disease. In a retrospective study of memantine in 18 children and adolescents with PDD, Erickson et al. found that there was improvement primarily in social withdrawal and inattention [70]. A total of 61% of patients were judged to be responders to memantine based on a rating of ‘much improved’ or ‘very much improved’ on the CGI scale.
Owley et al. conducted a prospective, 8 week open-label trial of memantine in improving cognitive functioning and behavioural symptoms in 14 children with PDD [71]. The study suggested that memantine improved hyperactivity, lethargy and irritability. There was significant improvement from baseline on the Children's Memory Scale Dot Learning Subtest. Chez et al. observed 151 patients with autism and PDDNOS in an open-label trial of memantine [72]. They found significant improvements in language function, social behaviour and self-stimulatory behaviour.
Memantine may be a promising agent in improving hyperactivity, inattention, social behaviour, language function, self-stimulatory behaviour and memory. Because current findings are based only on case series and open-label trials, randomized controlled trials are indicated.
Melatonin
Melatonin is an endogenous neurohormone secreted by the pineal gland that causes drowsiness. Melatonin levels increase rapidly after nightfall, peak in the middle of the night and decrease towards dawn. Melatonin has been increasingly used to manage sleep disorders in children with neurodevelopmental disorders. Wasdell et al. reported a randomized, placebo-controlled trial of melatonin treatment in delayed sleep phase syndrome and impaired sleep maintenance in 50 children with neu-rodevelopmental disabilities [73]. Overall, the therapy improved sleep of 47 children and was effective in reducing family stress.
Three open-label trials and one small randomized control trial of melatonin in the treatment of sleep disturbances in children with ASD have been reported. Paavonen et al. conducted an open clinical trial of melatonin in 15 children with Asperger's disorder [74]. The sleep patterns of all the children improved, and half of them had an excellent response to melatonin. Giannotti et al. conducted an open-label study of controlled-release melatonin in the treatment of sleep disorders in children with autism [75]. During treatment the sleep patterns of all 25 children improved. After discontinuation, 16 children returned to pre-treatment sleep patterns and read-ministration of melatonin was again effective. Treatment gains were maintained at 24 month follow up and no adverse effects were reported.
Andersen et al. conducted a large retrospective review of the use of melatonin for insomnia in 107 children with ASD [76]. After initiation of melatonin, parents of 25% of the children no longer reported sleep concerns and 60% reported improved sleep. Only thee children had mild side-effects, including morning sleepiness and increased enuresis. Garstang and Wallis conducted a small randomized controlled trial of melatonin in 11 children with ASD [77]. They reported that sleep latency, waking per night and total sleep duration improved in children receiving melatonin as compared to those receiving placebo. The use of melatonin may be considered in children with PDD with sleep disorders.
Secretin
Secretin is a gastrointestinal hormone produced in the duodenum. In 1998 Hovath first reported a case series of three children with positive responses to the administration of porcine secretin during endoscopy [78]. Interest in secretin as a possible treatment for autism resulted in several controlled studies of the effectiveness of i.v. secretin. Williams et al. conducted a Cochrane review of 13 randomized studies and found no evidence that single- or multiple-dose i.v. secretin is effective across a range of outcomes [79]. It was concluded that secretin should not be recommended or administered as a treatment for autism.
Conclusion
Pharmacological management does not address the core symptomatology of PDD. When dysfunctional behaviours or comorbid mental health disorders are present, behavioural management strategies should be considered together with the implementation of pharmacological intervention. There is a range of available pharmacological agents for the management of various dysfunctional symptoms in PDD. Broadly speaking, these agents help in the management of repetitive stereotyped behaviours, anxiety, aggression/irritability/self-injurious behaviour, hyperactivity/inattention and in sleep (see Tables 2–5 for selected studies). There is a paucity of systemic, well-conducted trials on the use of pharmacological agents in the management of PDD. More research in this area is warranted.
