Abstract
Heavy menstrual bleeding (HMB) is defined as excessive blood loss (>80 ml per cycle) during regular intervals of menstruation [1]. The impact of HMB on women has been well established and includes interference with physical, emotional, social and material quality of life [1]. The prevalence of HMB varies from 4 to 27%, with lower estimates based on objective measures of menstrual bleeding and higher estimates based on subjective measures of bleeding, respectively [1]. Up to 30% of women will consult with a healthcare provider regarding HMB during their reproductive years [2]. Approximately 50% of women who are evaluated for HMB have no organic pathology implicated as a cause [3]. Of those women with a discernible underlying HMB cause, uterine fibroids and polyps are among the most commonly reported pathologies in women with HMB [4].
The pathophysiology of HMB in women with fibroids is unknown; however, several theories have been proposed. These theories include increased endometrial surface area, increased uterine vascularity, impaired uterine contractility, endometrial ulceration by submucosal fibroids and compression of the venous plexus within the myometrium leading to endometrial venule ectasia and resulting in congestion of the myometrium and endometrium [5].
Tranexamic acid (TA) has been shown to diminish the increased fibrinolytic activity observed in the endometrial tissues of women with HMB [6]. A modified-release oral formulation of TA (Lysteda®, Ferring Pharmaceuticals Inc., NJ, USA) was approved by the US FDA for the treatment of cyclic HMB in patients in the USA. TA is a nonhormonal, competitive plasmin inhibitor that does not appear to affect fertility and is administered only during each menstrual cycle in women with HMB. It is available as a 650-mg oral tablet, and two tablets are taken three-times per day for up to 5 days during menstruation. The novel, modified-release TA formulation results in controlled dissolution of the drug, which reduces the gastrointestinal adverse events (AEs) that limit patient tolerability of immediate-release TA; in Phase I studies, these formulations were demonstrated to be bioequivalent [7].
Two pivotal Phase III studies have shown TA to be an effective nonhormonal treatment for HMB [8,9], and long-term data have demonstrated the tolerability of TA, with the most frequent AEs being headache, nasal and sinus symptoms, and back pain [10,11]. This pooled analysis of the two pivotal Phase III TA studies evaluated the efficacy and safety of TA in women with HMB and fibroids [8,9].
Materials & methods
Patients
Women aged 18–49 years were enrolled in each study if they had regular menstrual cycles (21–35 days apart) with each menstruation lasting <10 days for the preceding 6 months, >60 ml of menstrual blood loss (MBL) during the first pretreatment cycle and a mean baseline MBL of ≥80 ml, as measured by a validated alkaline hematin method. Women were required to have no abnormal findings on pelvic examination, normal results on cervical cytology screening and normal findings on transvaginal ultrasonograms. In these studies, fibroids or simple ovarian cysts were not considered to be abnormal findings. Women with anovulatory dysfunctional uterine bleeding, metrorrhagia, menometrorrhagia or polymenorrhea were excluded from the study. Those with significant renal or hepatic disease and those with other medical conditions that might have confounded the study or been detrimental to the subject (e.g., uncontrolled diabetes or hypertension, thromboembolic disease and clinically significant arrhythmia, among others) were excluded.
The use of concomitant medications (e.g., anticoagulants and hormonal contraceptives) was not permitted during the study period, with the exception of acetaminophen, opioids and oral iron (at any time), and NSAIDs and COX-2 inhibitors during the intermenstrual phase. Women using medications to treat HMB before screening underwent a washout period of up to 8 weeks before participation in the study. Enrolled subjects were required to use an acceptable nonhormonal form of contraception.
Study design
Both clinical studies were randomized, double-blind, placebo-controlled, parallel-group Phase III studies that were conducted at multiple sites within the USA. There were 63 participating sites in the first study and 40 sites in the second, which consisted of both university-affiliated and community-based sites. In the first pivotal Phase III study, after two pretreatment menstrual cycles, women were randomized (2:2:1) to receive TA 1.95 g/day (one 650-mg tablet three-times daily), 3.9 g/day (two 650-mg tablets three-times daily) or placebo for up to 5 days per menstrual period for three cycles. In the second pivotal Phase III study, after two pretreatment menstrual cycles, women were randomized to receive either TA 3.9 g/day (two 650-mg tablets three-times daily) or placebo for up to 5 days per menstrual period for six cycles. The study protocol was approved by each site's institutional review board. All participants provided written informed consent.
