Abstract
Studies have consistently reported that schizophrenia patients suffer from higher morbidity such as cardiovascular, diabetes, and respiratory diseases compared with the general population [1–4]. In particular, in addition to suicide, cardiovascular disease (CVD) is considered the most significant cause of premature death in patients with schizophrenia [5–7]. A study by Casey indicated that patients with schizophrenia were twice as likely to die from CVD as the general population [8]. Another study by Osby et al. in Sweden even found that male and female schizophrenia patients had an 8.3-fold and five-fold increase in cardiovascular mortality, respectively, compared to the general population [9]. Given the increased incidence of mortality from CVD in people with schizophrenia, efforts should be made to understand the incidence or the risk of developing CVD-related diseases in this particular population, for the purpose of developing effective prevention strategies. But although numerous studies have examined the incidence or prevalence of mortality from CVD among schizophrenia patients over the past decade, little attention has been given to investigating the risk of acute myocardial infarction (AMI) for schizophrenia patients.
Only two studies to date have explored the association between schizophrenia and AMI, with one reporting a negative association between schizophrenia and AMI incidence (incidence rate ratio = 0.77) [10], while the other failed to establish any relationship between schizophrenia and AMI [11]. Both studies did not use survival analysis techniques to correct for the excess mortality risk among schizophrenia patients and the duration of follow-up.
The aim of the present study was therefore to estimate the risk of AMI among patients receiving a first hospital diagnosis of schizophrenia for a 6 year follow-up period. We used Taiwan's National Health Insurance Research Database (NHIRD). Taiwan initiated its National Health Insurance (NHI) programme in March 1995 to finance health care for all its citizens. This nationwide, population-based dataset allows us to trace all medical service utilization history for all citizens, and it provides a unique opportunity to examine the association between schizophrenia and the subsequent risk of AMI, while adjusting for clinical and sociodemographic factors.
Methods
Database
This study used data from the NHIRD in Taiwan for the years 1995–2006. The dataset includes all inpatient and ambulatory care claims data from Taiwan's NHI programme, a single-payer payment system that provides health care for all Taiwanese citizens, offering unrestricted access to any health-care provider of the patient's choice. With >22 million enrollees in the NHI programme, the NHIRD is one of the largest and most complete nationwide population-based datasets in the world. It thus offers a unique opportunity to identify the risk of AMI occurring among schizophrenia patients.
Because the NHIRD consists of de-identified secondary data released to the public for research purposes, this study was exempt from full review by the Internal Review Board.
Study sample
The present study included a study cohort and a comparison cohort. The study cohort consisted of all patients who were hospitalized with a principal diagnosis of schizophrenia (any ICD-9-CM 295.XX code other than 295.7-schizoaffective disorder) between January and December 2000 (n = 19 999). We excluded those patients under 18 years of age (n = 179) to limit the study sample to the adult population. In addition, in order to avoid the potential confounding factors of institutionalization and chronicity, those patients who had been hospitalized or who received ambulatory care treatment for schizophrenia during the previous 5 year period were excluded from the study cohort, as were patients previously diagnosed with AMI (ICD-9-CM code 410.XX; n = 12 467). Ultimately, the study sample included 7353 schizophrenia patients.
The comparison cohort was extracted from a database released by the Taiwan National Health Research Institute (TNHRI) in 2000. This database consists of 1 000 000 random subjects, approximately 5% of all enrollees in the NHI programme. It was created by the TNHRI using a systematic sampling method of the entire database to randomly select a representative sample. After excluding subjects who had been diagnosed with both AMI and serious mental disorders (organic mental disorder, schizophrenia, affective disorder, and paranoid states, any ICD-9-CM codes 294.XX, 295.XX, 296.XX, or 297.XX), the comparison cohort was chosen by randomly selecting 22 059 enrollees (three for every schizophrenia patient) from the Registry for Beneficiaries of this representative dataset corresponding with the study group in terms of gender and age (<35, 35–44, 45–54, 65–64 and ≥65 years).
