Abstract
The obesity epidemic in the USA threatens the gains that have been made in the prevention and treatment of stroke. Both obesity and stroke disproportionately affect women more than men. Understanding the effect of obesity on stroke risk in women may be a useful stepping stone to reducing the burden of stroke in this vulnerable population. This article reviews the association between stroke and general obesity, abdominal obesity and metabolic syndrome in women. All three factors have been shown to independently increase stroke risk in women.
Keywords
Although substantial gains have been made in recent years in identifying, treating and controlling vascular risk factors [1–3], rates of Type 2 diabetes [4] and obesity [5] have increased, potentially impeding reductions in stroke incidence. The prevalence of obesity is slightly higher in women than men; the proportion of obese women in the year 2007–2008 was 35.5%, compared to 32.2% among men [5]. Continuing with the trajectory of the past 30 years will result in 86% of Americans becoming overweight or obese by the year 2030 [6]. In the setting of the burgeoning obesity epidemic, it is vital to gain an understanding of the effects of obesity on stroke risk and to determine if there are sex differences in these effects.
Stroke, which is characterized by a disruption of the blood supply to a portion of the brain, is the leading cause of disability and the fourth leading cause of death in the USA. Of all strokes, 87% are ischemic and the remainder are hemorrhagic (10% intracerebral hemorrhage and 3% subarachnoid hemorrhage) [7]. Ischemic stroke is caused by an occlusion or stenosis of an artery, reducing blood flow to the brain, while intracerebral hemorrhage is caused by blood vessel rupture, with subsequent bleeding into the brain parenchyma.
The public health burden of stroke in women is large. Due to women's greater longevity and the higher risk of stroke with age (the risk of stroke can nearly double each decade after 55 years of age), more women than men have stroke each year, and at least 55,000 more women than men die every year from stroke [8], which comprises 7.6% of all female deaths. It has been projected that by the year 2050, with the aging of the US population, stroke incidence will be 30% higher in women than in men [9]. In addition, there has been a recent increase in stroke prevalence among middle-aged women in the setting of stable stroke prevalence rates among similarly aged men [10–12].
Given the developing disproportionate prevalence of both obesity and stroke in women, an understanding of the effects of obesity on stroke risk in women may prove useful in mitigating the burden of stroke in women. This article will discuss the associations between general and abdominal obesity and metabolic syndrome (MetSD) and stroke. In addition, the influence of gender, age and race/ethnicity on the effects of obesity on stroke risk will be examined.
BMI & stroke risk
BMI is the most widely-used indicator for overall obesity. Among adults, a BMI of 18.5–24.9 kg/m2 corresponds to a healthy weight, a BMI of 25.0–29.9 kg/m2 is overweight, and a BMI ≥30.0 kg/m2 is obese. Numerous studies have found an association between elevated BMI and risk of stroke [13–21]. Although a few studies have shown that elevated BMI was no longer associated with increased stroke risk after adjustment for confounders [13,16,18], numerous studies have found that BMI >27 kg/m2 was associated with increased risk for stroke compared with normal weight subjects, even after adjusting for confounders such as Type 2 diabetes, hypercholesterolemia and hypertension [14,15,17,20,21]. There is a graded association between elevated BMI and stroke risk; the risk of total or ischemic stroke rises linearly with increasing BMI or increases in a stepwise fashion for higher BMI categories [17,19,21–23]. In fact, each 1 unit increase in BMI leads to a 5% increase in ischemic or general stroke risk in women [13,14,17]. One study found an inverse relationship between elevated BMI and ischemic stroke; the authors suggested this may have been due to low BMI among the elderly, heavy drinkers and smokers with stroke [24].
Elevated BMI and stroke in women.
