Abstract
Objective
Cervical cancer is the second most common cancer in Thai women; human papillomavirus (HPV) is the main cause. This study aimed to determine the clinical performance of HPV mRNA compared with HPV DNA testing.
Methods
Cervical specimens were collected from women aged 35 to 60 who attended the routine organized screening programme. We compared accuracy parameters of standalone HPV mRNA and HPV DNA tests, and those of triaging with liquid-based cytology or HPV genotyping and liquid-based cytology for those positive only for the less oncogenic HPV types. Test accuracy parameters were estimated using latent class analysis using Bayesian models.
Results
Of the 5046 women enrolled, 174 (3.4%) were HPV DNA positive and 141 (2.8%) HPV mRNA positive. Colposcopy compliance was 95.4% (n = 166) among HPV DNA-positive women and 94.3% (n = 133) among those HPV mRNA positive. The estimated sensitivity, specificity, and positive predictive value for detection of CIN2 or worse were 67.4%, 97.1%, 12.1% for HPV DNA testing, and 73.1%, 97.8%, 16.3% for HPV mRNA testing. These estimates for triaging of HPV DNA-positive women with liquid-based cytology were 64.4%, 98.8%, and 19.0%, respectively, and slightly better for liquid-based cytology triage of HPV mRNA-positive women, at 71.8%, 98.9%, and 22.1%.
Conclusion
A triaging strategy based on HPV genotyping and liquid-based cytology for those positive only for the less oncogenic HPV types had test characteristics comparable with that of liquid-based cytology triage. The HPV mRNA detection-based strategies had non-significant advantages over the HPV DNA detection-based strategies.
Introduction
Cervical cancer is the second most common cancer among women in Thailand. 1 Widespread implementation of cytology-based screening has substantially reduced cervical cancer incidence and mortality in Europe, North America, and Australia.2,3 In Thailand, since 2005, women aged 35–60 are offered free five-yearly cervical cancer screening under a universal health care coverage insurance scheme. Over the last two decades, cervical cancer incidence has been slowly declining. 4
Human papillomavirus (HPV) infection is the ‘necessary’ cause of cervical cancer. 5 Of nearly 200 HPV genotypes, 14 are causally associated with cervical cancer, and are therefore termed high risk- (hr-) genotypes. 6 Because women with persistent hr-HPV infection are at increased risk of developing high grade cervical intraepithelial neoplasia (CIN) and cervical carcinoma,6,7 detection of hr-HPV is recommended as a primary screening test for cervical cancer in women aged 30 or older. The majority of tests are based on the detection of deoxy-ribonucleic acid (DNA) of the L1 protein of the virus. 8 A major limitation of HPV DNA screening is its lower specificity, leading to a higher false positive rate than conventional cytology and more women without disease receiving further investigations and/or unnecessary treatment. Expression of messenger ribonucleic acid (mRNA) of the viral E6 and E7 oncogenes is strongly associated with cervical carcinogenesis,9,10 and the expression of these oncogenes is necessary for cell immortalization, neoplastic transformation, and the development of invasive cancer.11–13 The hr-HPV E6/7 mRNA test has a higher specificity, but similar sensitivity for the detection of CIN2 or worse disease (CIN2+), than HPV DNA tests in some of the earlier studies.14–19
Most management algorithms for HPV-positive women recommend cytology as the triaging test to reduce the referral to colposcopy. Many of the new HPV detection platforms provide additional information on the presence of the most oncogenic HPV types (e.g. HPV 16, 18, and 45), and this information can be used to triage HPV-positive women. In Thailand, women positive for HPV 16, 18, or 45 are directly referred to colposcopy, while those positive for other oncogenic types are advised cytology. In the second group, the women are referred to colposcopy only if the cytology is abnormal at a threshold of atypical squamous cells of unknown significance (ASCUS). Hereafter, we refer to this triaging approach as combined genotyping and liquid-based cytology (LBC) triaging.
