Abstract
In this article we aim to draw attention to the burden of cervical cancer in Africa for reproductive health and review strategies for prevention, including appropriate noncytology-based cervical screening and prophylactic human papillomavirus vaccination. We consider the heavy burden of disease attributable to human papillomavirus infection borne by developing countries, particularly in Africa. Following identification of the human papillomavirus as the infectious etiological agent and elucidation of the long natural history of cervical neoplasia, cervical cancer is now one of the most preventable of all cancers. Opportunities for primary prevention by prophylactic vaccination and secondary prevention by appropriate cervical screening are discussed, together with the importance of population coverage. Qualitative work on attitudes towards cervical cancer prevention, education needs, the creation of an environment for informed choice and uptake are essential aspects of effective prevention programs. Cervical cancer poses a huge health burden in Africa. It is a disease that is eminently preventable given political will, the availability of affordable vaccines, appropriate cervical screening and access to cheap, point-of-care human papillomavirus testing. There are a number of unanswered questions for the prevention of cervical cancer and a need for demonstration projects to address these and further develop prevention strategies.
Keywords
“Prevention of cervical cancer in Africa seems daunting, even overwhelming, but the scale of the problem means that we cannot ignore it.” [1]
Gijs Walraven (2003)
“More than any other cancer, cervical cancer reflects striking global health inequality. Of the 274,000 deaths due to cervical cancer each year, more than 80% occur in developing countries and this proportion is expected to increase to 90% by 2020. Affecting relatively young women, it is the largest single cause of years of life lost to cancer in the developing world. The deaths of women who are in their most productive years have a devastating effect on the wellbeing of their families.” [2]
Jan M Agosti, Sue J Goldie (2007)
“The human papillomavirus vaccine addresses a critical public health need and is one element of a comprehensive cervical cancer control strategy.” [3] WHO, UNFPA, Geneva Switzerland (2006)
These statements sum up the scale of the cervical cancer problem in Africa and the importance and urgency of prevention. Worldwide, approximately 500,000 new cases of cervical cancer are diagnosed annually. The actual figure may be considerably higher than this as data are either unavailable or of variable quality for many developing countries. Every year over 274,000 deaths occur as a result of cervical cancer, with 80% of these occurring in developing countries that have access to less than 5% of the global cancer treatment resources [4] and the proportion of deaths is expected to increase to 90% by 2020 [5]. The reality is that in many developing countries the incidence of cervical cancer equates to mortality in the absence of healthcare facilities to deal with the problem, reflecting the gross health inequalities between developed and developing countries. For example, Africa has nine-times the incidence of cervical cancer compared with the USA and 24-times the mortality. Moreover, the paucity of healthcare in general, and in particular an absence of palliative care, means that cervical cancer causes enormous suffering before it results in the death of a woman. The incidence of cervical cancer appears to be increasing in the developing world, whereas incidence is falling in developed nations, largely due to systematic screening activity [6].
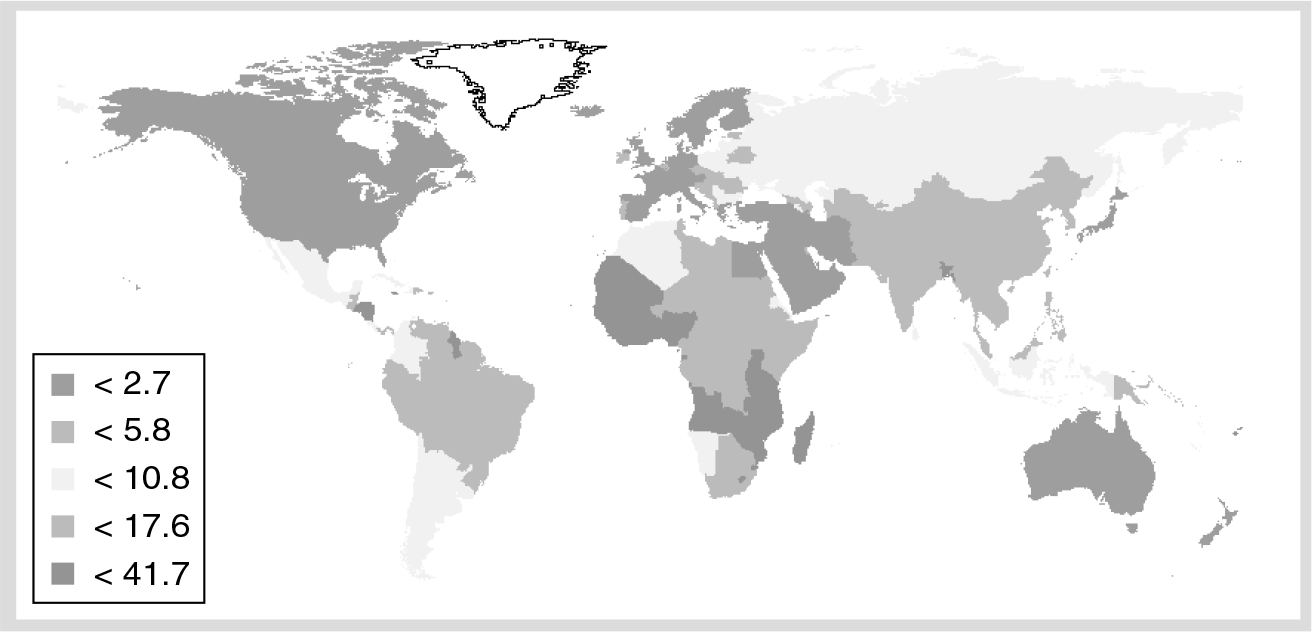
Global mortality map for cervical cancer (estimated age-standardized mortality rate per 100,000).
