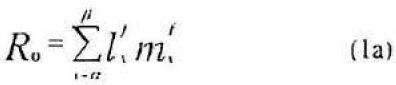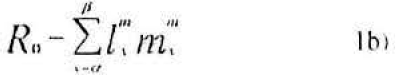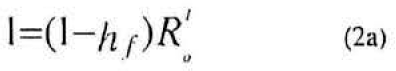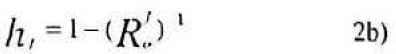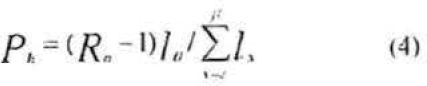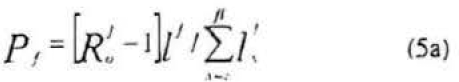Abstract
A mass rearing technique for the parasitoid C. stephanoderis on (Coffee Berry Borer) CBB immature stages was developed using the Cenibroca artificial diet (Portilla 1999). Evaluations were based on the number of emerging adult female and male CBB and its parasitoid progeny. 750 ml of Cenibroca diet was poured onto a rectangular plastic tray and divided into 1.500 small pellets using a grille squeezed into it while still hot. Using 55-60% diet moisture content at 27°C and 85% RH the emergence of CBB progeny adults started at day 26 after the date of inoculation of the borer adult female. Approximately 59.100 CBB stages in 750 ml of artificial diet (per tray) were obtained 40 days after diet infestation. In the parasitisation process a total of 2.500 immature stages were placed in a plastic rearing container, in which 60 wasps were placed. 1.500 female and male wasps per container were obtained between day 30 to day 45 after inoculation of female parasitoids.
Introduction
The use of mass reared insects in pest management dates back to the 1920s, when parasites were cultured on a large scale and released for establishing or augmenting biological control programs (Van den Bosh and Messenger 1973). Nowadays, a wide variety of insects are produced in millions and used as hosts and control agents in various pest control programs. Some examples are the boll weevil, Anthonomus grandis grandis; boll worm, Heliothis complex; pink boll worm, Pectinophora gossypiella; codling moth, Cydia pomonella; housefly, Musca domestica; cabbage looper, Trichoplusia ni; fall army worm, Spodoptera frugiperda; European corn borer, Ostrinia nubilalis; tropical fruit flies; mosquitoes; ladybird beetles; and several parasites (Singh and Moore 1985ab).
Early efforts on insect rearing have been developed to provide more efficient rearing methods (King and Leppla 1984, Knipling 1979, Singh and Moore 1985ab). However, there continue to be many problems, which remain to be solved. i.e. excess production sometimes must be discarded to reduce the colony size, and other times the colony is too small (Hsin and Wayne 1988). Thus, as evidenced by recent literature (Carey and Vargas 1985, Vargas et al. 1985 Whistlecraft et al. 1985, Plant 1986), more efficient rearing methods are always being sought (Hsin and Wayne, 1988).
Mass production of most parasitoid species requires host rearing. Therefore, one of the broad objectives for production is to optimally balance the number of host available for parasitisation with the number of adult parasitoid females held for renewal (Carey et al. 1988). In addition to specifying diet, rearing densities, and related rearing conditions, a properly designed rearing program must also specify the discard age and harvesting rates (e.g. Carey and Vargas 1985).
Mass rearing of the parasitoid C. stephanoderis is performed using its natural host (Villacorta and Barrera 1996). CBB has been reared by different systems: a) using coffee fruits naturally infested (Mendel 1934, Abraham et al. 1990); b) in coffee fruits artificially infested with the host (Barrera et al. 1991, Delgado and Sotomayor 1991), c) in parchment coffee artificially infested with the borer (Benavides and Portilla 1991, Portilla and Bustillo 1995 and Bustillo et al. 1996), d) in green coffee bean (without hull) artificially moistened and infested with borer; and e) in artificial diet infested with CBB (Brun et al. 1993, Villacorta and Barrera 1996). Based on results from the studies listed above the parchment coffee bean method has been the must successful for large-scale culturing of CBB and its parasitoid.
In Colombia, since the arrival of the CBB in 1988, CENICAFE has run many research programs with particular focus on management of the borer population using biological control. The parasitoid research program aims to develop methods for mass rearing of the parasitoids, to evaluate their behaviour and establishment in the coffee area plantations, and to study its compatibility with any other method of control (Orozco 1998). Colombia has been the only country that has achieved a reliable mass rearing system for this parasitoid using the parchment coffee as rearing medium (Benavides and Portilla 1991). Firstly, rearing was developed on a small-scale level that was used to release on local farms (Portilla and Bustillo 1995). Secondly, it was done at a semi-industrial level where the wasps were produced in co-operation with private industry (Bustillo et al. 1996).
At present, there are six laboratories producing these parasitoids in Colombia, five are privately owned. The parasitoids produced have been used as part of an aggressive program of introduction in most of the coffee production areas infested by the CBB (Bustillo et al. 1996).
Benavides et al. (1994) mentioned that parasitism by C. stephanoderis depends on population density of CBB, altitude, temperature and amount of parasitoids released. He found 29% parasitism in a plot at 1630 altitude and 19.9°C and 65% parasitism at 1080 altitude and 23.4°C but only immediately after release, percentages later declined. However, Salazar et al (1997) found similar parasitism and means stages per parasitoid (without significant difference) in plots with similar densities of CCB and greatly different amounts of parasitoids released.
Although of the parchment coffee method has provided a large number of parasitoids (approximately 1.400 million of parasitoids between 1994 to 1998), the wasps are expensive to produce (about 1000 wasp/1.5 U.S.$) (Orozco 1999).
Hence, this study was carried out to develop an inexpensive method for rearing large numbers of these insects. Life table statistics of CBB and C. stephanoderis were determined and, a conceptual model and method for mass rearing CBB and its parasitoid on the Cenibroca artificial diet can be developed.