In this study, we performed a direct comparison of the test characteristics of the HPV DNA and HPV mRNA detection tests, and evaluated the performance of triaging HPV-positive women with LBC alone, and a combination of HPV genotyping and LBC to detect CIN2 or worse (CIN2+) lesions, in a community setting.
Methods
Apparently healthy women aged 30–60 attending the 50 primary healthcare units in seven districts in Ubon Ratchathani province in North Eastern Thailand for cervical cancer screening under the national programme were eligible to participate. The study objectives and procedures were explained by primary care nurses, and all willing participants provided written informed consent. Recruitment began in July 2014 and was completed in January 2015. The study was approved by the ethics committee of the National Cancer Institute, Bangkok, Thailand.
After obtaining sociodemographic history, a trained nurse performed the screening procedure. The cervix was visually inspected for any abnormalities, cervical cells were collected using an Ayre’s spatula, and a conventional smear was prepared on a pre-labelled glass slide with spray fixative. The spatula was rinsed in a vial containing PreservCyt® solution (Hologic Inc., Marlborough MA, USA). A second cervical cell sample was collected using an endocervical brush, which was then rinsed and placed in the same vial containing the PreservCyt solution. The smears were stained with Pap stain at the cytology laboratory to be evaluated for conventional cytology.
Cervical specimens were stored at room temperature, and transported at room temperature to the National Cancer Institute (Bangkok), for HPV DNA testing using COBAS 4800 System (Roche Molecular Systems Inc., Branchburg, NJ, USA). The COBAS 4800 HPV test simultaneously detects 14 hr-HPV types: HPV16 and 18 individually, and pooled detection of another 12 hr-HPV genotypes (31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68). The HPV mRNA test from the PreservCyt sample was performed using APTIMA (Hologic Inc., Marlborough, MA, USA) technology, which could detect E6/E7 mRNA expression of 14 hr-HPV types. The samples positive on APTIMA were further tested for mRNA of HPV types 16, 18, and 45 by the APTIMA genotype assay, which does not differentiate between HPV 18 and 45. The adequacy of sample was confirmed by the detection of human beta-globin protein. All the samples positive on either of the HPV tests were tested with LBC (Hologic Inc., Marlborough, MA, USA) and interpreted by a cytopathologist at the NCI Bangkok. All HPV-positive women were also referred for colposcopy, irrespective of the reflex cytology or genotype results. HPV-negative women with abnormalities reported on conventional cytology were also referred for colposcopy. A trained gynaecologic oncologist performed the colposcopy and obtained directed biopsies from the lesions suspected on colposcopy. In cases of normal colposcopic findings, random punch biopsies were obtained.
We have previously reported on the performance of conventional cytology to detect CIN2+ lesions. 20 In the current analysis, we included women who had results on all three primary screening tests (conventional cytology, HPV DNA, and HPV mRNA tests). Characteristics of study participants were presented as numbers and proportions. In total, seven different screening modalities were assessed, the first three involving primary screening with conventional cytology or HPV tests, and the last four using different triaging strategies for HPV-positive women: modality 1- primary screening with conventional cytology; modality 2- primary screening with hr-HPV DNA detection; modality 3- primary screening with hr-HPV m-RNA detection; modality 4- triaging of HPV DNA-positive women with LBC; modality 5- triaging of HPV mRNA-positive women with LBC; modality 6- triaging of HPV DNA-positive women with combined HPV genotyping for HPV 16/18 and LBC; and modality 7- triaging of HPV mRNA-positive women with combined HPV genotyping for HPV 16/18/45 and LBC. LBC was considered abnormal if the result was ASCUS or worse. The final diagnosis (gold standard) was based on histology report and colposcopy diagnosis, if histology was not done, or was inconclusive.
