Abstract
Introduction
Since the early 1900s, obsessive–compulsive symptoms (OCS) have been consistently described as a distinguishing feature of patients suffering from psychosis (Gordon, 1926; Stengel, 1945; Pious, 1950; Rosen, 1957). Bleuler (1950), one of the contributors to the definition of schizophrenia, also mentioned OCS as a feature of schizophrenia. Demographic studies have also confirmed increased comorbidity of obsessive–compulsive disorder (OCD) and schizophrenia (Berman et al., 1995; Poyurovsky et al., 1999; Nechmad et al., 2003; Lee et al., 2006). In other words, OCS have been addressed as an important factor in explorations of psychotic-spectrum illnesses.
Exploratory studies on schizophrenia, with and without OCS, and on patients with OCD have tried to confirm the presence of neurocognitive impairments in visuospatial skills, delayed nonverbal memory, cognitive shifting, vigilance, and attention (Hwang et al., 2000; Lysaker et al., 2000; Shin et al., 2008). On the other hand, other research has suggested the absence of cognitive differences between patients with schizophrenia with and without OCS (Berman et al., 1998; Lysaker et al., 2002; Whitney et al., 2004; Öngür and Goff, 2005; Tiryaki and Ozkorumak, 2009). Furthermore, given that patients with schizophrenia and OCS have found to show severe levels of social withdrawal, longer hospitalization, and lower rates of marriage and employment (Berman et al., 1995), exploring the potential role of OCS in the developmental period preceding the onset of psychosis may have significant implications for clinical settings and interventions.
In addition to their role in research on schizophrenia, OCS also constitute an essential issue in research on individuals at ultra-high risk for psychosis (UHR). The presence of OCS has been classically presented as a prodromal sign for psychosis as well as a component of schizophrenia (Westphal, 1878). Kraepelin (1919) also commented on ‘the transition of obsession into other mental affections, especially in paranoia’. Indeed, the combination of OCS and poor insight is one of the symptom clusters that render discrimination between the prodromal phase of psychotic disorders and neurotic disorders difficult because many individuals at high risk for psychosis show OCS as prodromal symptoms (Tully and McGlashan, 2006). Unlike the situation with regard to schizophrenia, however, only a few studies have examined OCS in UHR individuals (Niendam et al., 2009; Fontenelle et al., 2011; Sterk et al., 2011). Recent studies have shown that OCS associated with various mood symptoms occur more frequently in UHR individuals than in the general population, but these studies have failed to find a significant link between OCS and conversion to schizophrenia. Moreover, to date, there has been no research investigating the effect of OCS on the neurocognitive functioning of UHR individuals.
The present study compared the clinical implications and neurocognitive characteristics of UHR individuals with OCS (UHR+OCS) with those of UHR individuals without OCS (UHR−OCS). The aims of this study were (1) to examine differences in the clinical profiles of these groups and (2) to investigate the differences between groups with regard to the nature and extent of neurocognitive impairments. We predicted that more severe clinical symptoms and neurocognitive deficits would be observed in the UHR+OCS group than in the UHR−OCS group and healthy controls.
Methods
Sample
Of 93 potential UHR subjects recruited from the Seoul Youth Clinic (Korea), 65 were enrolled in the study. Eighteen UHR individuals did not complete clinical and neuropsychological testing, and 10 were excluded due to the criteria described below. All referrals between November 2004 and August 2010 were screened via the Structured Clinical Interview for DSM-IV, Axis I Disorders, the Comprehensive Assessment of At-Risk Mental States (CAARMS), and the Structured Interview for Prodromal Symptoms (Yung et al., 2005; Jung et al., 2010). The UHR subjects had to fulfil at least one of the following criteria: (1) attenuated psychotic symptoms (n=60 in the study); (2) brief, limited, and intermittent psychotic symptoms (n=0); and (3) trait plus state risk factors (n=7). Two subjects met the intake criteria for both attenuated psychotic symptoms and trait plus state risk factors.