Materials and Methods
This study was carried out in the entomology laboratory facilities of CENICAFE in Colombia. The colonies of H. hampei and C. stephanoderis were rearing in controlled temperature environmental chambers and in constant temperature rooms. The relative humidity was controlled by saturated solutions of salt inside a container (26.5 g of KCl/100 ml of water) (Winston and Bates 1960)
Facilities and equipment required
Diet preparation and dispensing procedure
The rearing containers were designed to improve the handling preparation of the diet and save time in insect manipulation of the insects. They consisted of a rectangular plastic tray 50 x 30 x 2 cm with 6 lateral perforations of 1.5 cm diameter (3 per larger lateral side), that were made in order to avoid high humidity. The containers have inside a removable grill, with 1.500 divisions 0.85 x 0.85 x 10 mm that divide the diet into small pellets.
Dried green coffee bean of 12% moisture content were ground using a Provat Emmerich miller, which was calibrated to obtain a 2 mm coffee particle size. The ground coffee was autoclaved for 20 min at 120°C and 15 lb pressure. The industrial agar and water mix was also sterilised using an autoclave.
Sugar (8 g), casein (15 g), ground coffee (120 g) and powdered yeast (15 g) were combined and partially mixed and tipped into the blender together with melted agar (850 ml of water and 10 g of industrial agar). The mixture was blended for 10 min to achieve complete mixing. When it had cooled to 50°C, the nipagin (1 g), benomil (2 g), alcohol (75%) (10 ml) and Wesson salt (0.8 ml) dissolved in sterilised water were added. Figure 1 shows the dispensing procedure of the diet. Thus, after a final mixing the completed diet (750 ml) was poured into rearing containers for a depth of 5 mm. The diet then was divided into 1.500 small pellets using the grille squeezed into it while still hot. Each pellet was approximately 0,5 ml equivalent to 0,47 g after it is dried. Amounts greater than 0,5 ml waste diet material and smaller amount were found to dry out too rapidly.

Diet dispensing procedure showing the dividing process of the diet after dispensing.
The trays of solid diet were dried at 50°C for 12 to 15 hours in a heater until the diet reached 55-60% of moisture content. To analyse moisture content, a HG53 moisture analyser Mettler-Toledo was used. The diet that was not immediately infested, it was held for 10 to 15 days at room temperature. The trays were covered tightly with cling film to prevent desiccation.
The diet preparation room had to be scrupulously clean. Equipment, glassware, and rearing containers were washed with hot soapy water, disinfected with anti-microbial agent and covered until used.
Rearing and colony maintenance
Establishment of CBB colony
Coffee berries naturally infested in the field were collected from CBB infested plots. Benomil solution at 1,5% concentration was sprayed on the berries in order to prevent excessive microbial contamination. This material were placed in a tray 100 x 100 x 5 cm, with a wood frame and metallic mesh base that could hold approximately 20 kilograms of infested coffee berries. The tray was located inside a cage 100 x 100 x 100 cm, which was constructed with a wood frame and white nylon walls. The infested material was kept for four weeks to allow the pupae and larvae to become adults at a mean temperature of 25°C. 30 days later the infested material was sprayed with water and the room temperature was increased to 32°C using a electric heater to stimulate the emergence of CBB adults (Baker et al. 1992, Portilla and Bustillo 1995). The emerged adults, after a short flight, tented to land on of the nylon cage wall. This habit facilitated the collection process.
In order to renew the CBB colony, every six months new field collections were carried out.
Diet inoculation
Sodium hypochlorite solution at 2,55% concentration was sprayed on to adults to disinfect them, and then rinsed using sterilised water. Approximately 2000 active borers per rearing container were used to infest the diet pellets. These borers were sprinkled uniformly on the diet surface (Fig. 2).

Inoculation of the diet.
Development and collection of CBB immature stages
The trays with infested diet were fixed one on top of the other to reduce desiccation. These trays were placed in a controlled temperature environmental chamber at 23°C for 3 days. This temperature was chosen because the CBB had a low flight response needed to encourage establishment on the diet pellet.
They then were transferred to a constant temperature room at 27°C for 35 to 40 days (immature development room). Periodical examination of the diet rearing containers was done on 15 day and then every 8 days for 40 days, with 25 diet pellets randomly selected, dissected and their contents recorded. The rearing containers were kept under total darkness at all times.
After 25 days half the infested diet trays were transferred to the collection room at 27°C. The content of the rearing containers were placed in racks 35 x 25 x 3 cm with metallic mesh base (30 mesh screen), that could hold about 2000 infested diet pellets, which did not permit females of CBB to pass through. The rack was located inside a hermetically sealed plastic container 35 x 25 x 12 cm (Multiplasticos) to prevent desiccation of the diet pellets or fungus contamination. These infested pellets were left for 5 days in order to obtain the CBB immature stages, which collected at the bottom of the plastic container. The immature stages were collected once a day and used for establishing the C. stephanoderis colony.
The other half of the infested trays were left in the immature development room for approximately 40 days; these then were transferred to a collecting room. The content of the infested trays were placed in a cage with similar characteristics as described above in the adults collecting process and the same steps for adult collecting were also followed. However, to increase the relative humidity water was sprayed on the nylon walls cage. The collected adults of CBB were used to increase the stock colony and for experiments.
Establishment of C. stephanoderis colony
Parasitoids from the Cenicafe stock culture were used for establishing the colony. 60 newly emerged wasp females were placed with a total of approximately 500 eggs and early larvae and approximately 2000 second instar larvae, prepupa and pupae, which were sprinkled on to the paper lined surface in square plastic rearing containers 12 x 12 x 1.5 cm (Multiplasticos). The immature stages were obtained from the CBB immature separating process previously mentioned. The rearing containers were placed inside chambers at 27°C where they were left undisturbed for 25-30 days.
Adult C. stephanoderis collection
On day 25 the paper lined with the adhered wasp cocoons was removed and located in a transparent plastic cage. The back wall of the cage was oriented toward a bright light source such as a desk lamp at room conditions (27°C, 75-85% RH). A positively phototropic response caused the wasps to fly towards the light, facilitating the collecting process. Adult collection was realised using a pooter, which was connected to an electric car vacuum cleaner. This collection was done once a day for 20 days. The collected adult wasps were used to maintain the colony and for experiments, which were carried out under laboratory and field conditions.