We estimated positivity rates, detection rates (proportion of CIN2 or worse detected by a positive screening test among all women screened), sensitivity, specificity, and positive predictive values (PPVs) to detect CIN2 or worse for the screening and triaging modalities, participation to further assessment among the screen-positives, and performance characteristics of the each of the modalities. The positivity rates, detection rates, and PPVs, together with their 95% confidence intervals, were estimated using the exact binomial interval estimation methods.
Because colposcopy and/or biopsy were carried out only among women positive on any of the screening tests (conventional cytology and/or hr-HPV testing), the estimates of disease prevalence and performance characteristics were affected by verification bias. To allow for estimation of these parameters in the absence of disease verification for all participants, latent class analysis using Bayesian models was used. Three Bayesian models were constructed: the first for assessment of modalities 1, 2, and 3, the second for modalities 1, 4, and 5, and the third for modalities 1, 6, and 7. In each of these models, the assumption of statistical independence conditional on the true disease status of the HPV DNA and HPV mRNA based modalities was relaxed. In each Bayesian model, each level of the four observed variables (based on the findings of the three modalities and the final diagnosis) was constructed. Additionally, an unobserved or “latent” variable with two mutually exclusive categories, ‘diseased’ and ‘non-diseased’ was constructed and used as a measure of true disease.21–25 This unobserved variable was then used to estimate the corrected estimates of the disease prevalence, sensitivity and specificity of the screening test modalities. We used the exact McNemar’s Chi-square test of proportions for paired data to compare between screening tests the two test parameters (test positivity rates and detection rates) that we could calculate directly from the observed data.
All statistical analyses were performed using STATA 14.2 (StataCorp LP, Texas, USA) software and Just Another Gibbs Sampler (JAGS) software.
Results
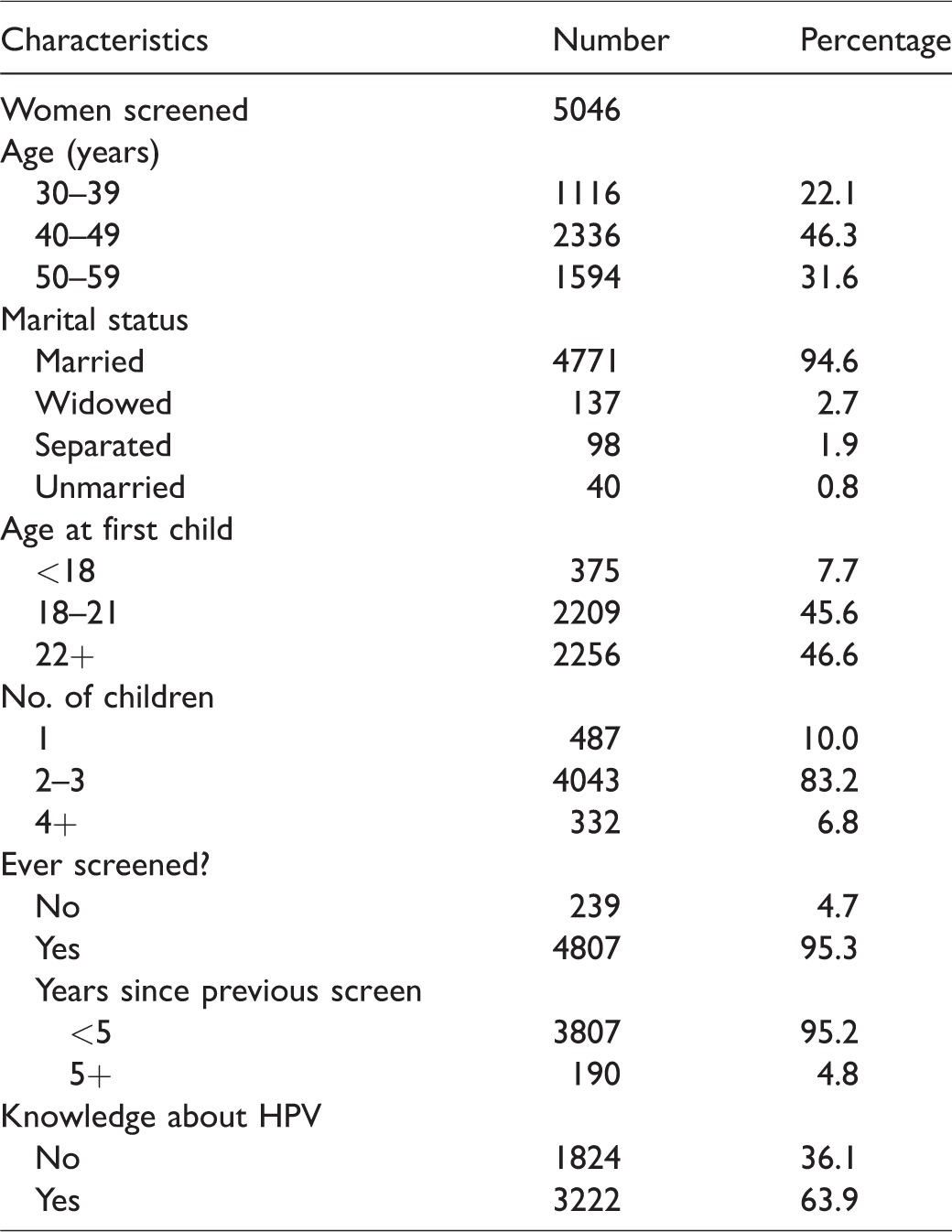
Table 1 describes the characteristic of the 5046 women included in the study. Prior screening was reported by 4807 (95.3%) of the women, and the majority (3807; 95.2%) had been screened in last five years with conventional cytology. Knowledge about HPV was reported in 3222 (63.9%) women.
Characteristics of women screened in the project.
