Abstract
Attention-deficit hyperactivity disorder (ADHD) is a common, early-onset, lifetime-impairing neuropsychiatric disorder [1] affecting 5–10% school-age children in Western countries [2] and 7.5% in Taiwan [3]. Literature has documented high psychiatric comorbidity in ADHD [4,5] including oppositional defiant disorder (ODD) [6], conduct disorder (CD) [6], mood disorders [7–9], anxiety disorders [10,11], learning disorder [12], tic disorder [13], initial use [14] and regular use of substances [14], and substance use disorders [15]. There is a lack of information, however about whether adolescents without persistent ADHD also have increased risks for other psychiatric disorders. Also, previous reports about ADHD and psychiatric comorbidity were mainly based on cross-sectional data [16] and only some have investigated psychiatric comorbidity longitudinally [6,17].
The predictors for comorbid substance use disorder in ADHD patients are CD [18], childhood CD/ODD symptoms [17], hyperactive/impulsive symptoms [14,19], childhood inattentive symptoms [17,19] and persistence of ADHD [17]. Childhood hyperactive/impulsive symptoms [20,21] and current ADHD symptoms [22] are associated with comorbid ODD/CD in ADHD patients. Comorbidity of tics is related to medications including methylphenidate [23,24] and atomoxetine [25]. Comorbid internalizing disorders are associated with an earlier age of onset of ADHD [26], more severe ADHD symptoms [26] and family loading for anxiety, depression and adversity [27].
Despite the extensive studies on ADHD comorbidity in Western populations, few have been conducted in Asian populations [28,29] and none of them has investigated predictors for comorbidity at adolescence or adulthood. To fill in the gap in the understanding of ADHD comorbidity in ethnic Chinese populations and to investigate the association between partially remitted ADHD and psychiatric comorbidity, the aim of the present study was to examine current psychiatric comorbidity among probands with and without persistent ADHD as compared to children and adolescents without lifetime ADHD; and to determine the factors predicting the risks of psychiatric comorbid conditions among adolescents with childhood diagnosis of DSM-IV ADHD.
Methods
Participants
We recruited 296 patients (male, 85.5%) aged 11–17, who had had overt ADHD symptoms since the mean age of 4.2 ± 1.6, and were clinically diagnosed with DSM-IV ADHD at the mean age of 6.7 ± 2.7, consecutively mainly from National Taiwan University Hospital (n = 242). Their lifetime and current (past 6 months) ADHD and other psychiatric diagnoses were confirmed by psychiatric interviews with the participants and their parents using the Chinese version of the Kiddie Epidemiologic Version of the Schedule for Affective Disorders and Schizophrenia (Chinese K-SADS-E) at the participants’ mean age of 12.9 ± 1.6. The patients who had psychosis disorders, autism spectrum, or a full-scale IQ < 80 were excluded. The ADHD subtype distributions at baseline were 221 (74.7%) for combined type, 65 (22.0%) for predominantly inattentive type, and 10 (3.3%) for predominantly hyperactive-impulsive type.
The school comparison participants (school controls), who were assessed without ADHD at the ages of 6-8 and at adolescence using the Chinese K-SADS-E interview at the mean age of 12.9 ± 1.5, were recruited from the same school districts as the patients with ADHD, yielding 185 school controls (male, 72.4%).
Measures
Chinese K-SADS-E
The Chinese K-SADS-E is a semi-structured interview scale for the systematic assessment of both past and current episodes of mental disorders in children and adolescents. The development of the Chinese K-SADS-E has been described elsewhere in detail [3,30]. The Chinese K-SADS-E is a reliable and valid instrument to assess DSM-IV child and adolescent psychiatric disorders, and has been used extensively in clinic-based [31] and community-based studies [32].