Data analysis
Reproductive potential and harvest rates
Insect quality was monitored continuously by keeping records of average oviposition and percent hatch. Routine observations of immature stage development and mortality and adult behaviour also were made. With the results obtained from this monitoring the basic population parameters were calculated for CBB and C. stephanoderis such as gross maternity (M), survival (L), net reproductive rate (Ro), finite rate of increase (λ), intrinsic rate of increase (rm), mean generation time (T), double time (DT). Definitions, symbols and equations for these parameters are described in Carey (1993) and Portilla (1999).
The harvest rates of CBB and its parasitoid were calculated considering the values of demographic parameters listed above with parasitoid males and females treated separately (Care, et al. 1988). Let lfx and lmx denote survival to age x of parasitoid females and males, respectively, and mfx and mmx denote a female parasitoid's production of female and male offspring at age x, respectively.
Therefore the per female production of daughter, denoted Rfo and sons, denoted Rmo, from their age of first reproduction to their discard age (b) is given as
Hence the harvest rate, hf of female parasitoids required to confer zero population growth in their population must be the solution to the equation
and the primary sex ratio (male: female), denoted S, is
The per female production rate of the host target stage, denoted Pt is
Where α denoted the age at which the target stages is harvested and β represents eclosion age. The interpretation of the equation 4 is this: The term Ro -1 gives per female number of hosts to be removed (harvested) for conferring exact replacement. Therefore this term multiplied by lα−1 yields per female number of female host age 0 to be removed for exact replacement. This product divided by the number of female-days in the standing cohort aged α to β (i.e., the denominator in equation 4) yields the number of females aged 0 to be removed for exact replacement (Carey 1993).
Following the same reasoning, the per capita adult female parasitoid rates for production of harvestable daughters and sons, denoted Pd and Ps respectively, are
Data on developmental time of immature stages of CBB was subjected to an analysis of variance (ANOVA) by the general linear model (GLM) procedure of SAS (SAS institute, 1988). Differences between least square means for all variables (calculated by the GLM procedure) were evaluated by the t-test.
Results
Rearing and colony maintenance
Development of CBB immature stages
Immediate attempts to feed by CBB female were indicative of the suitability of the physical consistency of the medium provided. In most rearing containers, egg laying started within the first 5 days after the infested diet container were transferred to the immature development room (27°C). The eggs were oviposited singly or in pairs on the surface grille. These eggs hatched and the larvae made a very superficial tunnel into the diet (< 1 mm). Every stage (eggs, larvae, prepupae, pupae and adult) developed on the diet surface (Fig. 3).
0.5 ml of Cenibroca diet pellet 30 days after infestation, showing the externa development of CBB immature stages.
This development behaviour was considered an important factor, which facilitated separating the immature stage process from the diet.
Figure 4 shows the trend in total brood production (gross fecundity) per female found in 0.5 ml of Cenibroca diet at various times after infestation. Eggs were found from 3 days after infestation (DAI). The presence of the first larval stage appeared at day 9, the second larval stage at 12 days, prepupae at day 16 and adults at day 22. Adult male offspring appear at the same time as female offspring. In the analysed samples the number of eggs per clutch was similar and in most of them only one clutch per borer were found.
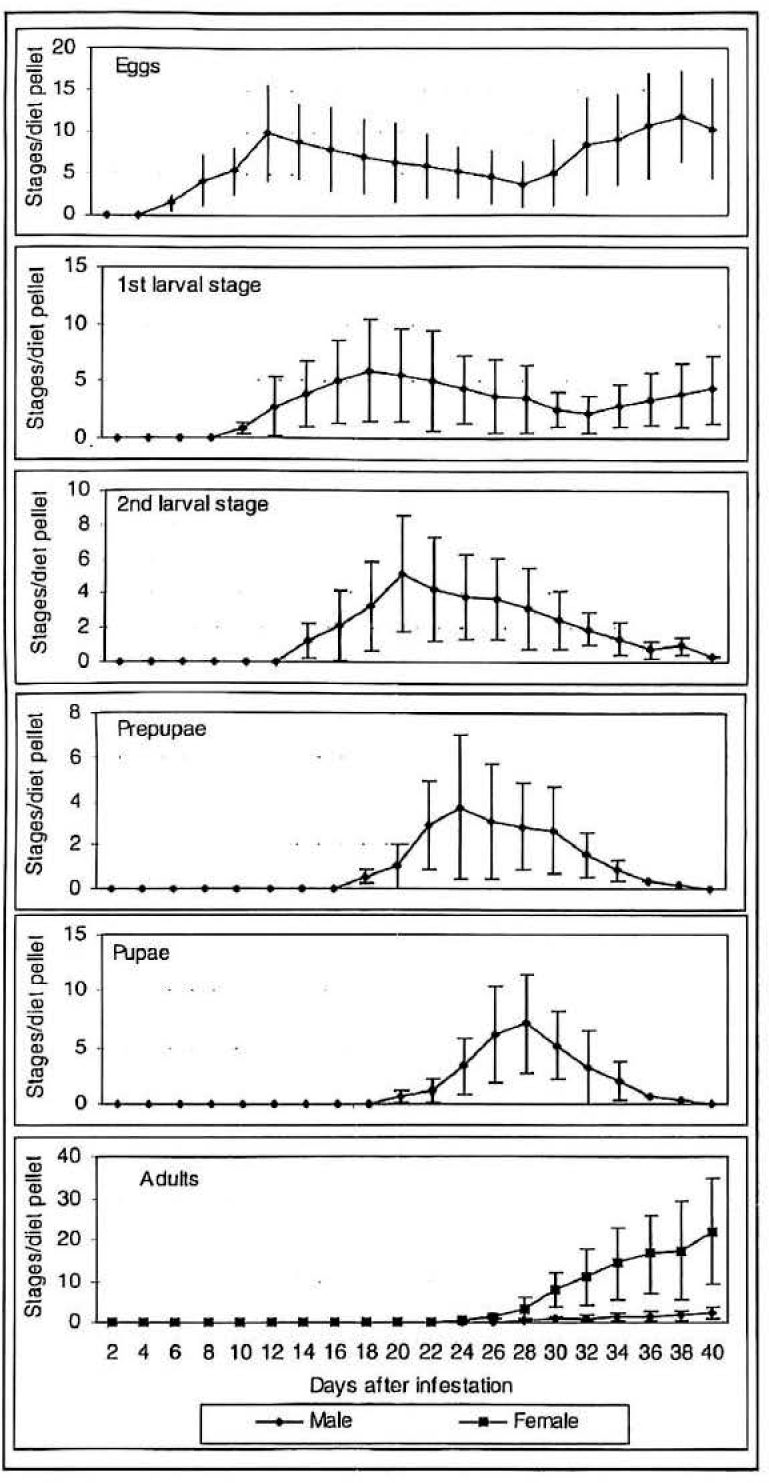
Mean number of stages per borer per 0.5 ml of Cenibroca artificial diet found at various times after infestation (27°C, 85%RH, 55-60% diet moisture content). 95% confidence limits of the mean (sample size n = 25 diet pellets per evaluation time).
