Abstract
This study examines the structure of epigeic and arboreal ant communities in fragments of Seasonal Semideciduous Forest (SSF) and surrounding matrices (soybean and corn crops and fallow fields) in the Brazilian Cerrado. Ants were collected using baited traps placed along transects perpendicular to the forest fragments. Species specificity and habitat fidelity were evaluated using the indicator species index (IndVal). Environmental variables such as fragment size, litter quantity, and edge proximity did not significantly affect ant richness in SSF. However, non-parametric multidimensional scaling revealed clear differences in community structure between habitats (soil and canopy in SSF, and soil in matrices), and among the three land use states in the matrix (soybean, corn, or uncultivated). Rarefaction curves showed higher ant richness in SSF fragments, in soil and trees. Species composition differed across land uses, in the agricultural matrices, although overall richness did not vary significantly. Based on IndVal, five ant species were identified as indicators of SSF soil, and four as indicators of matrix soil. These patterns demonstrate how land use influences ant communities. Since ants perform vital ecological functions, such as nutrient cycling, seed dispersal and soil structuring, changes in their composition can reflect underlying shifts in ecosystem processes. The presence or absence of specific ant species thus serves as a reliable signal of environmental quality. This reinforces the value of ants as effective bioindicators and highlights the importance of conserving native forest fragments amid expanding agricultural landscapes.
Introduction
The Brazilian Cerrado is rich in plant species with extensive spatial heterogeneity, which facilitates its categorization as the tropical savanna with the highest biodiversity, anywhere in the world (Strassburg et al., 2017). According to Coutinho (2006), the Cerrado biome consists of one grassland (cam po limpo) three savannic vegetations (Campo Sujo, Campo Cerrado and Cerrado
Habitat fragmentation is often defined as a process during which a large expanse of habitat is transformed into several smaller patches of smaller total area, isolated from each other by a matrix of habitats unlike the original (Carvalho et al., 2009; Fahrig, 2003). Thus, understanding forest fragmentation processes and evaluating their effects on ecological processes at different spatial scales has become one of the most significant challenges for conservation biology (Fahrig, 2017).
The composition and arrangement of environmental features at landscape scales are fundamental determinants of local community structure (Hoenle et al., 2023; Spiesman & Cumming, 2008). Tavares et al. (2019) highlighted the importance of understanding the role of the matrix as a secondary habitat and its influence on the biodiversity of forest fragments. This matrix can influence the dispersion of species in the landscape (Evans et al., 2019), the edge effect (Rodrigues et al., 2019; Sobrinho & Shoereder, 2007), and enhance disturbances in the fragment (Tavares et al., 2019). Furthermore, the environmental structure and composition, as well as their temporal modification, affect distribution, abundance, composition, and dynamics of species (Hoenle et al., 2023; Madureira et al., 2012; Rodrigues et al., 2019).
Fragmentation can affect forest dynamics both positively and negatively. Since most animal species are affected, certain groups can be used to measure the impact of such fragmentation (Assis et al., 2018). The search for appropriate rapid diagnostic tools to identify the intensity of anthropization in forest environments needs to be continually maintained, particularly to propose suitable conservation steps. Bioindicators can be a quick and precise method for diagnosing environmental conditions. Much research has been ongoing in recent years, utilizing these environmental bioindicators and gaining positive outcomes (Rodrigues et al., 2019).
Insects are reliable bioindicators of anthropic terrestrial disturbances (Oliveira et al., 2014). Many studies have indicated that among the various insect groups, ants have potential to function as bioindicators of environmental quality (Castro et al., 2020; Oliveira et al., 2014; Ribeiro et al., 2022; Rodrigues et al., 2019; Silva et al., 2024; Tiede et al., 2017). Due to their high abundance and species richness, ants have specialized taxa, wide geographical distribution, can be easily sampled and are easy to separate at the morphospecies level. Furthermore, ants are highly sensitive to environmental conditions changes (Bernardes Júnior et al., 2020; Hoenle et al., 2023).