Interviewer training
Four interviewers, who had undergone 1 year of intensive clinical and research training in child psychiatry before the Chinese K-SADS-E interview training, reached >90% agreement on all mental disorders assessed by the Chinese K-SADS-E (range = 98.25 ± 1.91–99.38 ± 1.06) against the rating of each item in the Chinese K-SADS-E by the first author for 30 clinical subjects before study implementation. The interrater reliability of the Chinese K-SADS-E among the first author and the four interviewers using 12 subjects was satisfactory for all mental disorders, with generalized kappa for each diagnosis ranging from 0.86 to 1.00. Their Chinese K-SADS-E interviews were audio-taped periodically and monitored by the first author, who was blind to the personal information of the participants, to ensure the quality of interviews.
Best-estimate diagnoses
The first author, trained in best estimate [33] for diagnoses during her doctoral study at Yale, was blind to the diagnostic status and name of the participant and was not involved in the direct Chinese K-SADS-E interview of any of the participants or their parents at follow-up. The first author made all the best estimates of each psychiatric diagnosis according to the data from the Chinese K-SADS-E interviews of participants and their mothers [34], medical records, and other self-administered questionnaires reported by the participants, parents, and teachers. The diagnostic coding was categorized into definite (meeting all DSM-IV diagnostic criteria), probable (either not meeting all DSM-IV symptoms criteria but more than half or no functional impairment), possible (some symptoms but no impairment), and no diagnosis. For mental disorders other than ADHD, those patients who received a rating as definite or probable on the best estimate were categorized as having a particular mental disorder.
Persistent and non-persistent ADHD
Patients with a childhood diagnosis of ADHD were categorized into (i) patients with persistent ADHD (186, 62.8%) if their current symptoms reached the definite category (meeting all DSM-IV ADHD diagnostic criteria including impairment) based on the best estimate; and or (ii) patients without persistent ADHD (110, 37.2%).
Procedures
The Research Ethics Committee of National Taiwan University Hospital approved the present study prior to implementation. We obtained written informed consent from both the participants and their parents. The Wechsler Intelligence Scale for Children–3rd edition was administered to all the participants to exclude those who had an IQ < 80. All the adolescent participants and their parents were interviewed independently by individual well-trained interviewers for the DSM-IV psychiatric diagnoses at baseline first to confirm childhood diagnosis, followed by current psychiatric assessments separately, using the Chinese K-SADS-E. Medication information was obtained by interviews and validated by medical records of prescription. All the interview data were cross-checked by research team members independently and the best estimate of psychiatric diagnosis of each participant was made by the first author blindly and independently.
Data analyses
Data analysis was conducted using SAS 9.1 (SAS Institute, Cary, NC, USA). The comparison groups were (i) patients with persistent ADHD; (ii) patients with non-persistent ADHD; and (iii) school controls without a lifetime diagnosis of ADHD. We used a logistic regression model to compare the rates of psychiatric disorders among the three groups controlling for age, sex, parental educational level, and current treatment with methylphenidate. Odds ratios (ORs) and 95% confidence intervals (CIs) were computed. For all the analyses, we started from the four-way interactions (age × sex × medication × group). We found that there was neither four-way nor three-way interactions for the group differences in psychiatric comorbid conditions. For the majority of statistical models, there were no two-way interactions between age, sex, medication, and the group (ADHD vs control) on the psychiatric comorbid conditions except that there was an interaction of age × group for ODD (regression coefficient, β = 0.19, p = 0.02). Therefore, we did not stratify by sex and age in all the analyses. We also tested the linear trend across the three groups from unaffected controls, to non-persistent ADHD, and persistent ADHD after model comparisons indicated that the group variable can be treated as a linear variable in the logistic regression model using the goodness of fit tests. The alpha was pre-selected at the level of p < 0.05.
Results
Sample description
Table 1 presents the demographic data, and medication and clinical history of the three groups. The two ADHD groups had a higher proportion of male participants and lower parental educational levels. Patients with persistent ADHD were found to have overt ADHD symptoms at a younger age than those without. There was no difference in maternal (p = 0.282) and paternal (p = 0.121) job types, age of clinical diagnosis of ADHD or medication history (Table 1).