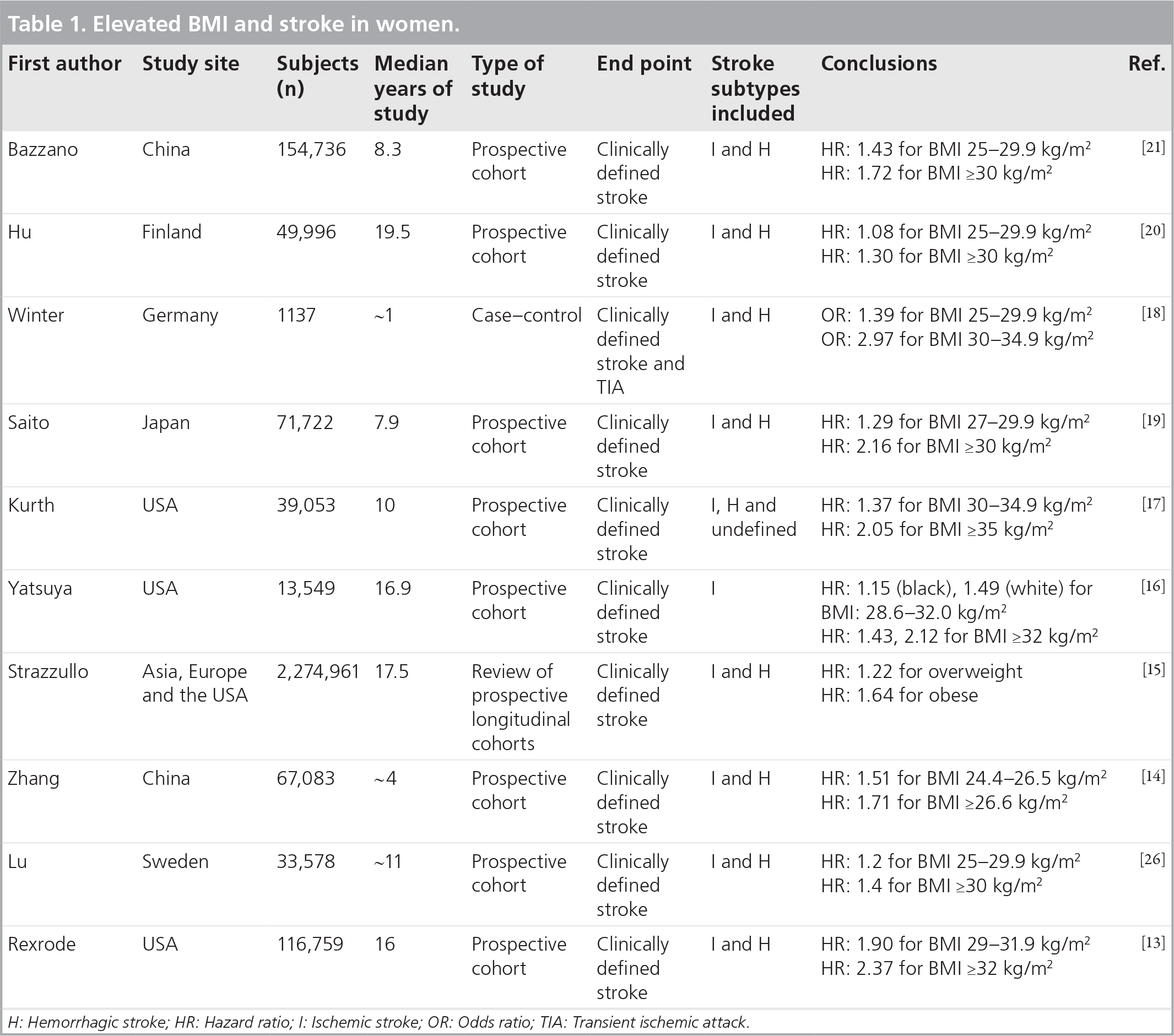
H: Hemorrhagic stroke; HR: Hazard ratio; I: Ischemic stroke; OR: Odds ratio; TIA: Transient ischemic attack; WC: Waist circumference.
Conflicting data exists with regards to sex differences in the effect of obesity on stroke risk. One study found a moderate correlation between BMI and stroke for men and no correlation in women [25], while another study came to the opposite conclusion [19].
The relationship between hemorrhagic stroke and BMI seems perhaps more complex, with conflicting studies showing no significant correlation [13,15,17,26], a positive correlation [14,19,21] and a U-shaped association in women [20].
Age is likely to influence the impact of obesity on stroke risk. A study of 154,736 individuals in rural China found that the effect of elevated BMI on stroke is greater in individuals who were <60 years [21]. Among 13,733 middle-aged women who participated in the National Health and Nutrition Examination Surveys, abdominal adiposity was independently associated with prior stroke [10,11].
