Abstract
Keywords
Heavy menstrual bleeding (HMB) is defined as excessive blood loss (>80 ml/cycle) during regular intervals of menstruation [1]. Excessive blood loss during menstruation is estimated to affect up to one-third of women of reproductive age [2]. Women who experience HMB will often present to a physician because of the subsequent negative impact on their daily lives and activities [3,4]. HMB has been shown to adversely affect mood, energy/vitality, work productivity, social interactions, family life and sexual functioning [5–7]. The effect HMB has on a woman's health-related quality of life (HRQL) derives from both the effort associated with managing menstrual bleeding and the consequences of excessive blood loss, such as fatigue and iron deficiency anemia [7].
Tranexamic acid has been shown to diminish the increased fibrinolytic activity observed in the endometrial tissues of women with HMB [8]. Immediate-release (IR) tranexamic acid is a nonhormonal, competitive plasmin inhibitor that is administered only during HMB. IR tranexamic acid has a well-established safety profile and does not appear to affect fertility [9,10]. A modified-release oral formulation of tranexamic acid (TA; Lysteda®, Ferring Pharmaceuticals, Inc., NJ, USA) was approved in November 2009 by the US FDA for the treatment of cyclic HMB in patients. In Phase I studies, TA was shown to be bioequivalent to IR tranexamic acid and was well tolerated [11]. Two pivotal Phase III studies have shown TA to be an effective nonhormonal treatment for HMB [12,13], and long-term data have demonstrated the tolerability of TA [14,15].
Numerous clinical trials investigating HMB have shown a reduction in menstrual blood loss (MBL) as the key efficacy end point, without measuring the effects of treatment on patient-reported outcomes. Some studies involving oral contraceptives, progestins and other pharmacologic therapies for HMB have demonstrated some improvement in patient HRQL; however, these improvements in HRQL were less than those noted with other treatments for HMB (i.e., ablation or hysterectomy) [7]. Another study indicated that HRQL improved in women with menorrhagia and abnormal laboratory hemostasis when they were treated with either tranexamic acid or intranasal desmopressin; tranexamic acid proved to be more effective [16]. Recent studies have shown that TA, in addition to reducing mean MBL per cycle, also has a positive impact on HRQL [12–14] as measured by the validated Menorrhagia Impact Questionnaire (MIQ) [17].
Using data from two pivotal Phase III TA studies [12,13], the objectives of this post-hoc analysis were to evaluate daily MBL in women with HMB and assess the relationship between individual daily MBL values and MIQ scores for patient perception of MBL, limitations in physical activity, and limitations in social/leisure activity during pretreatment and TA-treatment menstrual cycles.
Materials & methods
Patients
Women aged 18–49 years were enrolled in one of two Phase III studies if they had a mean baseline MBL of ≥80 ml (average MBL over two pretreatment menstrual cycles with the first MBL having a minimum of ≥60 ml) as measured by a validated alkaline hematin method. Women were required to have regular menstrual cycles (21–35 days apart) with each menstruation not lasting >10 days in at least four of the last 6 months prior to screening. In addition, women also had to have normal findings on pelvic examinations, cervical cytology screenings and transvaginal ultrasonograms. Although it is noted that fibroids are not considered a normal finding during pelvic examinations or on transvaginal ultrasonograms, for these studies, women with fibroids were not excluded unless, based on investigators' opinions, the fibroids required surgical management.
Women were excluded from the studies based on history or presence of anovulatory dysfunctional uterine bleeding, metrorrhagia, menometrorrhagia, polymenorrhea, endometrial polyps, endometrial hyperplasia, endometrial carcinoma, cervical carcinoma or any other clinically significant comorbid disease. Additional exclusion criteria consisted of clinically significant abnormalities that could confound the study or be detrimental to the patient as noted at physical examination. Women with a history of bilateral oophorectomy or hysterectomy, or women who were pregnant, breastfeeding, planning to become pregnant or became pregnant during either of the studies were also excluded.
Study design
Both randomized, double-blind, placebo-controlled, parallel-group Phase III studies were conducted at multiple sites within the USA [12,13]. In both studies, patients were randomized after two pretreatment menstrual cycles. In the first pivotal Phase III study, women were randomized (2:2:1) to receive 1.95 or 3.9 g/day TA or placebo for up to 5 days per menstrual cycle for three cycles [12]. In the second pivotal Phase III study, women were randomized to receive either 3.9 g/day TA or placebo for up to 5 days per menstrual cycle for six cycles [13].
