Abstract
Apoptosis, an active process of cell self-destruction, is associated with myocardial ischemia. The redistribution of phosphatidylserine (PS) from the inner to the outer leaflet of the cell membrane is an early event in apoptosis. Annexin V, a protein with high specificity and tight binding to PS, was used to identify and localize apoptosis in the ischemic heart. Fluorescein-labeled annexin V has been used routinely for the assessment of apoptosis in vitro. For the detection of apoptosis in vivo, positron emission tomography and single-photon emission computed tomography have been shown to be suitable tools. In view of the relatively low spatial resolution of nuclear imaging techniques, we developed a high-resolution contrast-enhanced magnetic resonance imaging (MRI) method that allows rapid and noninvasive monitoring of apoptosis in intact organs. Instead of employing superparamagnetic iron oxide particles linked to annexin V, a new T1 contrast agent was used. To this effect, annexin V was linked to gadolinium diethylenetriamine pentaacetate (Gd-DTPA)-coated liposomes. The left coronary artery of perfused isolated rat hearts was ligated for 30 min followed by reperfusion. T1 and T2* images were acquired by using an 11.7-T magnet before and after intracoronary injection of Gd-DTP-labeled annexin V to visualize apoptotic cells. A significant increase in signal intensity was visible in those regions containing cardiomyocytes in the early stage of apoptosis. Because labeling of early apoptotic cell death in intact organs by histological and immunohistochemical methods remains challenging, the use of Gd-DTPA-labeled annexin V in MRI is clearly an improvement in rapid targeting of apoptotic cells in the ischemic and reperfused myocardium.
Introduction
Apoptosis or programmed cell death, is an active process of self-destruction of cells and is associated with a number of disorders including neurogenerative diseases such as Alzheimer's disease, cerebral and myocardial ischemia, and organ rejection following transplantation. Apoptosis also seems to be common in advanced human atheroma and contributes to instability of atherosclerotic lesions [1]. In cancer therapy, the rate of apoptosis is directly correlated with tumor growth [2]. Thus, noninvasive detection and quantification of apoptosis might be a powerful diagnostic tool, both for monitoring the progression of the disease and for therapy.
Apoptosis is induced by disturbances in the local environment of the cells and is associated with characteristic morphological and biochemical changes, such as condensation of cytoplasmatic proteins, fragmentation of DNA, and molecular alterations in the cell membrane. One of the earliest events in apoptosis is the redistribution of phosphatidylserine (PS) from the inner to the outer leaflet of the cell membrane [3]. Expression of PS allows the removal of these cells by phagocytosis [4]. This behavior of PS makes it an attractive target for diagnostic imaging.
Several proteins with high specificity that bind tightly to PS were used to monitor apoptosis. One of these proteins is annexin V, a 36-kDa phospholipid-binding protein, which is an endogenous human protein that binds specifically to PS with nanomolar affinity (
Common disadvantages of nuclear imaging techniques in contrast to magnetic resonance imaging (MRI) are inferior spatial resolution and the lack of functional information such as flow, perfusion, diffusion, permeability, vascular structure, and cardiac contractility. Another limitation is the exposure to radioactivity. In view of these limitations of nuclear imaging techniques, our aim was to develop a high-resolution contrast-enhanced MRI method that allows rapid and noninvasive monitoring of apoptosis.
Recently, two PS-specific proteins (synaptotagmin-I and annexin V) have been labeled with superparamagnetic iron oxide (SPIO) particles for use as a specific MR contrast agent. Zhao et al. [18] have demonstrated the successful use of SPIO-labeled synaptotagmin-I in mice tumor MR imaging in vivo.
Schellenberger et al. [19] have used annexin V to synthesize iron oxide nanoparticles that target apoptosis. This compound has been used as an MRI contrast agent for use in in vitro labeling of apoptotic Jurkat-T cells [19]. Recently, this contrast agent was successfully applied by Sosnovik et al. [20] for imaging apoptosis in the reperfused infarcted beating mouse heart in vivo. This remarkable study described the feasibility of in vivo imaging of cardiomyocyte apoptosis using MRI for the first time.
Although all contrast agents affect both T1 and T2, superparamagnetic particles primarily reduce T2 and T2* and create negative contrast effects. SPIO induces strong field inhomogeneities and thereby decreases signal intensity in T2-weighted images especially at higher field strengths. This negative signal enhancement can be more prone to artifacts; however, it provides for the high sensitivity of SPIO-based imaging.