The high-risk HPV DNA and mRNA screening test positivity rates were 3.4% (174/5046) and 2.8% (141/5046), respectively, (significantly different, p-value <0.001). The HPV positivity rates among women aged 30–39, 40–49, and 50–60 were 4.8%, 2.9%, and 3.3%, respectively, for the DNA test, and 3.8%, 2.6%, and 2.4%, respectively, for the mRNA test. Figure 1 summarizes the study outcomes for HPV test results, reflex LBC results, and the final diagnosis based on colposcopy and histopathology. Compliance to colposcopy was 95.4% (166/174) for HPV DNA-positive women, and 94.3% (133/141) for those positive on HPV mRNA. Among HPV DNA or HPV mRNA-positive women with negative LBC results (<ASCUS), only six were detected to have CIN2+ lesions.
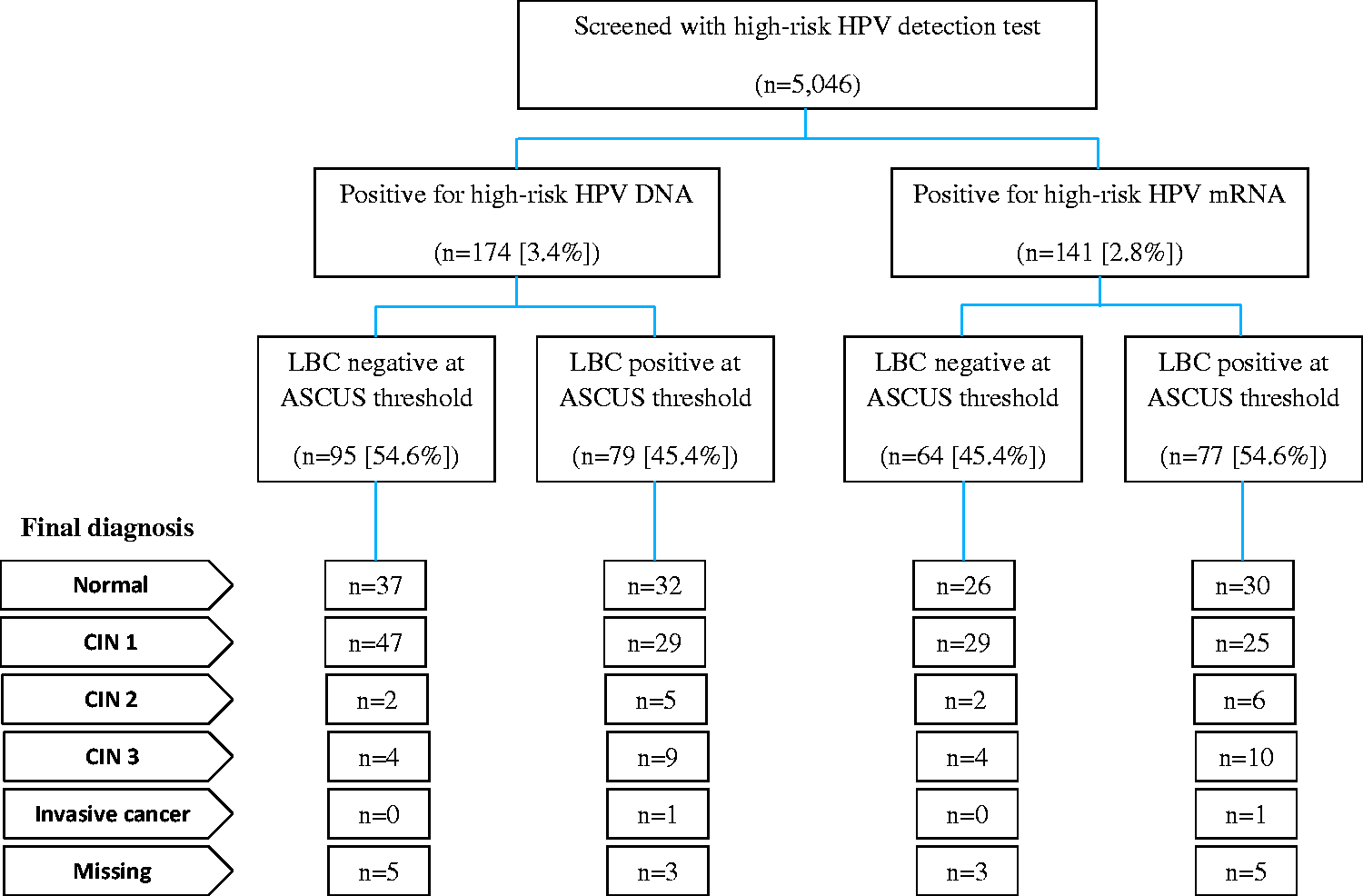
Flowchart of number of women screened, HPV genotyping and liquid-based cytology results, and final diagnosis among HPV-positive women.
In total, 22 cases of CIN2/CIN3 and one case of invasive cancer were detected in the screened women. Table 2 shows the histopathological outcomes of the women positive for hr-positive HPV DNA or HPV mRNA and hr-positive women triaged with LBC or triaged with HPV genotyping plus LBC. The mRNA test was negative in four women positive for HPV DNA 16, and three women positive for HPV DNA 18. None of these women had CIN2+ disease (data not shown).
Numbers of women tested, test-positives, and cervical neoplasia detected by screening and triaging modality used.