Patient assessments
Transvaginal ultrasonography was conducted at pretreatment visits during both studies to determine whether any fibroids were present. Women with fibroids were enrolled as long as the investigator determined that the number and size of the fibroids did not warrant surgical management. The alkaline hematin method was used to measure MBL at pretreatment visits during both studies. Baseline MBL was calculated as the mean of the two MBL measurements obtained during the two pretreatment visits in each study. Safety was assessed primarily by AE monitoring.
Statistical analysis
Data were reported for the combined intent-to-treat (ITT) populations from the 3.9 g/day and placebo treatment groups from each study and were analyzed according to fibroid status. The ITT population of each study included all randomized subjects who received at least one dose of study drug. Summary statistics were presented for all baseline demographic data. The statistical comparison between the 3.9 g/day (TA) and placebo groups was made by a χ2 test for categorical parameters (i.e., race) and an analysis of variance for continuous parameters (i.e., age and weight). For the efficacy analyses, descriptive statistics for MBL were calculated and statistical comparisons between the TA 3.9 g/day and placebo groups were made by analysis of variance. All efficacy results were analyzed according to fibroid status. Safety analyses primarily consisted of frequency counts of AEs. All statistical calculations were performed using SAS version 9.2 (SAS Institute Inc., NC, USA).
Results
Patients
A summary of the patient disposition has been provided in
Subject disposition.
Percentages are based on the all-randomized population.
Percentages are based on the ITT population.
Other events included an abnormal pap smear, menstrual cycle irregularities, fear of stroke, withdrawn consent and noncompliance.
ITT: Intent-to-treat; TA: Tranexamic acid.
A summary of patient demographics and baseline characteristics is provided in
Subject demographics and baseline characteristics, intent-to-treat population.
TA: Tranexamic acid.
Efficacy
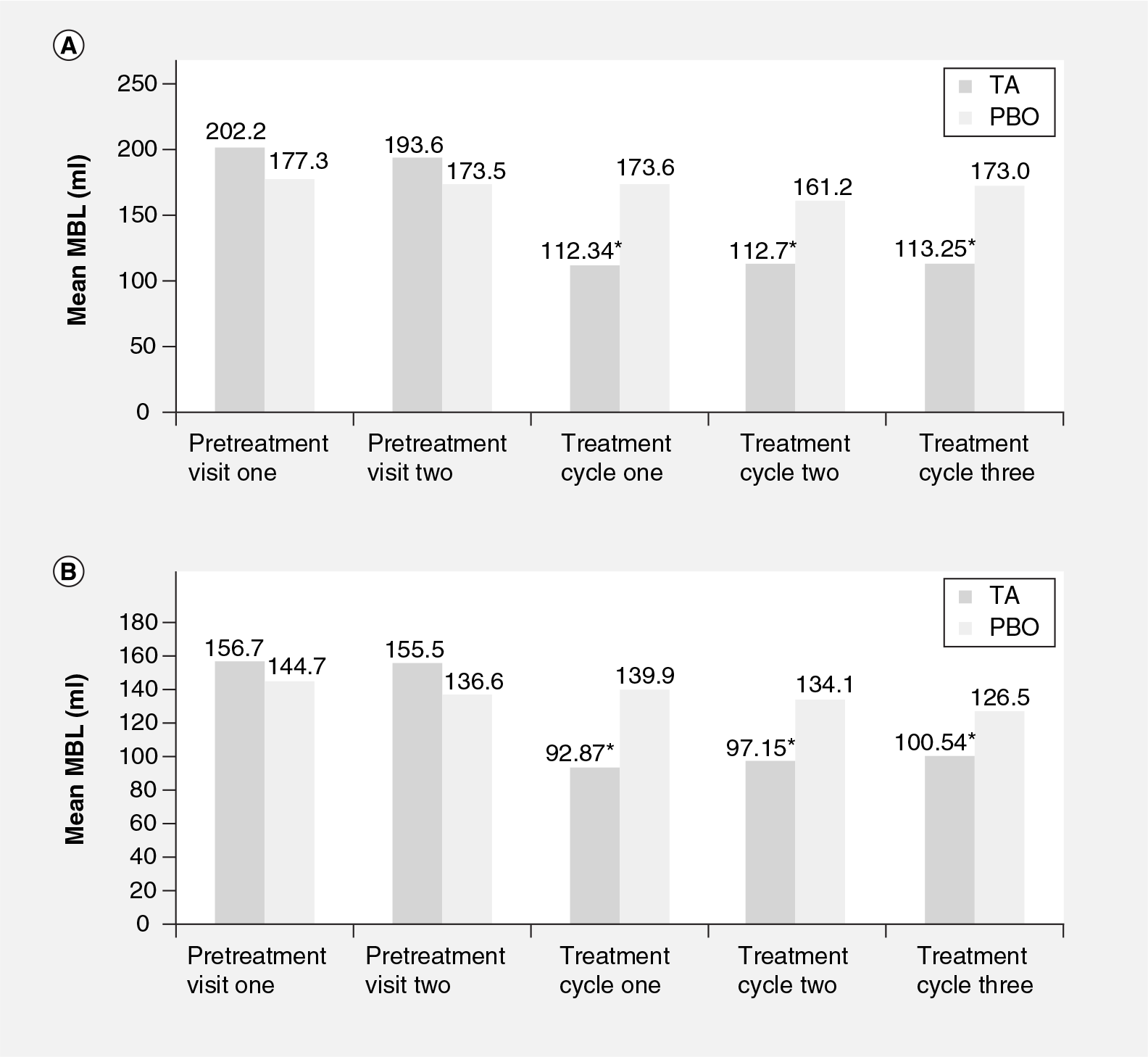
Regardless of fibroid status, mean MBL for women in the TA and placebo groups was comparable at each of the pretreatment visits (Figure 1). In women with fibroids and women without fibroids treated with TA, mean MBL was significantly (p < 0.001) reduced compared with placebo at the first visit, and the significant reduction in mean MBL was maintained across all three treatment cycles (Figure 1). For women with and without fibroids, the mean MBL in the TA treatment group was comparable across all three treatment cycles (Figure 1).