Each patient was tracked for 6 years from their index hospitalization in 2000, until the end of 2006, to identify whether or not the patient had an AMI during the follow-up period. In addition, to calculate the AMI-free survival time after index hospitalization for a 6 year period, the data were also linked to death certificate data in Taiwan, with cases censored if individuals died from non-AMI causes during that time (including 490 (7.71%) from the study cohort and 780 (3.54%) from the comparison cohort). Causes of deaths of schizophrenia patients included cancer (28%), non-AMI circulatory disorders (malignant essential hypertension, acute and subacute endocarditis and stroke; 12%), digestive disorders (20%), with unnatural causes (such as accidents or suicides) accounting for 22% of deaths, and miscellaneous natural causes combined totalling approximately 18%.
The regression modelling also adjusted for sociodemographic characteristics. The sociodemographic characteristics included gender, age, level of urbanization and the geographic location of the community in which the patient resided (Northern, Central, Eastern and Southern Taiwan), and monthly income (0, NT$1–15 840, NT15 841–25 000, ≥NT25 001) as variables. Urbanization levels, which have been recognized to have a general impact on CVD, are divided into five strata in Taiwan, with level 1 referring to the most urbanized and level 5 referring to the least urbanized communities. We selected NT$15 840 as the first income level cut-off because this is the government-determined minimum wage for full-time employees in Taiwan.
In addition, the regression modelling adjusted for substance abuse as well as comorbid medical disorders at the time of hospitalization index (hypertension, diabetes, hyperlipidaemia, and renal disease). Prior studies have consistently demonstrated that these physical comorbidities are major risk factors for AMI [12,13]. Some studies have also reported that patients with schizophrenia are more likely to have comorbidities such as hypertension, diabetes, and hyperlipidaemia than the general population [2]. In order to isolate the effects of these potential confounders on AMI, we adjusted for these medical comorbidities in the regression modelling.
