Abstract
Tranexamic acid, a synthetic lysine derivative, is an antifibrinolytic drug that prevents the breakdown of fibrin by competitively blocking binding sites of plasminogen. Tranexamic acid is often considered a first-line treatment for the management of heavy menstrual bleeding (HMB). A new oral formulation of tranexamic acid provides a nonhormonal HMB therapy that is safe, effective and well tolerated; is administered only during menstruation; addresses the excessive fibrinolysis implicated in many cases of HMB; and improves women's health-related quality of life by reducing limitations on physical, social and leisure activities. This article provides a summary of the clinical development, therapeutic efficacy and tolerability profile of this novel formulation of tranexamic acid for the treatment of HMB.
Heavy menstrual bleeding (HMB) is a major health problem that negatively impacts a woman's quality of life. The prevalence of HMB varies from 4 to 27% with lower estimates based on objective measures of menstrual bleeding and higher estimates based on subjective measures of bleeding [101]. The impact of HMB on women has been well established and includes interference with physical, emotional, social and material quality of life [101].
Selection of medical therapy for the treatment of HMB varies considerably, yet healthcare providers outside the USA have used immediate-release tranexamic acid for HMB for several decades because it is both effective and safe [1]. Support for the use of tranexamic acid in women with HMB includes a body of evidence showing increased fibrinolytic activity in menstrual blood of women with HMB [2,3]. Until recently, only the immediate-release form of tranexamic acid for HMB has been available worldwide. In November 2009, Lysteda™ (Ferring Pharmaceuticals, NJ, USA), a novel tranexamic acid formulation, was approved by the US FDA to treat cyclic HMB [102]. This novel oral formulation provides a higher per-tablet dose (650 vs 500 mg) and was designed to minimize gastrointestinal adverse effects compared with the immediate-release form. This article focuses on the clinical efficacy and safety results of Lysteda.
Overview of the treatment of HMB
Only two medications are approved by the US FDA for treatment of HMB: tranexamic acid (Lysteda) has been indicated for the treatment of cyclic HMB, and levonorgestrel-release intrauterine system (LNG-IUS [Mirena®], Bayer Healthcare Pharmaceuticals, Inc., NJ, USA), approved by the FDA in 2009, has been indicated for use as intrauterine contraception for up to 5 years and treatment of HMB for women who choose to use intrauterine contraception as their method of birth control. Important differences between these options include the impact on fertility (Lysteda has no impact, and LNG-IUS provides contraception), use of a hormone (Lysteda is nonhormonal, and LNG-IUS contains levonorgestrel) and patient use (Lysteda is user dependent for up to 5 days of menstrual bleeding, and LNG-IUS is user independent and is placed by a physician within the endometrial cavity for up to 5 years).
Other medications not approved by the FDA but used by clinicians worldwide for treatment of HMB include nonsteroidal anti-inflammatory drugs, combined oral contraceptives, progestogens (oral, injections and implants), gonadotrophin-releasing hormone analogue (GnRH-a) and synthetic testosterone derivatives. Currently there are ongoing clinical trials being conducted in the USA in women with HMB using a combined oral contraception containing estradiol valerate and dienogest, which is available in Europe as Qlaira® (Bayer Schering Pharma AG, Berlin, Germany) for an indication of HMB and in the USA as Natazia® (Bayer Healthcare Pharmaceuticals, Inc., NJ, USA) for the indication of birth control.
Introduction to the compound
Tranexamic acid is an antifibrinolytic drug that competitively inhibits plasmin activity. This inhibition prevents the breakdown of fibrin.
For more than three decades, immediate-release tranexamic acid, often given as two 500-g tablets every 6 h for up to 5 days (i.e., up to 40 tablets), has been used safely and effectively in Asia, Australia, Europe and Canada for the treatment of HMB. In addition, it has been used for the control of mucocutaneous bleeding in patients with bleeding disorders such as hemophilia and von Willebrand disease [4,5]. Tranexamic acid is a nonhormonal agent administered during menses that reduces menstrual blood loss (MBL) by inhibiting the dissolution of fibrin (by plasmin) within the endometrial vascular wall [6]. Oral immediate-release tranexamic acid use improves symptoms associated with HMB; however, this medication can be associated with a high level of gastrointestinal adverse effects [7]. Recently, Lysteda, designed to maximize efficacy while minimizing gastrointestinal adverse effects, was approved for the treatment of cyclic HMB in the USA.
