Abstract
Colorectal cancer (CRC) is the third most common cause of cancer in women. Screening for CRC increases early detection of cancer and premalignant polyps and decreases morbidity from this disease. However, adherence to the screening guidelines continues to remain inadequate both at the physician and patient levels. Several factors are of special importance to women. Presence of prior gynecological malignancies may increase the risk of CRC in women. Furthermore, new studies have shown other factors such as obesity and smoking to increase the risk of CRC in women. This article highlights issues unique to women with regards to CRC and outlines special considerations for determining screening intervals in women, identifies factors that make screening more difficult in women, and reviews studies that identify preventative strategies which, together with screening, may reduce the burden of CRC.
Keywords
Colorectal cancer (CRC) is the third most commonly diagnosed cancer in women in the USA, accounting for 10% of new cancer cases and 9% of cancer deaths [1]. The American Cancer Society estimates for women in 2010 include 70,480 new cases of CRC and 24,790 deaths from CRC [1,201]. The lifetime risk of being diagnosed with invasive CRC among women in the USA is currently 5.03% (one in 20) [1]. Stratification by age shows the risk of development of invasive CRC to be the highest in the age group 70 years or older (one in 24) followed by women from 60 to 69 years of age (one in 94) [1]. The overall CRC incidence and mortality rate is approximately 35% higher in men than in women. A study that looked at 10-year incidence and mortality of CRC among men and women at ages 50, 55 and 60 years found that women reached comparable levels of risk for CRC approximately 4–8 years later than men [2]. Studies have reported that the overall prevalence of advanced neoplasia among men and women with a negative fecal occult blood test (FOBT) with no family history of colon cancer in the 50–79 year age range was also higher in men (8.6%) than in women (4.5%) [3].
The incidence of CRC has decreased in both sexes between 1998 and 2006 with the reduction being 2.2% per year in women [1,201]. From 2001 to 2006, CRC incidence and mortality rates per hundred thousand women in the USA have been highest among African–Americans [1,202]. From 2002 to 2006 the overall incidence (per 100,000 population) in the USA in African–American women was 51.7 with an incidence of 42.6 in white subjects and 35.1 in Hispanic/Latinos. The higher incidence and mortality rates among African–American women suggests the presence of several possible factors that contribute to this increased risk [1,202]. These could include a genetic predisposition to developing CRC, the presence of increased risk factors such as obesity or smoking, or the impact of socio-economic or cultural factors including inequalities in access to or utilization of screening tests among African–Americans. Reduced access to quality treatment such as surgery and adjuvant chemotherapy may impact mortality in this group [1,4].
Identification of factors related to CRC that are unique to women is important as it might improve our current screening practices and suggest other strategies that could impact primary prevention of CRC specific to women. This article will focus on screening and prevention issues in CRC that are important to women. Discussion of all familial CRC syndromes is beyond the scope of this article.
CRC screening
Rationale
The rationale behind CRC screening is to remove premalignant polyps before they become cancer and to detect cancer at an early stage, thereby permitting early initiation of treatment and achieving better cure rates. Furthermore, it has been shown that removal of premalignant lesions will decrease the risk of CRC [5]. The incidence rate of CRC in women has been decreasing gradually since 1985 and has declined 2.2% per year from 1998 to 2006. A gradual decline in the mortality due to CRC in women had also begun since 1975. However, recently there has been a more dramatic decline in CRC-related mortality in women with a reported decline of 3.4% per year between 2001 and 2006 [1]. Historically, the period spanning the last three decades has seen a dramatic increase in the use of nonsteroidal anti-inflammatory drugs as well as HRT in woman, which could be contributing to the observed decline in incidence and mortality from CRC in women. Also, importantly, this period saw an increased utilization of CRC screening, which has meant improvement in early detection of CRC when cancers are in an earlier stage, and removal of premalignant lesions. Many different screening tools have been utilized. FOBT [6,7], screening flexible sigmoidoscopy directly [7,8] and colonoscopy indirectly [8] have all been shown to reduce mortality from CRC in the total population. Presently, computed tomography (CT)–colonography, although available in many locations, is not recommended as part of screening by the US Preventive Services Task Force (USPSTF) owing to its inability to detect colonic polyps of less than 6 mm in size [9,10], and to the concern regarding potential harm associated with the radiation dose from repeat CT examinations in the long run [11]. Discussion of other tests besides the above screening tests such as barium enema with air contrast and stool DNA test are beyond the scope of the subject of this review. These screening tests have been most recently reviewed in detail by Lieberman [12]. The current screening guidelines in the USA (same for both men and women) for an average-risk individual without a family history of CRC, multiple polyps or other factors increasing the risk for CRC are summarized in
Screening for colorectal cancer and adenoma detection for average-risk women and men aged 50 years and older.