Statistical analysis
The SAS statistical package (SAS System for Windows, version 8.2, SAS Institute, Cary, NC, USA) was used for statistical analysis. Pearson χ 2 tests were performed to examine the differences between the two cohorts in terms of sociodemographic characteristics and select comorbid medical disorders. The 6 year AMI-free survival rate was then estimated using the log–rank test to examine the differences in the risk of developing AMI between the two cohorts. Cox proportional hazard regressions were also carried out as a means of computing the adjusted 6 year survival rate, after adjusting for the variables mentioned here. A significance level of 0.05 was used to confirm significance.
Results
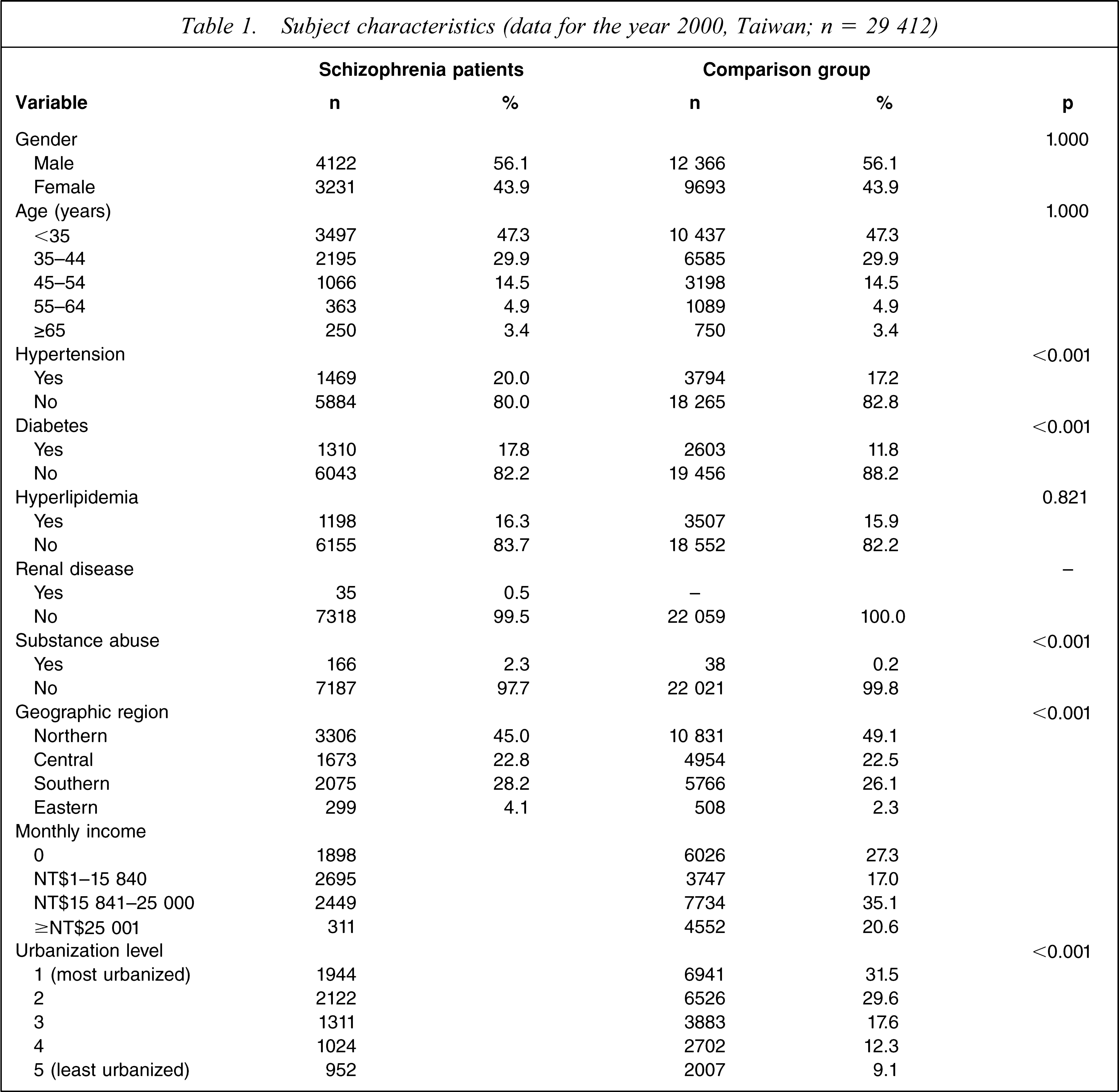
Table 1 shows the distributions of demographic characteristics and select comorbid medical disorders for these two cohorts. The majority of the sampled patients were <35 years of age (47.3%). Schizophrenia patients were more likely to have comorbidities, such as hypertension (p < 0.001) and diabetes (p < 0.001), at the time of the index hospitalization as compared to the comparison cohort. In addition, schizophrenia patients had a greater tendency to abuse substances (p < 0.001) than their counterparts in the comparison cohort. There were also significant differences between these two cohorts in terms of monthly income, geographic region and urbanization level of the community in which the patient resided (all p < 0.001).
Subject characteristics (data for the year 2000, Taiwan; n = 29 412)
Of the total sample of 29 412 patients, 130 (0.45%) experienced AMI during the 6 year follow-up period, including 30 (0.41% of the schizophrenia patients) from the study cohort and 100 (0.45%) from the comparison cohort. The log–rank test, however, showed that schizophrenia patients had significantly lower 5 year AMI-free survival rates than the patients in the comparison cohort (p < 0.001). Kaplan–Meier survival analysis is presented in Figure 1.