Each study was approved by the institutional review board at the study sites. Both studies were conducted in accordance with the ethical principles that have their origin in the Declaration of Helsinki and that are consistent with good clinical practice and the applicable regulatory requirements. Patients were required to provide written informed consent.
Patient assessments
In both Phase III studies, MBL was measured via the alkaline hematin method and HRQL was measured using the validated MIQ [17]. Patient perception of MBL was assessed by the question on the MIQ: ‘During your most recent menstrual period, your blood loss was …?’ Categorical responses consisted of ‘light’, ‘moderate’, ‘heavy’ or ‘very heavy’. Limitations in physical activity were assessed by the question on the MIQ: ‘During your most recent menstrual period, how much did your bleeding limit you in your physical activities?’ Categorical responses consisted of ‘not at all’, ‘slightly’, ‘moderately’, ‘quite a bit’ and ‘extremely’. Limitations in social/leisure activity were assessed by the question on the MIQ: ‘During your most recent menstrual period, how much did your bleeding limit you in your social or leisure activities?’ Categorical responses consisted of ‘not at all’, ‘slightly’, ‘moderately’, ‘quite a bit’ and ‘extremely’. Safety was assessed in each study primarily by adverse event (AE) monitoring.
Statistical methods
Assessments of MBL and MIQ scores were conducted using the combined intent-to-treat (ITT) populations from the 3.9 g/day TA treatment groups from each study. This post-hoc analysis examined MBL and MIQ scores obtained for two pretreatment menstrual cycles and the first three TA-treatment menstrual cycles. The ITT populations included all randomized patients who received at least one dose of study drug. Descriptive statistics were calculated for overall daily MBL and for daily MBL according to categorical MIQ scores. Safety data were reported for the ITT population from each study. Frequency counts were compiled for classification of AEs.
Results
Patients
Patient disposition and demographics for each study have been presented separately in previous publications [12,13]. Demographics and baseline characteristics are briefly summarized for this post-hoc analysis in
Patient demographics and baseline characteristics.
Daily MBL
The mean daily MBL for the two pretreatment and first three TA-treatment menstrual cycles are presented in
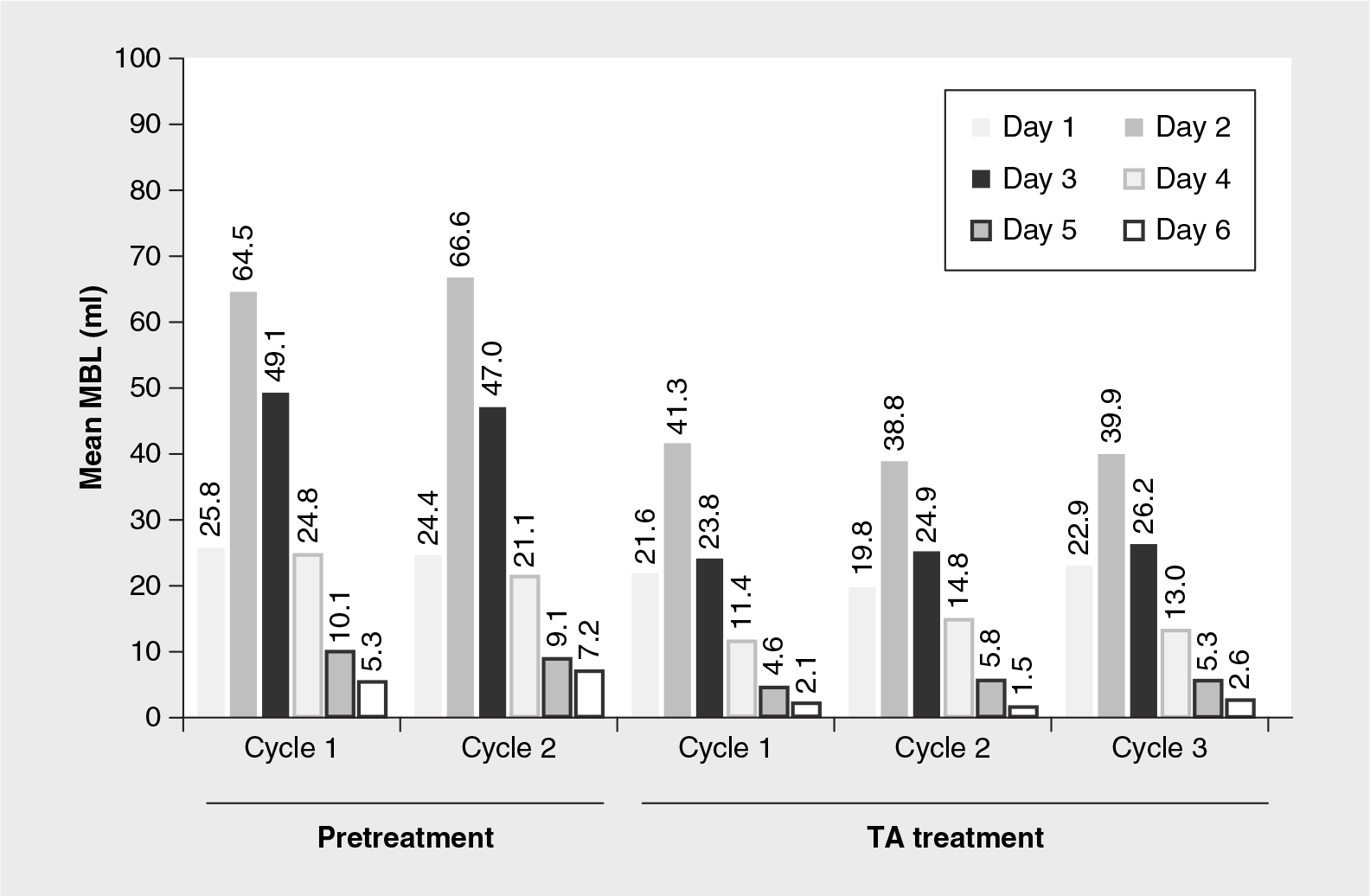
Overall daily menstrual blood loss for pretreatment and 3.9 g/day tranexamic acid treatment menstrual cycles.
Relationship between daily MBL & MIQ scores
Perception of MBL
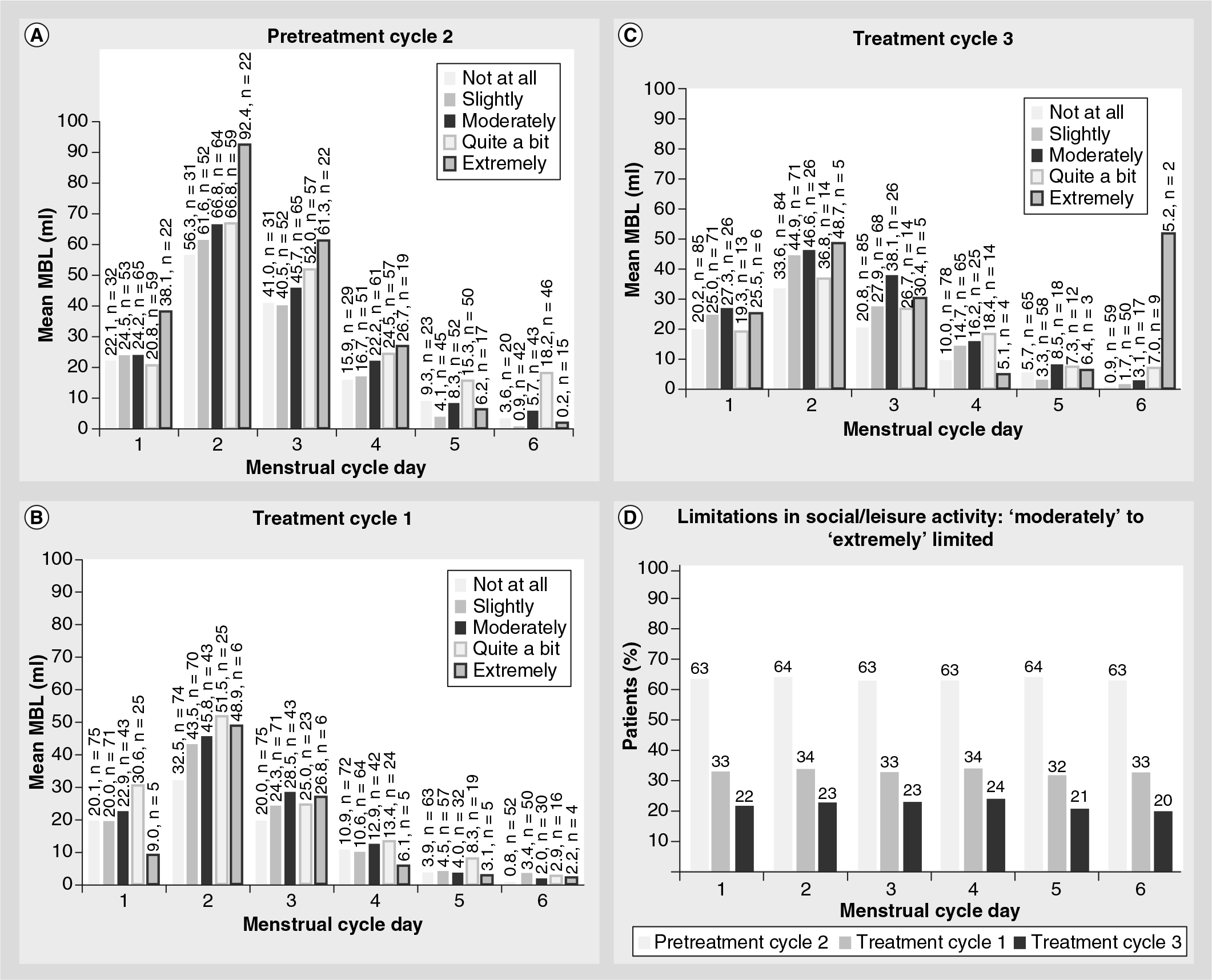
Values for daily MBL by perception of menstrual blood flow for the second pretreatment and the first and third TA-treatment menstrual cycles are presented in

Patient assessment of menstrual blood loss versus actual mean menstrual blood loss by menstrual cycle day.