Cervical cancer is the most common cancer affecting women in sub-Saharan Africa with over 75,000 new cases diagnosed annually (age standardized rate 31.7) and in excess of 50,000 deaths (age standardized rate 22.5). Yet, in the face of these statistics, cervical cancer is one of the most preventable of all cancers, following identification of the etiological infectious agent and elucidation of the long natural history of cervical neoplasia.
Africa has been chosen as the focus for this article, due to the lowest access to cancer care services, including diagnostic facilities such as pathology or imaging and basic treatment in terms of surgery, radiotherapy or chemotherapy. While some improvements have been made in recent years, 33 out of 56 countries in Africa have no radiotherapy services [7], nor prevention, screening, early diagnosis or palliative care programs. Even where facilities exist, these are concentrated in large cities and remain inaccessible to the majority of the population.
The role of human papillomavirus infection in cervical neoplasia
Human papillomavirus (HPV) infection plays a central etiological role in cervical neoplasia with 99.7% of cancers worldwide containing HPV DNA [8]. HPV infection is the most common viral infection of the lower reproductive tract. Approximately 40 different HPV genotypes infect the genital area of both sexes, including the skin of the penis, scrotum and vulva and the mucosa of the anus, vagina and cervix. These are classified according to their oncogenic potential into ‘low-risk’ and ‘high-risk’ (HR) genotypes. Two HR genotypes, HPV 16 and 18, are responsible for the majority of HPV-associated cancers of the ano–genital tract [9]. Two low-risk genotypes, HPV-6 and −11, cause a substantial proportion of low-grade cervical dysplasia and more than 90% of genital warts [10]. The peak prevalence of HPV infection occurs in the late teens and early twenties following onset of sexual activity [11]. HPV infection usually resolves spontaneously in 9–12 months, but may persist in a minority of individuals with the subsequent development of cervical intraepithelial neoplasia (CIN). If untreated, CIN precursor lesions may progress to invasive cervical cancer over a period of 10–30 years.
In addition to cervical cancer, HPV also causes 90% of anal cancers, 40% of vulval cancers and a proportion of head and neck cancers.
Global burden of human papillomavirus-related cancer.

HPV: Human papillomavirus.
Adapted with permission from [12].
Prevention of cervical cancer in developing countries
Given the burden of disease and lack of healthcare facilities to manage cervical cancer in Africa the imperative must be upon prevention, particularly given the introduction of new tools facilitating primary and secondary prevention.
Although cervical screening and now HPV vaccination are regarded as the main strategies for cervical cancer prevention in developed countries, it is important to note that health education and promotion, as well as behavioral change programs directed at HIV and other sexually transmitted infections will, if effective in changing sexual risk behaviors, also help to prevent cervical cancer [13–15].
Prophylactic HPV vaccines
Human papillomavirus vaccines contain the major capsid L1 protein, which self assemble into virus-like particles (VLPs) [16], morphologically and antigenically resembling intact HPV virions. These VLPs contain epitopes that stimulate the production of neutralizing antibodies.
Currently, there are two prophylactic HPV vaccines, both of which have received European licences Vaccine profiles are also summarized in
Human papillomavirus virus-like particles vaccine profiles.

Composition per 0.5 ml dose.
HPV: Human papillomavirus; VLP: Virus-like particles.
Gardasil®, a quadrivalent vaccine formulated with a conventional alum adjuvant (aluminium hydroxy-phosphate sulfate) containing HPV types 6/11/16/18, (Merck/Sanofi Pasteur MSD);
Cervarix®, a bivalent vaccine containing HPV16/18 VLPs formulated with a novel adjuvant known as ASO4, which is a combination of aluminium hydroxide and 3-deacylated monophosphoryl lipid A, (GlaxoSmithKline).