Stage structure within the production facility
Distribution of the pre-adult population rose sharply from the end of second week of reproduction (at 14 DAI) and until the end of the third week (at 22 DAI). This curve then remained at high levels until the end of the fourth week. Subsequently, this declined sharply over the 5-week period (Fig. 5). According to this result the percentage obtained between 22 to 28 days provided the harvesting period. However, on day 26 the adult distribution began to rise and the early stage distribution followed decreasing. The presence of the adults obstructs the immature stage separation process and the presence of early stages provides suitable host feeding in parasitisation. Hence, the advisable harvest period was considered between 23 to 26 DAI.
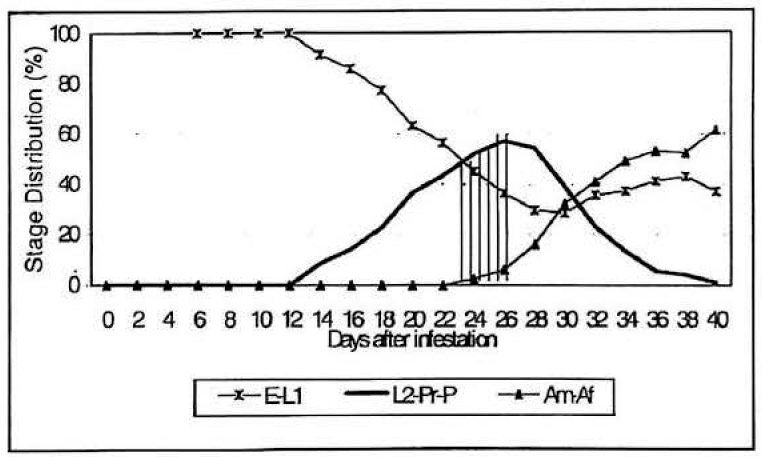
Percentage of the stage structure population for CBB on Cenibroca artificial diet at various times after infestation. E-L1 = Early stages of CBB, L2-Pr-P = Pre-adults, Am-Af = Female and male adults of CBB.
The percentage of the total brood colony of the six stages (eggs (E), first stage larvae (L1), second stage larvae (L2), prepupae (Pr), pupae (P) and adults female and male (Am-Af)) for CBB found at various time after infestation are presented in Table 1. Approximately 59.100 stages were obtained in 750 ml of Cenibroca diet 40 days after infestation (750 ml of diet / 2.000 CBB rearing container).
The presence of eggs and first stage larvae throughout the reproduction period facilitated the host feeding supply, which was carried out in the establishment of the C. stephanoderis colony.
Immature stages of CBB collection
Figure 6a shows the total production found in trays at 22, 24, 26 and 28 days after infestation (DAI) and the harvested amounts of immature stages of CBB obtained from these infested trays. 67% (approximately 20.000 immature stages of CBB) from the whole population was the greatest numbers harvested stages obtained from the trays at 22 DAI followed by 24, 26 and 28 DAI with 65% (19.658 CBB stages), 61% (18.886 CBB stages), and 60% (17.649 CBB stages). The harvested population decreased as pupal population rose, because it was difficult for pupal stages to pass through the mesh.
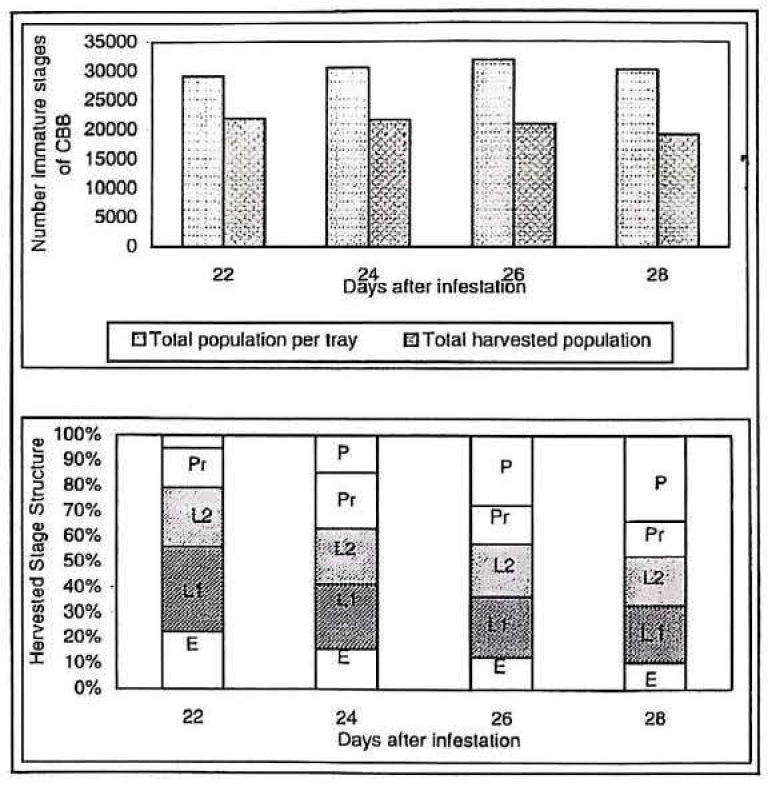
Immature stage population of CBB found in trays 22, 24, 26 and 28 days after infestation, and total harvested population obtained from them. Stage structure (%) E = eggs, L1 = first stage larval, L2 = second stage larval, Pr = prepupae, P = pupae.