Race/ethnicity also affects the impact of obesity on stroke risk. Most studies have relied on primarily Caucasian or European participants. In the rare studies including multiethnic populations, one study of over 2 million individuals found that excess body weight was a better stroke predictor in North American and European populations versus Asian populations, even though definitions of overweight and obesity were shifted downward for Eastern populations [15]. Nonetheless, even in Asian populations, BMI is a reliable predictor of stroke [16].
Abdominal adiposity & stroke risk
BMI may be an imprecise approximation of obesity, particularly because men and women have different body fat distributions even if they have equal total weights [27]. Several studies have suggested that abdominal adiposity has more significant health implications than overall adiposity and may more accurately indicate the degree of atherosclerotic or cardiovascular risk [28,29]. Abdominal adiposity can be measured using waist circumference (WC), waist-to-hip ratio (WHR), and waist-to-stature ratio (WSR). WC is the circumference of the abdomen around the top of the hipbone. Abdominal obesity is defined by WC >35 inches for women and >40 inches for men. WHR ratio is calculated by WC divided by the hip circumference; WSR is calculated by WC divided by height. Studies have investigated WC, WHR, and WSR in their abilities to predict stroke risk.
Numerous studies have found that elevated WC is an independent risk factor for total and/or ischemic stroke, after adjusting for potential confounders in multivariate models
Abdominal obesity and stroke in women.

H: Hemorrhagic stroke; HR: Hazard ratio; I: Ischemic stroke; OR: Odds ratio; TIA: Transient ischemic attack; WC: Waist circumference; WHR: Waist-to-hip ratio; WSR: Waist-to-stature ratio.
Several studies have compared WC to other abdominal obesity markers like WHR and WSR in predicting stroke risk. No difference in predictive value was found in comparison to WHR [14,16] and WSR [14], but one study found WC to be the worst predictor of the three [18].
Sex differences in the effect of abdominal obesity on stroke risk are ambiguous. Of the nine studies showing an association between abdominal obesity and stroke risk, six found this correlation in women [11,14,18,22,24,26]. A correlation between elevated WC and stroke risk was found in men but not women in two studies [16,20]. In addition, studies have found a trend suggesting that elevated WC increased stroke risk to a larger extent in women than in men [18,22].
The relationship between WC and stroke has been investigated in four multiethnic populations. Two studies found no correlation between elevated WC and stroke risk in a nonwhite population [16,24]. In a study of 67,000 women in China, even when the analysis was restricted to those with BMI <25 kg/m2, elevated WC was associated with an increased risk for total stroke [14]. This suggests that the relationship between WC and stroke is present regardless of whether it is accompanied by elevated BMI and may hold true for Asian populations that are generally less obese.
One of the common critiques of using WHR or WSR as diagnostic clinical tools is that both are harder to measure and standardize in comparison to BMI and WC [24]. Still, it seems that all measurements could potentially be useful stroke-risk predictors. Various studies have found a correlation between increased WHR and stroke risk in both men and women [14,16,18,20,24,26]. In studies that found this correlation in women, adjustment for potential confounders such as Type 2 diabetes, dyslipidemia and hypertension attenuated the effect in two studies [16,26]. Other studies found that every successive category of WHR increased ischemic or total stroke risk, even after adjustment for covariates [14,18,24]. The relationship between elevated WHR and hemorrhagic stroke is more obscure. While one study found that elevated WHR was correlated with hemorrhagic stroke [14], others have found no association [20,26]. Comparison of data are made difficult by the use of various categorizations for WHR, including dividing WHR measurements into tertiles [18], quartiles [20,24] or quintiles [14,16,26].
Regarding sex differences in the influence of WHR on stroke risk, one study found that men were more significantly affected by high WHR than women, and this difference was exaggerated when BMI was adjusted for [24]. In another study population of 33,578 women, adjustment for BMI further solidified the relationship between elevated WHR and stroke [26]. A single study demonstrated a stronger effect of WHR on stroke risk in women compared with men; this difference was thought to be a result of under representation of female stroke survivors [20]. One study found that the population attributable fraction, the proportion of stroke instances that could have been prevented if an individual was in the bottom 60% of WHR measurements versus in the top 40% of the population, was 21.7% [16]. This percentage was higher than that for BMI and WC, suggesting that treating high WHR in at-risk patients is of utmost importance. Other research has echoed the importance of WHR in assessing stroke risk by naming it the best predictor in comparison to WSR and WC [18,26], while further studies demonstrated that it was on par with other a bdominal obesity m arkers [14,16].