Limitations in physical activity
Values for the degree of limitations in physical activity by mean daily MBL for the second pretreatment and the first and third TA-treatment menstrual cycles is presented in

Limitations in social/leisure activity
The degree of limitations in social/leisure activity by mean daily MBL for the second pretreatment and the first and third TA-treatment menstrual cycles is presented in
Safety
Primary safety data from both Phase III pivotal studies have been reported previously [12,13]. Taken together, the majority of AEs across both studies were mild-to-moderate in intensity. The most commonly reported AEs in women treated with 3.9 g/day TA in the first Phase III pivotal study (occurring in ≥5% of TA-treated women and at twice the percentage of placebo-treated women) included viral upper respiratory tract infection (7%), musculoskeletal pain (5%) and myalgia (5%) [12]. The most commonly reported AEs in women treated with 3.9 g/day TA in the second Phase III pivotal study (occurring in ≥10 patients, irrespective of causality) included menstrual discomfort (61.5%), headache (56%), back pain (24%), nausea (15%) and anemia (10%) [13]. A total of six serious treatment-emergent AEs were reported by four women treated with 3.9 g/day TA; none were considered to be related to treatment [12,13]. In the first pivotal Phase III study, one woman treated with 3.9 g/day TA withdrew from the study due to treatment-emergent AEs; all events had resolved by the follow-up study visit [12]. In the second Phase III study, three patients treated with TA withdrew from the study due to AEs [13]. No deaths occurred among randomized women during the study periods in either pivotal study [12,13].
Discussion
Overall, mean MBL was highest on days 2 and 3 then lowest on day 6 for both pretreatment and TA-treatment menstrual cycles. These heavy- and light-flow days are also consistent with those days reported previously by women complaining of HMB [18]. As illustrated in
To the authors' knowledge, this study is the first to evaluate daily MBL in women with HMB and to establish relationships between patients' perceptions of daily MBL and identify how MBL is perceived to limit physical and social/leisure activities. Among women enrolled in the study, patient perception of menstrual blood flow generally coincided with mean daily MBL. However, anecdotal observations in practice often suggest that women perceive a greater MBL than can be objectively documented. The degree of limitations in physical activity was reflective of the mean daily MBL experienced during both pretreatment and TA-treatment menstrual cycles. However, TA treatment reduced the number of patients reporting ‘extremely’, ‘quite a bit’ or ‘moderately’ limited physical activities. In general, limitations in social/leisure activity were consistent with increased mean daily MBL experienced during both pretreatment and TA-treatment menstrual cycles. However, TA treatment reduced the number of patients reporting ‘extremely’, ‘quite a bit’ or ‘moderately’ limited social activities. For both limitations in physical and social/leisure activity, reducing MBL via TA treatment resulted in positive effects on these measures of HRQL.
Taken together, the relationships between daily mean MBL, patient perception of MBL, limitations in physical activity and limitations in social/leisure activity may influence the clinical management of HMB. The results of this study show that patient perceptions of daily MBL coincide with objective measurements of daily MBL, indicating that patient concerns regarding HMB should be assessed seriously. In addition, daily MBL is related to HRQL measures in that higher daily MBLs are associated with worse ratings of HRQL. Therefore, efforts to diagnose and decrease MBL should be a high priority for clinicians to improve the daily lives of women with HMB.