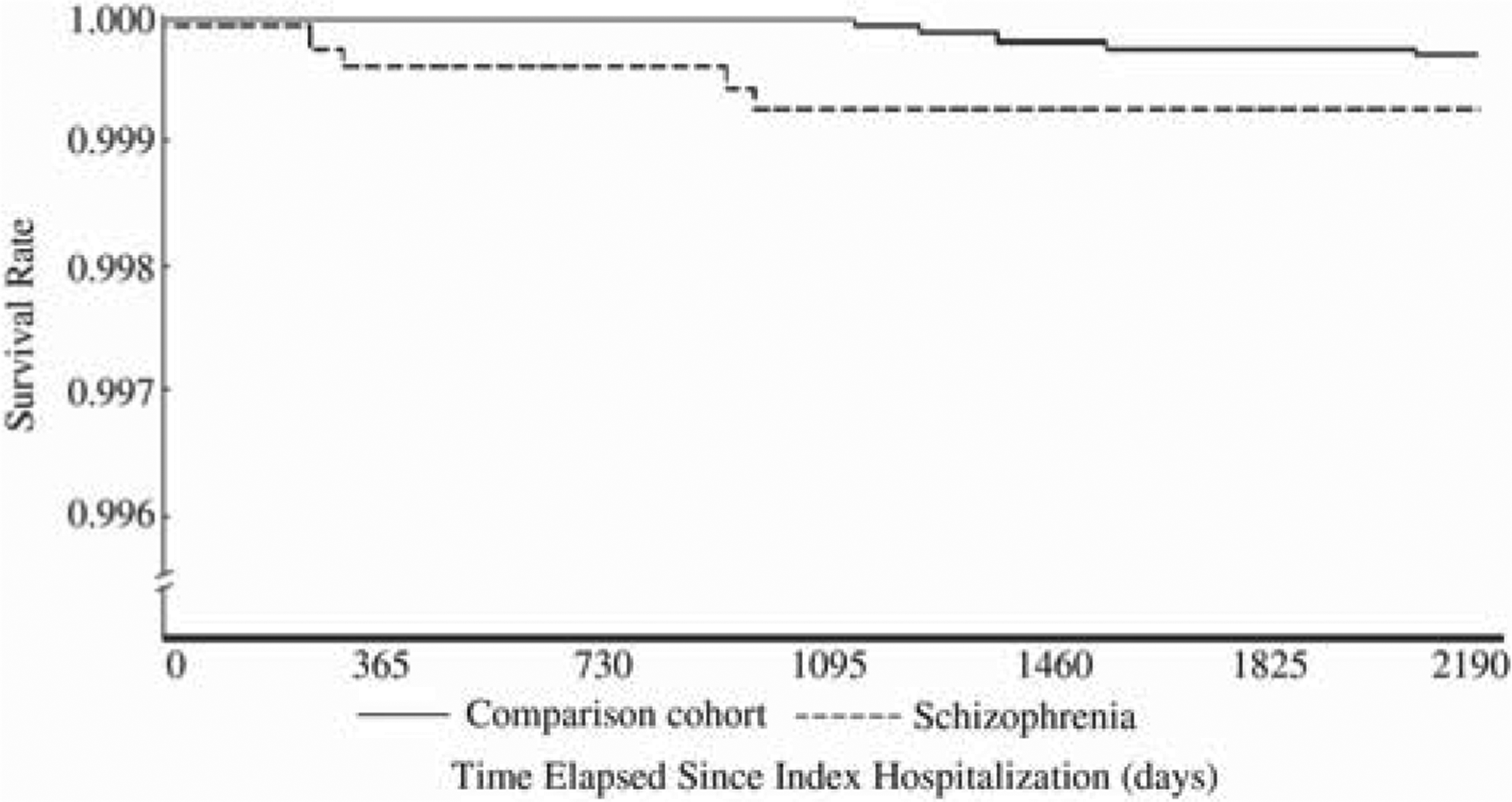
Acute myocardial infarction-free survival rates for (—) schizophrenia patients and (—) patients in the comparison cohort in Taiwan,
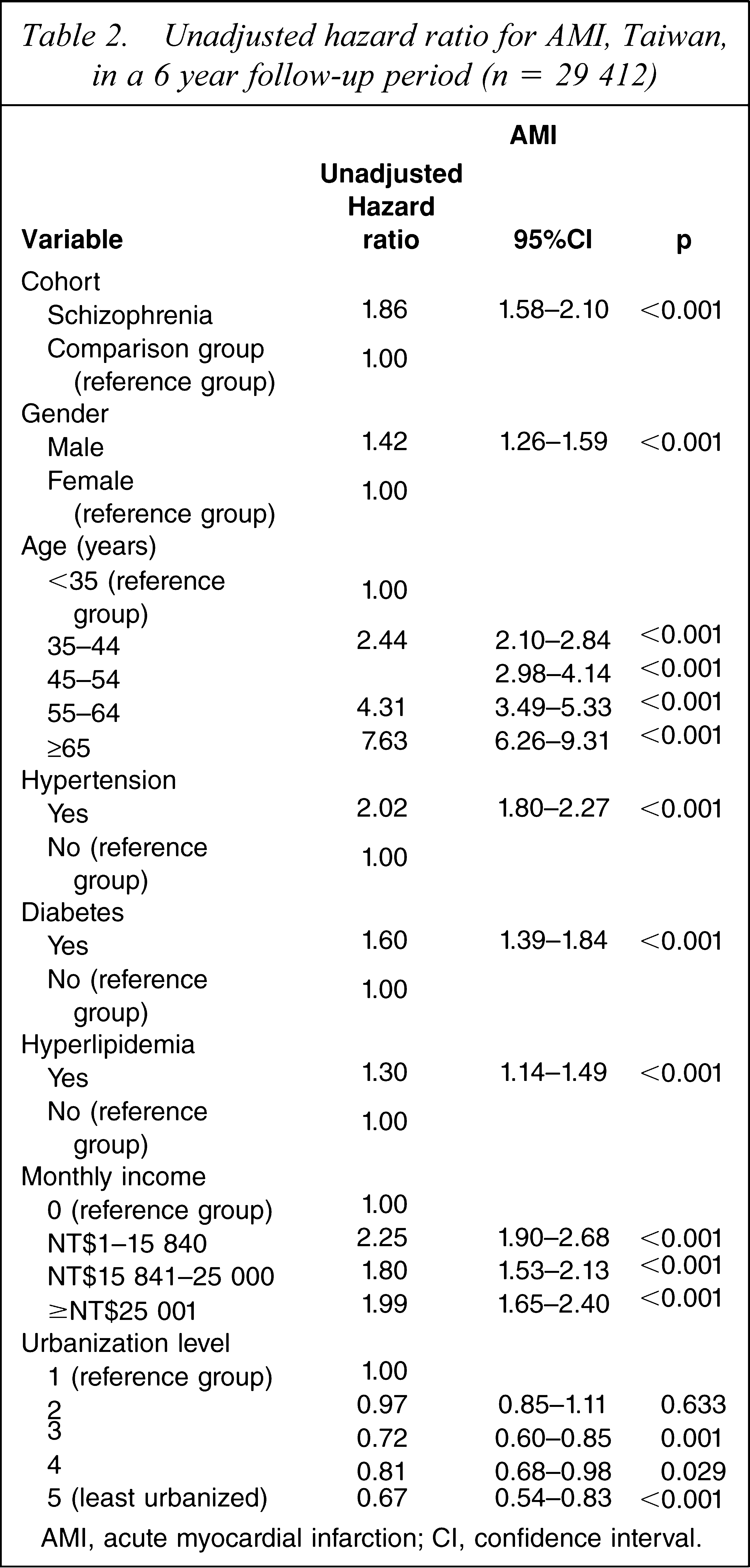
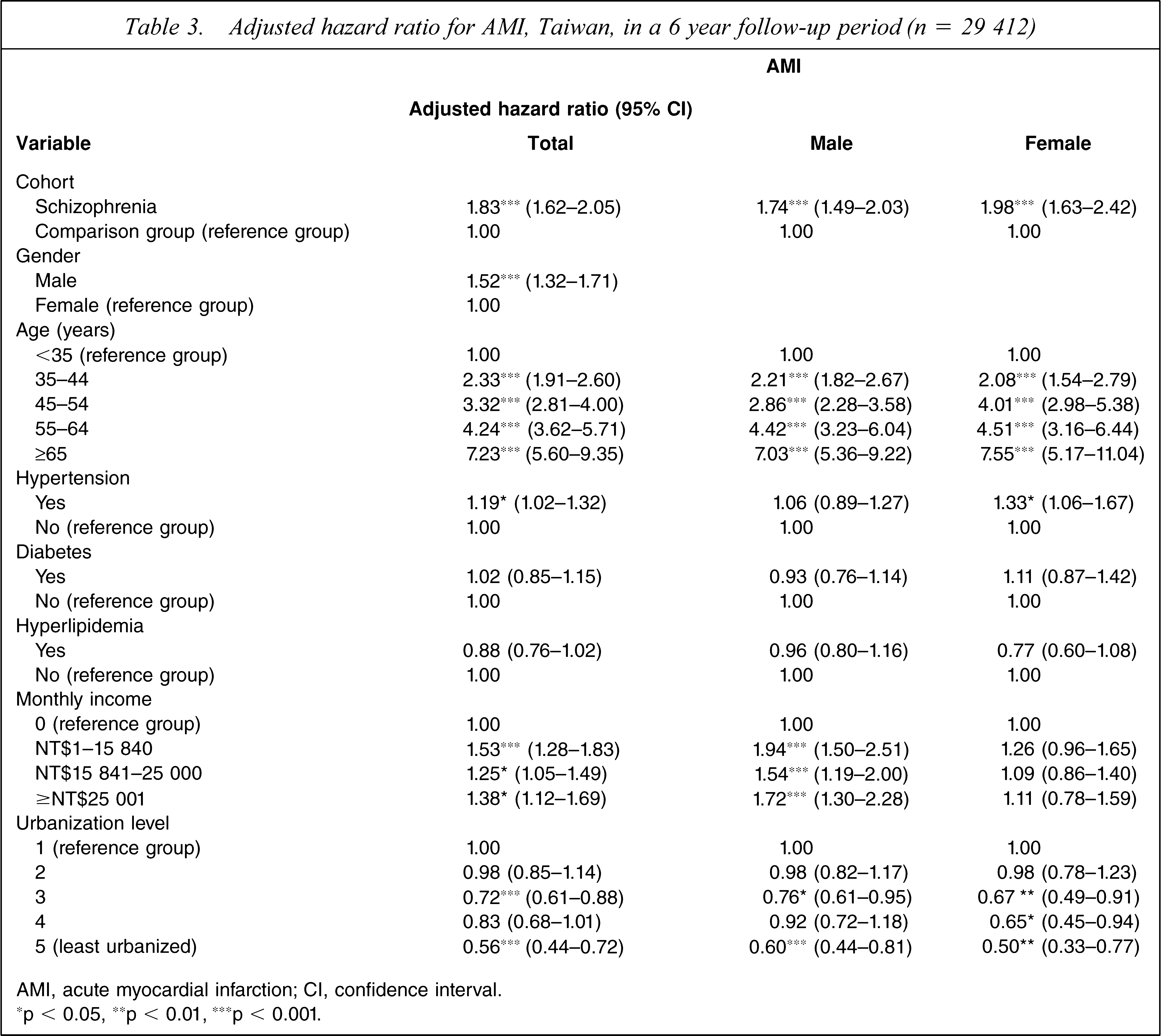
After censoring those patients who died from non-AMI causes, the unadjusted hazard ratios of AMI for schizophrenia patients were 1.86 (95% confidence interval (CI) = 1.58–2.10, p < 0.001) compared to patients in the comparison cohort (Table 2). Table 3 describes the adjusted hazard ratios for AMI by cohort. After censoring those patients who died from non-AMI causes and adjusting for the patients’ gender, age, monthly income, geographic region, level of urbanization and comorbid medical disorders, the hazard of AMI occurrence during the 6 year follow-up period was 1.83-fold greater for schizophrenia patients (95%CI = 1.62–2.05, p < 0.001) than for patients in the comparison cohort. Interestingly, the adjusted hazard ratio of AMI occurence also increased with urbanization level of the community in which the patient resided. Table 3 lists results from further analyses classified by patient sex. Among male and female patients, the adjusted hazard of AMI during the 6 year follow-up period was 1.74-fold (95%CI = 1.49–2.03, p < 0.001) and 1.98-fold (95%CI = 1.63–2.42, p < 0.001), respectively, greater for patients with schizophrenia than for those in the comparison cohort.