Figure 6b shows the harvested stages obtained from the rearing containers. Although, the greatest amount of the harvested population was found in tray 22 DAI, it was not considered as the optimal one, because 56,1% were eggs and first stage larval, suitable stages for feeding but not for oviposition. In contrast, 28 DAI contained the smallest amount of harvested population with its percentage structure of immature stages of 33,1% for early stages and 66,9% for pre-adult. From the latter population, 33,3% were pupae. From trays at 24 and 26 DAI were obtained the highest population percentage of prepupae and the second larval stage.
According to results shown in Table 1 the highest parasitation percentage on prepupae, the harvested stage structure obtained in trays 24 and 26 DAI was considerate the optimum for the immature collection procedure. The percentage of second stage larval is important, since these will become prepupae within a few days after parasitation.
Despite the pupae stage being the one preferred by C. stephanoderis for oviposition, it was not convenient to provide them because in the parasitisation process a high percentage of them became adult.
Percentage of the standing population produced for coffee berry borer on 750 ml of Cenibroca artificial diet at various times after infestation
Adults CBB collection
The external development behaviour facilitated the immature stage collection. However, the new generation adults had a good condition for tunnelling into the diet (35-40% diet moisture content), making it difficult to harvest adult. Hence, to stimulate the emergence of the adults, spraying water and an electrical heater were used in order to increase the humidity and temperature.
In spite of these difficulties approximately 30.000 CBB adults were collected from a trays 35 DAI (750 ml of diet/ 2.000 CBB adults/ tray). This amount was obtained by a harvesting period once per day during 15 days (Fig. 7). The biggest harvested population was obtained at 4-5 days after infested diet pellets were placed in the collecting cage (see materials and methods). The adults emergence distribution rose gradually during the first 3 days, it then increased sharply the next two days. The emergence declined drastically after 5 days. Thus, collecting emerging adults after the 8th day (43 DAI) is probably not cost effective.
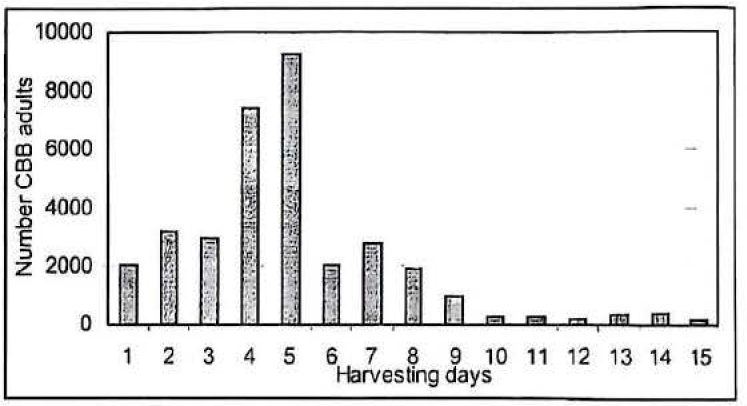
Harvested adults of CBB from 750 ml of Cenibroca artificial diet 35 days after infested.
Adults C stephanoderis collection
Once adults of C. stephanoderis were placed into the rearing containers these wasps begin exploring with their antennae for suitable host available for feeding or oviposition. The host selection procedure started by walking around the host, then grasping the host with her mandible and extending her ovipositor to sting it. After total paralysis of the host, which was achieved after various stinging, she finally laid her single egg in the host. Seemly, the provided amount of hosts and the area of the container did not allow competition between them for host feeding or host oviposition attacking. Thus, once the host was parasitized she grasped it with her mandibles and grouped it with other parasitized host making her own clutch (Fig. 8). However, in some containers casually only 1 or 2 big groups some times were found.

Grouped cocoons of C. stephanoderis in the rearing containers.
About 10.100 parasitoids were the total offspring obtained from 480 parasitoids, which were placed with 20.000 immature stages that were collected from a tray 24 DAI (Fig. 9). This figure shows the amount of adult wasps emerged during the next 20 days. This distribution rose gradually during the first week and decreased in the second week, thus, this time was considered as the harvesting period. As in the CBB adult collection process, the production of wasps obtained after 14 days collection (38 DAI) is not practical.
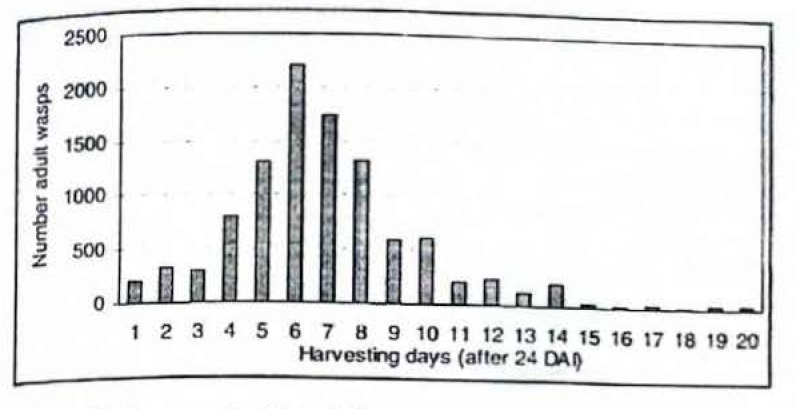
Harvested adults of C stephanoderis from immature stages of CBB developed on 750 ml of Cenibroca artificial diet.
Demographic parameters and harvest rates for CBB and its parasitoid
Table 2 presents the net reproduction (female offspring) by age for the coffee berry borer and the parasitoid C. stephanoderis and the associated harvest rates. f and hf. Therefore, from 5 to 30 days the net reproduction for C. stephanoderis increased by more than 6-fold from 4,62 to 28,76, while net reproduction for H. hampei increase proximately 21-fold from 0,67 to 14.37. The harvest rates give the numerical requirement for exact replacement in the CBB host and the female parasitoid, respectively. Thus, at 30 days both CBB and the parasitoids should be replaced.
