Abstract
Objective
We sought to evaluate the presentation and outcomes of patients with a diagnosis of colorectal cancer (CRC) at an academic Canadian center to identify strategies to improve the existing screening system for CRC.
Setting
Royal Alexandra Hospital, Edmonton, Alberta, Canada.
Methods
We performed a prospective cohort study. Data collected included: patient demographics, presentation, treatment, and outcomes 1 year after study completion.
Results
One hundred consecutive patients were included with a median age of 68 years (SD = 13.3). Most (58%) participants were male and 25% had a first-degree family history of CRC. Only 26% of CRC presentations were identified through screening. Of the screened patients, 81% had stage 0–2 disease, all underwent surgery and there were no deaths in this group 1 year after recruitment.
In contrast, 74% of patients presented with symptoms, including bleeding (26%), anemia (22%), and obstruction (19%). Thirty-six (49%) received elective surgery, 33 (45%) underwent emergency surgery, and 5 (7%) did not receive surgery. One year after recruitment, 21 patients (28%) in this group were deceased. Within the symptomatic cohort, 55% of patients were outside the age range recommended for screening, 22% did not have a family physician, and 50% had not been offered regular screening.
Conclusions
Despite an established screening program, a significant proportion of patients diagnosed with CRC at our center were not diagnosed via screening. Patients presenting with symptoms were more likely to have advanced disease, require more urgent surgeries, and experience worse outcomes compared to their screened counterparts. The current provincial approach to screening for CRC needs to be improved.
Introduction
Colorectal cancer (CRC) is one of the leading causes of cancer death in Canada. 1 Expected mortality is dependent on the stage of disease at diagnosis, ranging from a 5-year survival of 95% (Stage 1) to 14% (Stage 4). 1 Most CRCs develop via the adenoma-carcinoma pathway where adenomas take many years to develop into malignancies. 2 Screening protocols can identify and remove suspicious lesions through endoscopic polypectomy, reducing morbidity and mortality from CRC.3,4
In the province of Alberta (population 4.26 million), the Stop Colorectal Cancer through Prevention and Education (SCOPE) program is a centralized referral system, established in 2011, which coordinates CRC screening for patients aged 50 to 74 years of age.3,5 Alberta's CRC screening program utilizes fecal immunochemical testing (FIT) and colonoscopies. The FIT assay used in Alberta has a cutoff value of 75 ng hemoglobin/mL, with values above this cutoff registering as positive. Primary care providers (PCPs), which Alberta residents have access to free of charge, are encouraged to screen all average risk patients with a FIT test every 1–2 years. Albertans without a PCP can also order a FIT kit online; however, this is less well known. 6 FIT testing has also been deemed acceptable for testing patients aged 40–49 with one first-degree relative diagnosed with colon cancer at an age greater than 60 years as well as patients aged 75–84 after quality of life and life expectancy have been assessed. 7 Patients who have positive FIT tests are referred on for colonoscopy. Higher risk patients, such as those with known genetic syndromes or previous history of adenomatous polyps fall outside the general screening program and are followed with colonoscopy. Despite the availability of screening programs, advanced CRC at diagnosis remains significant in Alberta and across Canada.1,8–11 Complications associated with late stage presentation of CRC include bleeding, obstruction and perforation, which are associated with a higher proportion of emergency surgeries, higher morbidity and worse outcomes.12–14 Patients who initially present with visible rectal bleeding or other GI symptoms are deemed not appropriate for FIT screening and it is recommended that these patients are referred directly to gastroenterology by PCPs for consideration of colonoscopy.
To understand the outcomes of patients with CRC at our university-affiliated hospital, we performed an analysis of 100 consecutive patients diagnosed with CRC. Our objective was to understand the presentation and outcomes of patients diagnosed with CRC to identify areas for improvement.
Methods
We performed a prospective cohort study following STROBE reporting guidelines evaluating 100 consecutive patients with a new diagnosis of CRC at the Royal Alexandra Hospital (869 inpatient beds, one of two quaternary hospitals in the Alberta Health Services Edmonton Zone, zone population 1.3 million, catchment area >2 million) in Edmonton, Alberta between October 16, 2020, and July 29, 2021. 15 The final date for follow up was July 29, 2022.