Subject characteristics
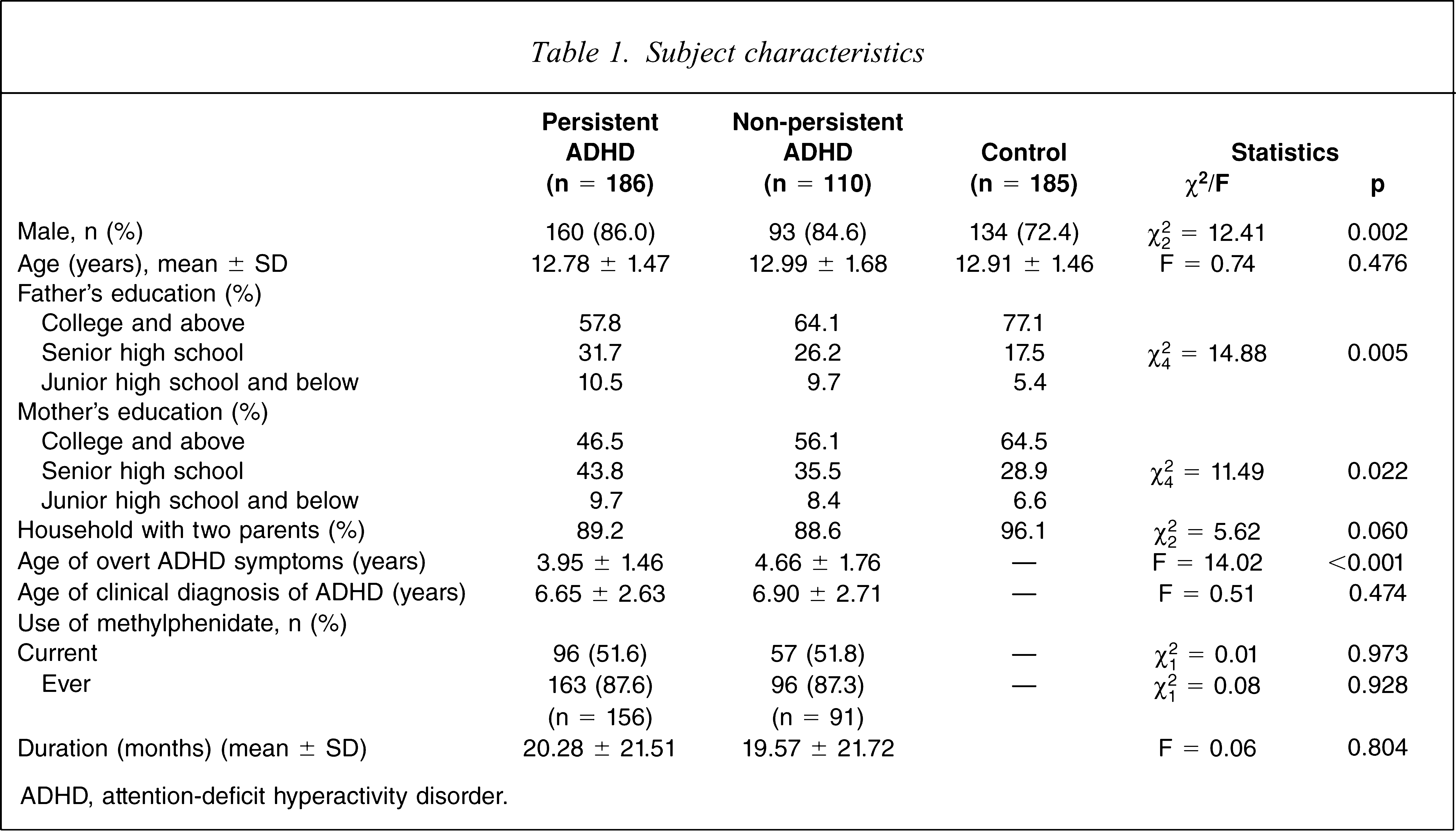
ADHD, attention-deficit hyperactivity disorder.