The UHR group was divided into two subgroups according to the presence of OCS: 24 participants were placed in the UHR+OCS group and 41 in the UHR−OCS group. The UHR+OCS group was selected using the Yale–Brown Obsessive Compulsive Scale (Y–BOCS) (Goodman et al., 1989) using a cut-off score of 8 as the criterion for admission (Lysaker et al., 2000; Whitney et al., 2004). The UHR+OCS group included eight individuals with a DSM-IV diagnosis of OCD at enrolment (33% of the 24 UHR+OCS participants). UHR individuals who lacked either obsessive thinking or compulsive behaviour were included in the UHR−OCS group.
To strictly limit borderline cases, both groups excluded participants who had some OCS items on the Y–BOCS but whose scores were below threshold (1 ≤ Y–BOCS ≤ 7) because we wanted to exclude the possibility that trivial symptoms might affect clinical or cognitive features. Following these exclusion criteria, 10 UHR individuals were not included in the analysis. Most UHR subjects had not received drug therapy (51/65, 78.5%) for at least 3 months before the study. Seven (10.8%) UHR individuals were taking antipsychotics, three (4.6%) were taking anxiolytics, two (3.1%) were taking antidepressants, and one (1.5%) was taking a mood stabilizer. Another six (9.2%) UHR individuals had taken antidepressants and one (1.5%) had taken only anxiolytics. No group differences were found with respect to medication use (χ2[6]=6.40, p=0.380). Fifty-three UHR individuals continued 1-year follow-up using the same clinical interviews.
Healthy controls were recruited from the community via advertising and screened with the Structured Clinical Interview for DSM-IV, Non-Patient Version (SCID-NP). Subjects were excluded if they had a history of either neurological or psychotic disorders, manic episodes, drug or alcohol abuse or dependence within the past 6 months, or an IQ below 70. Additionally, potential participants were excluded if they had any first- or second-degree relatives with a history of any psychiatric illness. Written informed consent was obtained from all participants after they had been completely informed of the study protocols. The Seoul National University Hospital Institutional Review Board approved the study protocol.
Clinical assessment
The baseline psychosocial functioning of all subjects was assessed using the Global Assessment of Functioning (GAF) (APA, 2000). The Y–BOCS was used to assess the severity of OCS based on time spent engaging in OCS, the degree to which OCS interfered with their lives, their level of resistance to OCS, and control over either obsessions or compulsions. Clinical symptoms were rated by trained psychiatrists using the Positive and Negative Syndrome Scale (PANSS) (Kay et al., 1987), the Brief Psychiatric Rating Scale (BPRS) (Overall and Gorham, 1962), the Hamilton Rating Scale for Depression (HAM-D), and the Hamilton Rating Scale for Anxiety (HAM-A) (Hamilton, 1959; Hamilton, 1960).
Neurocognitive assessment
The short form of the Korean Wechsler Adult Intelligence Scale (K-WAIS) was administered to estimate IQ based on scores on the Vocabulary, Arithmetic, Picture Arrangement, and Block Design subtests (Kim and Lee, 1995). The Trail-Making Tests (TMT) (Reitan, 1958), the Stroop task (Trenerry et al., 1989), the Category Fluency Test (supermarket and animal) (Kertesz, 1982; Mattis, 1988), the Controlled Oral Word Association Test (COWAT) (Benton, 1969), and the Wisconsin Card Sorting Test (WCST) (Heaton et al., 1993) were used to estimate executive function. Interference indices in both the TMT and Stroop task were calculated in terms of the following: (1) time differences in the TMTs were defined as time for completion of TMT-B minus time for completion of TMT-A; and (2) Stroop interference scores were defined as the time for the colour–word condition minus that for the word condition. Of the possible dependent variables provided by the WCST, we used (1) number of correct responses, (2) number of perseverative errors, and (3) categories completed.
We next administered the Korean version of the California Verbal Learning Test (K-CVLT) (Kang and Kim, 1997) and the Rey–Osterrieth Complex Figure (ROCF) test using the Boston Qualitative Scoring System (BQSS) (Stern et al., 1994) for verbal memory and visual learning/organizational ability, respectively. The K-CVLT is a measure of declarative memory related to both learning outcome and process. It yields qualitative scores for the immediate and delayed recall of 16 nouns learned verbally and for the strategies used for learning, such as semantic or serial clustering. The BQSS also provides comprehensive sets of qualitative scores as well as quantitative scores including response times for copying and immediate/delayed recall trials (Shin et al., 2006). The system used in this study examined the qualitative features of ROCF performance according to the following subscales: planning (the order in which the elements were drawn); fragmentation (whether the elements were drawn as entities or as pieces); perseveration (the repetition of a part or all of an element); and confabulation (the insertion of an unrelated shape). For the TMT, Stroop task, perseverative errors in WCST, and serial clustering index in CVLT, higher scores represent poor performance.