The SSF is present in regions where agriculture and cattle raising predominate, which the anthropization incidence due to factors such as the edge effect and natural disasters that result in alteration in the forest composition and structure (Mariano et al., 2024). In these conditions, these forests become good models of studies with bioindicator insects. Thus, this study aimed to characterize the epigeic and arboreal ant communities in SSF fragments in a region of Brazilian Cerrado, as well as the epigeic ant communities in agricultural matrices surrounding these same fragments. The structure of the ant communities in the different environments and at different times of occupation of the matrix by agricultural crops were compared. Furthermore, the metric variables of the SSF fragment were correlated with ant species richness.
Material and methods
Experimental area
The present study was conducted in six SSF fragments in the municipality of Ipameri, Goiás, Brazil. The geographical coordinates of the fragments and respective areas are as follows: F1: 17°43′46″S, 48°12′35″W, 20.9 ha; F2: 17°37′16″S, 48°10′20″W, 38.8 ha; F3: 17°45′42″S, 48°02′40″W, 4.22 ha; F4: 17°43′20″S, 48°07′01″W, 44.4 ha; F5: 17°36′00″S, 48°12′00″W, 33.4 ha; F6: 17°42′36″S, 48°18′28″W, 47.4 ha. The minimum and maximum distance between the fragments was around 4.7 and 31.5 km, respectively. These fragments were marked as strategically quite far from each other, thus ensuring sample independence. The matrices around all the SSF fragments were cultivated with identical cultures (soybean as the main crop and maize as a second crop immediately after soybean harvest).
Sampling and identification of ants
Ant sampling was carried out only with baited traps. First, a transect was established perpendicularly from a borderline point between the SSF fragment and its matrix area, where the bait traps were set up. As these traps (n = 30) were positioned at 15 m distances, 15 traps in total were set up in the matrix, apart from another 15 installed within the fragment. To attract the ants, a bait made of sardine oil and honey + sweet biscuit bran was placed in 250 mL plastic containers. Using metal clamps, the containers were affixed onto the ground to avert wind-induced movements; they were also attached to the tree trunks using small metal fasteners at 1.30 m above the ground surface. All the bait traps were in operation for two hours (between 8 and 10 am), highlighting that it is a quick and easy method of sampling ants (Baccaro et al., 2011), and the capture methodology followed that of Bernardes Júnior et al. (2020). The ants were gathered after this exposure period to the baits and transported for screening and identification to the laboratory. The ants were carefully identified employing the taxonomic keys reported by Baccaro et al. (2015); the specimens were also compared with specimens deposited at the Entomology Laboratory collection of the State University of Goiás.
Ants were classified into functional groups (guilds) according to the procedure proposed by Silvestre et al. (2003). Such a categorization into guilds gives precise data regarding the ecological functions, including, diet, nesting location, foraging competence, and related habitats (Castro et al., 2020; Fichaux et al., 2019; Tiede et al., 2017). The ant collections were performed once during three distinct stages of land occupation with the crops (corn and soybeans in the flowering period and when they were not raised in the matrix).
Litter sampling
Immediately after collecting ant traps in the SSF, litter was collected on the ground in plots of 1.0 x 1.0 m in the same places where the traps were installed. Stones and soil were removed from this material manually using 2.0 mm sieves. After drying naturally for 10 days, it was weighed. No litter sampling was performed in the matrices because in some areas of the matrices litter had been incorporated into the soil through harrowing, before planting the soybean and corn crops.
Statistical analysis
The species richness of the ants occurring in the different fragments surrounding the matrix and the time of their collection were verified using the Jackknife 1 estimator. The association between the ant richness and metric variables of fragment size, litter quantity and edge effect were studied through regression analysis. The IndVal (indicator value) of the individual species, developed by Dufrêne and Legendre (1997), was also employed. The data on species richness and frequency of occurrence were summarized using the Non-Metric Multidimensional Scaling (NMDS) ordination technique. In this technique, the samples are sorted based on similarity concerning the species composition and occurrence frequency in each habitat. Testing for significant differences between the samples was done using the analysis of factorial similarity (ANOSIM). For both analyses, the Jaccard index was used to ascertain the degree of similarity between the samples. In this study, the degree of specificity of a given species for a habitat combined with its fidelity within that status was measured by the percentage of its occurrence, according to McGeoch et al. (2002). Statistical analyses were conducted in R software (R Core Team, 2022), using the vegan (Oksanen et al., 2025) and iNEXT (Chao et al., 2014; Hsieh et al., 2022) packages. In addition, the ggplot2 package (Wickham, 2016) was used to represent the results.