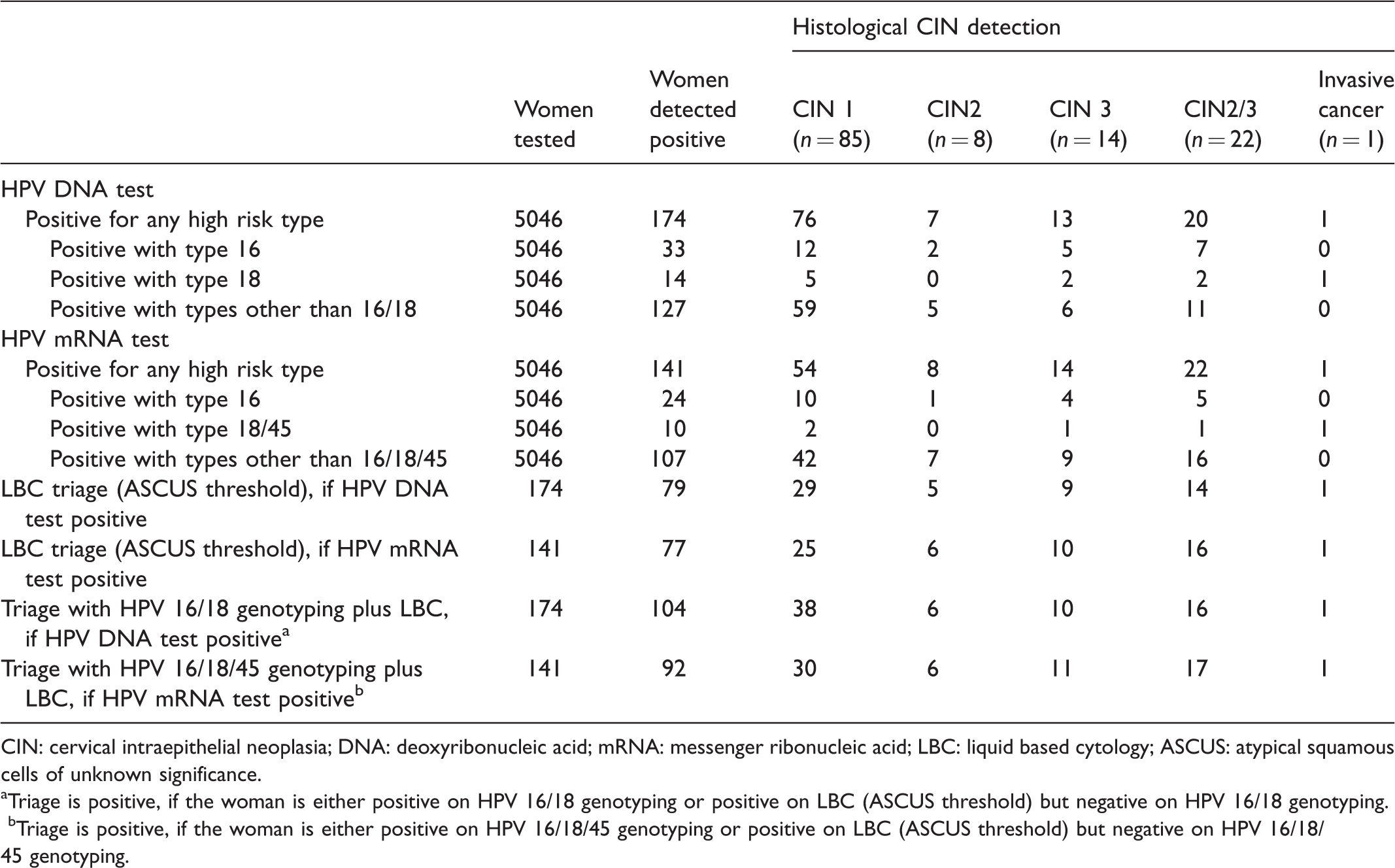
CIN: cervical intraepithelial neoplasia; DNA: deoxyribonucleic acid; mRNA: messenger ribonucleic acid; LBC: liquid based cytology; ASCUS: atypical squamous cells of unknown significance.
aTriage is positive, if the woman is either positive on HPV 16/18 genotyping or positive on LBC (ASCUS threshold) but negative on HPV 16/18 genotyping.
bTriage is positive, if the woman is either positive on HPV 16/18/45 genotyping or positive on LBC (ASCUS threshold) but negative on HPV 16/18/45 genotyping.