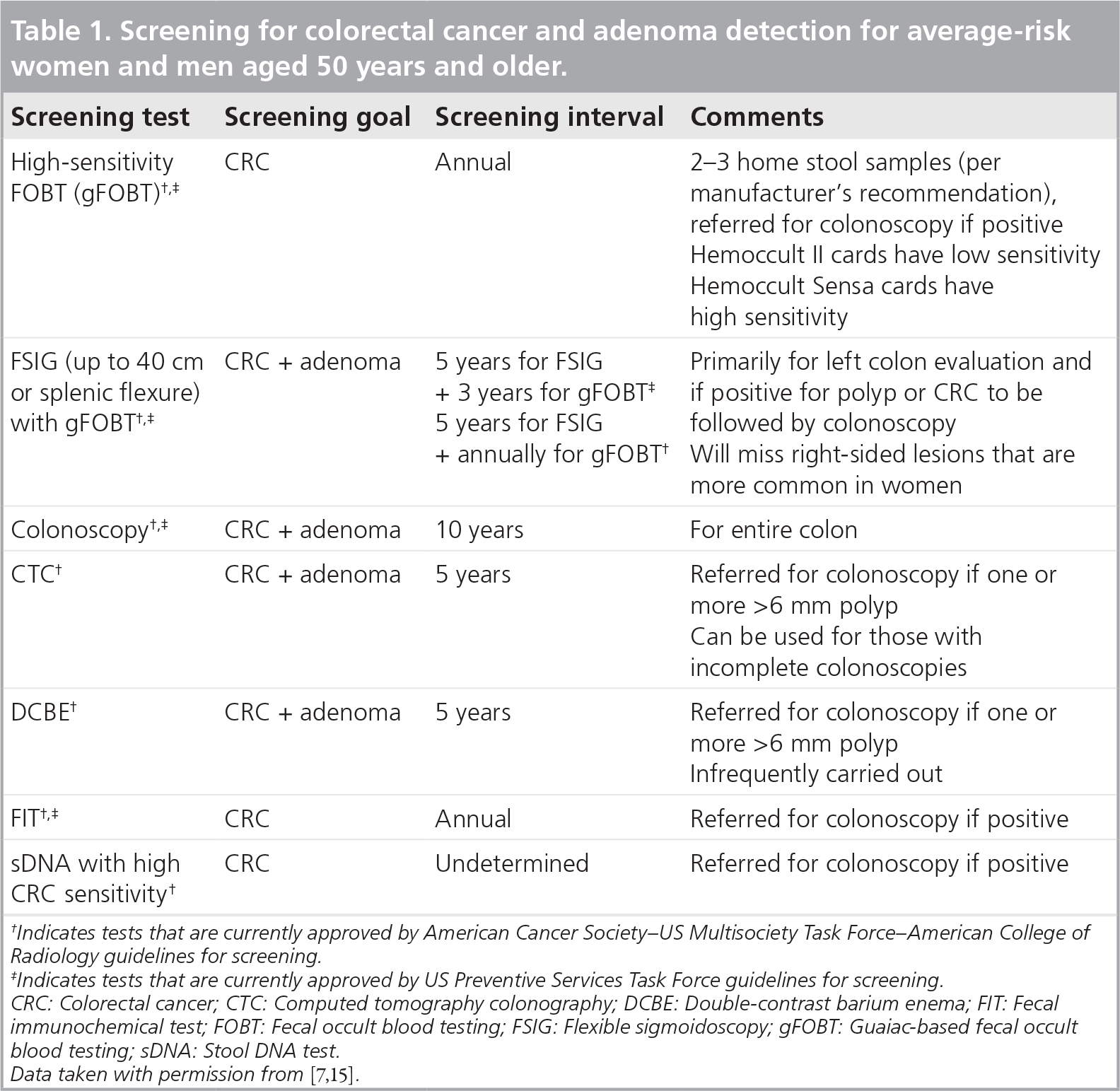
Indicates tests that are currently approved by American Cancer Society–US Multisociety Task Force–American College of Radiology guidelines for screening.
Indicates tests that are currently approved by US Preventive Services Task Force guidelines for screening.
CRC: Colorectal cancer; CTC: Computed tomography colonography; DCBE: Double-contrast barium enema; FIT: Fecal immunochemical test; FOBT: Fecal occult blood testing; FSIG: Flexible sigmoidoscopy; gFOBT: Guaiac-based fecal occult blood testing; sDNA: Stool DNA test.
CRC screening considerations for women
All of the screening modalities show a higher prevalence of polyps and malignant masses in men than in women, although at the older ages the rates are almost the same. The Clinical Outcomes Research Initiative showed that in average-risk individuals aged 50–59 years, the number needed to colonoscope to identify one person with a mass or polyp greater than 9 mm was 28 for women and 18 for men [16]. Eloubeidi et al. found that the likelihood of screening flexible sigmoidoscopy detecting a polyp or cancer was 4.84% in women compared with 8.47% in men [17]. Even though women have fewer CRCs and significant polyps than men, screening for CRC is just as important for women as men (reviewed in [11]) and will likely continue to impact the decreasing rate of CRC. At this time the age of initiating screening is the same for women and men, which in average-risk individuals with no family history of CRC is 50 years of age (45 years of age for African–Americans recommended by some) and in people with one first-degree relative (or two second-degree relatives) 40 years of age, or 10 years younger than the earliest cancer or colonic polyp (although recommendations vary). Whether or not initial screening at an older age for women will be recommended in the future will have to await further information. For a detailed overview of the current US screening guidelines, please refer to [5,13].
Increased risk for CRC occurs in asymptomatic (and symptomatic) women with a history of previous reproductive tract cancers and requires special consideration for the frequency of CRC screening [18]. Weinberg et al. found that endometrial cancer and ovarian cancer increase the risk of CRC. This increase occurs when uterine cancer was diagnosed before the age of 50 years (standardized incidence ratio [SIR]: 3.39). Similarly, ovarian cancer increases the risk of CRC when diagnosed before 65 years of age (from 50 to 64 years; SIR: 1.52) and increases the risk further if ovarian cancer is diagnosed before 50 years of age (SIR: 3.67). No increased risk for CRC occurred with a personal history of cervical cancer [18].