Waist-to-stature ratio is the least common mode of measuring abdominal adiposity, and the role of height as a factor in cerebrovascular risk has been controversial at best, with studies both demonstrating and rejecting a correlation [32]. Several studies found an association between elevated WSR and stroke risk [14,18,26]. One study of 67,000 women found a graded association between WSR and stroke risk; a 0.01 unit increase in WSR led to a 3% increased risk for total stroke, while a study of 33,578 women found that any individual with a WSR greater than 0.54 had excess stroke risk [14,26]. Further studies are required to clarify how WSR compares to other markers of obesity as a stroke risk predictor.
Metabolic syndrome & stroke risk
Metabolic syndrome, characterized by insulin resistance, abdominal adiposity, dyslipidemia and hypertension, is associated with an assortment of recognized stroke risk factors, and affects approximately one-fourth of the adult population in the USA [33]. Although there are numerous definitions of MetSD, all are consistently linked to cardiovascular risk [34]. MetSD may disproportionately affect black and Hispanic women [22,34].
There remains a debate surrounding whether MetSD components should be kept as separate entities that are clinically managed independent of one another, or if as a whole, MetSD confers added stroke risk and thus must be targeted as a single clustered disorder [34]. Supporting the latter, several studies have found that MetSD is linked with an increased risk of stroke, after adjustment for its components [31,35,36]. In addition, suffering from increasing numbers of MetSD components is associated with higher stroke risk [37,38]. Numerous studies have found an increased stroke risk in men and/or women with MetSD
Metabolic syndrome and stroke in women.
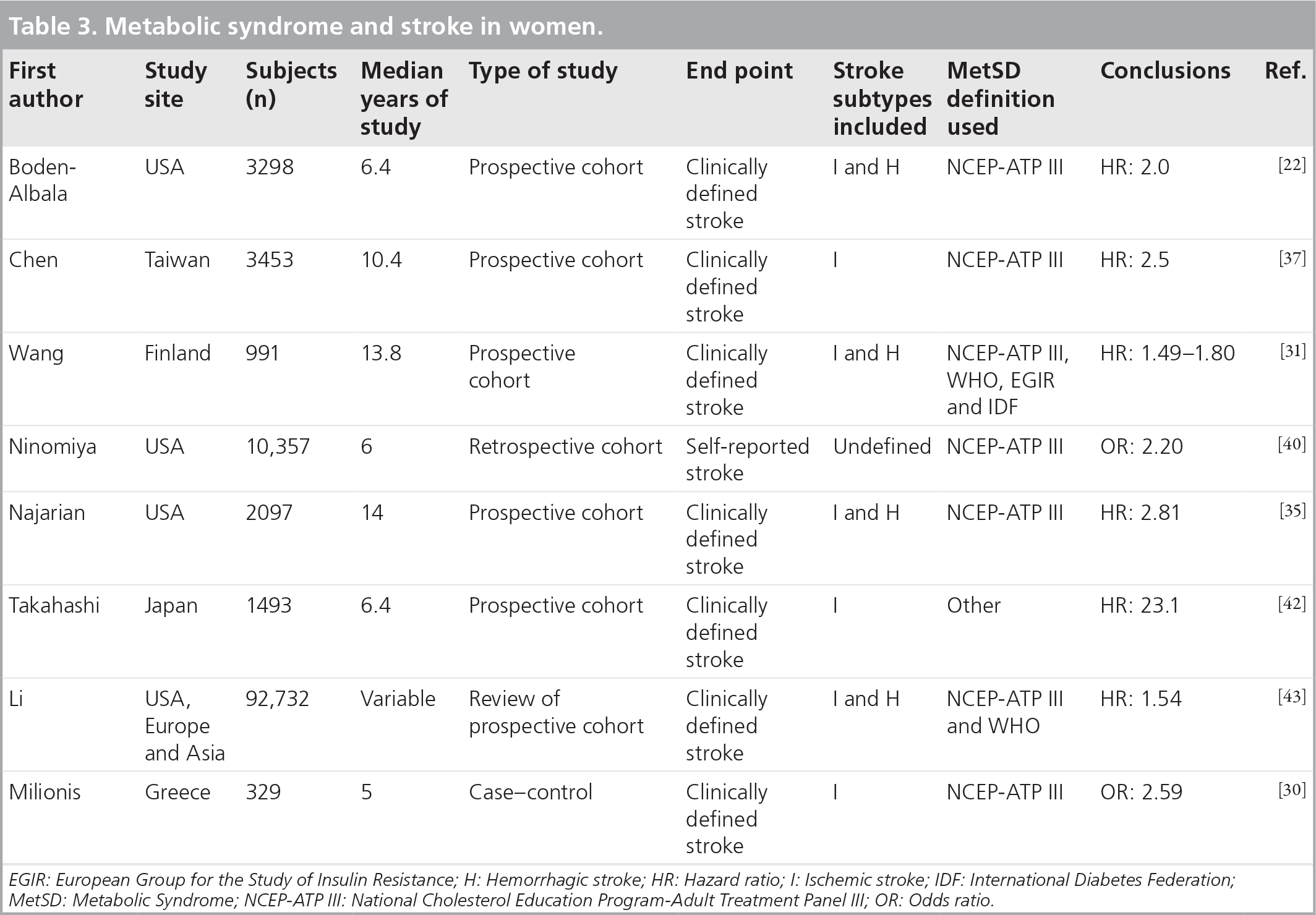
EGIR: European Group for the Study of Insulin Resistance; H: Hemorrhagic stroke; HR: Hazard ratio; I: Ischemic stroke; IDF: International Diabetes Federation; MetSD: Metabolic Syndrome; NCEP-ATP III: National Cholesterol Education Program-Adult Treatment Panel III; OR: Odds ratio.
The public health impact of MetSD may be more substantial than the influence of its individual parts. One study revealed that 19% of strokes could be avoided if MetSD was eliminated [22]. Another study revealed that MetSD components had a larger effect on the likelihood of stroke than MetSD [38]. To date, only one study has directly investigated the connection between metabolic syndrome and the prediction for second stroke [45]. Therefore, the value of screening stroke survivors for MetSD is undetermined.