Chemistry & pharmacodynamics
Mechanism of action
Tranexamic acid is a negatively charged synthetic antifibrinolytic lysine derivative that is the trans-stereoisomer of 4-(aminomethyl) cyclohexane carboxylic acid

Tranexamic acid.
Effect on fibrinolysis
Manifested by a reduction in the lysis of fibrin, tranexamic acid has been shown to decrease the activity of tissue plasminogen activator (tPA) and plasmin. Elevated concentrations of endometrial, uterine and menstrual blood tPA are observed in women with HMB compared with women with normal MBL [3]. This increase in fibrinolysis in women with HMB is most likely due to higher levels of endometrium-derived plasmin and plasminogen activators. The effect of tranexamic acid on lowering endometrial tPA activity and menstrual fluid fibrinolysis has been reported in women with HMB receiving oral doses of tranexamic acid for 5 days [3].
Pharmacokinetics & metabolism
The absolute bioavailability of tranexamic acid, as a standard immediate-release tablet, has been reported to be approximately 34% in healthy males [9]. Following administration of oral tranexamic acid, peak plasma concentrations are reached within 3 h. At therapeutic plasma concentrations, tranexamic acid is minimally bound to plasma proteins (3%), with a relatively small distribution volume. Less than 5% of a dose is metabolized. The main route of elimination of tranexamic acid is through the kidneys, with at least 95% of tranexamic acid excreted unchanged through the urine and an apparent elimination half-life of approximately 2 h [10]. Dosing of tranexamic acid should be reduced with renal impairment [6].
Clinical efficacy
Phase I trials
The pharmacokinetics (PK) of Lysteda were investigated in three randomized, single-dose studies: a bioavailability and bioequivalence study under fasting conditions in 28 women (mean age: 35 years) [11], a comparative bioavailability study under fed-to-fasting conditions in 28 women (mean age: 33 years) [12] and a comparative steady-state PK assessment under fasting conditions in 40 women (mean age: 33 years) [12]. A fourth study was performed as a single-dose, double-blind, four-way crossover study to evaluate the effect of Lysteda on ventricular repolarization in 48 healthy women (mean age: 32 years) under fasting conditions [10]. PK assessment in the adolescent population has not been reported.
The results of the Phase I studies are important as the PK profile of tranexamic acid has not been adequately characterized in women. PK parameters after single and multiple doses of Lysteda under fasting conditions are shown in
Pharmacokinetic parameters following single and multiple oral doses of Lysteda under fasting conditions.
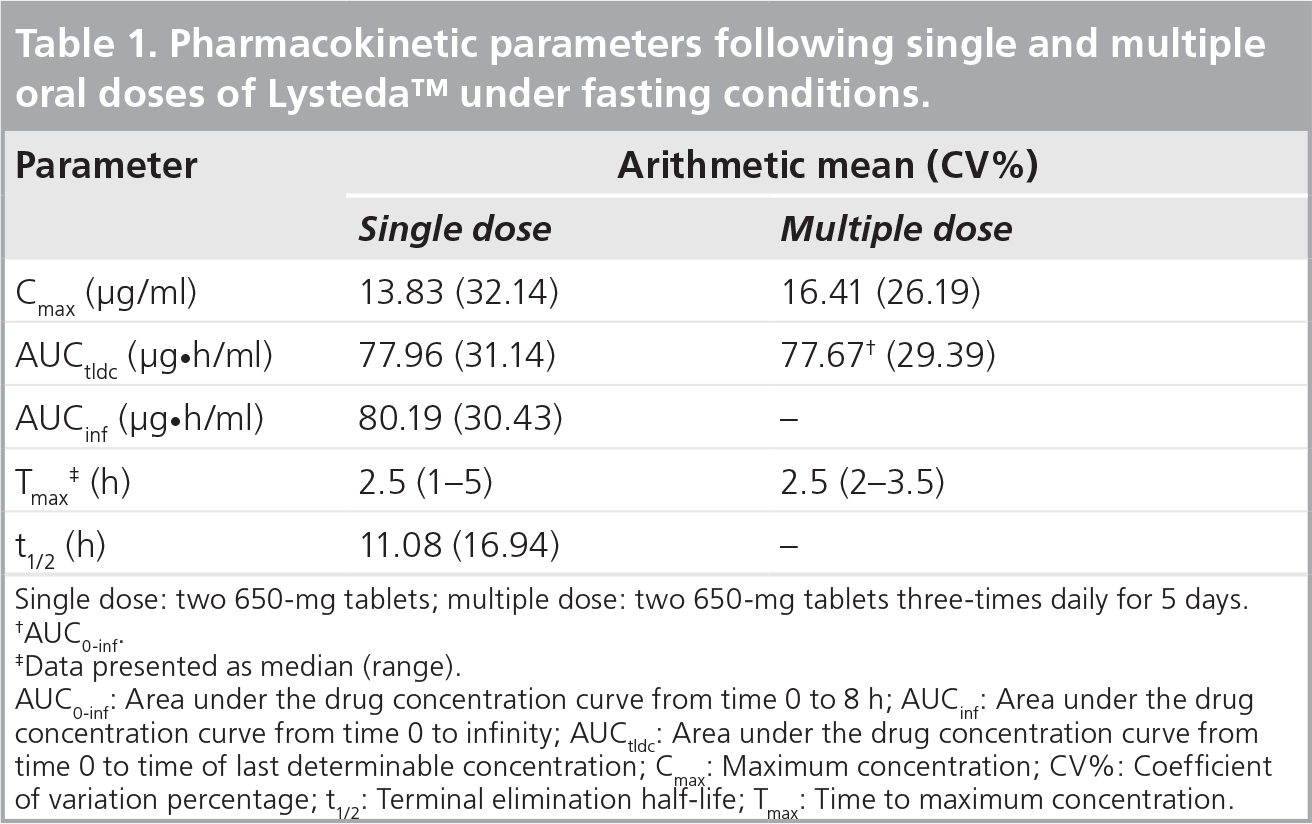
Single dose: two 650-mg tablets; multiple dose: two 650-mg tablets three-times daily for 5 days.
AUCo-inf.
Data presented as median (range).