Reports on the effect of breast cancer on the risk of a woman developing CRC or adenomatous polyps have been controversial. Two reviews in 1994 initially estimated the relative risk (RR) for CRC in women with breast cancer as 1.1 [19,20]. In 1998, a clinical expert panel concluded that the increased risk of CRC in a woman with a breast cancer history was similar to that in a person having a first-degree relative with CRC [21]. Subsequently, Newschaffer et al. used the Surveillance Epidemiology and End Results database from 1974 to 1995 to estimate the risk of CRC after breast cancer diagnosis [22]. They reported that there was no increased risk of CRC overall and in fact there was a reduction in rectal cancer, most prominent among African–American women. Recent studies of patients with a BRCA gene mutation for breast cancer did not find an increase in associated CRC cancer [23,24]. In a study of Ashkenazi Jews with BRCA1 and BRCA2, no increase in the incidence of CRC among relatives of carriers of these genes was noted [25]. More recently, Trentham-Dietz et al. examined the risk of a second neoplasm after the diagnosis of invasive breast cancer. The risk of developing CRC as the second neoplasm was decreased by postmenopausal hormone use prior to the cancer diagnosis (hazard ratio: 0.4; 95% CI: 0.3–0.7) and infrequent alcohol consumption (p = 0.01). There was a possible increase in the risk of CRC with a family history of two or more relatives with breast cancer (hazard ratio: 1.75; 95% CI: 0.81–3.79) but there were only seven women with CRC with such a history. No increase in CRC was seen in women with only one family member with a breast cancer [26]. Thus, current literature, although not conclusive, does not favor an association between breast cancer and an increased risk of CRC.
Special mention must be made of a genetic condition called Lynch syndrome, also known as hereditary nonpolyposis CRC (HNPCC). It is an autosomal-dominant disorder that runs in families, and is one of the most common inherited syndromes associated with colon cancer susceptibility. It is also associated with endometrial cancer (40%) and other cancers involving small bowel, ovaries, ureter, stomach, pancreas and renal pelvis. Current guidelines place individuals with a genetic or clinical diagnosis (based on a family history with or without a personal history) of HNPCC under a high-risk category for the development of CRC and recommends earlier and more frequent screening (colonoscopy every 1–2 years, starting at 20–25 years of age) [27]. However, identification of women at risk in an asymptomatic patient is challenging. Given the increased familial risk of CRC and other associated cancers in this condition, it is important to obtain a detailed family history to identify genetic syndromes requiring even more frequent screening. In 1999, the International Collaborative Group on HNPCC proposed the revised Amsterdam Criteria (Amsterdam II criteria) for diagnosis of Lynch Syndrome [28].
These criteria propose suspicion of Lynch syndrome in individuals with all of the following in three or more relatives with established diagnosis of Lynch syndrome-associated cancers, as mentioned above: one relative being a first-degree relative of the other two in whom other causes of colon cancer such as familial adenomatous polyposis is ruled out; presence of Lynch syndrome-associated cancers in at least two successive generations; and one or more relatives with early diagnosis of CRC (before age 50 years). Subsequently, with improvement in our understanding of the genetics of CRC, diagnosis of Lynch syndrome was defined to only include people with a mismatch repair gene mutation. However, only approximately 50% of individuals who met the Amsterdam II criteria were found to have Lynch syndrome based on the new definition. In fact, individuals who fulfilled the Amsterdam II criteria but did not have mismatch repair mutations appeared to have an overall lower risk of CRC [29]. Therefore, newer criteria were clearly needed for identifying individuals with Lynch syndrome. The revised Bethesda guidelines proposed in 2004 take into account the genetic component associated with Lynch syndrome [30]. The Bethesda guidelines recommend that CRC specimens from patients should undergo testing for microsatellite instability under the following circumstances [30]:
The diagnosis of CRC is made at less than 50 years of age;
The patient has either the presence of synchronous CRC or metachronous CRC, or other HNPCC-associated cancers (described above), regardless of age;
CRC is associated with the microsatellite instability high (MSI-H)-like histology (such as tumor-infiltrating lymphocytes) in a patient under 60 years of age;
CRC is diagnosed in an individual who has one or more first-degree relatives with an HNPCC-associated tumor (described above), with at least one cancer diagnosed under 50 years of age;
CRC is diagnosed in an individual with two or more first- or second-degree relatives with HNPCC-associated tumors (age independent). Screening of women with HNPCC should be based on the current guidelines [5].
From currently available information, women who have had an ovarian or endometrial cancer diagnosed at an age greater than 50 years without HNPCC and those women who have had a personal history of breast cancer should undergo average-risk CRC screening if there is no other reason that warrants more frequent screening [18,22,31]. Recently, Lansdorp-Vogelaar et al. addressed the question of whether screening guidelines should be individualized by race and sex. They used a MISCAN-colon microsimulation model to estimate life expectancy and lifetime CRC screening and treatment costs in a US cohort of black and white men and women at average risk for CRC. The various comparison groups were: no screening, uniform 10-year screening, uniform 8-year screening from age 50 years and individualized screening according to sex and race. The uniform 10-year strategy was dominated by the uniform 8-year strategy and individualized screening. Both the uniform 8-year screening group and the individualized screening by race and sex groups showed comparable costs and life expectancy gains, suggesting that individualized screening by race and sex brought only marginal benefits at the population level [32]. Frequent routine screening for endometrial and ovarian cancers in women with Lynch syndrome is essential and is discussed elsewhere [27].
CRC screening rates in women
Although the importance of screening for CRC is well known and various screening options are available, data derived from surveys have indicated that adherence to CRC screening guidelines in the USA is poor for both men and women, particularly in nonwhite groups. The National Health Interview Survey 2008 estimated that of women aged 50 years and older in 2008 only 9.7% had used home FOBT tests within the past year and 48.6% had an endoscopy test (including flexible sigmoidoscopy within the past 5 years or colonoscopy within the past 10 years). A total of 52% of women in this age group had either FOBT within the past year, flexible sigmoidoscopy in the past 5 years or colonoscopy in the past 10 years [33]. A state-wise difference was also observed with CRC screening in 2008, with Maine reporting the highest overall screening rates (combined for either FOBT in the past year or endoscopic screening within the past 10 years) in both sexes of 74% and Nevada reporting the lowest screening rate of 54.7% [33].
When broken down by sex, studies that assessed test-specific CRC screening adherence have found that 4–21% more men than women self-report having had a sigmoidoscopy or colonoscopy [34–36] and findings are similar for comparisons using medical record data [37]. However, several studies reported higher adherence to FOBT-based screening in women [38–40].