The subsequent question is which of the MetSD components independently confers the largest stroke risk? This is a practical research question since within a population with MetSD, it may be possible to isolate persons with an exaggerated stroke risk based on the MetSD criteria they meet. In one study of 10,357 individuals, hypertriglyceridemia had the highest odds ratio in both men and women [40]. In a study comparing MetSD definitions, components that were shown to separately predict stroke included insulin resistance defined by European Group for the Study of Insulin Resistance, central obesity defined by the National Cholesterol Education Program (NCEP), and impaired glucose tolerance defined by the WHO [31]. In another instance, the NCEP definition was employed and abdominal obesity and hypertension had the highest hazard ratios for ischemic and hemorrhagic stroke, respectively [38].
Studies have suggested that obesity is most central to the effect of MetSD on stroke risk. Some authors have claimed that the central obesity component is the most fundamental piece of the MetSD puzzle [39,42] since it can predispose individuals to Type 2 diabetes, hypertension and dyslipidemia [42]. One study of 599 individuals with Type 2 diabetes revealed that MetSD or combinations of its components was not associated with ischemic stroke, but WC was independently associated with stroke [46]. Another study revealed that in the presence of MetSD, elevated WC and BMI was associated with greater stroke risk [30]. In addition, one study found that the dose–response effect of MetSD on ischemic stroke worsened in those who were obese [37]. Similarly, another study suggested that if the MetSD definition included a measure of obesity, especially abdominal obesity, then MetSD predicted stroke [31]. Alternatively, one study revealed that elevated WC was the only component of MetSD that did not independently increase stroke risk [40].
Several studies have shown sex differences in the effect of MetSD on stroke risk. MetSD confers a higher stroke risk upon women than men [14,42,43] and MetSD accounts for a larger percentage of stroke events in women than in men (30 vs 4%) [22]. These sex discrepancies may be owing to higher general and abdominal obesity levels within women than men [5,12,47]. General obesity commands a larger stroke risk in women with MetSD [38] and women with MetSD are more likely to be abdominally obese than men [12,22].
Studies have found conflicting results regarding the effect of MetSD on stroke risk in different racial/ethnic populations. A study of 3298 individuals found that MetSD conferred a greater risk of stroke in the Hispanic population than in black and white populations [22]. Another study of 10,347 individuals found that non-Hispanic black individuals with MetSD had higher odds of stroke compared with non-Hispanic white individuals [40].