Statistical analyses
One-way analysis of variance (ANOVA) or χ2 tests were performed on demographic and clinical variables. Multivariate analyses of variance (MANOVA) with Bonferroni’s post hoc tests were conducted on the neuropsychological variables because of the possibility of correlations among the variables.
Results
Demographic data
We found no group differences in the demographic data including age (F[2,102]=1.37, p=0.900), sex (χ2[2]=0.18, p=0.916), education (F[2,102]=0.50, p=0.606), and IQ (F[2,102]=0.77, p=0.465) (Table 1). The mean score on the Y–BOCS was 18.21±7.87 in the UHR+OCS group. Those UHR individuals who had been followed for at least 1 year (18 UHR+OCS and 35 UHR−OCS) showed no difference in transition rates (three transits in UHR+OCS and seven in UHR−OCS; χ2[1]=0.09, p=1.000).
Demographic and clinical characteristics of the participant
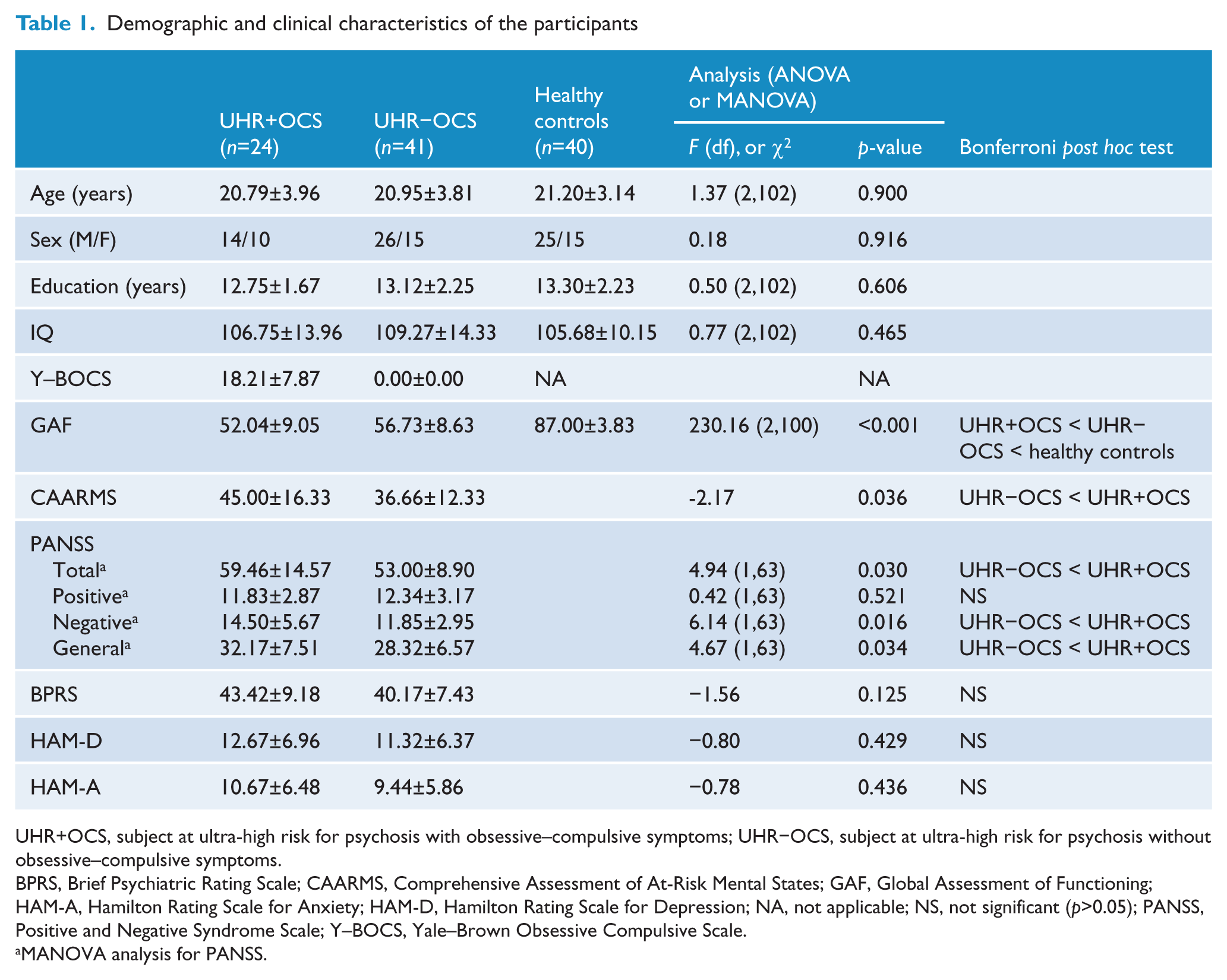
UHR+OCS, subject at ultra-high risk for psychosis with obsessive–compulsive symptoms; UHR−OCS, subject at ultra-high risk for psychosis without obsessive–compulsive symptoms.