Psychiatric comorbidity
Table 2 summarizes the rates and ORs with the 95%CIs of having other psychiatric disorders among the three groups. The ADHD groups were significantly more likely than the controls to have had any psychiatric disorders at adolescence including ODD, CD, tic disorder, mood disorders (major depression, bipolar disorders), ever use of nicotine, alcohol, betel nut, regular use of nicotine, substance use disorders (mainly nicotine use disorder), and sleep disorders (ORs = 1.84–25.34, Table 2). The significance of these results remained after further controlling for other comorbidities.
Psychiatric disorders at adolescence
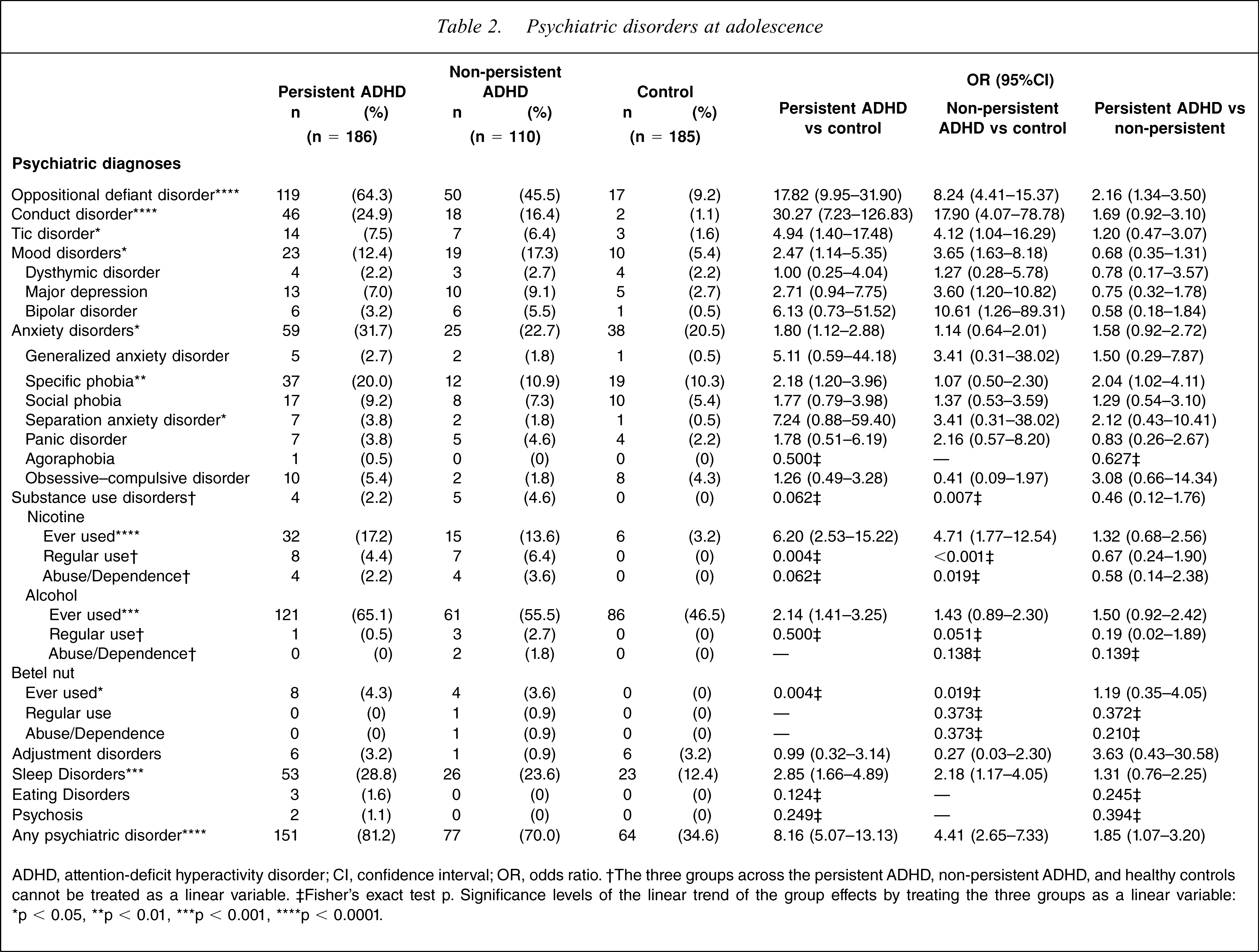
ADHD, attention-deficit hyperactivity disorder; Cl, confidence interval; OR, odds ratio. †The three groups across the persistent ADHD, non-persistent ADHD, and healthy controls cannot be treated as a linear variable. ‡Fisher's exact test p. Significance levels of the linear trend of the group effects by treating the three groups as a linear variable: ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001, ∗∗∗∗p < 0.0001.