Potential pathophysiologic mechanisms underlying the impact of obesity on stroke
The pathophysiologic mechanism by which general obesity increases stroke risk is poorly understood. One proposed mechanism is that obesity is associated with a prothrombotic and proinflammatory state that in turn raises levels of acute phase reactants like C-reactive protein. High levels of C-reactive protein may decrease endothelial cell production of nitric oxide; this may then instigate a cascade of events that leads to vasoconstriction, leukocyte adherence, platelet activation, oxidation and thrombosis [48,49]. This cascade results in hypertension, which consequently increases stroke risk [14,39,50,51]. Circulating coagulation factors [37] and attenuation of the protective effect of high density lipoprotein-cholesterol [30] owing to general obesity may also play a role.
The biological pathways by which abdominal adiposity increases stroke risk are also not yet understood, but platelet activation, inflammation, endothelial dysfunction, an overactive endocannabinoid system [52], or MetSD may all serve key roles in the process. In addition, increased V-LDL production due to the high lipolytic activity of abdominal adipose tissue [24,26] may increase stroke risk.
Similar to general and abdominal obesity, the avenues that confer to MetSD a stroke risk beyond those risks attributable to its components are as of yet unknown, but several pathways have been suggested. When it was first established, another common name for the MetSD was the insulin resistance syndrome, which emphasized it as a possible precursor for Type 2 diabetes [39,56]. If certain MetSD components combine to establish a prediabetic state, then its ability to increase stroke risk is plausible, given that Type 2 diabetes is an established stroke risk factor. MetSD may have a larger population impact than Type 2 diabetes since it is more widespread [35]. Other probable pathways include increased inflammatory markers and atherosclerosis resulting from MetSD [39,43,56].
Lifestyle interventions & stroke
Lifestyle factors such as a healthy diet [57–59], physical activity [60–65], abstinence from smoking [66–68], moderate alcohol intake [69–73] and maintenance of a healthy BMI (18.5–24.9 kg/m2) [17,74] reduce the risk of cardiovascular disease and mortality. Adherence to a combination of healthy lifestyle practices [75–82] reduces coronary events [75,77], ischemic stroke [78], all-cause mortality [76,81,82] and cardiovascular mortality [76,81].
Lifestyle intervention strategies to prompt changes in behavior vary widely in terms of success rates and specific methods used. Improving adherence requires a multifaceted approach, combining cognitive, behavioral and affective components [83,84]. Shared treatment decisions tend to be more effective, where patients incorporate risk-reduction options within their own personal resources and values [84,85]. It is important to provide information regarding personal risk to motivate patients to incorporate lifestyle change; the health belief model suggests that individuals' health-related behaviors can be explained by their perceived risk of a medical condition [86]. Finally, self-efficacy is essential; stroke survivors need to know that they can produce desired effects by their actions [87].
Although adherence to lifestyle advice ranges from 20 to 90% [88–90], several counseling-based interventions in nonstroke populations have been effective in promoting lifestyle change [84,91–93]. The Diabetes Prevention Program demonstrated a 58% reduction in diabetes incidence among nondiabetic persons with elevated fasting and postload plasma glucose concentrations [91]. A randomized controlled trial of lifestyle counseling for adult family members of patients hospitalized with cardiovascular disease resulted in improvement in diet score and exercise [93]. A Cochrane review of randomized/quasi-randomized trials of behavioral, pharmacological or multicomponent interventions to help patients stop smoking revealed that intensive counseling that begun during the hospital stay and continued with supportive contacts for at least a month postdischarge increased smoking cessation rates after discharge [94]. Finally, hospital initiation of secondary stroke prevention medications and diet/exercise counseling has been successful in improving vascular risk factors, recurrent events, and mortality for patients hospitalized with coronary artery disease [95] and stroke [96].
Conclusion
Both general obesity and abdominal adiposity are correlated with stroke risk in women. Elevated BMI, a marker of general obesity, is associated with increased stroke risk, even after adjustment for potential confounders such as hypertension, Type 2 diabetes or dyslipidemia. Elevated markers of abdominal obesity, such as elevated WC, WHR and WSR all independently increase stroke risk in women. Although BMI does not account for body fat distribution, there is no unequivocal evidence suggesting that abdominal obesity markers fare better as stroke risk predictors.