The test characteristics of the HPV screening modalities with or without any triaging estimated from the Bayesian models are presented in Table 3. The test positivity rates were highest for primary screening with the HPV DNA test alone (3.4%) and HPV mRNA test alone (2.8%). When triaging of the HPV positives with LBC was considered, these rates were reduced to 1.6% for the HPV DNA test, and 1.5% for HPV mRNA. The modality considering women with HPV DNA types 16/18 or those positive on both other non-16/18 types and LBC triage as positive reduced the positivity to 2.1%. Using HPV mRNA types 16/18/45 instead of HPV DNA 16/18 reduced the positivity rate to 1.8%. The detection rates for CIN2 or worse disease for the HPV DNA test alone and HPV mRNA test alone were 4.2/1000 and 4.6/1000 women screened, respectively (not significantly different with p-value = 0.50). These detection rates reduced to 3.0/1000 and 3.4/1000, respectively, when LBC triage was considered, and to 3.4/1000 and 3.6/1000, respectively, when HPV DNA types 16/18 plus LBC triage of other HPV DNA types and HPV mRNA types 16/18 plus LBC triage of other HPV mRNA types modalities were assessed.
Prevalence and screening test performance characteristics for detection CIN2 or worse disease obtained from the latent class analysis using Bayesian models.

aTriage is positive, if the woman is either positive on HPV 16/18 genotyping or positive on LBC (ASCUS threshold) but negative on HPV 16/18 genotyping.
bTriage is positive, if the woman is either positive on HPV 16/18/45 genotyping or positive on LBC (ASCUS threshold) but negative on HPV 16/18/45 genotyping.
cSensitivity and specificity have been adjusted for verification biases.
CIN: cervical intraepithelial neoplasia; DNA: deoxyribonucleic acid; mRNA: messenger ribonucleic acid; LBC: liquid based cytology; ASCUS: atypical squamous cells of unknown significance.
The sensitivity and specificity of the tests and the triaging strategies could not be estimated directly, as the verification tests (colposcopy and/or biopsy) were not performed on all test-negative women, but the statistical correction of verification bias allows an estimation of approximate sensitivity and specificity. The HPV mRNA test had slightly higher (but not statistically significant) sensitivity (73.1%) for CIN2+ than the HPV DNA test (67.4%). The specificity and PPV of the HPV mRNA test (97.8% and 16.3% respectively) were also higher than those of the HPV DNA test (97.1% and 12.1%, respectively), but the overlapping confidence intervals suggest no statistically significant difference. Among the different triaging strategies, HPV mRNA test followed by LBC triaging had the best balance of sensitivity (71.8%) and specificity (98.9%).
Discussion
Our study demonstrated comparable efficacy of the HPV mRNA and HPV DNA tests. Overall, both the tests showed good concordance (98.4%) to detect the presence or absence of HPV. The model estimated that there were about seven and six false-negatives, respectively, using the HPV DNA and HPV mRNA tests. Earlier studies observed higher specificity and PPV of the mRNA test, which can be explained by the fact that the E6/E7 mRNA is expressed in more advanced stage of carcinogenesis.26–29 However, in our study, 19 women were HPV mRNA positive but HPV DNA negative, and we observed that the mRNA test had slightly higher (but not statistically significantly) test characteristics than HPV DNA. This could be explained by the presence of interruptions or deletions in HPV DNA at the level of the L1 open reading frame. 30 In contrast, the E6 and E7 genes are almost invariably retained, as their expression is likely to be necessary for conversion to and maintenance of the malignant state.9,31–34
Cervical cytology is being replaced by HPV detection tests in many developed countries due to the higher sensitivity and objective nature of the HPV tests. In countries like Thailand, with inadequate number of cytopathologists and long turnover time for cytology reporting, a throughput test like HPV test has high relevance. The major drawback of the HPV detection test is its low specificity. Even though the HPV positivity reported in our current study is significantly lower than that reported in many Western countries, or even in China, the test positivity was higher compared with the cytology positivity reported in our earlier publication. 20 To reduce the number of colposcopy referrals, a triaging test is essential for HPV-positive women. We have demonstrated that any of the triaging strategies (reflex LBC or a combination of genotyping and LBC) can improve the specificity, without significant loss in sensitivity. The strategy based on HPV mRNA detection followed by LBC triage was the most effective of the strategies considered.