Women who undergo either Papanicolaou smears or mammography screening for breast cancer are more likely to undergo CRC screening than those women who did not undergo those tests [41]. Having had a mammogram within 2 years or a Papanicolaou smear within 3 years increased the likelihood of CRC screening from 24 to 60.6% and 33.3 to 56%, respectively [41]. In 2004 an increased rate of CRC screening in the over 16,000 women who participated in the Women's Health Initiative and who were on estrogen and progestin was seen compared with the rate in the general population [42]. It is encouraging that progress has been made recently in CRC screening but as of now, none of the 50 states met the American Cancer Society's 2015 goal of 75% screening of adults older than 50 years of age [33].
Barriers that prevent women from screening for CRC
There are several possible causes for why women do not undergo colonoscopy. Initial studies showed that a total of 30–48% of women prefer a female doctor [41,43,44]. This preference occurs more commonly in women who are employed (odds ratio [OR]: 2.53) and in women who have a female primary care physician (OR: 3.40) [43]. Women who prefer female endoscopists are more willing to wait for a procedure to be performed by a female endoscopist (79–89%) and are more likely than men to prefer the same doctor previously seen in an outpatient office visit to perform the procedure (43 vs 32%). However, only a small percentage of women (5%) would exclusively use a female endoscopist [43,44]. Recently, Schneider observed a greater gender preference amongst women compared with men (42.3 vs 21%; p < 0.001). Multivariate analysis showed that along with female gender, lower income level and history of physical/emotional abuse were found to be significant factors for gender preference. A history of abuse was shown to significantly increase the percentage of patients with a gender preference for the endoscopist (p < 0.001) [45]. Most recently, Denberg et al. studied whether sending out invitations to undergo CRC screening offering the option of a female endoscopist would affect the rates of screening. However, they found that although more women (44.2%) who received the invitation containing the option to request a female endoscopist were likely to request a female endoscopist than those who did not receive an invitation (4.8%; with p-values that were significant), the women who were offered a female endoscopist were not more likely to undergo screening than those who were not offered this option [46]. Thus, clearly, further studies are awaited to determine if screening rates in women could be affected by the sex of the endoscopist.
Several studies have looked at the barriers that limit screening for CRC. Among all people who have never been screened before, the common factors include a lack of knowledge regarding the importance of CRC screening for detecting and removing premalignant polyps, the knowledge that there is an improved chance of achieving cure if cancer is detected early, and the lack of awareness that there are several available testing options. Survey participants also cite reasons such as inconvenience and lack of time, cost of the procedure, fear of being diagnosed with cancer, and unpleasantness and embarrassment associated with the test [40,47,48,202]. Lack of effective communication between physicians and patients about CRC screening is considered to be an important barrier to achieving the goals of screening [46,49]. Lack of health insurance is also an important reason for avoiding CRC screening in both genders. With regards to women specifically, Stockwell et al. showed in a study based at five health centers in the Boston area that in those women who did not screen for CRC, the lack of a recommendation from their doctors was the most significant reason (36%). Other reasons given were fear of pain or discomfort (25%), embarrassment (14%), inconvenience (11%), cost (3%) and fear of results (2%) [41]. Previous history of sexual abuse may be another reason for women to avoid colonoscopy. A history of abuse may increase the anxiety related to the procedure, thereby increasing the requirement of more sedative medication during the procedure, and may contribute to patient satisfaction following the procedure. The colonoscopy itself may revive memories of abuse [Wolf JL, Unpublished Data].
Comparison of screening techniques in women versus men: flexible sigmoidoscopy & colonoscopy
Several studies have shown that flexible sigmoidoscopy and colonoscopy are easier to perform in men than women and are tolerated better by men. Flexible sigmoidoscopy is more commonly limited in women compared with men (45 vs 30.5%) [17]. Causes of this limitation are angulation of the colon and colonic spasm or pain [17,50]. Depth of insertion of less than 50 cm during sigmoidoscopy occurs in up to 23.5% of women, compared with 13.7% of men. Usage of an upper endoscope for screening sigmoidoscopy reduces spasm and pain and allows for increased insertion distance [51]. Similarly, women also tolerate colonoscopy less well than men. Multiple studies have reported that male sex and shorter duration of colonoscopy were associated with better patient tolerance and less pain [52–54]. Female sex was the only factor associated with significant discomfort during the procedure. Only 41% of men found the procedure very or moderately uncomfortable, whereas up to 63% of women found the procedure very or moderately uncomfortable. Men were found to be more willing to undergo colonoscopy without sedation [55]. The USPSTF recognizes the limitations of flexible sigmoidsocopy in identifying neoplasms as well as the seriousness of harms posed by flexible sigmoidoscopy and cautions that randomized controlled trials are awaited before the effects of flexible sigmoidoscopy on health outcomes including mortality from CRC will become clear [11]. Recently, in a large multicenter randomized controlled trial, Atkin et al. studied if one flexible sigmoidoscopy between ages 50 and 64 years would alter the incidence and mortality associated with CRC. They compared two groups (both men and women), an intervention arm that was offered screening by flexible sigmoidoscopy, and a control arm that was not offered screening. The median duration of follow-up was approximately 11 years. They noted that the incidence rate and mortality rate of CRC (per 100,000 person-years) were significantly reduced by 23 and 31%, respectively, in the intention to treat group and these differences were more profound in the per protocol analysis. No sex differences in outcomes were noted in this study [56].
With regards to colonoscopy, whether prior abdominal or pelvic surgeries have an impact on colonoscopy procedural outcome in women has been investigated. Marshall et al. found that in women with a previous hysterectomy, using a pediatric colonoscopy was successful in increasing the completion to cecum rate from 71.4% with the standard colonoscopy to 96.1% [57]. Although Anderson et al. were able to complete colonoscopies in 94.8% of women and 98.2% of men, of those with incomplete examinations 76.8% were women [58]. Hysterectomy and previous pelvic surgery are controversial as causes of incomplete colonoscopy in women. One factor for why colonoscopies are more difficult in women is the colonic length and redundancy of the colon. Saunders et al. estimated that the median colonic length was 10 cm more in women (155 cm) than in men [59]. Most of this was due to the increased length of the transverse colon with a marked increase in redundancy of the transverse colon (62 vs 26%). Sigmoid colon redundancy and possible adhesions due to endometriosis may also be important.