Both patients with and without persistent ADHD had higher risks for ODD, CD, tic disorder, mood disorders, ever and regular use of nicotine, ever use of betel nut, and sleep disorders than the controls (Table 2). In addition, patients with persistent ADHD were more likely than the controls to have anxiety disorders, particularly specific phobia (Table 2). Patients without persistent ADHD were more likely than the controls to have major depression, bipolar disorders, substance use disorder (nicotine abuse/dependence). Moreover, patients with persistent ADHD were more likely to have ODD, specific phobia, and any psychiatric disorders than their partially remitted counterparts (Table 2).
We found that the three groups were suitable to be treated as a linear variable in the majority of models except those having substance use disorder, regular use of nicotine, nicotine abuse/dependence, regular use of alcohol, and alcohol abuse/dependence (p < 0.05 for model comparisons). There were significant linear trends from the three groups on their prediction of any psychiatric disorders, ODD, CD, tics, anxiety disorders (particularly, specific phobia and separation anxiety), sleep disorders, ever use of nicotine, alcohol, and betel nut.
Correlates for psychiatric comorbid conditions
We further examined the factors influencing the risks for five major significant psychiatric comorbidities (Table 3). These predictors included childhood ADHD core symptoms, age of overt observable ADHD symptoms, treatment with methylphenidate, and demographics (sex, age, parental education as senior high school or below, household without two parents). Analyses indicated that inattention, hyperactivity, and impulsivity symptoms at childhood, and longer duration of treatment with methylphenidate significantly predicted ODD/CD. Ever treatment with methylphenidate significantly decreased and longer