BPRS, Brief Psychiatric Rating Scale; CAARMS, Comprehensive Assessment of At-Risk Mental States; GAF, Global Assessment of Functioning; HAM-A, Hamilton Rating Scale for Anxiety; HAM-D, Hamilton Rating Scale for Depression; NA, not applicable; NS, not significant (p>0.05); PANSS, Positive and Negative Syndrome Scale; Y–BOCS, Yale–Brown Obsessive Compulsive Scale.
MANOVA analysis for PANSS.
Clinical assessment
Both UHR groups experienced moderate levels of impairment compared with controls in terms of global functioning according to the GAF (F[2,100]=230.16, p<0.001 (Table 1). Moreover, the UHR+OCS group demonstrated more severe dysfunction than did the UHR−OCS group in daily life (healthy controls > UHR−OCS > UHR+OCS). Additionally, significant differences were observed between the UHR+OCS and UHR−OCS groups in most of the clinical scales. Scores on the CAARMS were higher in the UHR+OCS than in the UHR−OCS group (t=−2.17, p=0.036). On the PANSS scale (Wilks’ λ=0.85, F[4,60]=2.60, p=0.045), total scores (F[1,63]=4.94, p=0.030), scores on the negative elements (F[1,63]=6.14, p=0.016), and scores on the general subscales (F[1,63]=4.67, p=0.034) were higher in the UHR+OCS than in the UHR−OCS group. We found no significant differences between UHR groups in scores for the positive elements of the PANSS (F[1,63]= 0.42, p=0.521). Tendencies towards increased scores on the BPRS (t=−1.56, p=0.125), HAM-D (t=−0.80, p=0.429), and HAM-A (t=−0.078, p=0.436) were observed in the UHR+OCS group, but these were not significant. No correlations between Y–BOCS levels and scores on the clinical scales (GAF, r=−0.17, p=0.435; CAARMS, r=0.17, p=0.427; PANSS total, r=0.08, p=0.704; BPRS, r= 0.05, p=0.819; HAM-D, r=−0.18, p=0.399, HAM-A, r=0.05, p=0.804) were observed.
Neurocognitive assessment
The MANOVA revealed significant overall differences in the neurocognitive testing among the three groups (Wilks’ λ=0.50; F[44,162]=1.53, p=0.031) (Figure 1). A series of executive functioning tests revealed significant differences only for the Stroop interference index (F[2,102]=4.76, p=0.011). The UHR−OCS group showed a longer delay in the colour–word interference condition than did the control group (p=0.008), and the delay shown by the UHR+OCS group was similar to that shown by the control group (p=0.917). In the area of visual learning, we observed significant effects of the BQSS confabulation score on ROCF immediate recall (F[2,102]=3.28, p=0.042). The Bonferroni’s post hoc test revealed that when asked to immediately recall visuospatial stimuli, the UHR−OCS group showed significantly more confabulations than did healthy controls (p=0.036). Although the BQSS perseveration scores for the ROCF immediate recall were lower among the UHR+OCS group (F[2,102]=3.11, p=0.049), post hoc adjustments for multiple comparisons using Bonferroni’s test revealed no significant differences. With the exception of the aforementioned findings, we observed no significant difference in any index of executive function or verbal and visual memory. The performance of the three groups with regard to the neurocognitive tasks is presented in Table 2.