Both prophylactic HPV vaccines have shown excellent effectiveness against persistent HPV infection and related cervical lesions among HPV-naive women in Phase 2/3 studies [17–22].
In the per protocol population for the Females United to Unilaterally Reduce Endo/Ectocervical Disease (FUTURE) II study (women aged 16–26 years receiving all three doses of the quadrivalent vaccine and negative for HPV throughout vaccination until 1 month after the third dose), vaccine efficacy was 98% against HPV16/18-related high-grade cervical lesions for an average 3 years follow-up. In the intention-to-treat population (women with or without previous HPV infection) a vaccine efficacy of 44% for high-grade HPV16/18-related cervical lesions was observed. The estimated vaccine efficacy against all high-grade cervical lesions, regardless of causal HPV type in the intention-to-treat population was 17% [22]. In the FUTURE I study, vaccine efficacy was 100% against vulval intraepithelial neoplasia, vaginal intraepithelial neoplasia and genital warts due to vaccine-specific types [21].
The bivalent vaccine has demonstrated (in women aged 15–26 years and DNA negative for vaccine HPV types) 90.4% efficacy against HPV16/18 CIN2+ (two cases in the vaccine group and 21 cases in the placebo group). It should be noted that it is unlikely that the HPV 16 or 18 detected in the two cases in the vaccine group caused the CIN2+ since another oncogenic HPV type was present in the biopsy and preceding cytology samples [20].
Evidence for a degree of cross protection against nonvaccine HPV types has been shown for both vaccines [20,23]. The bivalent vaccine shows efficacy against abnormal cytological outcomes with any high-risk HPV type and particularly for HPV-45 and −31 [19], with protection against incident and persistent HPV infection of 88 and 60%, respectively for HPV45, and 54 and 36%, respectively for HPV31 [24]. Additionally, broad protection against 12-month persistent infections with 12 combined oncogenic HPV types, not including HPV-16 and −18, have been observed in the order of 27% [20].
Recent data for the bivalent vaccine shows protection against CIN2+ caused by the five most prevalent nonvaccine oncogenic HPV types in cervical cancer (i.e., HPV-31, −33, −45, −52, −58) of 53% in the according to protocol cohort [25].
A 32.5% reduction in CIN2+/AIS associated with ten nonvaccine (HPV-31, −33, −35, −39, −45, −51, −52, −56, −58, −59) HPV types have been shown for the quadrivalent HPV vaccine. The cross protection efficacy was driven by A9 species members: HPV31, −33, −35, −52 and −58 [26]. Cross-neutralizing antibodies against nonvaccine HPV types are generated at lower concentrations than the dominant type-specific neutralizing antibodies [27].
The duration of protection from HPV L1 VLP vaccines is not yet known, although follow up in clinical trials has currently extended in excess of 5 years. Seroconversion has occurred in 98% or more vaccines and there has been no obvious breakthrough of disease in the vaccinated cohorts in either Phase II or III trials. Both VLP vaccines result in high levels of serum neutralizing anti-HPV L1 IgG that at their peak are up to 1000-times and at 5–6 years up to 10–12-times those measured following natural genital HPV infections. Mathematical modeling of the kinetics of antibody decay suggests that antibody could persist for 30 years [28]. Immune memory is essential for successful immunization with evidence for the generation of immune memory after HPV VLP vaccination in the form of recall responses to antigen challenge for the quadrivalent vaccine at 5 years and detection of circulating B cells 1 month after the third dose of the bivalent vaccine [29]. At the present time, duration of protection cannot be predicted with certainty and postvaccine surveillance and monitoring are essential. The vaccines require refrigeration and maintenance of a cold chain. Vaccine shelf life is estimated at 3 years from the date of manufacture.
Trials of both vaccines and postmarketing data to date have revealed no apparent safety concerns. Common side effects are minor and can include pain, swelling or redness at the injection site. Such side effects generally persist a day or two. Fainting after HPV vaccination is more common among teenagers than among young children or adults. However, fainting amongst teenagers is most often a response to the injection process rather than a side effect of the vaccine. Serious adverse events for both vaccines are extremely rare and are no more common than for other vaccines. Events involving hospitalization, disability, life-threatening illness or other medically important conditions account for approximately three per 100,000 events reported for Gardasil in the USA. There does not appear to be any significant difference in the number or severity of adverse events between the two HPV vaccines. There are no reports of deaths caused by the vaccines [101].