Pharmacological management for anxiety and repetitive behaviours
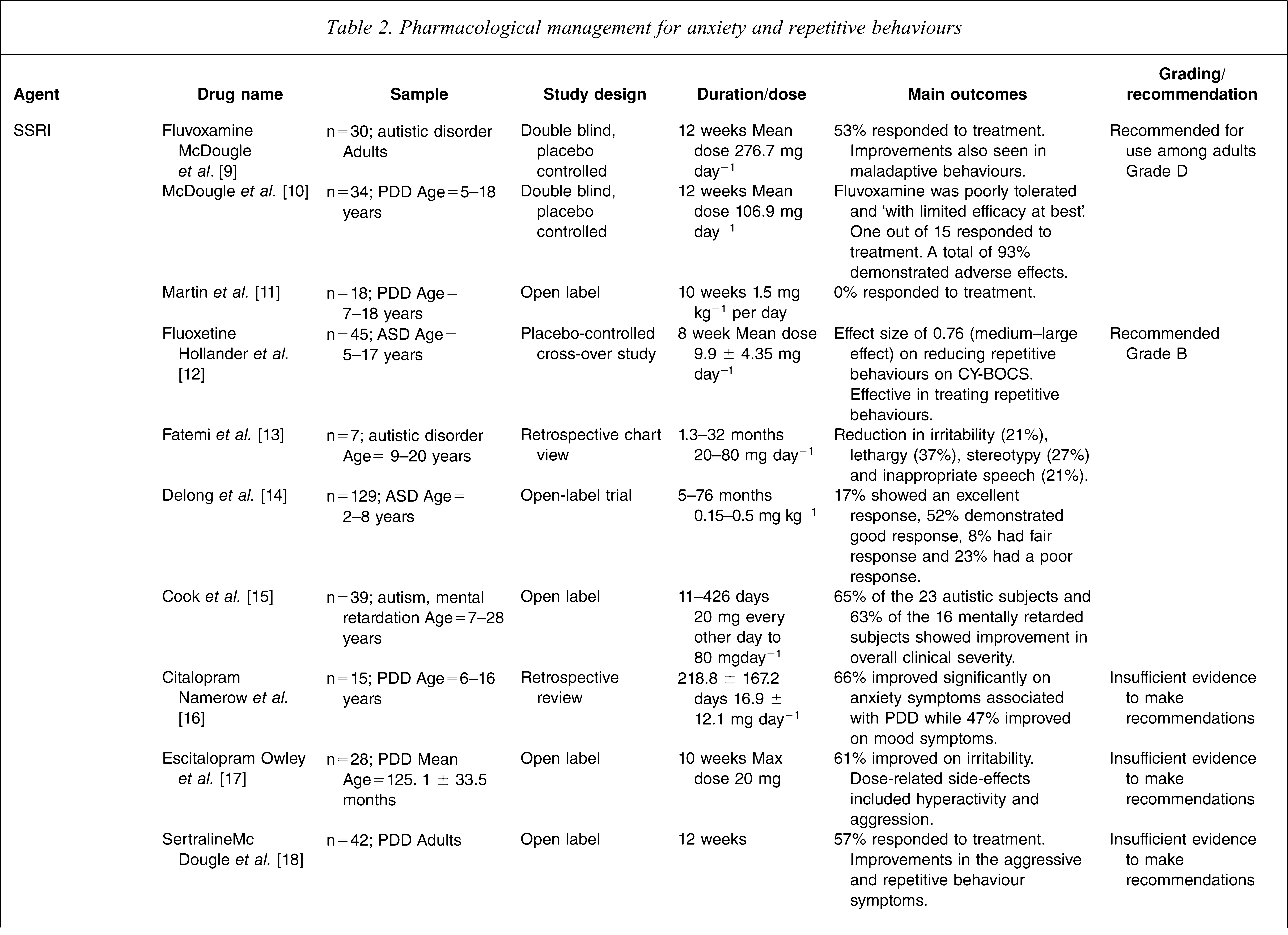

ASD, autism spectrum disorder; CY-BOCS, Children's Yale–Brown Obsessive–Compulsive Scale; PDD, pervasive developmental disorder; SSRI, selective serotonin re-uptake inhibitor.