Net reproduction, by age for H. hampei and C. stephanoderis associated with their harvest rates
Seven demographic parameters are summarised in table 3. Hence, using this rearing method these obtained parameter values express that the growth rate per generation of C. stephanoderis was of 28,8 female and 6.0 male wasps per newborn female in a mean time of 36.2 days. Its daily growth rate was 1.10 daughter females per female, so the population could double every 6,5 days. Whereas, the growth rate per generations of H. hampei was of 14.4 female wasps per newborn female in a mean time of 40,8 days. Its daily growth rate was 1.07 daughter females per female and its population could double every 10.2 days.
Life tables statistic for C. stephanoderis and H. hampei
The per capita adult female of CBB rates for production of harvestable daughters, was 1.12 (females and male per day) and the per capita adult of C. stephanoderis rates for production of harvestable daughters, Pd was 0.93 for female wasps and 0.19 for male wasps, Ps. Figures 10 and 11 give diagrammatic sketches of a 4 week production cycle for CBB and C. stephanoderis. The harvesting rates and demographic parameter values showed similarities between them.
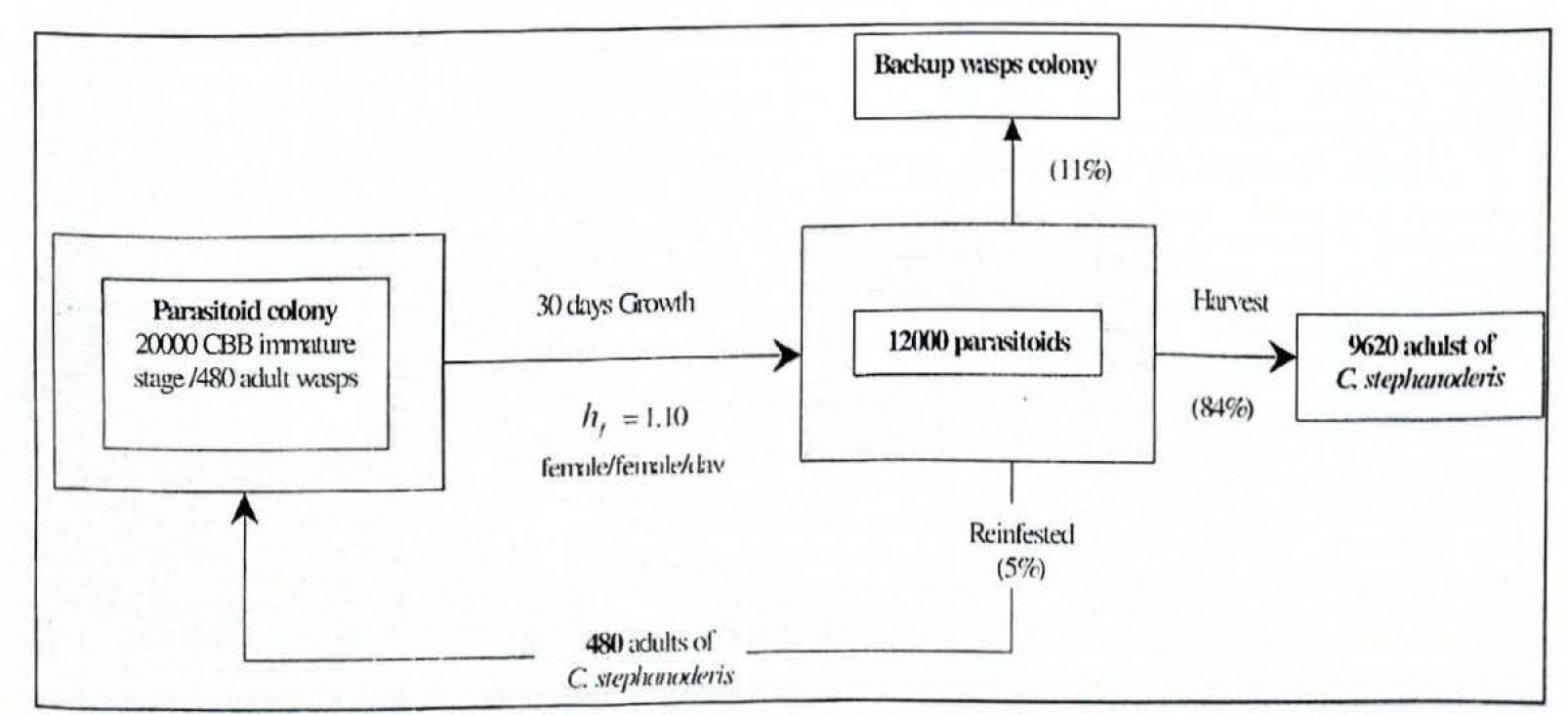
Diagram of production cycle for Cephalonomia stephanoderis.