Patients were identified by referral to the study by general surgery, gastroenterology, emergency room and internal medicine physicians. Our study targeted all patients with CRC diagnosed at the Royal Alexandra Hospital including those referred for elective surgery. Patients unable to consent and patients with recurrent CRC were excluded.
After pathologic confirmation of CRC, patients were contacted, consent obtained, and a questionnaire was completed by the research team through discussion with the patient (Supplemental Data Sheet 1). Areas of interest included demographics, presentation, diagnosis, treatment, and outcomes. Medical records were reviewed to confirm accuracy.
Demographic data included the age, sex, body mass index (BMI), comorbidities, socioeconomic status (SES), smoking history, country of birth, education status, family history of CRC, and personal history of polyps.16,17 Patients were stratified into two groups: patients diagnosed on screening (asymptomatic/screening cohort); and patients who had presented following signs or symptoms related to CRC (symptomatic cohort).
Regarding outcomes data, we defined anemia as <120 g/L for females and <135 g/L for males, according to our provincial lab reporting system. Bowel obstruction/perforation was defined based on computed tomography imaging or intraoperative confirmation by the general surgery team.
Data were anonymized and entered into a software system managed by Research Electronic Data Capture (REDCap, Vanderbilt University, Nashville, TN, USA). 18 Statistical analysis was completed using SPSS version 25. 