Human papillomavirus vaccines are not recommended for pregnant women, although no statistically significant increase in spontaneous abortion rates or congenital abnormalities has been observed for either HPV vaccine in women inadvertently vaccinated in early pregnancy, although data are limited. As HPV vaccines are not live vaccines, they can be safely administered to HIV-infected individuals. However, it is not clear how effective the vaccines are in triggering an immune response in HIV-infected or otherwise immunocompromised individuals and this is currently being investigated in clinical trials.
Human papillomavirus vaccination programs
In 2009, the WHO recommended routine HPV vaccination of 9–13-year-old girls [30], in countries where cervical cancer prevention constitutes a public health priority, vaccine introduction is programmatically feasible, sustainable financing can be secured and the cost–effectiveness of vaccination strategies are considered. However, the delivery of HPV vaccination programs face significant challenges, requiring innovative and multidisciplinary approaches to communication, strengthening of the local infrastructure and national immunization programs in Africa. Preadolescent girls are not routinely targeted by immunization schedules or other healthcare interventions and only some may attend school. HPV vaccines, therefore, require new approaches to program delivery and multiple stakeholders need to be involved in advocacy and social communication. Sexual and reproductive health communities are working with a vaccine directed against a sexually transmitted infection that is linked to a common, but under-reported, genital cancer, raising issues that are distinct from experience with other vaccines (e.g., rubella and tetanus). It will, therefore, be crucial for countries to strengthen existing services and use them as entry points for new interventions, looking for maximum synergy [3].
Major issues for adolescent immunization in developing countries include obtaining parental consent, reaching girls in school and those out of school, motivating school teachers and principals and obtaining endorsement by faith leaders or community elders for example.
Prevention of cervical cancer by vaccination against HPV may be a cost effective way of reducing the burden of cervical cancer and other anogenital diseases related to HPV infection in developing countries. Whilst it has been difficult to establish and maintain effective cervical screening programs in many developing countries, these same countries have in many cases developed comprehensive vaccination programs, which with appropriate adjustment could prove to be instrumental in cervical cancer prevention. As these vaccines are prophylactic, preventing initial infection with HPV, they are required to be given prior to contact with the virus and, therefore, sexual debut. The age of sexual debut may vary between countries but it is generally considered that HPV vaccines should be targeted towards adolescents aged 9–12 years, bearing in mind that even in countries with a relatively late average age of sexual debut, there will be some girls who debut early. The cervix in pubertal girls may be particularly vulnerable to HPV infection due to the physiological changes occurring during puberty. Additionally, it has been shown that neutralizing antibody titers are greater in prepubertal children compared with older teenagers [31,32].
Currently, prophylactic HPV vaccines are prohibitively expensive for use in low-resource settings, although in 2008 the Global Alliance for Vaccines and Immunization (GAVI) prioritized support for HPV vaccines for the poorest countries, providing funding is found. Encouragingly, vaccine prices have already started to drop in low- and middle-income countries compared with those charged in high-income countries and previous experience following the introduction of the hepatitis B vaccine has shown large reductions in cost over time.
Cervical screening in low-resource settings
Screening using cervical cytology is not feasible in most developing countries due to the financial, technical and infrastructure investments required and, therefore, other screening modalities require consideration, for example, visual inspection with acetic acid (VIA), visual inspection with lugol's iodine (VILI) and HPV testing.
Visual inspection with acetic acid
Several studies inspecting the cervix have demonstrated that direct VIA 3–5% is a reliable, reasonably sensitive and cost-effective screening strategy [33–36]. It has been shown to have a sensitivity in the range of 56–80% and a specificity of 64–98% for detecting high-grade CIN in several studies [36–44]. This does mean, however, that visual inspection techniques may miss a quarter to a half of true positives, as well as needing access to skilled colposcopy if screen positives are to be assessed further. VIA with magnification (VIAM) and VILI have also been assessed [44,36]. VILI was on average 10–30% more sensitive and equally specific, whilst VIAM showed similar results to VIA alone. These techniques require a lower level of infrastructure compared with cytological screening, providing immediate results and the possibility of treatment in a single visit. However, they have relatively low specificity, possibly related to the high prevalence of other sexually transmitted infections, and these are subject to the attendant problems of overtreatment [45]. Another unknown factor is how well VIA performs when genital schistosomiasis is prevalent. Furthermore, standardized initial training and continuing education programs with supervision and mechanisms for quality control will be required if widespread visual inspection screening is to be successfully implemented.