MetSD has consistently been shown to be associated with increased stroke risk and likely confers stroke risk beyond its components. While more evidence is required, it appears MetSD may increase stroke risk to a larger extent in women than men.
Although the pathophysiological mechanisms underlying the effects of obesity on cerebrovascular disease are poorly understood, potential causal pathways include proinflammatory states, elevated levels of C-reactive protein, accelerated atherosclerosis, insulin resistance, platelet activation and endothelial dysfunction.
Maintaining certain lifestyle factors such as a healthy diet, adequate exercise, a healthy BMI, restraint from smoking, and moderate alcohol intake can all contribute to the reduction of stroke risk. Types of interventions including physician-assisted, hospital-initiated and collaborative strategies are ever-evolving fields that continue to uncover effective methods for behavior modification.
Future perspective
As the obesity epidemic continues gaining momentum and people are forced to deal with the resulting health effects, health professionals need to tackle obesity through implementation of lifestyle management strategies and employment of pharmaceutical interventions. Ideally, more accurate stroke risk-assessments will be developed. Since stroke risk is closely related to the presence of MetSD, prevention of MetSD could act as a defense against stroke [36,97]. Although lowering weight via reduction of caloric intake and increase in daily physical activity is optimal, bariatric surgery is being utilized more regularly for those who are morbidly obese [98]. Those with a high risk in the short term will benefit more from drug therapies that treat elevated blood pressure, insulin resistance, prothrombotic and proinflammatory states [97].
Areas of active research include understanding the neuroendocrine regulation of obesity and genetic determinants of this regulation [99,100]. Although studies have consistently found a correlation between abdominal obesity and increased stroke risk, the effects of inflammation in fat are independent of BMI [101]. Recent studies utilizing adipose tissue biopsies have demonstrated possible links between decreased adipogenic and lipogenic profiles of abdominal fat, abdominal cell differentiation, and insulin resistance [102,103]. Although laborious and impractical for regular clinical use, these surgical biopsies are nonetheless important analytic tools for research concerning the pathophysiological effects of obesity [104]. In addition, visceral adipose tissue is thought to have higher vascular risk than the subcutaneous adipose tissue sampled in these biopsies.
As the obesity pandemic continues, novel ideas concerning the treatment of obesity will certainly arise. Interventions to reduce obesity will likely require a multidisciplinary approach, including community-based participation. Thus far, research regarding the effects of weight reduction on stroke risk is lacking [105].
New pharmacological therapies will also surely arise, and a new line of promising compounds may help reduce weight by targeting serotonergic systems [106]. Some compounds that are currently being studied as potential treatment options for the components of MetSD include metformin, magnesium, pioglitazone, telmisartan, niacin and topiramate [36]. The success of metformin for treatment of Type 2 diabetes has sparked interest in the development of other compounds that also activate AMP protein kinase for treatment of metabolic disorders [107]. Despite ongoing research on drug treatments, researchers continue to stress the importance of lifestyle modifications as the ideal method of treatment for the metabolic syndrome [108].
Genomics research is also in full force and this will undoubtedly influence our understanding of stroke in the coming years. Genome-wide association study approaches are currently being applied to stroke [109]. Many studies have already isolated potential loci that could lead to genetic predispositions for cardiovascular disease, proposing a future where biomarkers make highly personalized treatment of atherosclerosis a possibility [110]. A recent study suggests that the heritability of ischemic stroke is much higher in women than in men [111].
In conclusion, in the setting of the obesity pandemic, a multifaceted approach, incorporating lifestyle interventions and medications will likely be needed to begin to curb the effects of obesity on cardiovascular health in women.
Executive summary
Women with a BMI of >27 kg/m2 have an increased risk of stroke.
Stroke risk rises with higher BMI values.
Elevated BMI increases stroke risk in all racial/ethnic groups.
Elevated waist circumference, waist-to-hip ratio, and waist-to-stature ratio are each associated with an increase in stroke risk, even after controlling for covariates.
Metabolic syndrome is associated with an increase in stroke risk among women.
Metabolic syndrome may confer stroke risk beyond its components.
Pathophysiologic mechanisms underlying the effect of obesity on stroke risk.
Novel therapeutic interventions for preventing stroke in obese individuals.
The mechanisms underlying sex differences in the effect of obesity on stroke risk.
Footnotes