Mean MBL was highest for women with and without fibroids on days 2 and 3 of both pretreatment and treatment cycles (Figure 2). Regardless of fibroid status, mean MBL was reduced in women treated with TA compared with women treated with placebo across all days of all treatment cycles (Figure 2). Within the TA treatment group of women, regardless of fibroid status, mean MBL was reduced across all days of each treatment cycle compared with each pretreatment cycle (Figure 2).

The greatest reductions in mean MBL in women in the TA and placebo groups with and without fibroids were noted on days 2 and 3 of each treatment cycle, with some treatment differences in mean MBL reaching statistical significance (p < 0.001; Figure 2). There were more statistically significant (p < 0.001) reductions in MBL in women with fibroids treated with TA compared with placebo than in women without fibroids (Figure 2).
Safety
Primary safety data from both of the Phase III pivotal studies have been reported previously [8,9]. Taken together, the majority of AEs across both of the Phase III pivotal studies were mild-to-moderate in severity. There were no statistically significant group differences in reported treatment-emergent AEs in either of the pivotal studies. The most commonly reported AEs in women treated with TA 3.9 g/day in the first pivotal Phase III study (occurring in ≥5% of TA-treated women and at twice the percentage of placebo-treated women) included viral upper respiratory tract infection (7.0%), musculoskeletal pain (5.2%) and myalgia (5.2%) [6]. The most commonly reported AEs in women treated with 3.9 g/day TA (occurring in more than ten subjects irrespective of causality) in the second pivotal Phase III study included menstrual discomfort (61.5%), headache (55.6%), back pain (23.9%), nausea (14.5%) and anemia (10.3%) [9].
A total of six serious treatment-emergent AEs were reported in four women treated with 3.9 g/day of TA across both studies; none were considered to be related to treatment [8,9]. In the first pivotal Phase III study, four women treated with TA withdrew from the study owing to treatment-emergent AEs; all AEs had resolved by the follow-up study visit [9]. In the second Phase III study, three subjects treated with TA withdrew from the study due to AEs [8]. No deaths occurred among randomized women during the study periods in either pivotal study [8,9].
Discussion
In this study, mean MBL was reduced in women treated with TA compared with placebo, regardless of fibroid status beginning with the first treatment cycle and throughout all three treatment cycles. Thus, regardless of the pathophysiology of HMB in women with fibroids in this study, TA was effective in reducing MBL through its impact on the coagulation cascade as a competitive plasmin inhibitor.