AUC0-inf: Area under the drug concentration curve from time 0 to 8 h; AUCinf: Area under the drug concentration curve from time 0 to infinity; AUCtldc: Area under the drug concentration curve from time 0 to time of last determinable concentration; Cmax: Maximum concentration; CV%: Coefficient of variation percentage; t1/2: Terminal elimination half-life; Tmax: Time to maximum concentration.
A thorough QT prolongation study was performed to evaluate the effect of therapeutic and supratherapeutic doses of Lysteda on ventricular repolarization [10]. Lysteda (1.3 and 3.9 g), positive-control moxifloxacin and placebo-control doses were administered at each period and plasma drug concentrations and PK profiles were evaluated simultaneously over 24 h postadministration. The QTc interval for both doses of Lysteda was not prolonged compared with the placebo group. There was no relationship between adjusted QTc intervals and plasma tranexamic acid concentrations for either dose. Overall, cardiac repolarization was not influenced by Lysteda at the doses studied.
Phase III trials
The efficacy of Lysteda has been evaluated in two Phase III, double-blind, placebo-controlled, randomized trials in the treatment of HMB [13,14]. Generally healthy women, aged 18–49 years, with cyclic HMB (defined as mean MBL of at least 80 ml/cycle) were randomized to receive Lysteda or placebo. One study spanned three consecutive menstrual cycles and included 297 women who were assigned to receive at least one dose of Lysteda 3.9 g/day (n = 115), Lysteda 1.95 g/day (n = 115) or placebo (n = 67). The second study spanned six consecutive menstrual cycles and included 189 women who were assigned to receive at least one dose of Lysteda 3.9 g/day (n = 117) or placebo (n = 72). Study medication was self-administered three-times daily during menstruation for up to 5 days/cycle (maximum of 15 doses).
The primary efficacy variable was a change in MBL as measured by a validated alkaline hematin method that quantitatively assesses the blood component of menstrual loss although not menstrual fluid volume [15]. The three-part primary end point was achieved if mean MBL reduction from baseline with Lysteda treatment was significantly greater than that of placebo, mean MBL reduction from baseline exceeded a threshold that was perceived as meaningful to subjects and the mean change in MBL with Lysteda treatment exceeded 50 ml/cycle.
A receiver operating characteristic curve analysis was performed to determine the minimal reduction in MBL that would be considered meaningful or important to women with HMB; it was determined that reducing MBL by 36 ml/cycle (approximately 22% from baseline) was considered to be a meaningful improvement [16]. In addition, a validated patient-reported outcome instrument, the Menorrhagia Impact Questionnaire, was used to measure changes in health-related quality of life aspects. Specific parameters that were measured included limitations of social or leisure activities, physical activities, work outside or inside the home and perceived MBL reduction [17].