The decision to select the appropriate algorithm for cervical cancer screening is based on the effectiveness (number of tests required, referral rate to colposcopy, rate of detection of disease, and number of cancers and deaths averted) and efficiency (trade-off between benefits and harms) of the different strategies. A micro-simulation modelling study based on the evidence from different randomized controlled trials has demonstrated that five-yearly HPV test starting from age 30 is the most efficient, with the most aceptable balance between benefits and harms. 35 The same modelling study observed that primary HPV testing followed by triaging with HPV 16/18 genotyping and cytology (referral to colposcopy for women positive for HPV 16/18 genotype and cytology triage for those positive for other oncogenic HPV) required 12–14% higher colposcopy referrals compared with primary HPV testing followed by cytology triage, though the second strategy was associated with a 1–2% increased life-time total testing. Our study, based on a real programme setting, validates the observations of the modelling analysis. Triaging of HPV-positive women with cytology has the lowest test positivity (hence lowest colposcopy referral rate) at the cost of requiring more cytology tests to be performed compared with triaging with a combination of genotyping and cytology. The non-significant difference between sensitivity and specificity of the different triaging strategies indicates programmes should select strategy based on the existing resources, availability of the technology, and cost. The data from our study will allow a proper health technology assessment of the different strategies in the context of Thailand.
The efficacy of triaging with HPV genotyping depends on the prevalence of the high-risk HPV genotypes in the CIN2+ lesions, which shows geographic variation. HPV 52 and HPV 58 are more common in cervical cancer and precancerous lesions in East Asia than in other regions.36–38 Although HPV 16, 18, and 45 are the most common genotypes associated with CIN2+ lesions globally, the study by Khunamornpong et al. 39 has confirmed that HPV 52, 16, and 58 were the three most common genotypes among women with histologically confirmed CIN2+ in Chiang Mai, Thailand. This may explain our observation that triaging with a combination of genotyping and LBC is no better than triaging with LBC alone. However, a smaller number of women need to be recalled for collecting samples for cytology with the first strategy compared with the second.
A major limitation of our study is that the screen negative women did not have the gold standard test, and so test accuracy measures may be affected by the verification bias. It is possible that the test sensitivities were under-estimated due to the statistical adjustments, as the estimated sensitivities in our study are lower than the previously reported sensitivities of the tests. The strength of this study is the direct comparison of clinical accuracies of two different HPV diagnosis technologies, with or without triaging applied, on an unselected population within a routine screening programme.
Conclusion
The HPV mRNA and HPV DNA tests have comparable test characteristics. Triaging with either reflex LBC or a combination of genotyping and LBC works equally well for HPV-positive women. The second triaging strategy may be more efficient, as the genotype test results are available concurrently, it can be performed on self-collected samples, and fewer cytology tests would be necessary. Thailand has to adopt a suitable approach based on the availability of technology, feasibility of implementation, and assessment of cost implications to reverse the current rising trend of cervical cancer. 40
Footnotes
Acknowledgements
The authors gratefully acknowledge all women who participated in this screening project. We also thank the staff of Ubon Ratchathani Provincial Public Health Office and Ubon Ratchathani Cancer Hospital, and from the IARC Screening Group, Mrs Krittika Guinot, for her help in preparing and submitting the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimer
Where authors are identified as personnel of the International Agency for Research on Cancer/World Health Organization, the authors alone are responsible for the views expressed in this article and they do not necessarily represent the decisions, policy or views of the International Agency for Research on Cancer/World Health Organization.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partly supported by National Cancer Institute foundation.