Similar to the recently published results by Lukes et al. [12], mean MBL in this analysis was also highest on days 2 and 3 of each menstrual cycle, regardless of treatment or fibroid status. TA treatment resulted in a greater number of statistically significant differences in mean MBL compared with placebo on days 2 and 3 in women with fibroids compared with those without fibroids. This greater number of statistically significant treatment differences in women with fibroids may have been due to the increased MBL often observed in patients with fibroids.
These results that demonstrated the efficacy and safety of TA for the treatment of HMB in women with fibroids, have the potential to impact clinical practice. Treatment options for women with HMB and fibroids currently include invasive surgical procedures, including hysteroscopic, laproscopic, robotic and abdominal myomectomy [1]. However, the use of TA may decrease the need for these invasive surgeries to alleviate HMB often experienced in women with fibroids via the decreased MBL demonstrated in this analysis. Furthermore, for those patients requiring myomectomy, TA may prevent blood transfusions needed for these procedures by allowing for natural improvement in hemoglobin prior to surgery. TA has been shown to increase both ferritin and hemoglobin levels in women with HMB [13].
Limitations of this study include the pooled nature of the data analysis, which was required to have a sufficient number of women with and without fibroids for comparison. Also, the effect of TA treatment in women with HMB due to other etiologies or in combination with other treatments cannot be determined from these data owing to the exclusion criteria and medication restrictions imposed by the study. The results of this analysis may not be generalizable to women with markedly enlarged fibroids that require surgical management.
Conclusion
This post hoc analysis of the results from two pivotal Phase III studies of TA revealed that TA was effective in significantly reducing mean MBL in women with fibroids, as well as in women without fibroids beginning with the first treatment cycle and lasting throughout all three treatment cycles. The assessment of mean daily MBL revealed more significant reductions in mean daily MBL on days 2 and 3 for women with fibroids than for women without fibroids treated with TA compared with placebo. TA was well tolerated by subjects in both the Phase III pivotal studies.
Financial & competing interests disclosure
S Eder has disclosed that he has received research support from Xanodyne, Warner-Chilcott, Bayer and Teva. J Baker has received research support from Ferring Pharmaceuticals Inc. J Gersten has received research funding from Pfizer, Organon, Ortho-McNeil, Wyeth, Galen Holdings, Warner Chilcott, Lilly, Sepracor, TAP, Barrier Therapeutics, Xanodyne, Roche, BioSante Pharmaceuticals, Duramed, Berlex, Johnson & Johnson, Amgen, Perrigo Pharmaceuticals, Boehringer Ingelheim, Bayer, Teva, Schering-Plough, Ferring Pharmaceuticals Inc. and Merck. TL Adomako is an employee of Ferring Pharmaceuticals Inc. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Executive summary
A total of 371 patients were included in the combined intent-to-treat population (3.9 g/day: n = 232; placebo: n = 139). Fibroids were present for 96 women treated with tranexamic acid (TA; Lysteda®, Ferring Pharmaceuticals Inc., NJ, USA) and 51 women in the placebo group.
Across treatment groups by fibroid status, the majority of women were Caucasian with mean ages ranging from 37 to 41 years old.
In both women with fibroids and those without fibroids, mean menstrual blood loss was significantly (p < 0.001) reduced compared with placebo at the first visit; a significant reduction in mean menstrual blood loss was maintained across all three treatment cycles.
For women with and without fibroids, the mean menstrual blood loss in the TA-treatment groups was comparable across all three treatment cycles.
For women treated with TA compared with placebo, regardless of fibroid status, mean menstrual blood loss was reduced in women treated with TA across all days of all treatment cycles.
The greatest reductions in mean menstrual blood loss in women with and without fibroids were noted on days 2 and 3 of each treatment cycle, with some differences in mean menstrual blood loss reaching statistical significance (p < 0.001).
More statistically significant (p < 0.001) reductions in menstrual blood loss per day occurred in women with fibroids treated with TA compared with placebo than in those women without fibroids.
Taken together, the majority of adverse events across both of the Phase III pivotal studies were mild-to-moderate in severity.
Professional writing assistance was provided by L Miesle, PharmD, CMPP, of The JB Ashtin Group Inc. (MI, USA). This assistance was funded by Ferring Pharmaceuticals Inc.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