19 Categorical data are expressed as frequencies and proportions while continuous data are expressed as median ± interquartile range. Chi-square tests were used to compare two categorical variables and Fisher's exact tests were used when the cell frequency was less than 5. Independent t-tests were used to determine statistically significant differences between continuous values. Independent sample median tests were used to compare the median between two variables.
Results
Demographics and presentation
During the study period, 140 patients were diagnosed with CRC, of whom 112 met the inclusion criteria. Twelve patients declined to participate (Figure 1). Of the 100 remaining patients, 26 patients were identified by screening while 74 patients were symptomatic from their CRC and underwent colonoscopy or emergency surgery that confirmed their cancer (Table 1). There were no statistically significant differences between the two cohorts with respect to age, gender or comorbidities. Compared to patients in the symptomatic cohort, patients in the screening cohort were more likely to have a family physician (p = 0.031) and more likely to have a higher education status (p = 0.039).
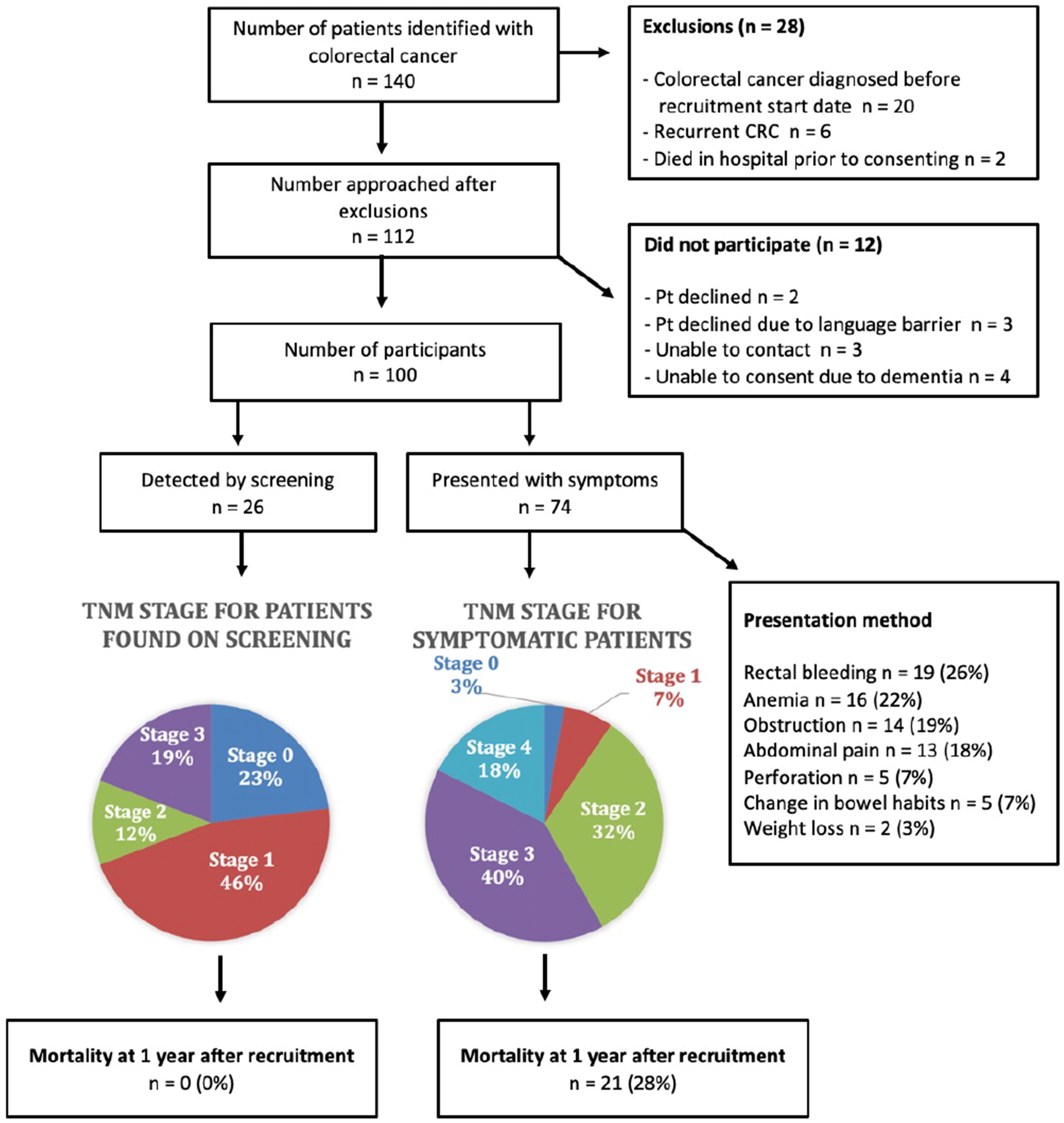
Inclusion and exclusion diagram for 100 consecutive patients identified with colorectal cancer at a tertiary Canadian hospital from October 16, 2020 to July 29, 2021.
Characteristics of recruited patients (n = 100).
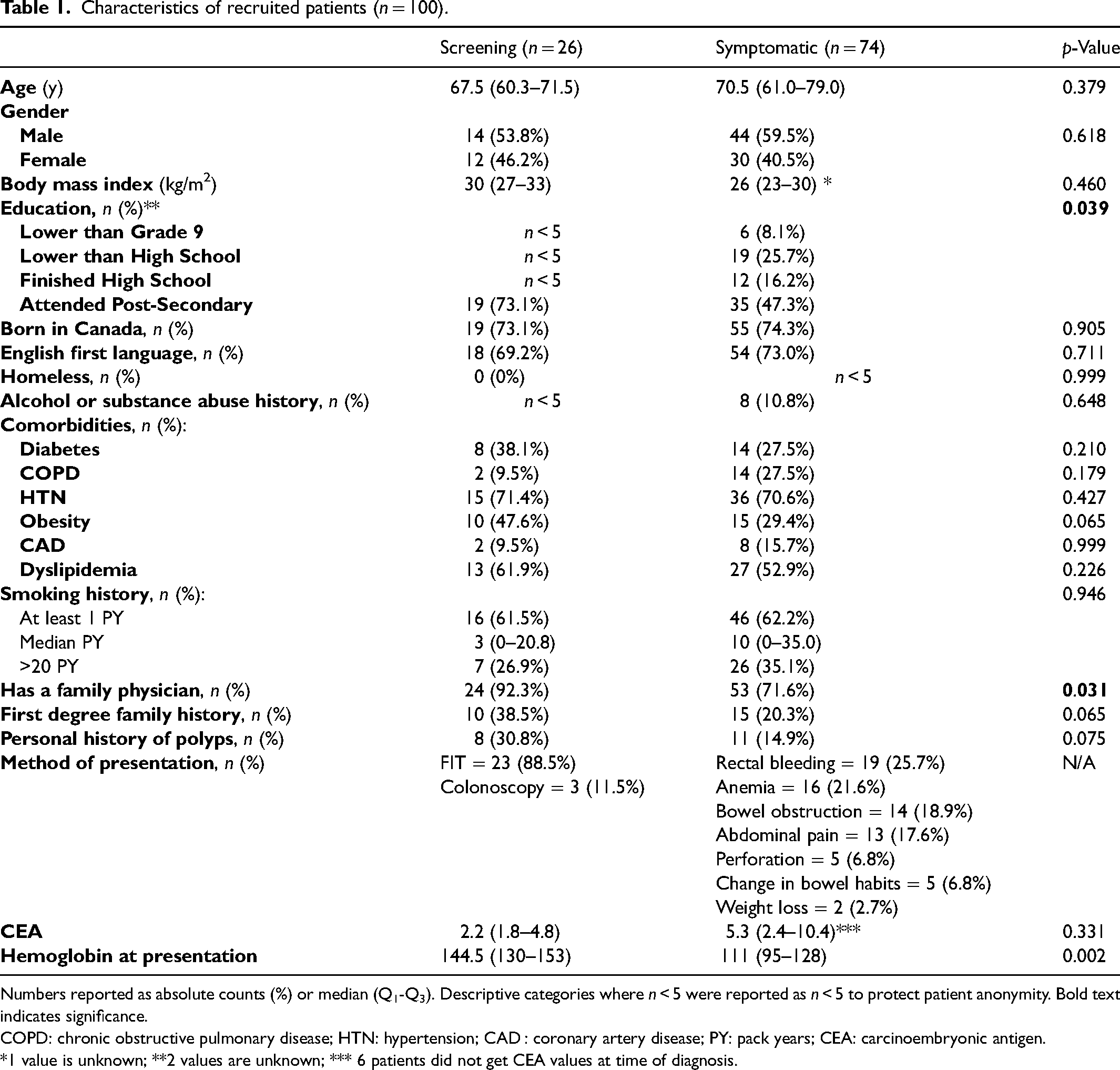
Numbers reported as absolute counts (%) or median (Q1-Q3). Descriptive categories where n < 5 were reported as n < 5 to protect patient anonymity. Bold text indicates significance.
COPD: chronic obstructive pulmonary disease; HTN: hypertension; CAD : coronary artery disease; PY: pack years; CEA: carcinoembryonic antigen.