treatment duration of methylphenidate significantly increased the risk for tics. Older age and comorbidity with CD significantly predicted the risk for mood disorders and substance use disorders. Female gender and longer duration of methyl phenidate treatment significantly increased the risk for specific phobia.
Prediction of baseline ADHD symptoms, medication, and demographics of adolescent psychiatric outcomes
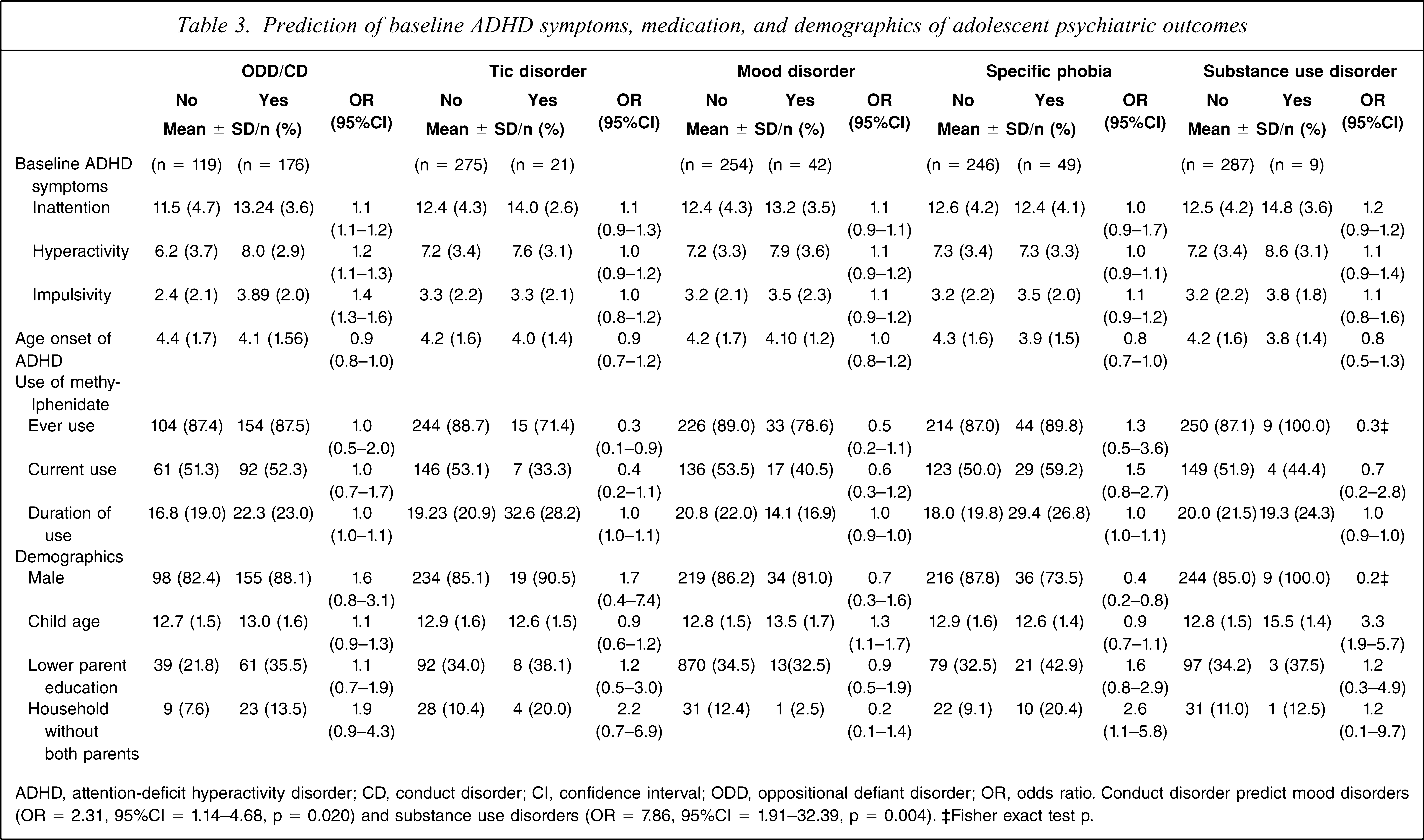
ADHD, attention-deficit hyperactivity disorder; CD, conduct disorder; Cl, confidence interval; ODD, oppositional defiant disorder; OR, odds ratio. Conduct disorder predict mood disorders (OR = 2.31, 95%CI = 1.14–4.68, p = 0.020) and substance use disorders (OR = 7.86, 95%CI = 1.91-32.39, p = 0.004). ‡Fisher exact test p.
We then used backward elimination in the model selection to identify the most associated correlates for the following comorbid conditions. We found that childhood hyperactivity (OR = 1.09, 95%CI = 1.01–1.25; p = 0.004) and impulsivity (OR = 1.29, 95%CI = 1.14–1.47, p < 0.001) significantly predicted ODD/CD. Longer duration of methylphenidate treatment (OR = 1.03, 95%CI = 1.01–1.05, p = 0.018) and younger age (OR = 0.60, 95%CI = 0.39–0.93, p = 0.023) predicted the risk for tic disorder. Older age (OR = 1.35, 95%CI = 1.08–1.70, p = 0.009), and comorbidity with CD (OR = 2.31, 95%CI = 1.12–4.80, p = 0.024) increased and ever treatment with methylphenidate (OR = 0.42, 95%CI = 0.19–0.97, p = 0.043) decreased the risk for mood disorders. Male gender (OR = 0.39, 95%CI = 0.19–0.82, p = 0.013) decreased and having a household without both parents (OR = 2.70, 95%CI = 1.18–6.22, p = 0.019) increased the risk for specific phobia. Older age (OR = 3.31, 95%CI = 1.81–6.06, p < 0.001) and comorbidity with CD (OR = 12.24, 95%CI = 1.99–75.25, p = 0.007) were associated with an increased risk for substance use disorders.