Pharmacological management for ADHD symptoms
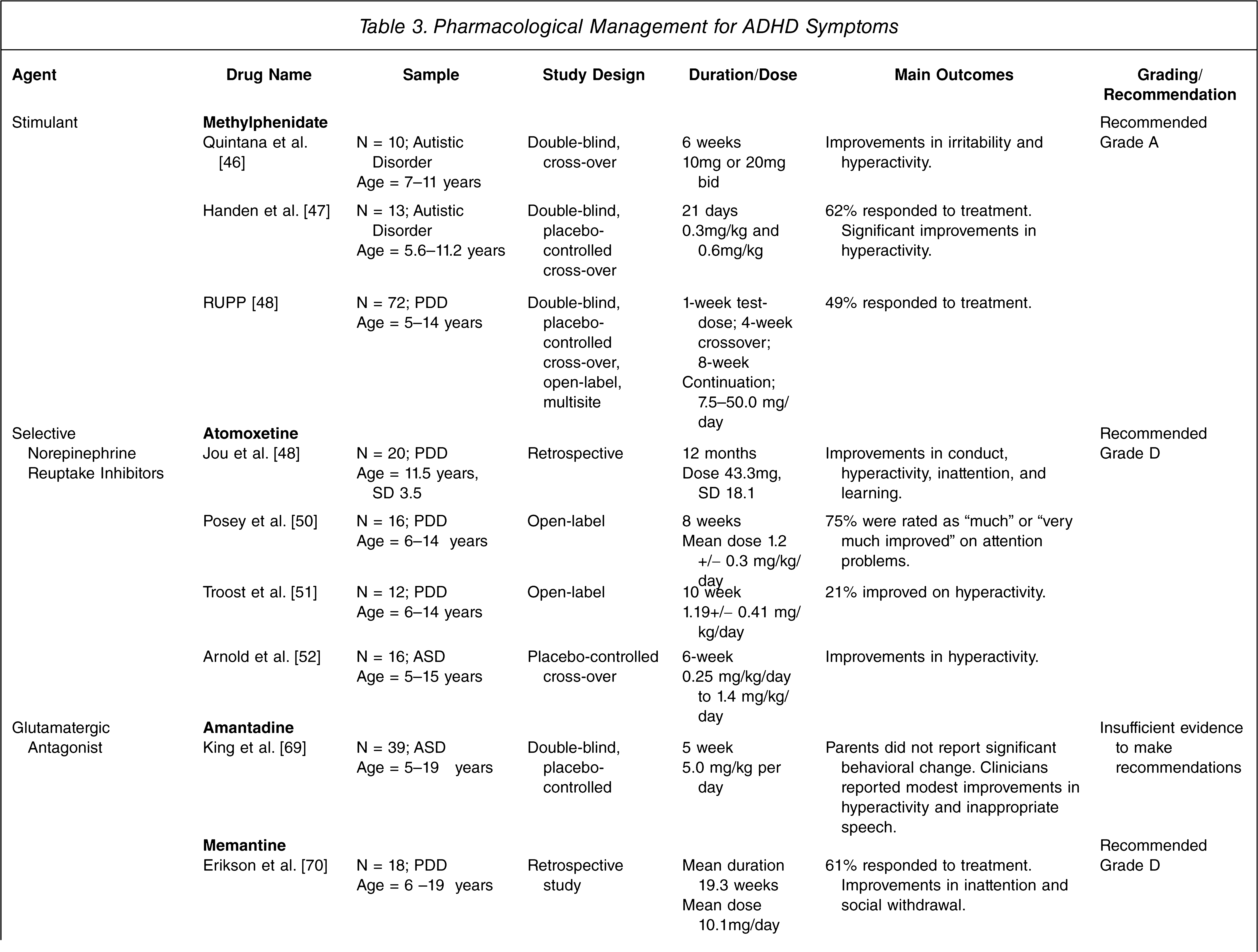

ASD = Autistic Spectrum Disorder, PDD = Pervasive Developmental Disorder.