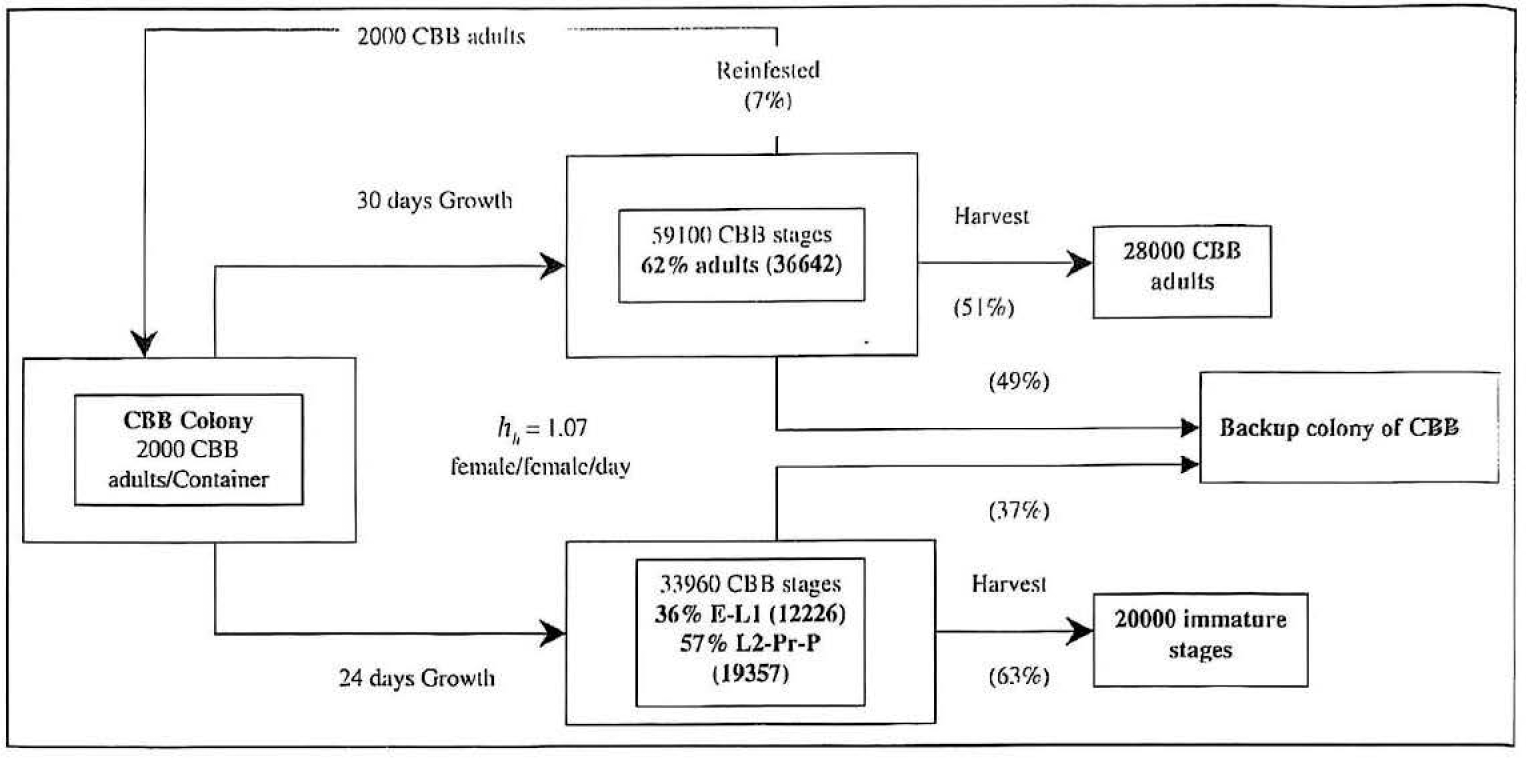
Diagram of production cycle for Hypothenenus hampei per rearing container. E = Eggs, L1= First stage larval, L2 = Second stage larval, Pr. Prepupa, P = Pupa.
The elements associated with the production of H. hampei and C. stephanoderis such as development of artificial diet for rearing CBB, colony establishment and maintenance, design of the rearing laboratory, research and development of techniques, quality control and production provided the construction of the conceptual model for rearing these insects using artificial diet (Fig. 12).
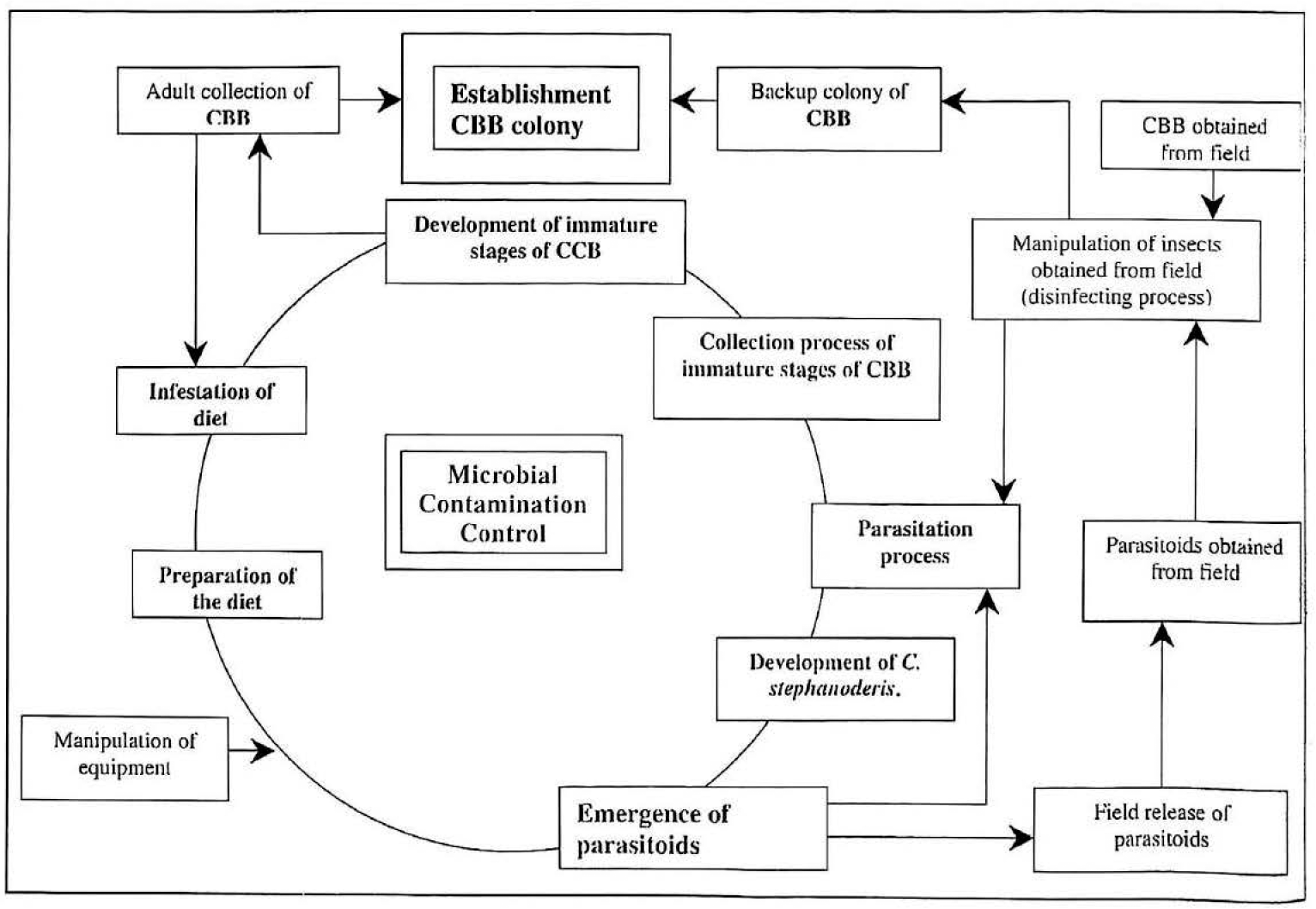
Conceptual model for mass rearing CBB and its parasitoid C. stephanoderis.