Sankaranarayanan et al. address the effect of VIA on cervical cancer incidence and mortality in a cluster-randomized trial in Tamil Nadu, India [46]. Women randomized to screening underwent a ‘once in a lifetime’ screen by VIA. Screen-positive women had colposcopy, directed biopsy and, where appropriate, immediate treatment. The primary outcome measures were cervical cancer incidence and mortality. In the screened group the incidence hazard ratio was 0.75 (95% CI: 0.55–0.95) and mortality hazard ratio was 0.65 (95% CI: 0.47–0.89). The study used nurses who underwent a 3-week training course, using IARC manuals [47,48] to develop skills in VIA, colposcopy, biopsy, discussion of results, cryotherapy, follow-up and referral of women with lesions unsuitable for treatment in the community or requiring cancer therapy. Doctors were trained to supervise nurses and undertake more complex treatment.
Sankaranarayanan and colleagues conclude that VIA is a simple, feasible and effective method to prevent cervical cancer and death among deprived populations but emphasizes the importance of high-quality training of providers, continuous quality assurance and monitoring for success. Similar conclusions were reached in a Tanzanian study [36]. The Indian study showed the greatest reduction in hazard ratios in the 30–39-year age group [46], indicating the age group within which to concentrate screening activity.
HPV testing in cervical screening
Human papillomavirus vaccine testing as a screening modality has greater objectivity and reproducibility than visual inspection and does not depend on the visibility of the transformation zone. It is less demanding in terms of initial training but HPV DNA testing is relatively expensive with a number of methodologies available. Several of these are based upon the PCR, which is a fairly simple technique generating large amounts of target DNA. One limitation is that it is a very sensitive technique and the contamination of specimens can be a problem. The sensitivity of PCR also means that further evaluation of what constitutes clinically significant positive results is required. In the future it may be possible to combine HPV testing with other biomarkers to indicate women with clinically significant disease or those with HPV infections likely to progress to high-grade disease. In this way biomarkers have the potential to improve the specificity of HPV testing. Likewise, restricting HPV testing to women over the age of 30 years will improve performance since HPV infection is extremely common in younger women and it is a persistent infection with oncogenic types that leads to disease. Simple, more affordable, rapid, user-friendly and accurate HPV tests suitable for use in low-resource countries are eagerly awaited.
The first such test, CareHPV, has been developed by Qiagen in collaboration with PATH for use in low-resource settings. It is a signal-amplification assay that detects DNA from 14 HR types of HPV. The test requires no running water or mains electricity and can be carried out by technicians in approximately 2.5 h. The short assay time allows for testing and clinical follow-up +/- treatment all in the same day. CareHPV has been evaluated in a cross sectional study in rural China [49]. Using CIN2+, the sensitivity and specificity of CareHPV were 90 and 84.2%, respectively, for cervical specimens compared with 41.1 and 94.5% for VIA and 97.1 and 85.6% for hybrid capture (HC) 2.
Another factor to be taken into account in HPV testing is whether the more aggressive natural history of HPV16/18 infections warrants the introduction of type-specific HPV tests into screening and management.
In a landmark study enrolling a total of 131,746 women aged 30–59 years, Sankaranarayanan and colleagues compared the effect of VIA, cervical cytology, HPV testing (using hybrid capture II) and standard care (controls) in a single round of screening on the incidence of cervical cancer and associated deaths over 8 years of follow-up. This was a cluster-randomized trial of 52 villages in Osmanabad district, India, randomly assigned into one of four clusters of 13 villages each [50]. The results are summarized in
Results of once in a lifetime cervical screening in rural India.

FU: Follow up; HPV: Human papillomavirus vaccine; N/A: Not applicable; VIA: Visual inspection with acetic acid.
Human papillomavirus vaccine testing reduced the number of advanced cervical cancers and deaths, while no significant reductions were seen in the cytology or VIA groups compared with the control group. In an accompanying editorial Schiffmann and Wacholder concluded that the implications of the Indian study were immediate and global: that HPV testing should be adopted for widespread implementation and that low-resource countries did not need to establish cervical cytology [51].
It is worth noting that the reduction in mortality hazard ratio seen with VIA was less than that witnessed in the Tamil Nadu study described previously [46]. This may be due to the larger numbers screened in the more recent study and the difficulty of ensuring continuing training and quality assurance using VIA.
In the South African context, Denny and colleagues undertook a randomized controlled trial (RCT) of two noncytology screen and treat approaches, VIA and HPV (HC2) testing, with a delayed evaluation (control) group [52]. The study involved a total of 6555 nonpregnant women aged 35–65 years with screen-positive women treated using cryotherapy. Both approaches were shown to be safe and resulted in a lower prevalence of CIN2–3 compared with the control group at 6 and 12 months. HPV testing was associated with a 77% reduction of CIN2+ at 6 months compared with a 37% reduction seen with VIA. No increase in HIV-seroconversion rates was observed following randomization. In this trial, screening and treatment occurred over two visits several days apart and outside such trial conditions lower follow-up rates might be expected.