*1 value is unknown; **2 values are unknown; *** 6 patients did not get CEA values at time of diagnosis.
Among the screening cohort, 23 patients had colonoscopies following a positive FIT test and 3 were identified on screening colonoscopy without FIT. The median time from a positive FIT to colonoscopy in the screening cohort was 124 days (Supplemental Table 1). In the symptomatic cohort, the most common presenting complaints were rectal bleeding (26%), anemia (22%) and bowel obstruction (19%) (Table 1). The median duration of symptoms was 150 days (Q1–Q3 = 30–365). Fifty-five percent (41 patients) of the symptomatic cohort were outside the screening age (30 [41%] patients above 75y; 11 patients [15%] below 50y).
Looking at patients who were lost to follow up: 5 patients in the symptomatic group had positive FIT within the previous 5 years and did not receive colonoscopy. Looking at false negatives of our screening tests, three patients in our study had negative FIT within 18 months of their diagnosis and 1 patient had received colonoscopy within 3 years of their symptomatic presentation at diagnosis.
Management and outcomes
In this study, 95 patients underwent surgery: 84 underwent resections with curative intent while 11 received palliative procedures (Table 2). Five patients were not offered surgery, three due to patient factors and two due to disease factors. Symptomatic patients had a lower hemoglobin at presentation (110.0 vs 144.5, p = 0.002), with a higher proportion of anemia (hemoglobin <120 g/L in females; hemoglobin <135 g/L in males) compared to the screening cohort. This finding was observed irrespective of gender (Supplemental Table 2).