Results and discussion
The highest ant species richness was observed in the soil than in the trees, with greater richness 1 during the dry season (including the period lacking the crop), with likely food shortage and probably more exploitation of the baits in the traps (Figure 1 A and B). Data on ants captured on the ground and tree extracts from Semideciduous Seasonal Forests (SSF) and matrices in the soil surrounding SSF fragments are presented in Table 1. In total, 43 morphospecies were captured.
Delabie and Fowler (1995) reported that in the tropical forests, 50% of the myrmecofauna were related to leaf-litter (on the soil surface), which is also confirmed in our study. Widenfalk et al. (2016) highlighted that environmental filters such as temperature and humidity variations, even over short distances in the litter, significantly affect the structure of soil insect communities. Between the litter and arboreal gradients, a greater range of variation of these environmental filters, including luminosity, can be considered; these filters possibly influenced the significant changes in the composition and richness of ant species found in our study.
According to Hoenle et al. (2023), Oliveira et al. (1995) and Tews et al. (2004), monoculture matrix environments are simplified, and generally, greater species richness is associated with greater habitat heterogeneity. According to Suguituru et al. (2011), largely anthropized environments (such as the matrices in our study) are unlikely to conserve most primary forest species, such as groups of specialist predators and nomadic species, which show specialized biology. Furthermore, Majer et al. (1984) noted a correlation between the ant species richness and that of the plants, meaning the recolonization of the regions anthropized by particular plants is followed in sequence by the recolonization of the ant species. Besides the heterogeneity, Battirola et al. (2005) and Büchi and Vuilleumier (2014) pointed to the fact that the ability of different species to coexist in the same habitat is dependent upon the ecological breadth that each of them possess within the community.
In the present study, no significant relationship was found between the ant richness (observed and estimated) and fragment size of the SSF (observed: F = 0.09826; Adjusted R-Squared = 0.220; p-value = 0.7699) (estimated: F = 1.402; Adjusted R-Squared = 0.07436; p-value = 0.302). This result contrasts with the basic conceptual model that more extensive fragments support a larger number of species (MacArthur & Wilson, 1967). It is important to point out that in the region covered by the present study, there were no areas of SSF considerably larger in size than those shown here, which may have governed the relationship between richness and fragment size. An identical observation can be considered for the non-edge effect seen in our study for the soil ants (observed: F = 8.11; Adjusted R-squared = 0.0547; p-value = 0.2014) (estimated: F = 3.613; Adjusted R-squared = 0.1443; p-value = 0.0897) and tree ants (observed: F = 3.75; Adjusted R-squared = 0.1642; p-value = 0.0748) (estimated: F = 3.794; Adjusted R-squared = 0.1664; p-value = 0.0735). The smallest fragment (F3) in the present study was only 4.22 ha, in which similar or even greater ant richness was found than that identified in the most significant fragments (Table 1). This also implies that when many ant species coexist in a relatively small fragment like this, it is not related to the territory size alone. Highlighting the structural attributes that are easily measurable is important, although they may not fully reflect an ecosystem's dynamic properties that reveal its current performance (Mahon et al., 2023; Palmer & Febria, 2012). However, with the evolution in knowledge about habitat loss and fragmentation, the importance of the matrix for maintaining species richness and abundance in the fragments was widely recognized (Laurance, 2008; Rodrigues & Nascimento, 2007). According to Tavares et al. (2019), there is a strong interaction between habitat remnants and the other units that make up the landscape.
No significant relationship was identified in the present study between ant richness and litter quantity in the fragment (observed: F = 1.743; Adjusted R-squared = 0.1294; p-value = 0.2572) (estimated: F = 1.726; Adjusted R-squared = 0.1269; p-value = 0.2592). Mean and standard deviation of litter mass present in the SSF at the three different times of ant capture were: SSF when in a matrix occupied by soybeans (0.80 ± 0.46 kg), corn (0.71 ± 0.36 kg), without agricultural cultivation (1.13 ± 0.46 kg). No litter sampling was performed in the areas surrounding the fragments, because in some areas of the matrices, litter had been incorporated into the soil through harrowing, before planting the soybean and corn crops. In general, higher quantities of litter suggest greater richness of the edaphic ants in the forest fragments (Bernardes Júnior et al., 2020).