Comparison of screening techniques in women versus men: FOBT, double-contrast barium enema & stool DNA test
Immunological-based FOBT (iFOBT) and guaiac-based FOBT (gFOBT) are widely used for early detection of CRC. Recently, Brenner et al. showed that among 1157 male and 1167 female participants in the German screening colonoscopy program for different qualitative iFOBTs and gFOBT, the sensitivity and positive predictive values were substantially higher, and specificity and negative predictive values were substantially lower among men than among women [60]. When applying the value of test positivity of 2 μg/g stool recommended by the manufacturer of the quantitative iFOBT, the values for sensitivity, specificity and the positive and negative predictive value were 47.6, 85.0, 33.0 and 91.3% among men, and 30.7, 89.5, 19.3 and 94.1% among women. p-values comparing men and women for the above findings were statistically significant (p < 0.05). Sex differences of the predictive values were mostly accounted for by sex differences in the prevalence of advanced colorectal neoplasms (which was found to be higher in men than in women in the study). iFOBT (also called fecal immunochemical test) uses antibodies specific to human blood components and is less prone to false-positive tests than gFOBT. A quantitative iFOBT has high sensitivity for advanced neoplasia (approximately 67%) [61]. It is becoming more popular as a testing method because of higher sensitivity for CRC detection compared with gFOBT and because it uses one-time testing instead of three-time testing used by the standard gFOBT (reviewed in [11]). Two other tests, double-contrast barium enema (to detect colonic polyps) and stool DNA test (mutated DNA derived from tumor that is shed in stools) are also included in screening guidelines. No sex-specific differences with regard to screening outcomes have been described for either and therefore we will not discuss them further.
Should the method of screening for CRC be similar for men and women?
The best screening modality for CRC is the test that maximizes detection while minimizing costs at the same time. The major limitation of flexible sigmoidoscopy is that its range of detection is limited to the left side of the colon. Recently, several studies have studied the location of polyps and CRC in men and women separately and found differences in the localization of polyps and cancer [3,62–64]. McCashland et al. noted that women had a greater risk of developing right-sided and pure right-sided polyps and tumors when compared with men. Overall, however, men developed more polyps and CRCs than women [62]. Schoenfeld et al. examined the predictive value of distal-colon neoplasia for advanced neoplasia in the proximal colon [3]. The findings in women were compared with those in men from the VA Cooperative Study 380 [64]. Both studies had the assumption that if a small adenoma was detected in the distal colon by sigmoidoscopy, a follow-up colonoscopy would have been performed and would have detected any proximal neoplasia. It was found that in women, if flexible sigmoidoscopy had been the only test performed, advanced neoplasia would have been detected in 1.7% and missed in 3.2% of women. Only 35.2% of women would have had their advanced neoplasms detected by sigmoidoscopy alone compared with 66.3% of matched men from the VA Cooperative Study 380 [3,64]. The prevalence of advanced neoplasia among men and women with a negative FOBT and no family history of colon cancer was lower in women compared with men aged 50–59 (2.9 vs 4.7%), although this result was not significant (p = 0.15). This difference achieved significance at 60–69 years of age (5% in women vs 10.6% in men). Again, no significant difference was observed in the age group 70–79 years (11.8% in women vs 10.6% in men). Their conclusion was that even though there were fewer polyps in women than men in the 50–59-year-old group, more polyps would be missed by flexible sigmoidoscopy in women than men and that flexible sigmoidoscopy would be a poor screening tool for women [3].
In other words, colonoscopy is a better tool for CRC screening in women. Recently, in a Canadian study, Singh et al. looked at sex differences and predictors of CRC occurring after a negative colonoscopy [65]. Their study, which included 45,985 individuals (27,379 women) who had an initial negative screening colonoscopy, found that the proportion of CRC, which was located in the proximal colon, after negative colonoscopy was higher in women (62%) compared with men (50%; p = 0.03) in the follow-up period ranging from 6 months to over 5 years after initial colonoscopy. They noted that independent predictors for early or missed CRC defined as within 3 years of a negative colonoscopy were older age of subjects and performance of index colonoscopy by nongastroenterologists [65].