Limitations
Women with HMB in each of these studies were treated with TA, thus results may not be generalized to other women with HMB treated with other medications, devices or procedures. Furthermore, this was an exploratory post-hoc analysis to assess daily MBL in women with HMB and examine any relationships between objective measures of daily MBL and subjective measures regarding patient perceptions of MBL and its subsequent impact on HRQL. Future studies specifically designed to assess the statistical significance of these relationships would be beneficial.
Conclusion
This post-hoc analysis based on data from two pivotal Phase III studies of TA revealed that mean daily MBL values coincided with patients' perceptions of MBL, and that mean daily MBL was related to the degree of limitation in both physical and social/leisure activities. TA was well tolerated in women in both Phase III pivotal studies.
Future perspective
These results point to the importance of assessing HRQL when evaluating women with HMB as the two are closely linked. Treatment of HMB should include not only reducing MBL, but also improving the patient's overall quality of life during menses. We also look forward to future studies regarding the impact of TA on HRQL and other functional outcomes.
Executive summary
The objective of this post-hoc analysis of data from two Phase III studies was to evaluate daily menstrual blood loss (MBL) in women with heavy menstrual bleeding and assess the relationship between individual daily MBL values and Menorrhagia Impact Questionnaire (MIQ) scores for patient perception of MBL, limitations in physical activity, and limitations in social/leisure activity during pretreatment and tranexamic acid (TA)-treatment menstrual cycles.
MBL was measured via the alkaline hematin method and health-related quality of life was measured using the validated MIQ.
Effect-size analyses for daily MBL and MIQ scores were not conducted.
Safety was assessed primarily by the incidence of spontaneously reported adverse events (AEs).
Mean daily MBL was highest on day 2 and lowest on day 6 of each pretreatment as well as TA-treatment menstrual cycle. The mean MBL on all cycle days was markedly reduced during the TA-treatment menstrual cycles compared with the pretreatment cycles, with the largest reduction occurring on day 2 when MBL was highest.
Mean daily MBL values corresponded to patients' perceptions of their menstrual blood flow.
Across pretreatment and TA-treatment menstrual cycles and on any given menstrual cycle day, patients who had the highest mean daily MBL responded that their physical activities were either ‘extremely’ limited or limited ‘quite a bit’.
Across pretreatment and TA-treatment menstrual cycles and on any given menstrual cycle day, patients who had the highest mean daily MBL responded that their social/leisure activities were either ‘extremely’ limited, limited ‘quite a bit’ or ‘moderately’ limited.
The majority of AEs across both studies were mild-to-moderate in intensity.
Across studies, the most commonly reported AEs in women treated with 3.9 g/day TA were menstrual discomfort, headache, back pain, nausea, anemia, viral upper respiratory tract infection, musculoskeletal pain and myalgia.
Footnotes
Financial support for data analysis and manuscript development was provided by Ferring Pharmaceuticals, Inc. Authors were not compensated for their work on this manuscript. AS Lukes has received funding from Abbott Pharmaceuticals, American Medical Systems, Bayer, CDC and Prevention/Association for Prevention Teaching and Research, Duramed, Endoceutics, Ethicon, Hologic, Interlace Medical, Luitpold, Merck, National Improvements for Women's Healthcare, NIH, Smith and Nephew, Trent Foundation and Xanodyne Pharmaceuticals. She has also been a consultant and/or has served as a speaker for Abbott Pharmaceuticals, AMAG (4 years ago), American Medical Systems, Bayer, Boehringer-Ingelheim, Daichii, Ethicon, Ferring Pharmaceuticals, Inc., Hologic, Interlace Medical, Microsulis (5 years ago), Myriad, Watson Pharmaceuticals and Xanodyne Pharmaceuticals. J Baker has received research support from Ferring Pharmaceuticals, Inc. S Eder has received research support from Bayer, Boehringer-Ingleheim, Church and Dwight, Graceway Pharmaceuticals, Teva, Warner-Chilcott and X anodyne Pharmaceuticals. He has also served as a consultant for Everett Laboratories and as a speaker for Ferring Pharmaceuticals, Inc. and Merck. TL Adomako is an employee of Ferring Pharmaceuticals, Inc. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Writing assistance was funded by Ferring Pharmaceuticals, Inc. (NJ, USA). Professional writing assistance was provided by L Miesle of The JB Ashtin Group Inc. (MI, USA).
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