Discussion
Insect rearing management is the efficient utilisation of resources for the reproduction of insects of standardised quality to meet programme goals (Singh and Moore 1985ab; Krebs 1972; Mendel et al. 1987). Leppla (1984), Schwalbe and Forrester (1984) and Fisher (1984) have discussed organisations in insect management, insect production management and the role of the insectary manager respectively. In this chapter six major elements were associated with the production of H. hampei and C. stephanoderis. These are: development of artificial diet for rearing CBB, colony establishment and maintenance, design of the rearing laboratory, research and development of techniques, quality control and production.
On rate of increase and demographic parameters of CBB, the only available information comes from studies realised by Baker et al. (1992) and Ruiz et al. (unpublished) but carried out under field conditions. Gómez et al. (1992) and Villacorta and Barrera (1996) gave some techniques for rearing of the parasitoid on CBB using small glass tubes with Ecobrovill-160 artificial diet. These authors obtained approximately 3000 parasitoids per litre of diet. However, no one has estimated the population parameter for these species under laboratory condition using artificial diet.
In this work the demographic parameter values, which were calculated with the populations obtained within the rearing laboratory facilities expressed that the growth rate per generation of C. stephanoderis was of 28.8 female and 6.0 male wasps per newborn female in a mean time of 36.2 days. Its daily growth rate was 1.10 daughter females per female, so the population could double every 6.5 days. Whereas, the growth rate per generations of H. hampei was of 14.4 female wasps per newborn female in a mean time of 40.8 days. Its daily growth rate was 1.07 daughter females per female and its population could double every 10.2 days. The per capita adult female of CBB rates for production of harvestable daughters, was 1.12 (females and male per day) and the per capita adult of C. stephanoderis rates for production of harvestable daughters, was 0.93 for female wasps and 0.19 for male wasps,.
Hence, approximately 41,000 adults of coffee berry borer could be harvested per litre of Cenibroca artificial diet 40 days after infestation. Whereas, at 24 days after infestation approximately 34.000 immature stage could be harvested, which produced 20.000 parasitoids. Thus, the cost of 1000 parasitoids will cost U.S. $ 0.25 using the rearing method developed in this work. The cost of production to rear this parasitoid has been reported by some authors. In Mexico, to produce 1000 parasitoids costs about 5.4 dollars using coffee berries artificially infested (Barrera 1994). In Colombia 1000 wasps costs about 1.5 dollars (Orozco 1997). Villacorta and Barrera (1996) reported that the cost of the Ecobrovill-160 is about U.S$ 10 per litre, thus the cost of producing 1000 borers is about $ 0.25 dollars and the cost of 1000 wasps is $ 3.5 dollars.
The experiments and analysis carried out in this study must be considered as an important step towards a comprehensive understanding of the behaviour, feeding habits, life history and population parameters of these insects, which are an essential prerequisite to colony management both H. hampei and C. stephanoderis. However, it not resolve all problems associated with developing of this insects using artificial diet. Thus, recommend investigations on improvement of microbial control without adverse effects on the insect. To achieve the balance between optimum growth of the insect(s) and control of microbial growth would be test levels of feeding inhibitors. Determine the probable nutritional feeding stimulants for the insect and physical requirements for an acceptable diet that dose not affect the external oviposition behaviour but that provide the optimum reproductive potential of CBB should be studied.
Research into developing improved methods of mechanisation of the immature collection process must be carry out, which could reduce the current cost of this insect and allow a large increase in capacity. On the other hand I suggest carry out intensive laboratory studies on the bethylid Prorops nasuta and the eulophid Phymastichus coffea to adapted this rearing system in order to obtain others possible options for controlling CBB. This, because at present all this parasitoids are viable biological control and there are a mass rearing method to reproduce them.
To rear P. nasuta could be adapted the rearing method developed in this study using all steps: a) preparation of the diet, b) inoculation, c) recollection process of CBB immature stage and d) recollection process of CBB adults. Whereas, for P. coffea only the steps (a) and (b) could be used because this parasitoid needs only adults for its breeding. So, the production cost for rearing P. nasuta could be the same or probably higher than C. stephanoderis but for P. coffea it could be cheaper. This may be the next challenge for the future, to see how cheaply P. coffea can be produced and how effective it might be used in the field. In Colombia this parasitoid has been released but it is still too early to confirm its establishment (CABI 1997).
Footnotes
Acknowledgement
This was a collaborative project funded by National Federation of Colombian Coffee growers FNCC through the national research centre CENICAFE and the British Overseas Development Administration (ODA) through CABI Bioscience. I am especially grateful to all staff at CABI Bioscience for their help and the unlimited use of the facilities at Headquarters. Thanks are due to all of the CENICAFE team. Thanks are especially to Alex Bustillo for his scientific and technical contribution. Many thanks to my supervisors John Mumford and Dave Moore for their helpful criticism, understanding and knowledge through this study. I would also like to thank my advisor, Peter Baker, for his many useful comments and suggestions.