Interval of repeat CRC screening
The current screening interval recommendation following a negative sigmoidoscopy or colonoscopy in men or women is the same. Should men and women be screened differently after an initial negative screen? Limited data are available to predict the comparative risk of the occurrence of a benign or malignant neoplasia following a negative colonoscopy. Rex et al. reported that 5 years after a negative colonoscopy in asymptomatic average-risk individuals, the occurrence of adenomas is 18% in women and 32% in men [66]. However, this result was not statistically significant (OR: 1.92; 95% CI: 0.82–4.53). Imperiale et al. examined the 5-year risk of colorectal neoplasia among 847 persons who had no neoplasia on baseline screening colonoscopy [67]. The RR of developing an adenoma in men versus women was 1.73 (95% CI: 1.22–2.45; 19 vs 11%), whereas the risk of an advanced neoplasia was markedly increased to 2.6% in men compared with a small increase of 0.3% in women with a RR of 8.89 (95% CI: 1.17–67.7). No CRC was seen in this study. A follow-up study by Imperiale et al. was reported in which 1256 subjects with no identifiable adenomas at baseline screening colonoscopy were rescreened after a mean of 5 years [68]. They found no cancers but found that 16% of patients had one or more adenomas. Of the adenomas, 1.3% of subjects had advanced adenomas, of which 52.6% were distal to the splenic flexure. The risk of advanced adenoma, although low, was found to be higher among men than women (RR: 3.31; 95% CI: 1.02–10.8). Most recently, Brenner et al. looked at the 10-year risk of developing colorectal neoplasia among 5181 persons with no neoplasia on baseline screening colonoscopy in Germany [69]. They observed no cases of CRC and noted no difference in the standard prevalence ratio of advanced adenomas in men and women over a mean of 11.9 years postcolonoscopy compared with 8.4 CRC cases that were expected during this time period based on age- and sex-specific prevalence among participants who had not received a colonoscopy. The prevalence of advanced adenoma was also lower overall among subjects who had previous colonoscopies (standardized prevalence ratio [SPR]: 0.42). Adjusted prevalence ratios for detecting an advanced adenoma were 0.34 between 6 and 10 years (significant), 0.38 between 11 and 15 years (significant) and 0.53 beyond 15 years (not significant) among participants with a negative colonoscopy compared with those with no previous colonoscopies. Risk reduction was found to be more pronounced for distal (SPR: 0.28; significant) than for proximal neoplasms (SPR: 0.82; not significant) [69]. Clearly, more data need to be analyzed by sex and ethnic group in order to determine if recommendations for follow-up screening colonoscopies should be the same for everyone with average risk for CRC and whether repeat screening should be 10 years or later after the initial colonoscopy.
Preventative strategies for reducing the risk of CRC in women
The low screening rates for CRC have spurred interest recently in identifying risk factors that could be modified by lifestyle changes for primary prevention of CRC. Furthermore, the concept of chemoprevention has gained momentum recently. The factors that have been shown to modify the risk of colorectal neoplasia in women are summarized in
Factors associated with increased risk of colorectal cancer in women.
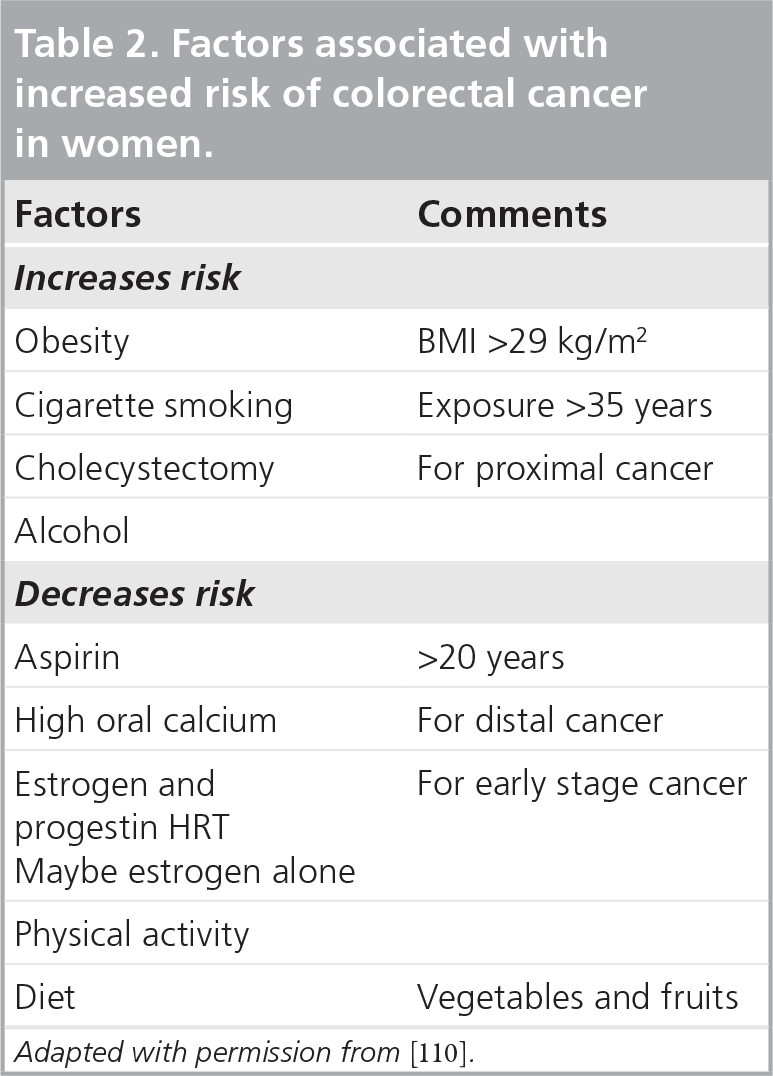
Adapted with permission from [110].
Multiple studies have found an association between colorectal neoplasia and cigarette smoking in both sexes. In addition, an association between smoking and adenomas has been found. In the USA, as many as one in five combined cases of CRC is attributable to cigarette smoking (reviewed in [77]). Lieberman et al. used a multivariate model that included multiple possible risk factors for CRC including smoking and showed that the OR of the effect of smoking on advanced neoplasm (OR: 1.85; 95% CI: 1.33–2.58) was comparable to the OR of developing advanced neoplasm in an individual with a first-degree relative with CRC (OR: 1.66; 95% CI: 1.16–2.35) [78]. The induction period between initial exposure and diagnosis of CRC has been shown to be approximately three to four decades [77]. In a study from the Women's Health Initiative, a pooled analysis of participants in the observational study and three clinical trials, the risk of rectal cancer was increased with longer duration of smoking and older age at smoking cessation. The risk of colon cancer was not found to be increased in this study. However, in this study, the rate of cigarette smoking was lower than age-matched women from the general US population [79]. Similarly, high amounts of alcohol intake have been associated with increased risk of CRC [80].