Treatment with Lysteda 3.9 g/day exceeded all three prespecified primary end points in both studies
Analysis of end points in Phase III, randomized controlled trials.

LPA: Limitation in physical activities; LS: Least squares; LSLA: Limitation of social or leisure activities; MBL: Menstrual blood loss.

Menstrual blood loss levels over duration of therapy.
Health-related quality of life aspects were improved in subjects taking Lysteda 3.9 g/day beginning with the first treatment

Health-related quality of life using select items from the Menorrhagia Impact Questionnaire.
Treatment with Lysteda 1.95 g/day did not meet the prespecified tripartite primary end point; notably, the mean reduction in MBL was not 50 ml or greater [14]. As a result, the 3.9 g/day dose is considered the lowest effective dose of this novel formulation of tranexamic acid for this indication of HMB.
Safety & tolerability
The safety of Lysteda was assessed in eight clinical studies (four in healthy volunteers and four in patients with HMB). These studies included four Phase I studies: a bioavailability study [11], a fed-and-fasting bioavailability study [12], a steady-state PK study [12] and a thorough QT study [10]. In addition, three Phase III studies were also included: a three-cycle efficacy study [14], a six-cycle efficacy study [13] and a 27-cycle, open-label safety study [19]. A nine-cycle, open-label extension study that enrolled subjects from the three-cycle and six-cycle efficacy studies has been completed. Although results have not yet been published, data on safety and tolerability appear to be comparable with the other Phase III studies.
The combined safety results from the Phase I clinical studies indicate a favorable adverse event (AE) profile for Lysteda. A total of 308 AEs were reported by 70 of 123 subjects in the three PK studies. The AEs reported in more than 10% of subjects in these studies were headache (n = 30), dizziness (n = 22) and nausea (n = 19). None of these reports were significant compared with placebo. Dyspepsia was less common with oral Lysteda (3%) versus intravenously administered Lysteda (11%; p = 0.02). No deaths or serious AEs occurred during the three PK studies. Nausea was reported less frequently with Lysteda (zero of 27 subjects) compared with an immediate-release formulation administered orally (two of 27 subjects) [11]. One subject was withdrawn from the fed-and-fasting bioavailability study due to AEs (vaginal infection, loose stools, vomiting and nausea) that were considered to be unrelated to study drug.
A total of 125 AEs were reported by 37 subjects in the thorough QT study. The most frequently reported AEs were headache (n = 21 subjects), nausea (n = 10) and vomiting (n = 7). One subject was withdrawn from the thorough QT study due to a serious AE of endometriosis that was considered to be unrelated to the study drug.
The incidence of treatment-emergent AEs reported in the three-cycle and six-cycle studies was similar among subjects receiving Lysteda and placebo. In the three-cycle study, the most frequent treatment-emergent AEs were viral upper respiratory tract infection (n = 8), musculoskeletal pain (n = 6) and myalgia (n = 6). In the six-cycle study, the most frequent treatment-emergent AEs included sinus headache (n = 9), abdominal pain (n = 8), musculoskeletal pain (n = 8) and pain in extremity (n = 8). Summaries of the most common AEs reported for subjects treated with Lysteda during either the three-cycle or six-cycle efficacy study are provided in
Adverse events reported by more than 5% of subjects treated with Lysteda and more frequently in Lysteda-treated subjects.

Includes headache and tension headache.
Nasal and sinus symptoms include nasal, respiratory tract and sinus congestion, sinusitis, acute sinusitis, sinus headache, allergic sinusitis and sinus pain and multiple allergies and seasonal allergies.
Abdominal pain includes abdominal tenderness and discomfort.
Musculoskeletal pain includes musculoskeletal discomfort and myalgia.
Arthralgia includes joint stiffness and swelling.
Treatment-emergent adverse events occurring in 3% of subjects in any treatment group by system-organ class and preferred term.

The safety and tolerability profile in the open-label safety study of Lysteda in women with HMB was conducted over 27 cycles. This is the longest safety study of a tranexamic acid formulation published to date. A total of 723 women, aged 18–49 years, with a history of cyclic HMB were assessed. Nearly a third (n = 239) remained in the study throughout all 27 on-treatment menstrual cycles. Primary reasons for withdrawal included failure to return (n = 156), subject request unrelated to study (n = 117), other event (n = 112) and AE or serious AE (n = 97). The majority of AEs were mild or moderate in intensity. Treatment-emergent AEs most commonly reported across the duration of the 27-cycle study included headache (n = 437), menstrual discomfort (n = 360) and back pain (n = 227). The incidence of treatment-related gastrointestinal treatment-emergent AEs was low and no evidence of ocular toxicity or prothrombotic effects was observed.