Two dominant guilds were evident in the three collection environments, namely, the soil dominant (SD) and omnivorous (O) ones (Figure 1 B and Table 1), predominantly represented by the genera
The composition of the ant community in SSF fragments is different from that observed in surrounding matrices (ANOSIM, at p < 0.001) (Figure 2 A). The species rarefaction curve by sampling units (Figure 2 C) highlights a significant and greater richness of ant species in SSF areas. Particularly for epigeic ants in matrices, the composition of the ant community is different for the different moments of occupation with crops; however, no significant differences in species richness are observed (Figures 2 B and D). Within a community, the arrangement of the ant species is affected by the distribution of resources they exploit (Bernardes Júnior et al., 2020). In the matrices, in particular, the resources showed variations in terms of quantity, type, and temporality, which is the reason for the compositions of the different soil ant groups when their habitats were planted with soybean and corn or left without any agricultural cultivation.
According to Palmer and Febria (2012), the results of this study reiterate the significance of focusing on the presence or absence of particular species, as well as monitoring all the alterations in the assemblage and the way they affect the food chain and other processes of the ecosystem. The present study did not address the effects of climatic variations on the ant community structure. However, as reported by Costa et al. (2018), the climatic variables exert a strong influence on the foraging pattern of the ants, implying that they can affect the visitation of ants to the attractive capture baits; also, it is important to highlight, the characterization of the plant community of the SSF fragments was not the object of the study, they certainly influence the dynamics of the ant community.
Using IndVal,
In the present study, none of them simultaneously indicated the different habitats, thus implying that they are reliable bioindicators. Species strongly related to conserved natural areas, just like the SSF fragments in this study, need to remain focused on the conservation and monitoring measures, according to Nascimento et al. (2007).