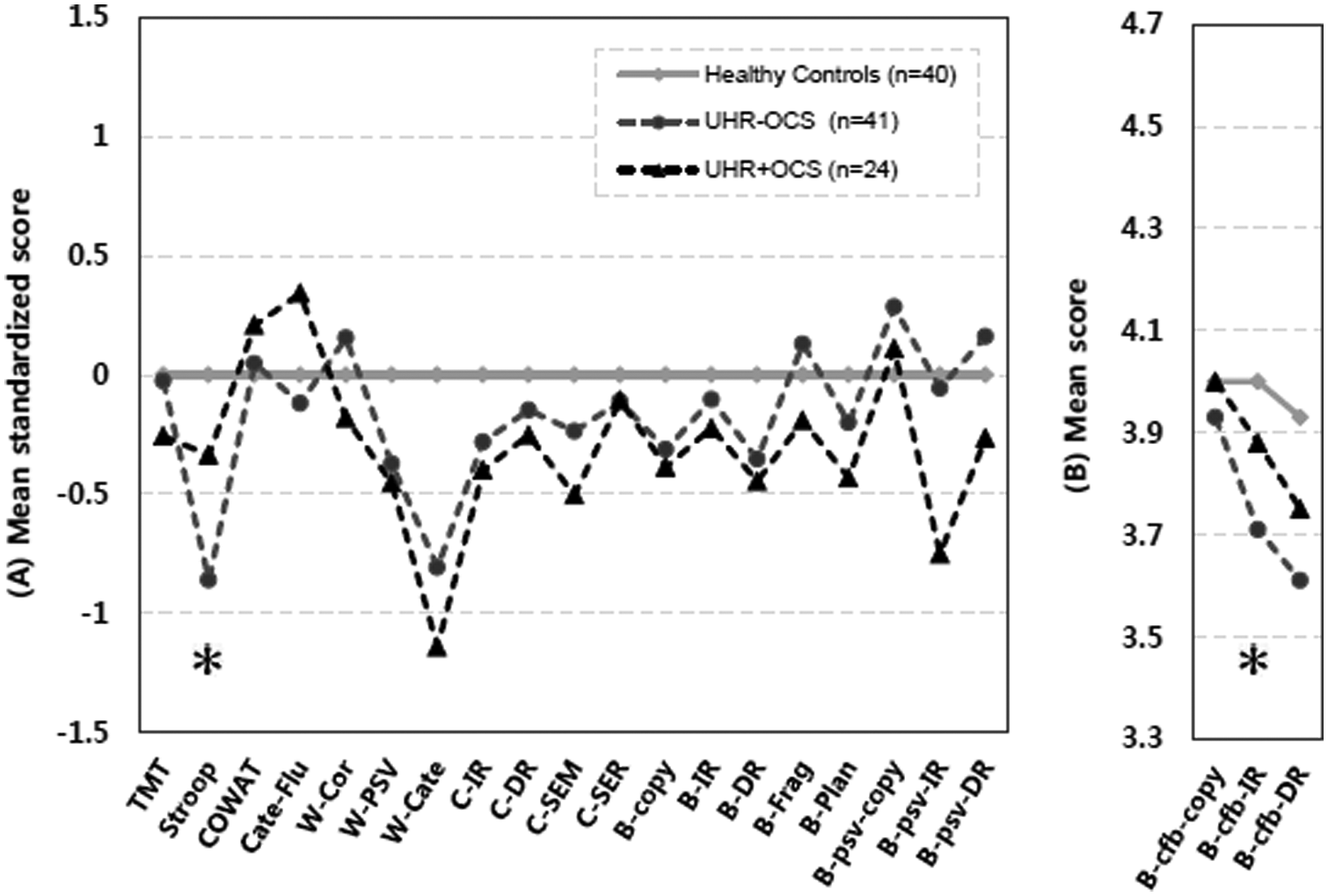
Performance across neurocognitive tests of healthy controls and ultra-high risk with and without obsessive–compulsive symptoms. (A) Group means of neurocognitive scores standardized to the control mean: Z-scores for neurocognitive variables were computed based on the mean and standard deviation of the controls. Plus/minus sign of TMT, Stroop, W-PSV, and C-SER were reversed to adjust the severity profile in the graph. (B) Group means of neurocognitive raw scores: the Boston Qualitative Scoring System-Confabulation score could not be transformed because the distribution of controls showed uniformed distribution (standard deviation of 0).