A total of 1276 women received Lysteda in the eight clinical trials leading up to the approval of the product. A total of 13,116 menstrual cycles were managed with Lysteda. No thrombotic or thromboembolic events were reported in any of the eight clinical studies in women who received Lysteda. One incident of deep venous thrombosis (DVT) occurred in the six-cycle study in a woman who received placebo.
Taken together, the safety data generated using the novel formulation of tranexamic acid suggest that a dose of Lysteda 3.9 g/day (dosed 1.3 g three times a day or every 8 h) for up to 5 days during menstruation is safe in women with HMB associated with menorrhagia for up to 27 menstrual treatment cycles.
Postmarketing surveillance
A pediatric study is currently underway assessing the PK of Lysteda in healthy adolescent patients, aged 12–17 years, with HMB. The US FDA provided a waiver on a pediatric study for patients aged 0–11 years, because Lysteda is not indicated for use in premenarcheal children. A pharmacoepidemiologic study is to be initiated to assess the patterns of concomitant use of Lysteda and hormonal contraceptives, including an evaluation of the ages of women using both products as compared with women using Lysteda alone.
Regulatory affairs
Lysteda is approved in the USA for the treatment of cyclic HMB. Immediate-release tranexamic acid is approved in more than 80 countries and, in addition to treating HMB, it is also approved to treat hemorrhage following surgery (e.g., cardiac surgery, liver transfusion, orthopedic surgery and cesarean section), hemophilia patients with factor VIII or factor IX deficiency who have oral mucosal bleeding or are undergoing oral surgery, and hereditary angioedema and localized bleeding secondary to hyper fibrinolysis including epistaxis and hyphema [103].
Conclusion
Lysteda is an effective and safe nonhormonal treatment for HMB that is used during menstrual bleeding only. The novel oral formulation provides reduced gastrointestinal side effects (i.e., less dyspepsia) and allows for less frequent dosing (four times a day to three times a day) given the higher per-tablet dosage. Even though it is technically a hemostatic agent that can theoretically induce hypercoagulability, present experience has not been associated with an increased risk of thrombosis.
Future perspective
Given the other worldwide uses of immediate-release tranexamic acid [7,103], this novel formulation of tranexamic acid has widespread potential for other indications.
We look forward to the results of the PK assessment of Lysteda in the adolescent population and we are indeed interested to see whether its efficacy and favorable side-effect profile is the same in adolescents with HMB. We also welcome studies of its use in intuitively more advanced cases of HMB associated with underlying bleeding disorders such as von Willebrand disease. Recent results suggest that the standard immediate-release formulation of tranexamic acid can be effective in this often difficult-to-manage population [20].
Executive summary
Tranexamic acid diminishes the dissolution of hemostatic fibrin by saturating the lysine receptor sites of plasminogen and preventing binding to fibrin monomers, thus preserving and stabilizing fibrin's matrix structure.
Plasma concentrations of tranexamic acid reached putative levels (≥5 μg/ml) within 1.5 h after dosing with Lysteda.
Minimal accumulation is observed after repeated dosing of Lysteda to steady-state conditions.
Lysteda is an efficacious and durable therapy for heavy menstrual bleeding and improves women's health-related quality of life.
Menstrual blood loss is reduced by 39–40% with Lysteda 3.9 g/day compared with 2–8% reductions with placebo in Phase III controlled trials.
Adverse events frequently reported in clinical studies were consistent with those observed with the use of an immediate-release formulation of tranexamic acid and included menstrual discomfort, headache and back pain.
The majority of adverse events, with short- or long-term exposure to Lysteda, were mild or moderate in intensity.
No drug–drug interaction studies were conducted with Lysteda.
Caution should be exercised with concomitant use of Lysteda and hormonal contraceptives, factor IX complex concentrates or anti-inhibitor coagulant concentrates, tissue plasminogen activators and
During menstruation, the recommended dose of Lysteda is 1.3 g (two 650-mg tablets) taken three-times daily for a maximum of 5 days without food or drink restrictions.
Footnotes