Treatment details for study patients who received surgery (n = 95)*.

Numbers reported as absolute counts (%) or median (Q1-Q3).
LOS: length of stay; TNM: tumor, node, metastases stage.
5 patients were unable to receive surgery.
Median test comparison.
Fisher's exact test.
The rate of ostomy formation was 45% in the symptomatic cohort (55% temporary) compared to 27% in the screening cohort (71.4% temporary) (p = 0.742). The median length of stay was 6.5 days (Q1-Q3 = 4–10.8) for the screening cohort and 12 days (Q1-Q3 = 7–27.5) for the symptomatic cohort (p = 0.007, Figure 2). At 1 year follow up there were no deaths in the screening cohort, while 21 patients (28%) in the symptomatic cohort were deceased. Of note, although there was no significant p-value for a difference in age amongst the two groups, there was a larger range for ages amongst individuals in the symptomatic group, and the symptomatic group included more individuals over the age of 75, which could impact mortality rate (Figure 1, Supplemental Table 3). Among the 26 patients in the screening cohort, 18 (69%) patients had stage 0 (23%—adenocarcinoma in polyp) or stage 1 (46%) disease. In contrast, 30 patients (40%) in the symptomatic cohort were diagnosed with stage 3 disease and 13 patients (18%) were diagnosed with stage 4 disease (Figure 1).
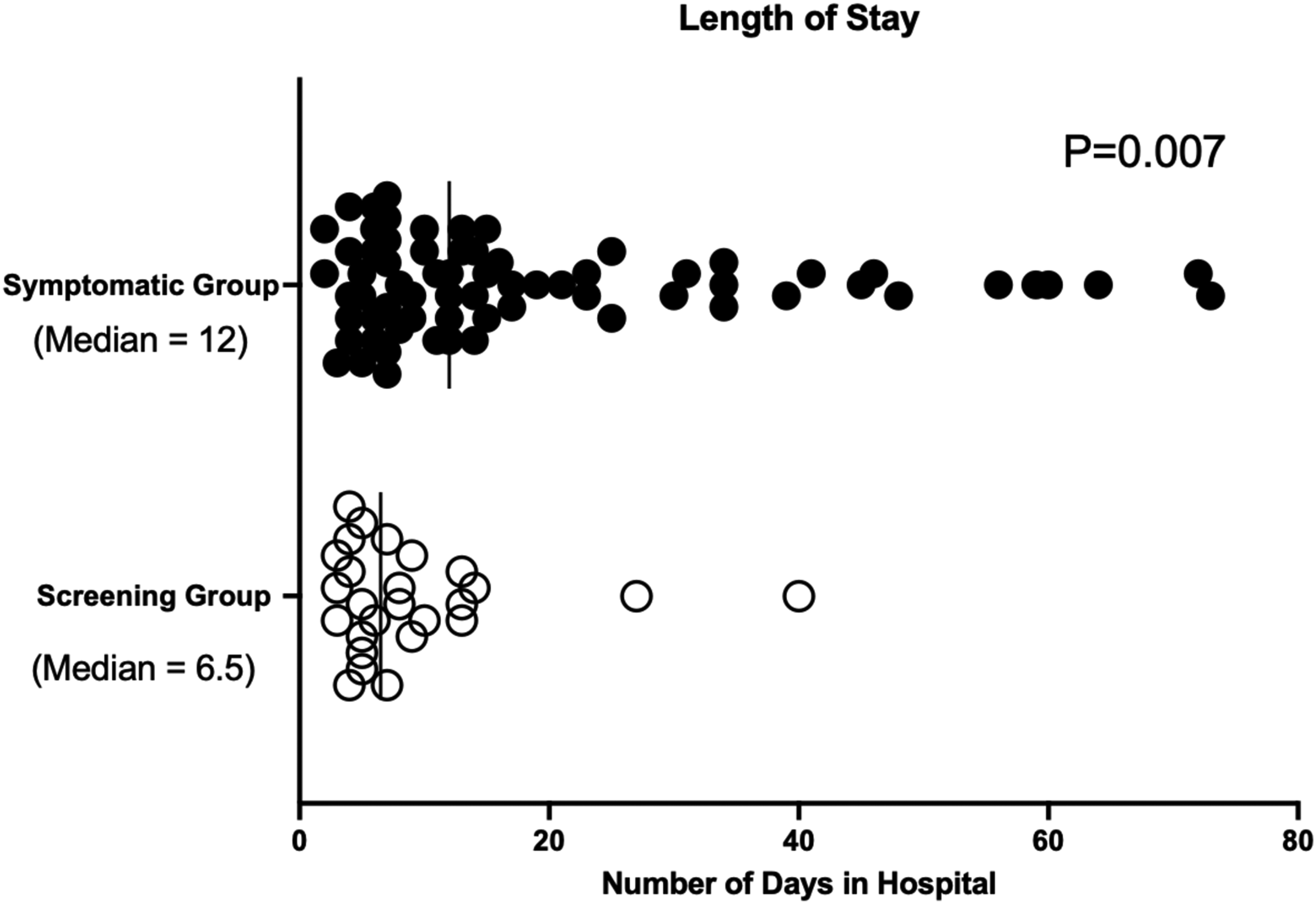
Post-operative length of stay when segregated by screening versus symptomatic group.