Discussion
Many Western studies have reported high rates of psychiatric comorbidity in ADHD, but this is the first to investigate the psychiatric outcomes at adolescence in an Asian population. Similar to Western studies in children and adolescents with ADHD [4,5,35,36], the present major findings are that a childhood diagnosis of ADHD was significantly associated with the risks for ODD, CD, tics, mood disorders, and ever and regular use of tobacco and betel nut, regardless of persistent ADHD. Although Biederman et al. reported that psychiatric comorbidity is related to persistent ADHD [37], the present study demonstrated that these psychiatric disorders except anxiety disorders also present in children and adolescents without persistent ADHD. This suggests that psychiatric comorbid conditions at adolescence cannot be fully explained by the persistence of ADHD symptoms. There was a significant linear trend in the order of school controls, non-persistent ADHD, and persistent ADHD in terms of the prediction of any psychiatric disorders, ODD/CD, tics, anxiety disorders, sleep disorders, ever use of nicotine, alcohol, and betel nut. The novel finding is the increased risk for specific phobia in adolescents with persistent ADHD.
Oppositional defiant/conduct disorders
The rate of ODD in the ADHD group (57.3%) has ranged from 32% [38] to 77% [37] across studies. The rate of CD (21.7%) is higher than that in epidemiological samples (12%) [38], but lower than that in clinical samples (30–50%) [4]. Consistent with previous studies [22], we also found a significant relationship between current ADHD symptom severity and current diagnosis of ODD/ CD. A gradient relationship across the three groups regarding the rate of ODD/CD suggests a dose-related linear relationship between current ADHD symptoms and an ODD/CD diagnosis, consistent with a recent study [39]. The present findings also indicate that a childhood diagnosis of ADHD predicts a higher risk for ODD/CD at adolescence [40], regardless of the persistence of ADHD. The present findings lend support to the notion that the severity of childhood hyperactive and impulsive symptoms is the most predictive factor for adolescent ODD/CD [20,21]. The significant effect of childhood inattention on adolescent ODD/CD, however, can be explained by hyperactive/impulsive symptoms.
Surprisingly, we found an association between longer duration of methylphenidate treatment and increased risks for ODD/CD at adolescence. Methylphenidate treatment was not designed to be randomized in the present study, so confounding self-selective factors such as ADHD symptom severity and comorbidity, may determine continued medication use [41]. Hence, treatment may indeed stand as a proxy for severity of ADHD [42,43]. Corresponding to previous studies, there were neither gender [44,45] nor age [3] differences in the risks for ODD/CD in ADHD.
Tic disorder
The rate of tic disorder in the ADHD group (7.1%) was lower than in previous reports, ranging from 10.9% (MTA study) [46] to 33% [47], with the average around 20% [36,48]. Unlike some [49] but corresponding to others [23,24], the present findings demonstrated that duration of methylphenidate treatment predicted tics, after taking confounding factors into consideration, suggesting that long-term use of methylphenidate may increase a risk for tics.
Mood disorders
The lower rate of bipolar disorder (4.1%) in the present report than in previous reports of 16% [8] to 23% [37] may be partially explained by the younger age of the present sample. Consistent with previous studies [35], ADHD is highly comorbid with major depression as a function of age [3], despite the fact that its rate is in the lower end of 3–75% [4]. The present findings, however, did not provide evidence to support an association between persistent ADHD and adolescent major depression [22], compared with non-persistent ADHD. Moreover, consistent with some studies, CD, not ODD, is associated with an increased risk for mood disorders [4]. Due to the relatively young age range and short follow-up period in the present study, further study of this sample with a longer follow-up period to clarify the prediction of ADHD to mood disorders is warranted. Identification of risk factors and correlates for mood disorders, which was not clarified significantly in the current study, will also be our next step.