Previous experiments by our group and others have shown that labeling of apoptosis in the intact heart by histological or immunohistochemical methods remains challenging [21]. Therefore, the aim of our study was the detection of early apoptotic events after reperfusion injury in the intact heart by using a “positive” label. For this purpose, a gadolinium (Gd) complex that enhances signal in spin–lattice or T1-weighted intensity images was tested. Imaging of apoptosis was performed by using an isolated rat heart model of myocardial ischemia followed by reperfusion. This isolated rat heart model bears the potential to study cardiomyocyte apoptosis under specific and controlled conditions. Several pathophysiologically important parameters such as the composition of perfusate and hemodynamics can be manipulated easily. This model system eliminates a variety of problems: respiratory motion, difficulties arising from large blood pools in the ventricular cavities, and inhomogeneity introduced by air–tissue interfaces. In addition, imaging of the isolated rat heart in a high-field magnet allows for very high resolution imaging, including imaging of perfusion variations and coronary vessels and the visualization of the myocardial microstructure [22–28].
Materials and Methods
Contrast Agent Synthesis
For the detection of the molecules of interest, biotinylated annexin V (Sigma, Deisenhofen, Germany) was linked via avidin by covalent binding with gadolinium-diethylenetriamine pentaacetate (Gd-DTPA)-labeled biotinylated liposomes (MBT, Munich, Germany). The paramagnetic Gd ions were incorporated into the membrane (Figure 1) and can therefore interact with surrounding protons. The Gd content of the coated liposomes as determined by atomic absorption spectroscopy was 1.4 mM. The diameter of the liposomes, determined by dynamic laser light scattering by the manufacturer, was about 40 nm. In the following, the contrast agent Gd-DTPA–annexin V liposomes is abbreviated as Gd–annexin V.

Schematic representation of a paramagnetic apoptotic marker that is composed of Gd-labeled biotinylated liposomes and biotinylated annexin V. PS, phosphatidylserine.
Experimental Protocol
All experiments were performed in accordance with the European guidelines for the care and use of laboratory animals. Hearts (
The same experimental protocol was used for the control experiments (
MRI of Apoptotic Cells
1H MR microscopy was performed in an 11.75-T wide-bore magnet (AMX 500, Bruker, Karlsruhe, Germany) using a gradient system with a maximum strength of 660 mT/m. Transmission and reception of the MR signal was achieved with a quadrature birdcage resonator (RAPID Biomedical, Wuerzburg, Germany) tuned to the 1H frequency of 500.15 MHz. T1 maps were obtained by using an inversion recovery snapshot fast low-angle shot (FLASH) sequence. After inversion by an adiabatic 180° pulse, a series of 16 snapshot FLASH images [30,31] was acquired in the short-axis view to observe the T1 relaxation (field of view [FOV] = 20 × 20 mm, echo time [TE] = 1 msec, repetition time [TR] = 3.6 msec, α = 3, matrix 64 × 128, zero-filled before Fourier transformation to 1282 data points, in-plane resolution 140 μm2, slice thickness 1.5 mm). Eight averages were performed within a total acquisition time of less than 1 min, depending on heart rate. T1 maps were obtained by calculation of T1 for each pixel by a single exponential fit from the time course of the 16 signals after correction for the acceleration of relaxation by the FLASH pulses [32]. For T2* imaging, a 2-D gradient-recalled multiecho pulse sequence was used as described previously [33]. Imaging parameters were as follows: FOV = 20 × 20 mm, matrix 256 × 256, spatial resolution = 78 μm in-plane, and slice thickness = 250 μm. The minimum echo time was 2.05 msec. After each radio frequency excitation, 16 echoes were acquired with an interecho delay of 3.36 msec. T2* maps were obtained by fitting the relaxation time course to a monoexponential function.
Histology and Immunohistochemistry
We used the TdT-mediated X-dUTP nick-end labeling (TUNEL) technique as the standard technique for the localization of DNA fragmentation [34]. This is the most widely used technique for the detection of apoptotic cells. Staining was performed according to the manufacturer's instructions (Apoptag, Chemicon, Hofheim, Germany).
Data Analysis
A midmyocardial region of interest was defined manually within the ischemic zone in the left ventricular free wall (250–400 pixel). Use of the isolated heart model allows for easy and reliable identification of the ischemic zone, which can even be observed macroscopically on the epicardial surface. Mean values for T1 of each heart were obtained by averaging the values in the region of interest.
All values are expressed as mean ±
Results
Magnetic Resonance Imaging
In preliminary experiments (
After coronary occlusion, regions within the infarcted area showed a significant decrease in T1 from 2.504 ± 0.102 sec before to 1.938 ± 0.219 sec after application of the targeted contrast agent (
Figure 3 shows a T2* map of the same isolated rat heart in the short axis view. The map showed a corresponding decrease of T2* of about 20 msec in the infarcted myocardium compared with the remote myocardium.