According to the definition of the US National Cholesterol Education Program Adult Treatment Panel III (2001) and further revised by American Heart Association, metabolic syndrome is characterized by three out of the following factors: central adiposity (waist circumference >102 cm in men and 88 cm in women), hypertension (blood pressure ≥130/85 mmHg), low high-density lipoproteins (<40 mg/dl in men or <50 mg/dl in women), hypertriglyceridemia (150 mg/dl or greater) and concomitant presence of impaired glucose tolerance (fasting serum glucose >100 mg/dl). Several studies have shown an association between colorectal neoplasia with markers of dysregulation of the glucose and insulin metabolic pathways [81,82]. An earlier study had found that women with a BMI greater than 29 kg/m2 had a RR of colon cancer of 1.45 (95% CI: 1.02–2.07) compared with women with a BMI less than 21 kg/m2 [72]. Recent studies have consistently found that the increase of colorectal neoplasia with obesity is approximately 50% in both men and women combined, with the risk being greater in men than women [83]. A meta-analysis by Ning et al. identified 56 studies that included more than 7 million subjects, including 93,812 cases of CRC [84]. They found that a BMI over 23 was associated with increased risk for CRC, with the increase in risk being 14% for a BMI of 23.0–24.9, 19% for a BMI of 25.0–27.4, 24% for a BMI of 27.5–29.9 and the highest increased risk of 41% for a BMI equal to or greater than 30.0 kg/m2. Among subgroups, Asian and premenopausal women had the highest rise in risk beyond a BMI over 23. This association was significantly stronger for colon than rectal cancer, and greater in men than women [84]. Sato et al. studied 15,380 subjects (8225 women) in Japan with mean age 47.3 ± 8.5 years and found that the OR of detection of polyps in subjects with a BMI of 25 or higher versus subjects with BMI less than 25 was significantly higher in men (OR: 1.34) but not clinically significant in women (OR: 1.13) [85]. A study from Korea by Kim et al. that included 1744 asymptomatic patients (798 women) who underwent colonoscopy for cancer screening found a significant positive association between BMI and the prevalence of colonic adenomas and advanced polyps only in relatively young subjects of both sexes (men in their thirties and women in their forties), and in premenopausal but not postmenopausal women [86].
Along the same lines, a recent study found increased prevalence of colorectal neoplasm among patients with newly diagnosed coronary artery disease [73]. However, this increased risk of CRC related to coronary artery disease was not found to be related to sex. An association between greater levels of physical activity and decrease in the risk of CRC has been reported by several studies and was reviewed in detail recently [87]. In the Nurses’ Health Study (NHS), women whose weekly energy expenditure was greater than 21 metabolic-equivalent task hours had a RR of colon cancer of 0.54% (95% CI: 0.33–0.90), compared with women whose weekly energy expenditure was less than 2 metabolic-equivalent task hours per week [88]. A recent meta-analysis that included 52 studies which studied the relationship between physical activity and CRC found that physically active individuals had a 20–30% lower risk of colon cancer compared with individuals who were less active physically [89].
There are several factors that have been found to decrease the risk of CRC. In contrast to encouraging initial results, neither fiber [90,91] nor vitamin E [92,93] appeared to change the incidence of CRC in women. Aspirin usage was shown to be associated with significant reduction in the risk of CRC. In the NHS, women had a significant reduction in risk of CRC after 20 years of consistent aspirin use (RR: 0.56; 95% CI: 0.36–0.90) [94]. An update to the NHS published in 2005 showed that in women taking more than or equal to 2 tablets of 325 mg per week, the adjusted RR for CRC was 0.77 (95% CI: 0.67–0.88) compared with nonregular users. Significant reduction was observed only after 10 years or more of aspirin usage and appeared to be dose related, with the maximum reduction of risk being observed in those taking more than 14 aspirins per week (RR: 0.47; 95% CI: 0.31–0.71). For other nonaspirin NSAIDs, a similar dose–response relationship was observed. However, the risk of major gastrointestinal bleeding was also related to aspirin dose [95]. By contrast, no reduction in risk of CRC following more than an average of 10 years of aspirin usage was noted by the Women's Health Study [96]. It could be argued that the lower dose of aspirin of 100 mg every other day taken by subjects in this study could have partially contributed to this observation.
It was also observed that after taking a multivitamin containing folate for 15 years, women had a decreased risk for proximal colon cancer (RR: 0.16; 95% CI: 0.06–0.52) and distal CRC (RR: 0.37; 95% CI: 0.15–0.90) but not for rectal cancers [97]. Among many substances contained in the multivitamins, folate was believed to be the substance most likely responsible for the observed reduction in CRC. However, recent findings of the Aspirin–Folate Prevention Study have challenged this notion. In this study, patients with a recent history of colonic adenomas were randomized to receive either folate or placebo or further randomized to receive aspirin or placebo. Follow-up colonoscopies showed that folic acid intake was associated with higher risk of having three or more adenomas despite adjustment for age, sex, smoking, alcohol use, BMI, baseline plasma folate and allocation to the aspirin arm [98]. However, it has been argued that the study was carried out over the time when folate fortification of food supply had intensified and therefore resulted in increased basal folate levels in the placebo group as well, thereby nullifying the effect of the treatment arm.
Calcium has also been shown to decrease both the risk of adenoma [99] as well as CRC [100]. In the Calcium Polyp Prevention Study, subjects (including both men and women) taking 1200 mg of supplemental calcium per day had a 20% reduction in risk of recurrent CRC adenomas over 4 years (31 vs 38%) [99]. An inverse association appeared to exist between higher total calcium intake in distal colon cancer in women taking greater than 1250 mg of calcium per day compared with those taking less than 500 mg of calcium per day (RR: 0.73; 95% CI: 0.41–1.27). There was no association found between calcium intake and proximal colon cancer [100]. In this study it was found that supplemental calcium was associated with a lower risk of distal CRC in women only when compared with a woman with a dietary calcium intake of less than or equal to 700 mg per day. Contrary to the above findings, in the Women's Health Initiative study, supplementation with calcium and vitamin D for 7 years was noted to have no effect on the incidence of CRC [101]. However, it could be argued that at enrollment, the women in both groups had a mean calcium intake of 1151 mg and a vitamin D intake of 367 IU, which was almost the recommended dietary intake. Recently it was reported that the protective effect of calcium supplementation on risk of adenoma recurrence extended for up to 5 years after stopping calcium supplementation [102].