Discussion and conclusions
Effective prevention and screening are key to decreasing CRC mortality.4,20 This study demonstrates that despite having established screening programs for CRC in our province, we continue to see low screening rates and a significant amount of advanced disease at presentation. In 2018, the Canadian Cancer Society reported that 49% of CRC cases were identified at stage 3–4 compared to 28% for breast cancer and 20% for cervical cancer.21,22 In our study, symptomatic patients were more likely to be diagnosed with advanced disease, require emergency surgery, and have lower survival at 1 year. We found that our CRC patients diagnosed by screening had a 0% 1-year mortality compared to a 28% 1-year mortality in patients presenting with signs and symptoms related to their CRC. New strategies are needed for improving CRC screening at the patient, physician, and community levels.
Rates of screening for CRC in eligible, average risk Canadians aged 50–74 remain low, previously quoted at 16–51%.23–26 The low uptake of screening leads to a significant number of unnecessary CRC-related deaths.27,28 Our study identifies that patients undergoing screening had a higher level of education and increased accessibility to a family physician compared to patients who presented with symptomatic CRC. To our knowledge, this has not been previously shown in a Canadian population. 29 From a systems point of view, screening rates could be improved by ensuring that: (1) All Canadians have access to a primary care physician (PCP). (2) PCPs consistently take a family history and recommend CRC screening following guidelines. (3) Patients are compliant with screening recommendations.
A significant barrier to improving CRC screening rates in Canada is the shortage of PCPs. 30 One of the differences between the screened versus unscreened population in our study was access to a family physician. 9 Approximately 4.6 million Canadians over the age of 12 reported not having a PCP in 2019 and American literature has demonstrated that physician recommendation is one of the biggest motivators driving patient participation in CRC screening.31–33
Due to the of lack of access to primary care an argument can be made that screening should be taken out of the primary care domain. Differences in the CRC screening process exist across Canada, with some provinces having mail-based CRC screening programs. In the United States, the Kaiser Permanente Northern California (KPNC) group was able to improve the proportion of patients with up-to-date CRC screening from 39% to 83% with a direct-to-patient annual FIT program over a 15-year period, with an accompanying decrease in CRC incidence and mortality. 34 Direct-to-patient programs would ensure that all patients over 50 years of age are offered screening for CRC, which may be more effective than relying on access to an ever shrinking pool of PCPs.