In the control group (untargeted, biotinylated, Gd-labeled liposomes), the images obtained after the injection showed no binding of the control agent to apoptotic cardiomyocytes in the ischemic myocardium (Figure 4). T1 was 2.371 ± 0.158 sec before vesicle infusion and 2.399 ± 0.175 sec after vesicle infusion (

T1 maps representng an isolated rat heart (group 1; short axis view) after left coronary occulation followed by reperfusion (A) before (IZ 2.3 ± 0.018; RM 2.35 ± 0.022 sec) and (B) post Gd–annexin V infusion (25 μg; IZ 1.8 ± 0.025; RM 2.41 ± 0.02 sec). LV, left ventricle; RV, right ventricle; IZ, ischemic zone; RM, remote myocardium (control area).

T2* map post Gd–annexin V infusion. IZ, ischemic zone.
The postinfusion T1 value in the infarct zone after application of the active annexin-labeled probe (1.938 ± 0.219 sec) was significantly different from the postinfusion T1 value in the infarct zone of the hearts perfused with the untargeted control probe (2.399 ± 0.175 sec,

T1 maps representing an isolated rat heart (group 2; short axis view) (A) before, (B) during, and (C) after application of the untargeted Gd-labeled liposomes.
Histology
To prove the hypothesis that our contrast agent is mainly able to detect cells in the early phase of apoptosis, hearts were stained by use of the TUNEL technique to visualize DNA fragmentation in the ischemic myocardium (Figure 5). DNA fragmentation is considered to be a hallmark of ongoing apoptosis. This well-established method cannot distinguish between the nucleosomal cleavage of apoptosis and the nonspecific DNA degradation of necrosis. Therefore, TUNEL staining is not able to visualize apoptotic cells in the early phase of ischemia.
Discussion
Recently, Sosnovik et al. [20] successfully imaged apoptosis in the reperfused infarcted mouse heart in vivo by the use of annexin-V-bound iron oxide nanoparticles. This study described the feasibility of in vivo imaging of cardiomyocyte apoptosis using MRI for the first time. Here, we demonstrate for the first time the feasibility to rapidly target apoptotic cell death in acute reperfusion injury by use of a “positive” Gd-labeled annexin V contrast agent. Compared to previously used larger liposomes, the liposomes used in this study were relatively small. Although this may limit the sensitivity, the smaller size facilitates extravasation of the probe in the context of leaky vessels in the ischemic area, and therefore promotes targeting. Although the sensitivity of our agent may be lower due to lower Gd loading, nevertheless we were able to demonstrate that the sensitivity was sufficient to detect apoptotic cardiomyocytes.
Similar to the experiments previously performed by Dumont et al. [11], who showed that fluorescently labeled annexin V binds to apoptotic cells in vivo, in our experiment we chose a reperfusion time of about 90 min. This setup resulted in a marked increase in annexin-V-labeled cardiomyocytes, representing the PS externalization [11]. Kajstura et al. [35] reported that apoptosis was the major form of cell death during the first few hours of the evolution of myocardial infarction in a rat model, whereas necrotic myocyte cell death follows apoptosis later on in the time course after infarction. The administration of Gd–annexin V led to a significant increase in imaging contrast in those regions containing cells in the early phase of apoptotic cell death. These findings are in agreement with the known temporal sequence of apoptosis, in which one of the earliest events is the externalization of PS, followed by DNA fragmentation.