Given the increased risk of cervical neoplasia in HIV-infected women [53] and the high HIV prevalence rates in many African nations, the performance of cervical screening in these cohorts is important. HPV testing may be as sensitive, but less specific given the high prevalence of multiple HPV infections in immunocompromised women [54]. In the South African RCT described above, 956 women were HIV infected. In the control group, HIV infected women had higher rates of CIN2+ detected by 36 months (14.9%) than HIV-uninfected women (4.6%; p = 0.0006). Screen-and-treat utilizing HPV DNA testing significantly reduced CIN2+ through 36 months in both HIV-infected (relative risk = 0.20, 95% CI 0.06–0.69) and HIV-uninfected women (relative risk = 0.31, 95% confidence interval 0.20–0.50). Reductions in the VIA group were less marked. Complications of cryotherapy were mostly minor and did not differ in frequency between HIV-infected and HIV-uninfected women [52,55].
In a Ugandan study VIA and VILI used as a sole method for cervical cancer screening resulted in significant false-positive results, since HIV-infected women were found to have a high prevalence of inflammatory cervical lesions [56]. Further work is required to optimize cervical screening in HIV-infected women.
Further studies are required to evaluate more fully the alternative approaches to cervical screening [33]. The most clinically and cost-effective strategies appear to be those that improve linkage between screening and treatment, through a reduced number of visits or effective follow-up [57]. Prophylactic HPV vaccination will take two or more decades to establish its effect on cervical cancer burden and in the meantime effective screening strategies in developing countries are crucial. Additionally, screening will remain essential to identify precursor lesions caused by nonvaccine-targeted oncogenic HPV types – current HPV vaccines potentially prevent 70–80% of cervical cancers – and for those women who have not been vaccinated. Operational research is required to investigate the possibility of offering HPV vaccination to adolescents (or infants) and screening to mothers during vaccination sessions.
Low-intensity cervical screening modeling
Modelling studies of a low intensity, once in a lifetime screen for women aged between 30–45 years have shown a reduction in cervical cancer burden of 30% [57,58]. In Goldie's model, a once in a lifetime screen at the age of 35 years using VIA or HPV DNA testing reduced the lifetime risk of cervical cancer by 25–36%, at a cost of less than US$500 per year of life saved [57]. Two screenings in a lifetime reduced lifetime risk of cancer by approximately 40%, although incremental benefits of more than two screenings were much smaller.
HPV DNA testing of health worker-obtained cervical samples has sensitivity for detection of high-grade lesions or cervical cancer that is equivalent or superior to that of cytology, [44,59–62]. Since the prevalence of transient HPV infection is high, especially amongst younger women, HPV testing is less specific for cervical neoplasia than cytology, although its presence may have prognostic value [63,64]. A South African study indicated that HPV testing of self-administered vaginal swabs was as sensitive as cytological screening for detecting high-grade cervical disease in women aged 35 years and over [65]. Preliminary data from a small study in HIV-infected women indicates that HPV detection in urine samples is possible, removing the need for gynecological examination [66]. Larger studies are required to verify these findings.
The characteristics of screening tests are summarized in the model of Goldie et al. [57] in
Characteristics of screening tests.

The importance of coverage
Population coverage is extremely important for both cervical screening and HPV vaccination. In England, for example, cervical screening was introduced in 1964 and although a large number of smears were taken (over 40 million), it became obvious that the program was not reaching those women who were at greatest risk and some individuals with positive results were not followed up successfully. In 1988 the Department of Health reorganized cervical screening, requiring all district health authorities to introduce a computerized call–recall system and recommending that women aged 20–64 years should participate in cervical cancer screening every 3–5 years. Following the reorganization, cervical cancer rates progressively declined from 1990. Organized, systematic screening programs have been shown to reduce the incidence of cervical cancer by up to 75–80% [67].
The ultimate contribution of HPV vaccination to cervical cancer prevention in different countries depends upon screening efficacy, involving coverage and quality of screening and uptake of vaccination, especially amongst women who are inadequately screened [68]. If the proportion of cervical cancer prevented by HPV16/18 vaccination prior to contact with the virus is assumed to be 75% then potential avoidable cervical cancers are shown in
Cervical cancer avoidable in different countries by screening efficacy and vaccination coverage.
