Abstract
Lapatinib is an oral, small-molecule, dual kinase inhibitor that targets both HER2 and the EGF receptor. Lapatinib was approved in June 2008 in Europe for the treatment of advanced HER2-positive breast cancer. Promising results in trastuzumab-refractory metastatic breast cancer were obtained from Phase I, II and III studies in combination with chemotherapy. Diarrhea and rash are the most common side-effects and are mostly moderate and treatable. Cardiac toxicity occurs rarely and mostly as an asymptomatic and reversible decrease of left ventricular ejection fraction. Unlike trastuzumab, some data show that lapatinib could cross the blood–brain barrier, with some evidence of activity in treating or preventing brain metastases. Its evaluation is actively ongoing, in combination with trastuzumab and in the adjuvant setting.
Breast cancer is a significant health concern, with over 181,000 new cases diagnosed in 2008 in the USA and 40,000 deaths [1]. Most women diagnosed with invasive breast cancer have early-stage disease, and treatment-associated surgery, with eventual adjuvant chemotherapy and/or radiotherapy. However, even with optimal adjuvant therapy, a significant number of women develop recurrent disease. The evolution of systemic treatments for advanced or metastatic breast cancer (MBC) were the introduction of hormonal treatment in the 1940s, combined chemotherapy in 1969, and anthracyclines in 1972. In the 1990s, taxanes, gemcitabine, vinorelbine and capecitabine also entered clinical care [2]. More recently, the area of targeted therapies led to the development of HER2-targeted molecules, which have become a standard of care for patients with breast cancer.
HER2 is a member of the EGF-receptor (EGFR) family, which includes EGFR, HER2, HER3 and HER4. HER2 transduction is involved in the process of cell proliferation, cell invasion and angiogenesis. Approximately 20% of breast cancer patients have HER2 gene amplification, which results in glycoprotein overexpression [3].
HER2 amplification or overexpression has been associated with poor prognosis and is the major predictive factor for efficacy of trastuzumab, a human anti-HER2 monoclonal antibody [4].
In clinical trials, in association with chemotherapy, trastuzumab demonstrated that it significantly improved disease-free survival (DFS) and overall survival (OS) in patients with breast cancer overexpressing HER2 in the metastatic and adjuvant setting [5–9]. Unfortunately, some patients do not benefit from trastuzumab treatment, and almost all women with MBC eventually progress during trastuzumab therapy.
Several hypotheses have been raised to explain the mechanisms of resistance to trastuzumab: PTEN loss [10]; high level of extracellular cleaved HER2 (p95) [11], activation of alternative pathways (IGF-1 receptor) [12]; receptor masking or inaccessibility [13]; overexpression of ligands of the HER family (TGF-α) [14]; and/or expression of EGFR/HER3 heterodimers.
These last data suggest that combined inhibition of EGFR and HER2 can be synergistic against HER2-overexpressing breast cancer. Lapatinib is a small-molecule, reversible, dual kinase inhibitor of the tyrosine kinase activity of HER1 and HER2.
This review focuses on clinical efficacy and safety results of lapatinib, and the new perspectives in MBC.
Mechanism of action
Lapatinib is a compound developed from the quinazoline core that is able to reversibly inhibit the tyrosine kinase activity of both HER1 and HER2. It completes for the ATP-binding site on the tyrosine kinase domains of both EGFR and HER2, thereby preventing the use of ATP as a cofactor for phosphorylation of the tyrosine residue, a critical step during receptor activation. These interactions prevent the phosphorylations of tyrosine residues on signaling proteins and the subsequent signal transduction of both Ras/Raf mitogen-activated protein kinase and the phosphoinositol-3-kinase/Akt pathways, leading to an increase in apoptosis and decreased cellular proliferation [15,16].