Unadjusted hazard ratio for AMI, Taiwan, in a 6 year follow-up period (n = 29 412)
AMI, acute myocardial infarction; CI, confidence interval.
Adjusted hazard ratio for AMI, Taiwan, in a 6 year follow-up period (n = 29 412)
AMI, acute myocardial infarction; CI, confidence interval.
∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001.
Discussion
The present results indicate that, after censoring those patients who died from non-AMI causes, the hazard of AMI was 1.83-fold greater for schizophrenia patients than for patients in the comparison cohort during the 6 year follow-up period, after adjusting for sociodemographic characteristics and comorbid medical disorders.
The present findings differ from a prior study by Curkendall et al., in which the incidence of AMI in the population with schizophrenia was not found to be significantly different from that in a comparison group during a follow-up period from January 1996 through March 1999 in Saskatchewan, Canada [11]. That study, however, did not adjust for the excess mortality risk among schizophrenia patients, and the overall effect of such survivor bias might be to underestimate the magnitude of associations between schizophrenia and AMI.
The present study findings also contradict a study by Jakobsen et al., who reported a statistically significant lower risk of AMI for schizophrenia patients compared to a control cohort, with the incidence rate ratio being 0.77, based on data from the Danish Psychiatric Central Research Register [10]. Their study, however, failed to adjust for important confounding factors such as age, hypertension, diabetes and hyperlipidaemia, which are associated with increased risk of AMI [12,14,15], and this could cast doubt on their findings.
Although previous studies did not find an association between schizophrenia and increased risk of AMI, others have documented higher rates of CVD among schizophrenia patients [8,9]. These studies have reported that factors such as obesity, poor diet, diabetes mellitus, dyslipidaemia, smoking, hypertension and the use of antipsychotic drugs contribute to the increased risk of CVD in schizophrenia patients [16–18]. We have adjusted for hypertension, diabetes and hyperlipidaemia. Other mechanisms contributing to the association between schizophrenia and AMI identified in the present study have not been clearly established. The actual prevalence of the aforementioned medical conditions that contribute to increased risk of CVD among patients with schizophrenia may exceed the number of patients currently being treated for them, as not all schizophrenia patients are properly diagnosed and many resist medical treatment in Taiwan [19]. Underestimation of this sort may lead to an apparent lack of significant associations between comorbid medical diseases and AMI in the present regression analysis.
Schizophrenia patients have a tendency to unhealthy lifestyles, including poor diet, a lack of exercise and obesity, largely attributable to their social disadvantages and schizophrenia symptoms [18]. In order to avoid the potential confounding factors of institutionalization and chronicity that may contribute to unhealthy lifestyles, we selected only those schizophrenia patients who had not been hospitalized or who did not receive ambulatory care treatment for any type of mental illness within the previous 5 year period before the index hospitalization. The increased risk of AMI remained evident in the follow-up period after an acute episode of schizophrenia.