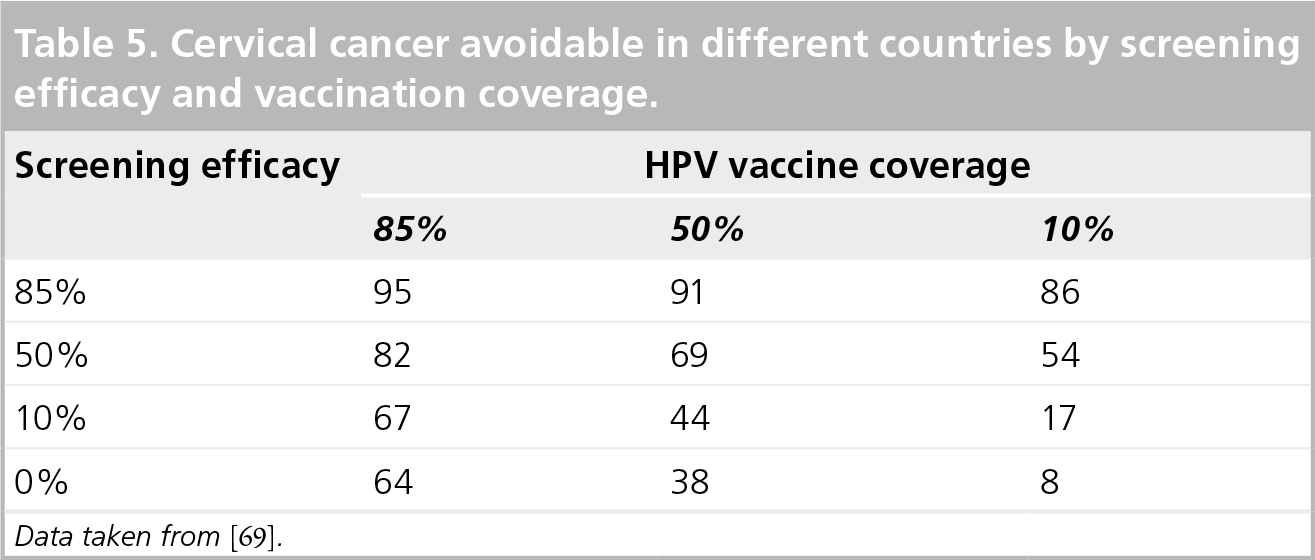
Data taken from [69].
In countries with high screening efficacy, such as Nordic countries and other high-income countries, cervical cancer prevention is high as a result of screening and is less affected by HPV vaccine coverage. In countries with poor screening efficacy, the level of protection against cervical cancer is critically affected by HPV vaccine coverage. In intermediate-and low-resource countries where cervical screening efficacy is poor or nonexistent, approximately two-thirds of cancers could be prevented if 85% vaccine coverage is achieved. This has important consequences for the cost–effectiveness of cervical cancer prevention programs.
Challenges for the prevention of cervical cancer in Africa
There are important challenges in the prevention of cervical cancer in Africa. First, cervical cancer has to be accepted as an important public health problem alongside competing healthcare priorities. Resources are required to deliver prevention programs, which need to be both sustainable and of high quality. Programs require evaluation to demonstrate what does and does not work, to allow for improvement and development. Low levels of awareness of cervical cancer and the potential for prevention need to be addressed along with ensuring access and acceptability for target groups. Effective assessment and treatment for precursor lesions with low complication rates is essential for screen-positive women.
The formative research undertaken by PATH on the introduction of HPV vaccine into Uganda summarizes findings into a HPV vaccine delivery strategy; a HPV vaccine communication strategy and a HPV advocacy strategy [102]. The research recognizes the need to address gaps in cold chain and logistics capacity; the need for public education and the development of positive messages about HPV vaccination; and the incorporation of HPV vaccination into existing policy and cancer plans. PATH is also evaluating strategies to introduce HPV vaccination into three other countries through demonstration projects in India, Peru and Vietnam [103].
Unanswered questions
Unanswered questions concerning both the primary and secondary prevention of cervical cancer in Africa include how to promote an environment for informed choice and ensure acceptability; how to identify target groups for vaccination and screening and record participation; whether there is a need for vaccine booster doses; the extent of cross protection against nonvaccine HPV types in Africa; possible type replacement and the optimal strategy for integration of vaccination and screening. The importance of male vaccination has recently been raised again by Michels and zur Hausen [69], and although not currently cost effective [70] this may require reconsideration once vaccine costs diminish or poor vaccine coverage is demonstrated amongst girls in low-resource settings.
Consideration should be given to targeting infants for HPV vaccination where robust infrastructures already exist in many countries for childhood immunization. This would require the demonstration of HPV vaccine safety, efficacy and lack of interference with other childhood immunization and continued protection from infection following exposure during adolescent and young adulthood, many years later. An alternative strategy could utilize children entering school, alongside other vaccine boosters or healthcare interventions.