Mean scores and standard deviation of neurocognitive test in ultra-high risk group with and without obsessive–compulsive symptoms.
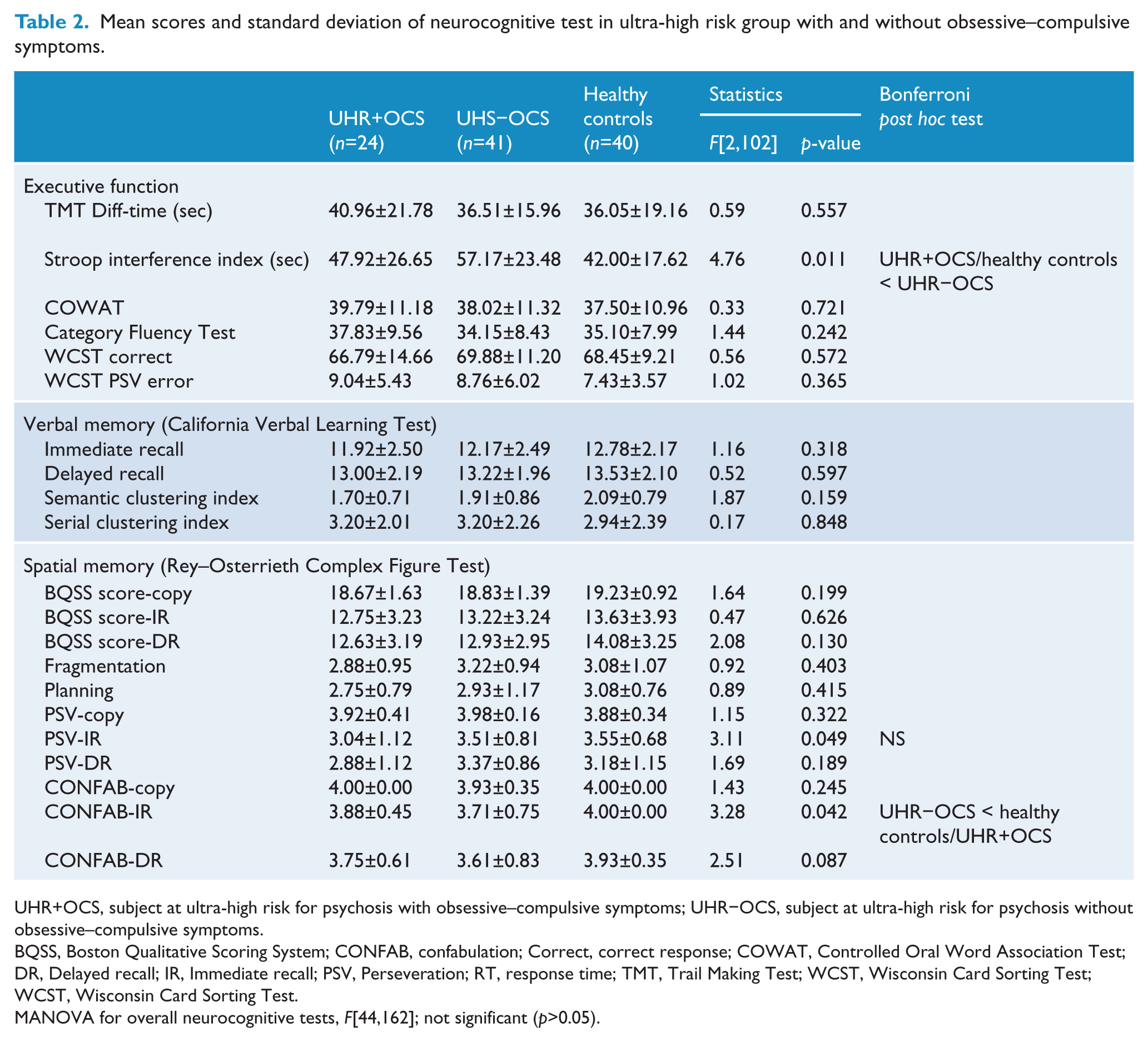
UHR+OCS, subject at ultra-high risk for psychosis with obsessive–compulsive symptoms; UHR−OCS, subject at ultra-high risk for psychosis without obsessive–compulsive symptoms.