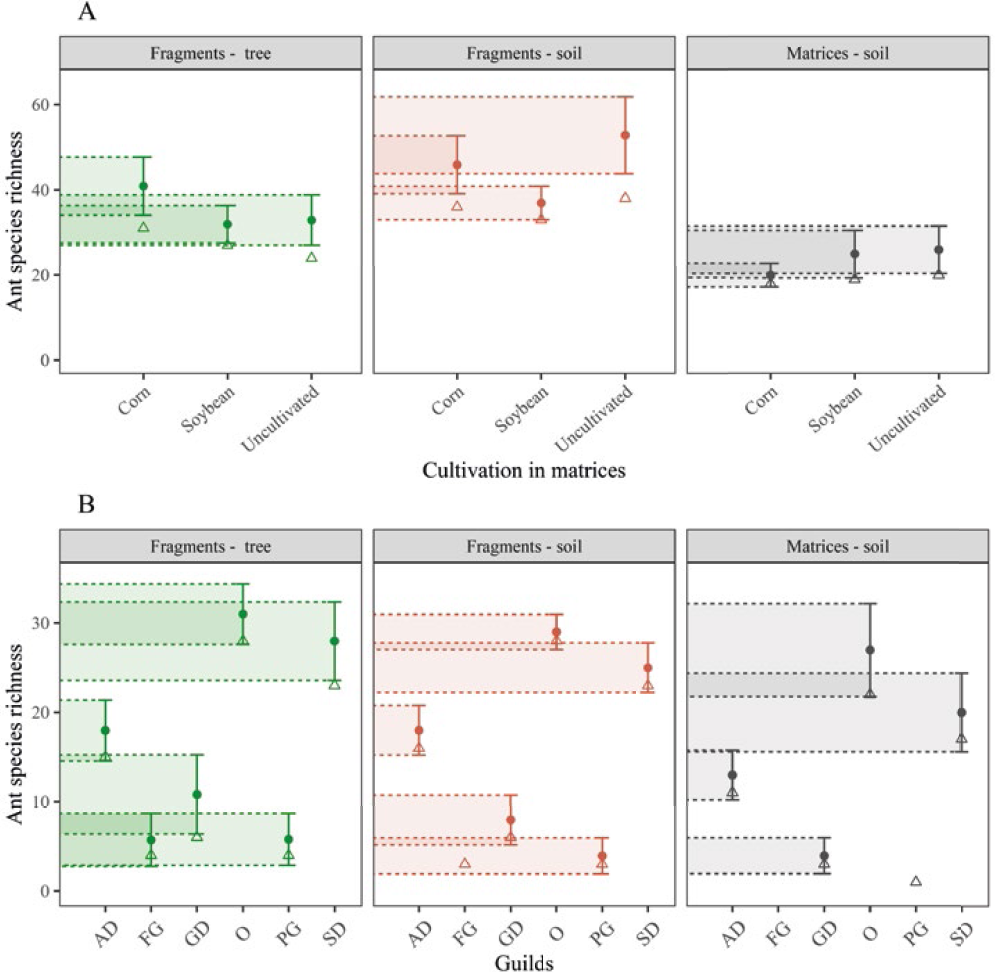
A: Estimated richness of ant species (•) and observed richness (A) in the seasonal semideciduous forest (SSF) fragments and the matrices that surround them. B: Comparison of the ant species richness in the different environments by guilds, which included CF = Cultivators of fungi; SD = Soil dominants; O = Omnivorous; AD = Arboreal dominants; GD = Generalist dominants; Soil dominants; PG = Predatory generalists; FC = Cultivators of fungi. The bars indicate the confidence interval as given by the estimator Jackknife 1.
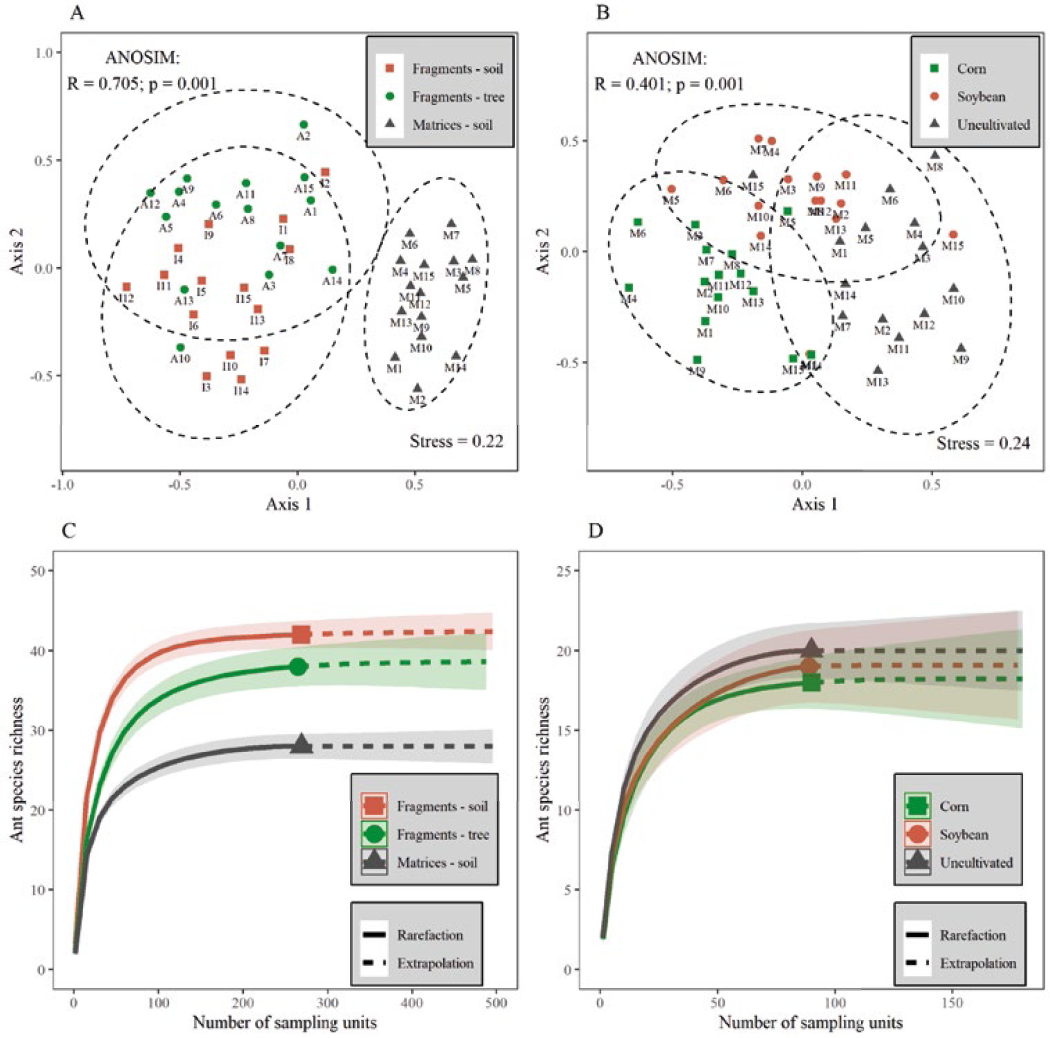
A: Scores ordered according to the Non-Metric Multidimensional Scaling (NMDS), drawn from the data on the composition of the ant morphospecies, noted at the points sampled in a fragment area of a Seasonal Semideciduous Forest (SSF) and its surrounding matrix. The dotted lines indicate the confidence ellipses for the treatments evaluated, which were later compared by applying the similarity test (ANOSIM; p <0.05). A1 to A15 indicate the collection points in the trees; 11 to 115 represent the collection points on the soil within the fragment, while points M1 to M15 refer to the collection points on the soil within the matrices. B: Ants from the soil in matrices, recorded at the sampling points, during the collection periods cited, harvests (soybean and corn), and without cultivation. The M1 to M15 represents the total number of collection points. C and D: Estimated ant species richness with rarefied and extrapolated samples (iNEXT), with 95% confidence intervals (shaded areas).
Ants captured in the soil and tree extracts from the Seasonal Semideciduous Forests (SSF) and Matrices in the soil surrounding the SSF fragments.
Guild or specialization: SD = Soil dominants; GP= Generalist predators; AD = Arboreal dominants; O = Omnivores; CF = Cultivators of fungi. F1 to F6 = Fragments of investigated SSF; M1 to M6 = Matrices of the respective investigated SSF fragments.