At the specialist level, we must ensure timely endoscopy. This has been an ongoing challenge throughout Alberta over the past decade. 35 In Edmonton, the SCOPE program has helped centralize CRC screening. The SCOPE program has demonstrated improvement in access to endoscopy since conception; however, in our study the time from positive FIT to colonoscopy was a median of 124 days, outside the two-month window recommended by guidelines, highlighting the need for further improvements. 36
At the administration level, Alberta has implemented high quality screening methods including the FIT. 37 However, not all patients who have FIT recommended complete the test, and a proportion of patients do not receive colonoscopy following a positive FIT.36,38–41 Previous literature suggests patients more likely to adhere to screening were older, higher SES, more highly educated, and married.42,43 These factors may explain the advanced rate of non-screened advanced cancers found in patients of lower SES neighborhoods which we have seen in our previous geospatial analyses (Supplemental Figure 1).44,45
Most Canadian guidelines for average risk patients recommend CRC screening for ages 50–74. In recent years, patients are being diagnosed earlier with CRC, driving the US Task Force to suggest CRC screening start at age 45.46–50 In our study, 11 patients were below the age of 50. A recent study that reviewed 2473 cases of early onset CRC demonstrated that 25% of cases met criteria for early screening and that 98% of these cases would have had screening initiated at an age younger than the age of CRC diagnosis had guidelines been followed, reinforcing the need for appropriate screening in these populations. 51
Guidelines also suggest stopping CRC screening at the age of 75. Many patients in our study had not received screening starting at age 50, making it problematic to assume patients over 75 have been screened at a younger age. Literature shows that properly selected patients over 75 have decreased mortality from CRC screening.48,52 In our study we found 30 patients diagnosed at an age greater than 75. The majority of our elderly patients when faced with the decision between an invasive, high-risk intervention or palliation, agreed to and even demanded surgery. We believe that the decision to discontinue screening after 75 should be approached on a case-by-case basis. 53
Our study is not without limitations including its sample size and single hospital setting. Our patient recruitment method was dependent on provider referral, but inpatient lists were reviewed every two days to minimize patient loss. Our screening versus symptomatic cohorts had some demographic differences at the baseline, which could bias our results. Part of data accrual did overlap with the early waves of the COVID pandemic during which PCP visits were decreased. There was a short pause in screening colonoscopies during COVID in Alberta, approximately 1 month, and afterwards we resumed screening with high-risk patients (including those who were FIT positive). Interestingly, a study from Alberta found that diagnosis of CRC through screening decreased by 6.5% during the pandemic (Walker, 2021). This study estimated that pre-pandemic approximately 32% of cases of CRC in Alberta were detected by screening so a decrease by 6.5% corresponds to 25.5%, similar to the number found in our study. 54 It has been shown that the COVID pandemic has been correlated with lower screening rates globally.54–56
In conclusion, this study found a low rate of screening-detected CRC and poor outcomes for cancers in symptomatic patients. These results suggest that the current approach to screening for CRC in our region is failing. We suspect that the situation is similar in many other provinces across Canada. We propose the following solutions for policy makers: 1—All citizens should have access to a primary care physician. 2—An emphasis should be made on educating the public and PCPs about the importance of CRC screening. 3—Adoption of a direct-to-patient CRC screening outreach program should be trialed.
Supplemental Material
sj-docx-1-msc-10.1177_09691413251332588 - Supplemental material for Advanced disease at presentation for Canadian patients with colorectal cancer despite provincial screening programs: A call to action
Supplemental material, sj-docx-1-msc-10.1177_09691413251332588 for Advanced disease at presentation for Canadian patients with colorectal cancer despite provincial screening programs: A call to action by Kieran Purich, Courtney Streu, Sunita Ghosh, Ram V Anantha, Clarence Wong and Daniel Schiller in Journal of Medical Screening
Footnotes
Author contributions
KP and DS were involved in all stages of the article—study design, patient recruitment and data accrual, data analysis and manuscript writing and editing. CS and SG were involved in data collection and analysis & approved the final manuscript. RA was involved in data analysis & approved the final manuscript. CW was involved in study design, patient recruitment and approved the final manuscript.
Data availability
Raw data not included in the published article are available upon request from the primary author.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
This study was approved by the Health Research Ethics Board of Alberta Cancer Committee (HREBA-CC) under the Study ID of HREBA.CC-20–0166.
Consent to participate
All participants provided formal signed (if data obtained in person) or verbal (if data obtained by phone) consent to participate in the study.
Consent for publication
The consent form agreed to by all participants explicitly explained that the study results would be published. All authors have also consented to the publication of the final manuscript and accompanying data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