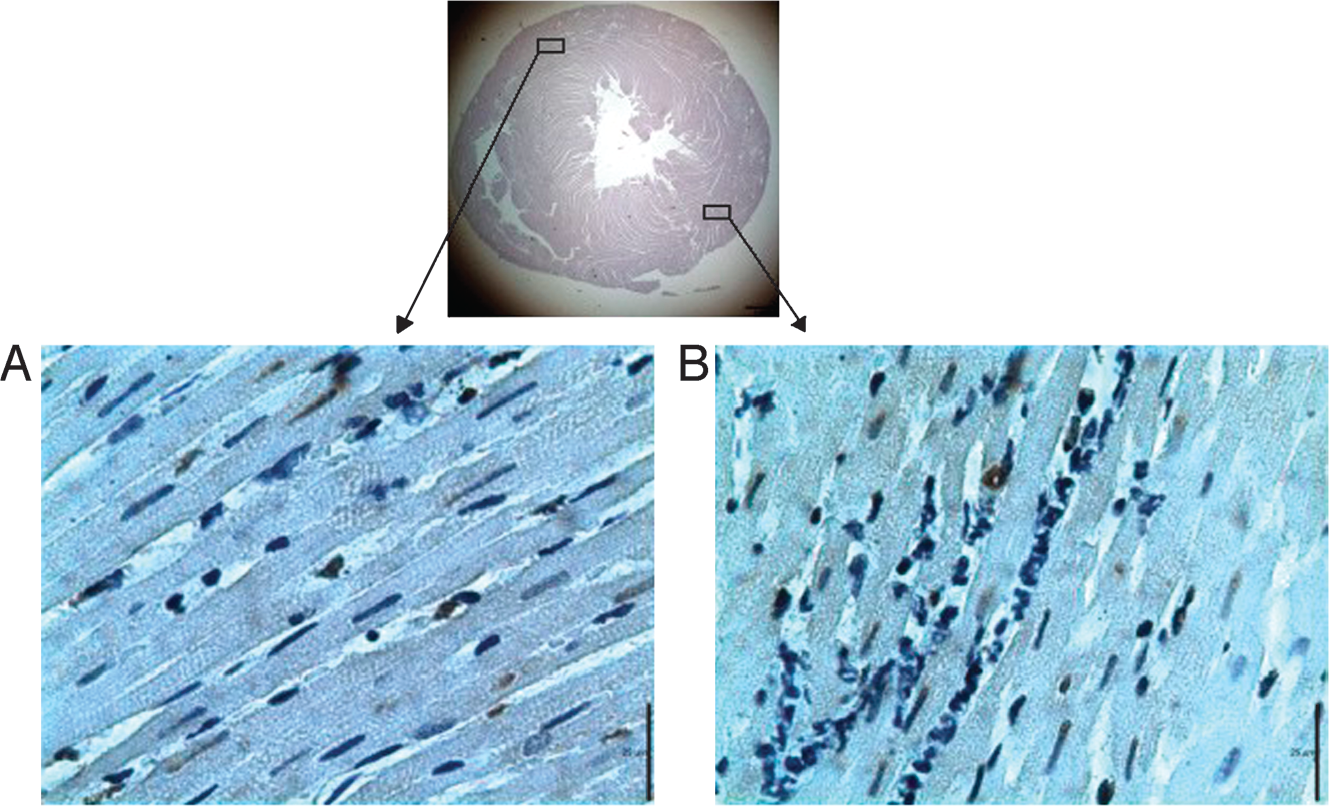
Typical results of a TUNEL stain of (A) remote region and (B) ischemic region of the left ventricle after 2 hr of reperfusion. Apoptotic cells showed brown nuclei.
The specificity of the contrast agent used was demonstrated in the control experiments: The untargeted vesicles showed no unspecific binding in the area with apoptotic cells. Comparison between the average postinfusion T1 value in the ischemic area of the hearts perfused with the active annexin-labeled probe and the average postinfusion T1 value in the ischemic area of the hearts perfused with the unlabeled control probe showed a significant difference. Also, in preliminary experiments, there was a dose-dependent effect of the labeled Gd–annexin V on T1 (data not shown). These findings indicate active binding of the annexin-labeled probe to PS on the apoptotic cardiomyocytes. In some hearts, probe accumulation of the active probe was observed in the right ventricle. This raises the speculation that the probe accumulation in the right ventricle might be caused by right ventricular damage (apoptosis) induced during surgery.
In principle, annexin V can be used to label cells in various stages of apoptosis ranging from the early phase without morphological changes of the nuclei to the late phase with pyknotic nucleus and condensed cytoplasm. In our case, after 90 min of reperfusion we expected a high amount of annexin V-positive cells, but only a few TUNEL-positive cells. It is known that the TUNEL technique detects only cells in the later stage of apoptotic cell death [16,36]. Our data show that in contrast to annexin V MRI, only a small number of apoptotic cardiomyocytes in the early phase of apoptosis were detected by the TUNEL technique.
In conclusion, only molecular MRI allows the detection of cells in an early apoptotic stage by using a PS-targeted contrast agent.
The use of the isolated rat heart model presented here allows one to study cardiomyocyte apoptosis under specific and controlled conditions such as composition of perfusate or given hemodynamic parameters. This model also allows for high-resolution imaging of perfusion, vessel morphology and geometry, flow, and myocardial microstructure. Frequently encountered problems of cardiac MRI of the intact animal such as respiratory motion or the influence of air–tissue artifacts could be avoided.
We envisage that the new Gd-DTPA-labeled annexin V may be valuable for in vivo MR detection of early apoptotic cell death and might contribute to the diagnostic process of diseases, such as acute myocardial infarction, heart failure, atherosclerosis or cancer, to assess prognosis and to evaluate accordant therapies and treatment. Other promising fields of investigation with Gd-DTPA-labeled annexin V MRI might be the detection of cardiac allograft rejection, inflammation, and autoimmune disorders [15].
Footnotes
Acknowledgments
The authors thank Toralf Peymann and Brita Schulze from the Munich Biotech AG for the creation and donation of the liposomes. We also thank Sabine Voll, Yvonne Vogt, and Brigitte Schmidt for their expert technical assistance. This work was supported by Deutsche Forschungsgemeinschaft, Sonderforschungsbereich “Pathophysiologie der Herzinsuffizienz” SFB 355/A8.