Furthermore, cigarette smoking is consistently reported as one of the most important risk factors for AMI [19,20]. The smoking rate in the general population is around 21%, but the prevalence of tobacco use among patients with schizophrenia is as high as 80% [21]. In addition, schizophrenia patients are inclined to smoke more cigarettes daily than smokers in the general population [22]. This may contribute to the increased risk for AMI among schizophrenia patients.
Obesity is also an independent risk factor for AMI [23]. One study by Allison et al. reported that 42% of schizophrenia patients have a body mass index (BMI) > 27 compared to approximately 25% of the general population [24]. In particular, the weight gain induced by the use of antipsychotics has been seen as contributing significantly to the risk for numerous diseases, including insulin resistance, lipid abnormalities and hypertension [25], all of which can result in vascular events such as AMI. In addition, psychiatric drugs, antipsychotics in particular, may have direct cardiac side-effects [26]. The possibility of AMI developing under the use of psycho-tropic medications should be a concern for both patients and clinicians alike.
Recently, in another study we found that schizophrenia patients younger than 45 years of age had a twofold increased risk of stroke [27]. The increased risk for cardiovascular and cerebrovascular events among schizophrenia patients might imply a common mechanism that puts this disadvantaged population at higher risk of morbidity and mortality.
The major strength of the present study is its large nationwide population-based dataset of patients covered by the NHI programme, allowing us to trace all medical history and use of medical services for these patients. In addition, the strengths of the study include the availability of data on patients with a wide range of sociodemo-graphic characteristics and adjustment for comorbidities associated with the risk of AMI.
Despite these strengths, the findings need to be interpreted within the context of four limitations. First, the psychiatric and AMI diagnoses, which rely on administrative claims data reported by physicians or hospitals, may be less accurate than diagnoses made according to a standardized procedure. The NHI Bureau, however, randomly samples a fixed percentage of claims from every hospital each year, to verify diagnosis validity and quality of care through chart review by an independent group of doctors. Fines for fraud are 100-fold the amount of the false claim charged to the NHI Bureau. To ensure the validity of the schizophrenia diagnoses in the present study, we confirmed that all of the study cohort patients had at least two consensus schizophrenia diagnoses after the index hospitalization. Furthermore, virtually all hospitals in Taiwan capable of admitting AMI patients are equipped with electrocardiography or cardiac echo, which considerably increases the validity of AMI diagnosis. Second, the NHIRD database may represent patients who had sought treatment for schizophrenia and AMI. The comorbid medical or mental illnesses were presented based upon physician or psychiatrist diagnosis. For patients in the study and comparison cohorts, the codings or diagnoses of medical and mental illnesses should be parallel as long as they had sought treatment under the universal NHI programme. The third limitation is that the NHIRD lacks some variables such as dietary habits, cigarette smoking and BMI, which are likely to be associated with AMI occurrence. Last, although we have excluded subjects who had been diagnosed with both AMI and serious mental disorders, patients in the comparison cohort may have other CVDs apart from AMI or other mental health conditions. The present comparison cohort may not necessarily be representative of the general population.
In summary, the present study found that during a 6 year follow-up period, the adjusted hazard ratio of AMI occurrences was 1.84-fold greater for schizophrenia patients than the comparison cohort. We believe that there is an imperative for more comprehensive research that is designed to better understand the pathophysiological mechanisms underlying schizophrenia, and their association with AMI. Additionally, the present results draw attention to the need for clinicians to more actively monitor and treat schizophrenia patients with well-recognized risk factors associated with AMI.
Footnotes
Acknowledgements
This study is based in part on data from the National Health Insurance Research Database provided by the Bureau of National Health Insurace, Department of Health and managed by National Health Research Institutes. The interpretation and conclusions contained herein to not represent those of Bureau of National Health Insurance, Department of Health or National Health Research Institutes.