Individual indication value (IndVal) of the ants sampled using attractive baits, from the interior soils of the Seasonal Semideciduous Forest (SSF) fragments and from the interior soil of the matrices of these fragments.
Conclusions
Our results highlight significant differences in species composition and richness in specific environments and strata of SSF fragments and surrounding matrices. Greater ant species richness was found in the SSF fragments than in the surrounding matrices, which supported the crops. Although no significant relationship was identified in this study between the ant richness and metric variables like fragment size, quantity of litter, and spatial disposition of the ants (edge effect), the findings suggest that the structural composition of the ant community and a few species of the community can act as reliable environmental bioindicators. Our results, obtained relatively easily and quickly, may guide decision-making related to Cerrado conservation since ants are an ecologically dominant group that provides several ecosystem functions and is a bioindicator of changes affecting wider groups of organisms.
Origin and Funding
The present work originated from the line of research in genetic resources and biodiversity of the Post-Graduate Program in Plant Production at the Universidade Estadual de Goiás.
Author's Contribution
first, second and sixth authors. conceived the idea of the study. First, second and fourth authors helped in organizing the experimental material for further recording the data designed the experiment. Third, fourth, fifth and sixth helped in data analysis, supported in write up and review the manuscript.
Conflict of Interest
The authors have no conflicts of interest.
Footnotes
Acknowledgements
The authors express their gratitude to the Coordination for the Improvement of Higher Education Personnel (CAPES) for the financial assistance through the Social Demand Program (PDPG Emergencial de Consolidação Estratégica dos Programas de Pós-Graduação - Processo n. 88881 849302/2023- 01, Programa PDPG-Consolidação-3-4) and to the reviewers of the final manuscript for their comments and suggestions.