Anxiety disorders
The rate of anxiety disorders in the ADHD group was similar to those reported in epidemiologic and clinical samples of ADHD, ranging from 25% [4] to 33% [50]. The magnitude of association, however, between persistent ADHD and anxiety disorders is not compatible with the estimate based on a meta-analysis (OR = 3.0, 95%CI = 2.1–4.3) [35]. One possible explanation is the higher prevalence rate of anxiety disorder in the control group (20.5%), which was higher than in Western (5–15%) [51] and Taiwanese epidemiological studies [3]. Despite the increased risk of anxiety disorders in adolescents with persistent ADHD, no such relationship was found in adolescents without persistent ADHD. This finding implies that anxiety disorders are likely to be related to current ADHD symptoms rather than a childhood diagnosis of ADHD [52].
Surprisingly, persistent ADHD is related to the risk for specific phobia, particularly in girls, a novel finding warranting further investigation. This finding is similar to some studies that girls are at a greater risk than boys regarding anxiety disorders [44,45] and separation anxiety disorder [45]. The present finding can also be explained by a higher prevalence rate of specific phobia in female gender [20]. The finding of an increased duration of methylphenidate treatment predicting specific phobia corresponds to some clinical trials showing that anxious and phobic symptoms are related to the adverse effects of methylphenidate [53]. Similar to earlier studies showing the relationship between parental divorce/ separation and comorbidity with anxiety disorder in ADHD [27], the present results indicate that a household without two parents predicts specific phobia.
Substance use
The present study not only lends evidence to support the prediction of persistence of ADHD to the risk for substance use in adolescence as shown in Western studies [17] and a Taiwan study [32], but also demonstrated an association between non-persistent ADHD and substance use in adolescents. This implies that a childhood diagnosis of ADHD may increase the risk for substance use, regardless of persistent ADHD at adolescence. Within the ADHD group, consistent with the majority of studies, older age [54] and comorbidity with CD [32], not ODD [32], were the predictors for substance use disorders. Lack of association between methylphenidate treatment and substance use disorder in adolescence is compatible to the findings of a 10 year follow-up study by Biederman et al. [55]. Unlike in several other studies [17,19], we did not find that childhood ADHD symptom severity predicted substance use disorders at adolescence. The relatively younger age at assessment in the present study than the other studies may have precluded a sound prediction of ADHD for substance use disorder at late adolescence or young adulthood.
Limitations
The major limitations of the present study were that questionable external validity needs to be examined further; that despite the clinical assessment of ADHD at childhood with detailed medical records, the Chinese K-SADS-E interview on childhood diagnoses were conducted at adolescence, resulting in possible recall bias; and that the follow-up period was not long enough to include more cases of mood disorders and substance use disorders for a more powerful examination of the prediction of ADHD and its comorbid conditions for these two disorders in adolescence and young adulthood.
The strengths of the current study were that the sample was of adequate sample size from a relatively less studied population; comprehensive assessments were done of ADHD and other psychiatric disorders at childhood and adolescence on clinical and psychiatric interviews of the participants and their parents; and collection was done of complete information about demographic data, baseline and current ADHD symptoms, age at onset of ADHD, and medication treatment history, which were validated by detailed medical records.
Conclusion
As in Western studies, Taiwanese adolescents with persistent ADHD are indeed more likely to have comorbid psychiatric disorders, and the onset of mood disorders and substance use disorders is a function of age, and comorbidity with CD. In addition, the increased likelihood of psychiatric comorbidity exists even in those without persistent ADHD, suggesting that the improvement of ADHD symptoms at adolescence may not result in decreased risks for psychiatric comorbidity. The present findings imply that identifying and eliminating factors contributing to the pathway from ADHD to other psychiatric disorders such as ODD, CD, anxiety disorders, mood disorders, and substance use disorders and recognizing age-specific comorbid patterns throughout the developmental stage is likely to be an important preventive strategy to offset the long-term adverse psychiatric outcomes among children with ADHD.