Several studies have shown the benefit of HRT in reducing CRC risk and mortality [42,103,104]. The data on the type of HRT that can accomplish this reduction are somewhat controversial. The data from the Women's Health Initiative showed that there was no decrease in the incidence of CRC in women taking equine estrogen alone [105]. However, in women who took estrogen plus progestin there was a decrease in CRC incidence, with an annualized percentage of 0.1 versus 0.16% (hazard ratio: 0.61; 95% CI: 0.42–0.87), and of invasive CRC, with an annualized percentage of 0.09 versus 0.16% (hazard ratio: 0.56; 95% CI: 0.38–0.81) [42]. This reduction appeared to be statistically significant for reduction of colon cancer but not statistically significant for reduction of rectal cancer. However, a negative factor in this study was that although the risk of CRC was reduced by estrogen plus progestin intake, there were an increased number of lymph nodes containing CRC in those women who received hormone therapy versus placebo. A recently reported large retrospective case–control study based on the Surveillance Epidemiology and End Results data found a 20% reduction in CRC risk (95% CI: 0.6–0.9) associated with usage of estrogen plus progestin preparations but not in women who were on estrogen alone preparations [106]. Most of the studies published have noted beneficial effects in reducing CRC risk only with estrogen–progesterone combination preparations. However, a recent study by Hildebrand et al., which used data from the Cancer Prevention Study II Nutrition Cohort to assess associations of CRC with estrogen only versus estrogen plus progesterone usage, found that among current users of estrogen but not estrogen plus progesterone users for over 20 years, there was an associated 45% reduction in risk of CRC (RR: 0.55; 95% CI: 0.36–0.86) [107]. Despite all the support for HRT as chemopreventive agents, currently they are not recommended for CRC chemoprevention because of risks associated with their usage such as increased risk of breast cancer, venous thrombosis and cardiovascular events.
Statins have also been studied as possible chemopreventive agents against CRC after an initial study in 2005 showed an adjusted RR reduction of 0.47 for CRC after 5-year use of statins. However, subsequent studies have not been able to validate this finding [108]. Current evidence therefore does not support the hypothesis that prolonged statin use is protective against CRC.
Recently interest has been generated in exploring the role of diet and risk for CRC. In this regard, Miller et al. compared the dietary pattern of 431 men and women with CRC with that of 726 age-matched controls who did not have CRC and found that a largely plant-based diet with higher intake of fruits, vegetables, whole grains, nuts, seeds, vegetable oils and low-fat dairy in women and fish in men was associated with a reduction in the risk of CRC by 62% in men and 65% in women [109].
Conclusion
The overall prevalence of CRC is greater in men than women with the onset of CRC in women lagging approximately 8 years behind that of men. However, current guidelines for CRC screening do not differ between men and women with screening starting at 50 years of age in asymptomatic average-risk individuals. Issues specific to women that place them at higher risk for CRC should be considered to make screening for CRC more effective. Current literature suggests that women with endometrial or ovarian cancer at a young age should be screened more frequently. Overall screening rates for women remain inadequate. Improvements to the CRC screening rates for women can be achieved by improving the education for primary care physicians such that they adhere to the screening protocols and refer their patients appropriately. Other means of improving screening in women might include the availability of more women colonoscopists and improvement in access to healthcare. Women tolerate colonoscopies and flexible sigmoidoscopies less well compared with men and are more likely to have an incomplete examination than men. Improvements in equipment, such as variable flexible colonoscopes, have helped improve completion of the examination. More data are needed on the recurrence of adenomas in women versus men in order to assess the correctness of identical recommendations of screening intervals in women and men. Women can potentially reduce the risk of colon cancer by stopping smoking, exercising, losing weight (if overweight or obese), taking adequate calcium, ingesting folate (if basal levels of folate are low) and, perhaps in postmenopausal women, by hormonal (estrogen and progestin) therapy.
Future perspective
To reduce the burden of CRC in women, studies must be directed towards better understanding the disease process in women, especially in relation to its association with other gynecologic conditions and the role of hormonal factors as risk factors or protective agents against CRC. Such information can also determine how screening should differ for high-risk women. More research is needed to determine if the delay in onset of polyps and lower risk of polyp recurrence in women compared with men can safely be relayed into a delay in the initial screening and longer follow-up screening intervals. If validated this change in screening could have a potential positive impact on money spent on healthcare. Newer screening tools are also on the horizon and studies looking at their utility in women are awaited. Further identification of genetic factors impacting CRC will be expected to improve our understanding of how the disease develops and thereby offer opportunities to develop strategies for primary prevention. Attention to strategies for weight loss, and new and optimized chemoprevention may help to further erode the rate of CRC.
Executive summary
The overall prevalence of colorectal cancer (CRC) is higher in men than women and the onset of CRC in women lags approximately 8 years behind that of men.
Current screening recommendations for individuals with average risk for CRC are the same for men and women starting at 50 years of age.
Current literature favors earlier and more frequent screening of women with endometrial or ovarian cancer, both of which have been associated with increased risk for CRC.
Individuals with Lynch syndrome require earlier and more frequent CRC screening than average-risk individuals.
Improving education of primary care physicians about screening recommendations is important to improve screening rates in women.
The best tool for screening for CRC in women remains undetermined and further studies are awaited in this regard. However, colonoscopy appears to be the best endoscopic screening option.
Lifestyle modification such as weight reduction, physical activity, smoking and alcohol cessation and chemoprevention with aspirin and calcium are associated with reduction of risk for CRC.
The role of hormone replacement in modifying the risk for CRC remains unclear.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