BQSS, Boston Qualitative Scoring System; CONFAB, confabulation; Correct, correct response; COWAT, Controlled Oral Word Association Test; DR, Delayed recall; IR, Immediate recall; PSV, Perseveration; RT, response time; TMT, Trail Making Test; WCST, Wisconsin Card Sorting Test; WCST, Wisconsin Card Sorting Test.
MANOVA for overall neurocognitive tests, F[44,162]; not significant (p>0.05).
Discussion
The presence of OCS is an important issue in the evaluation and treatment of psychosis because of its frequency in psychotic disorders and its effect on social relationships, employment, and global functioning (Fenton and McGlashan, 1986). The present cross-sectional evaluation of UHR subjects with and without OCS demonstrated that those with OCS showed significantly greater dysfunction in daily living and increased clinical symptoms while retaining neurocognitive functioning. They even performed better on the Stroop task and showed fewer confabulation errors on the ROCF recall tests than did those without UHR. Although the UHR+OCS group showed more perseverative errors on the ROCF, the differences were not significant after post hoc tests. The possibility that OCS could serve a protective function with respect to neurocognitive impairments should be studied, especially in the context of a previous study that regarded OCS as buffer that prevented the emergence of psychosis during prodromal periods (Pious, 1950). The distinctive differences between UHR individuals with and without OCS suggest that UHR individuals with OCS should attract the same sort of clinical interest as do those with OCS and schizophrenia (Berman et al., 1997). Our study may provide ideas for clinical interviews with and research on the heterogeneous UHR group.
The clinical implications of OCS in our sample of UHR individuals were revealed on a variety of psychiatric scales. GAF scores showed clear discrepancies in the global functioning of the three groups (healthy controls > UHR−OCS > UHR+OCS). These findings related to GAF scores were undetected in a previous study (Niendam et al., 2009). It seems that this discrepancy was a result of the larger sample size of the present study. Similar to the results related to GAF scores, data from the CAARMS, which provided comprehensive information about the psychopathology in UHR individuals, showed significantly higher scores in the UHR+OCS as compared with the UHR−OCS group. The PANSS total, negative, and general scores were also significantly higher in the UHR+OCS as compared with the UHR−OCS group.
We included UHR individuals with both OCD and OCS in our analyses; thus, the severity of OCS versus that of OCD could be called into question. However, we found no linear relationship between scores on the Y–BOCS and those on other clinical scales. That is, the occurrence of OCS could be a crucial factor for UHR individuals, even though it may be seemed trivial.
The current findings suggest that UHR+OCS individuals may suffer more severe clinical problems than do those with UHR−OCS. These clinical findings indicate compromised daily functioning and suggest additional clinical symptoms, and they therefore have implications for patients’ quality of life and for prognostic issues. Previous studies have considered that the GAF scores obtained at baseline are prognostic indicators for psychotic disorders (Lehmann and Cancro, 1980; Kendell, 1983; Berman et al., 1997). Additionally, Diener et al. (1984) reported links between decreased GAF scores and vulnerability to mental disorders. Given the decreased GAF scores in UHR+OCS individuals, their higher CAARMS and PANSS scores, which together represent the level and range of psychopathology, also highlight the possibility of a poor prognosis and compromised quality of life for these individuals (Packer et al., 1997; Karow et al., 2005; Yung et al., 2005). It is interesting to note that the transition rates did not differ between groups despite group differences in the GAF and other symptoms scales. This discrepancy might have been due to the protective effect of OCS against the transition to psychosis (Pious, 1950). Niendam et al. (2009) also suggested that OCS may be a risk factor for mood symptoms and suicidality in UHR individuals, but they defended the contribution of OCS in the conversion to psychosis. Intriguingly, results on the PANSS total, negative, and general scores similar to those in the present study have been found in previous studies of schizophrenia patients with and without OCS (Hwang et al., 2000; Lysaker et al., 2002); those previous studies also failed to confirm increased positive scores in individuals with OCS. We should be cautious not to over-analyse data obtained from studies on schizophrenia, but should consider these findings in planning future research on the potential link between suppressed positive symptoms and OCS in psychosis.