Further study is required in HIV-infected individuals and into second-generation vaccines. Surveillance of HPV types in populations and in cancers will be important in evaluating cervical cancer prevention programs. Just how soon HPV vaccination becomes affordable for low-resource countries remains to be seen.
Future perspective
Cervical cancer is perhaps the most preventable of all cancers and in future it could be virtually eradicated in Africa through prophylactic vaccination and HPV-based, single-visit cervical screening using a see, assess and treat approach. Whether this becomes reality will depend on political will, philanthropy, public education and affordable tools, in terms of HPV vaccines and point-of-care HPV tests. Further research will refine the tools and strategies.
Executive summary
Cervical cancer represents a huge disease burden in Africa and the paucity of healthcare in general and, in particular, an absence of palliative care means that cervical cancer causes enormous suffering before it results in the death of a woman in Africa.
Worldwide, approximately 500,000 new cases of cervical cancer are diagnosed annually with 274,000 deaths: 80% occurring in developing countries that have access to less than 5% of global cancer treatment resources.
The incidence of cervical cancer appears to be increasing in the developing world, whereas incidence is falling in developed nations largely due to systematic screening activity.
Despite the statistics, cervical cancer is perhaps most preventable of all cancers due to the long natural history of precursor disease (cervical intraepithelial neoplasia [CIN]) and the discovery that human papillomavirus (HPV) infection plays a central etiological role in cervical neoplasia.
The high-risk HPV types 16 and 18 are responsible for 70–80% of cervical cancers.
Cervical cancer prevention strategies include health education and promotion aiming to change sexual risk behaviors; primary prevention through prophylactic HPV vaccination and secondary prevention by cervical screening and treatment of precursor lesions.
These are currently produced from the HPV major capsid protein, which self assembles into ‘virus-like particles’ morphologically and antigenically resembling intact HPV virions.
Currently, there are two prophylactic HPV vaccines: Gardasil®, a quadrivalent vaccine formulated with a conventional alum adjuvant (aluminium hydroxy-phosphate sulfate) containing HPV types 6/11/16/18, (Merck/Sanofi Pasteur MSD); and Cervarix®, a bivalent vaccine containing HPV16/18 VLPs formulated with a novel adjuvant known as ASO4, which is a combination of aluminium hydroxide and 3-deacylated monophosphoryl lipid A (GlaxoSmithKline).
Both prophylactic HPV vaccines have shown excellent effectiveness against persistent HPV infection and related cervical lesions among HPV-naive women in Phase 2/3 studies.
Evidence for a degree of cross protection against nonvaccine HPV types has been shown for both vaccines.
The duration of protection from HPV L1 VLP vaccines is not yet known, but is longer than 6 years.
Trials of both vaccines and postmarketing data to date have revealed no safety concerns.
It is not clear how effective the vaccines are in triggering an immune response in HIV-infected or otherwise immunocompromised individuals.
The delivery of HPV vaccination programs face significant challenges in terms of the infrastructure and personnel required, access to preadolescent girls and the current high cost of the vaccines.
Multiple stakeholders need to be involved in advocacy and social communication.
Screening using cervical cytology is not feasible in most developing countries and other screening modalities require consideration such as visual inspection methods and HPV testing.
Several studies have demonstrated that direct visual inspection of the cervix with acetic acid 3–5% is a reliable, reasonably sensitive and cost-effective screening strategy but that it requires high-quality training of providers, continuous quality assurance and monitoring for success.
HPV testing as a screening modality has greater objectivity and reproducibility than visual inspection but is relatively expensive and lacks specificity.
Single-visit ‘screen, assess and treat’ reduces loss to follow-up.
Randomized controlled trials of cervical screening in South Africa and India have shown HPV testing to be the most effective at reducing the incidence of high-grade CIN or advanced cervical cancer, respectively.
Modelling studies of a once in a lifetime screen for women aged between 30–45 years have shown a reduction in cervical cancer burden of 30%.
Cervical cancer still requires recognition as an important healthcare priority.
Sustainable and high-quality resources to deliver cervical cancer prevention programs with identification of, and access to, target populations are required.
Public education to ensure acceptability of program is also required.
Effective assessment and treatment of precursor lesions presents a challenge.
High population coverage is needed for effective prevention of cervical cancer.
The evaluation and development of future programs and studies will address the remaining unanswered questions.
Footnotes
The author has been on advisory boards and received honoraria for both human papillomavirus vaccine companies (GlaxoSmithKline and Sanofi Pasteur MSD). The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