Multiple in vitro studies have examined the activity of lapatinib upon inhibition of EGFR and HER2 activity. Biochemical kinase activity assays show lapatinib to be a potent inhbitor of EGFR and HER2 activity in which the 50% inhibitory concentration (IC50) of lapatinib was 10.8 and 9.2 nM for EGFR and HER2, respectively [17]. Moreover, lapatinib interacts synergistically with trastuzumab in vitro: studies based on cell lines demonstrated that lapatinib in combination with trastuzumab enhances apoptosis in HER2-overexpressing breast cancer cell lines and in cells resistant to trastuzumab monotherapy [18].
Pharmacokinetics
The pharmacokinetic parameters of lapatinib have been characterized previously in both healthy participants and in patients heavily pretreated with EGFR and/or HER2.
Lapatinib is administered as an oral drug and its absorption is incomplete and variable. After ingestion of an oral dose, measurable lapatinib concentrations appeared in plasma after a 15-min time lag, and they reached a maximum value at 4 h post-dose. Lapatinib elimination half-life increased with dose from 6 h to 14 h after single doses that ranged from 10 to 250 mg, and it increased with time after repeated dosing to an effective accumulation half-life of 24 h [19].
More than 99% of lapatinib is bound to albumin and α-1 acid glycoprotein. Lapatinib undergoes both hepatic and intestinal metabolism by cytochrome P450 (CYP) isoenzymes CYP3A4 and 3A5 [20]. The concomitant use of strong CYP3A4 inhibitors (e.g., ketoconazole, itraconoazole and clarithromycin) or strong inducers (e.g., dexamethasone, phenytoin and carbamazepin) should be avoided. Grapefruit may also increase plasma concentrations of lapatinib and should be avoided too. It is also a substrate and inhibitor of the efflux transporters breast cancer resistance protein (BCRP; ABCG2) and P-glycoprotein (Pgp; ABCB1) [21]. Less than 2% of the dose is excreted in urine.
The bioavailability of lapatinib is susceptible to variation when administered with food. A single 1500 mg oral dose of lapatinib was administered to 27 patients with advanced solid tumors on each of three occasions: after an overnight fast, with a low-fat breakfast, and with a high-fat breakfast [22]. The low-fat breakfast produced mean increases in lapatinib area under the concentration–time curve (AUC) of 167% (2.67-fold) and maximum concentration (Cmax) of 142% (2.42-fold). The high-fat breakfast produced mean increases in lapatinib AUC of 325% (4.25-fold) and Cmax of 203% (3.03-fold) compared with the fasted state. The results of this study indicate that administration of lapatinib 1500 mg with food dramatically increased its relative bioavailability and the associated absolute variability in AUC. Patients should be informed of the importance of taking lapatinib at least 1 h before or 1 h after a meal, in contrast with capecitabine, which should be taken with food or within 30 min after food.
Lapatinib in metastatic refractory breast cancer
Efficacy
Evidence for the efficacy of lapatinib in MBC was derived from trials of monotherapy and combination therapy involving patients with trastuzumab-refractory MBC.
Monotherapy
The first Phase II clinical trial was conducted by Blackwell in 2004 [23]. In this nonrandomized, open-label, multicenter, Phase II study, 78 patients with HER2-overexpressing MBC and progressive disease on prior trastuzumab-containing regimens were treated with lapatinib daily at a dose of 1250 mg or 1500 mg. Complete response (CR) and partial response (PR) were 7.7% for investigators and 5.1% for independent committees and clinical benefit rates were 14.1% and 9%, respectively. Median time to progression was 15.3 weeks and median OS was 79 weeks.