Pharmacological management for aggression, irritability, and self-injurious behaviours
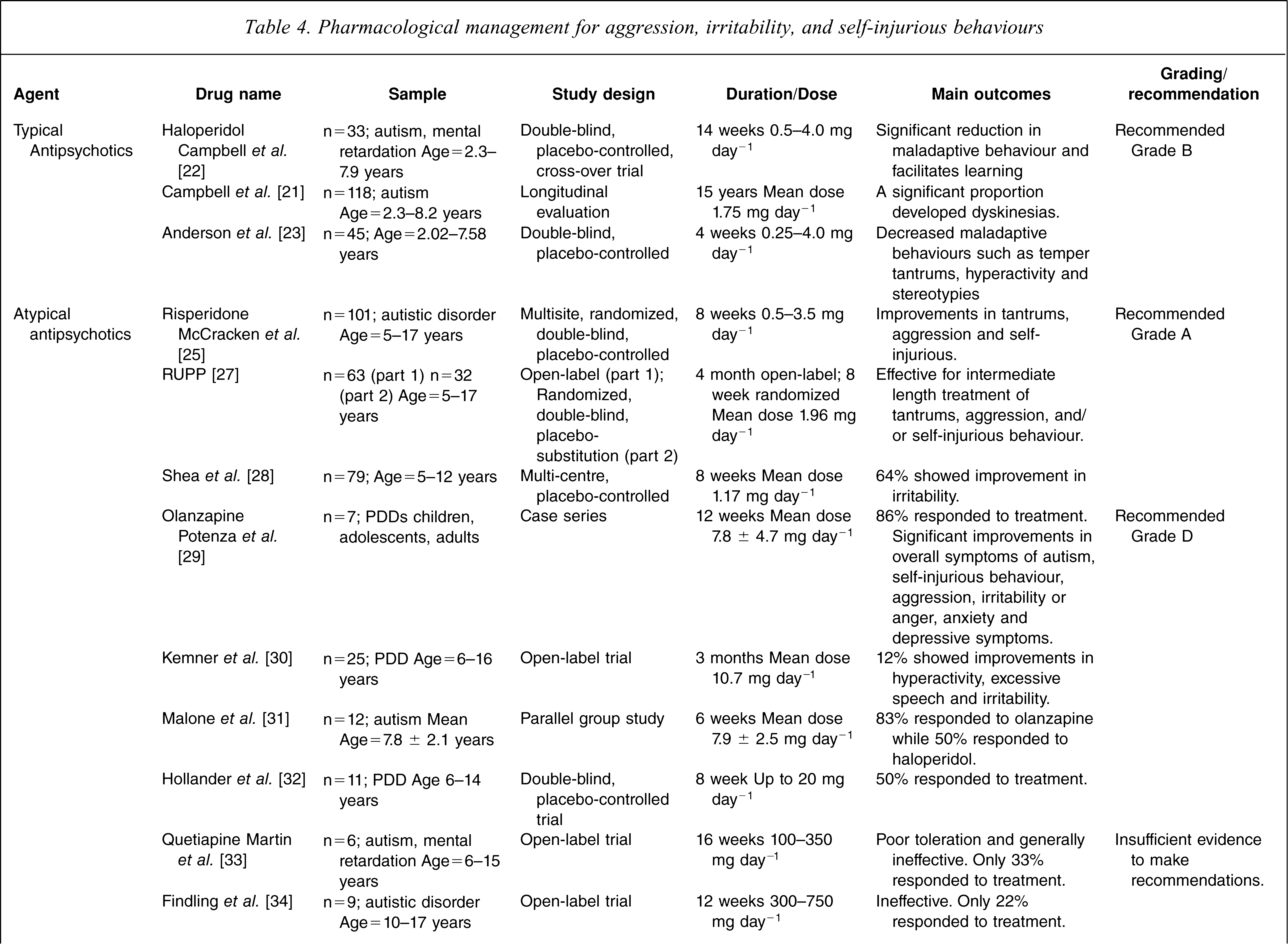
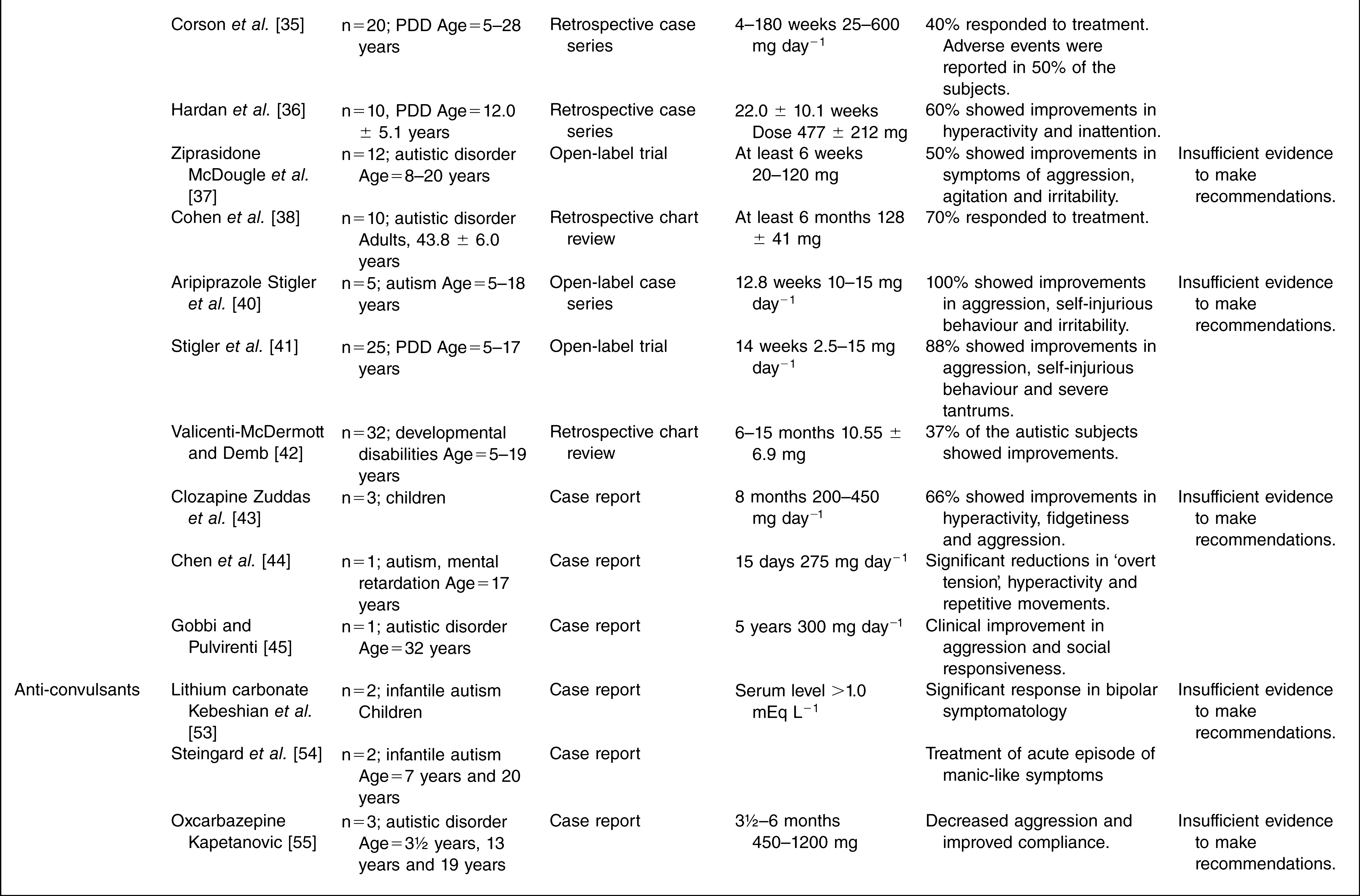
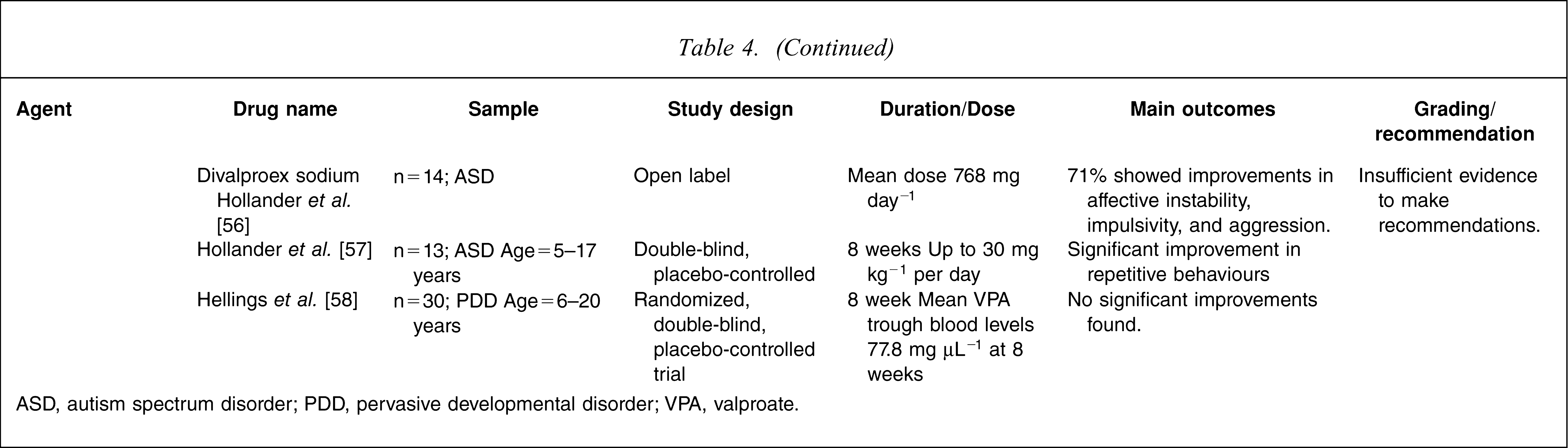
ASD, autism spectrum disorder; PDD, pervasive developmental disorder; VPA, valproate
Pharmacological management for sleep

ASD, autism spectrum disorder.
Footnotes
Acknowledgements