Despite the increased clinical symptoms in the UHR+OCS group, several differences in neurocognitive functioning between the groups were attributable to the poor performance of the UHR−OCS group. The UHR+OCS group showed no signs of impairment in neurocognitive tests including the Stroop interference index and the error pattern during the ROCF immediate recall test, whereas UHR−OCS individuals performed poorly in these tasks. With the exception of the above, we found no significant group differences related to executive function, quantitative (immediate and delayed recall) or qualitative (semantic and serial recall) measures of verbal learning, and quantitative measures (copying, immediate, and delayed recall) of spatial learning. Nevertheless, despite these overall insignificant results, several neurocognitive differences should not be overlooked because the results of the Stroop and ROCF recall tasks clearly reflect the features of OCS. Indeed, we focus next on the interpretation of differences between the two groups in Stroop scores and error patterns in the ROCF recall tasks, which represent neurocognitive functions such as behavioural inhibition, sustained attention, monitoring, and executive retrieval functions.
First, the UHR+OCS group performed better than the UHR−OCS group on the Stroop task. The distinctive pattern of the response times under the incongruent and congruent conditions of the Stroop task seemed similar to that found in previous studies on schizotypal or schizophrenia patients with OCD (Spitznagel and Suhr, 2002; Lee et al., 2009). This research reported that schizophrenia-spectrum patients with OCD performed better than patients without OCS in the Stroop task. Given that OCD patients may perform even better than healthy controls in the same tasks (Moritz et al., 2002), it is clear that further research is needed to clarify the relationship between OCS and performance on neurocognitive tasks such as the Stroop task. Second, UHR−OCS subjects obtained lower scores for the ROCF immediate recall in the BQSS confabulation scores. In terms of the error patterns of the ROCF recall task, OCS in the UHR individuals may be understood as a protective factor against confabulation errors. Previous studies have noted differences between confabulators who suffer from ‘pathological certainty’ and OCD patients who suffer from ‘pathological uncertainty’ (Hirstein, 2005; Glowinski et al., 2008). That is, a pattern of excessive checking or a tendency to seek certainty among those with UHR+OCS may suppress inappropriate confabulation responses. However, this understanding requires further support because one study reported a greater number of confabulation errors in patients with pure OCD (Shin et al., 2004). Finally, the current results suggested a significant discrepancy between the clinical and neurocognitive profiles of the UHR+OCS group. Some previous studies also have suggested that the severity of psychiatric symptoms is irrelevant to cognitive impairment in UHR individuals, first-episode psychosis patients, and even adolescents with early-onset schizophrenia (Rund et al., 2004; Rhinewine et al., 2005; Niendam et al., 2006). Interestingly, a recent study showed an inverse correlation between subjective quality of life and neurocognitive functioning (Kurtz and Tolman, 2011), unlike previous studies (Ritsner, 2007; Savilla et al., 2008). Further study is needed to explore the discrepancy between clinical and neurocognitive profiles in UHR+OCS and UHR−OCS individuals.
This study has several limitations. Similar to the previous study on UHR+OCS and UHR−OCS (Niendam et al., 2009; Sterk et al., 2011), we found no differences between the UHR groups in the rates of transition to psychosis 1 year after enrolment in the study. Although many studies have suggested that UHR individuals are usually judged to have converted within in 1 year (Yung et al., 2004; Cannon et al., 2008), further study with larger sample sizes and longer follow-up periods is necessary to clarify the transition risk in UHR+OCS individuals. If the follow-up period had been extended, the rate of transition to psychosis in these groups may be affected. An example of such work is a recent study with a 13-year follow-up period (Lin et al., 2011). Another limitation of this study concerns the undifferentiated OCS dimension. This subgroup could not be divided in our study due to the limited sample. If this clinical condition had been considered with greater precision, we would have been able to provide more detailed explanations about UHR individuals. Further research allowing for investigation of the OCS dimension by using a large UHR sample needs to be performed.
Footnotes
Funding
This work was supported by the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea (A090096).
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