Burstein et al. reported on a multinational, open-label, Phase II trial of the efficacy and tolerability of lapatinib monotherapy in patients with advanced or refractory MBC who had previously been treated with an anthracycline, a taxane, capecitabine and trastuzumab [24]. The study EGF20008 included 229 patients with either HER2-positive (n = 140, cohort A) or negative (n = 89, cohort B) MBC. The patients received lapatinib 1500 mg/day. The majority of patients (76%) had received four or more previous lines of therapy and 95% had stage IV disease. The median time of previous trastuzumab exposure in patients with HER2-positive tumors was 73 weeks. No objective response occurred according to the response evaluation criteria in solid tumors (RECIST) in HER2-negative tumors. In the HER2-positive cohort, there were three PRs and three CRs for a response rate of 4.3% by investigators' assessment or 1.4% by independent review. A clinical benefit occurred in 5.7% of HER2-positive patients according to both the investigators' and independent review. The response rate in this trial is very low but must be examined in the context of refractory MBC.
Similarly, preliminary results from an open-label, Phase II trial in Japan in a cohort of 45 patients with HER2-overexpressing MBC refractory to anthracyclines, taxanes and trastuzumab indicated a clinical benefit of 33.3% with lapatinib monotherapy 1500 mg daily [25].
Combination
The most convincing results involving lapatinib are from a Phase III trial. Based on the encouraging results of the Phase I trial of lapatinib combination with capecitabine [26], a Phase III, randomized, open-label study comparing lapatinib in combination with capecitabine with capecitabine alone was conducted (EGF100151 study) [27]. This trial randomized 399 patients with HER2-positive, locally advanced or MBC that had progressed after treatment with anthracycline-, taxane- and trastuzumab-containing regimens, to receive either a combination therapy (lapatinib 1250 mg/day continuously plus capecitabine 2000 mg/m2 on days 1 through 14 of a 21-day cycle) or monotherapy (capecitabine alone at a dose of 2500 mg/m2 on days 1 through 14 of a 21-day cycle). The primary end point was time-to-progression (TTP) in the intent-to-treat (ITT) population (n = 324), and a prespecified blinded statistical interim analysis by an independent committee reported superiority of the combination therapy to monotherapy. There were 49 disease-progression events in the combination group and 72 in the monotherapy group and median TTP was 8.4 and 4.4 months, respectively. The hazard ratio (HR) for TTP with combination therapy was 0.49 (95% CI: 0.34–30.71; p < 0.001). Combination therapy was also associated with a higher response rate (22 vs 14%). At the time of the interim analyses, there were no differences in OS between groups. However, the number of events was not sufficient to draw conclusions from with reference to this end point. As a result, further enrolment of patients was discontinued, and patients receiving capecitabine alone were given the option of initiating lapatinib therapy. The US FDA approved lapatinib based on these results. An update of efficacy data and biomarker analysis from this pivotal trial that included 399 randomized women was recently published [28]. The addition of lapatinib prolonged TTP from 4.3 to 6.2 months with a HR of 0.57 (95% CI: 0.43–40.77; p = 0.00013) and provided a trend toward improved overall survival (HR: 0.78; 95% CI: 0.55–51.12; p = 0.177). As concluded by the authors, the results of this study support the use of lapatinib and capecitabine in women with progression of HER2-positive breast cancer after treatment with trastuzumab, anthracyclines and taxanes.
Lapatinib toxicity
There are considerable data regarding the safety and adverse effect profile of lapatinib. Similar to other EGFR small-molecule tyrosine kinases inhibitors, lapatinib is well tolerated and grade 3 or 4 adverse events are rare.
In the largest Phase III study published to date, the most common adverse event were diarrhea, hand-foot syndrome, nausea, fatigue, vomiting and rash. In the Cameron update of EGF 100151, the grade 3 diarrhea rate was 13% in the lapatinib arm versus 10% in patients receiving only capecitabine, and the grade 3 palmar plantar erythrodysesthesie rate was 12 versus 14%, respectively [28]. Prevalence of side effects leading to discontinuation was similar between patients receiving lapatinib and capecitabine (14%) and those patients receiving only capecitabine (14%). The adverse effect profile of lapatinib derived from Phase I and II trials is similar.
Diarrhea-related events owing to lapatinib have been underlined in a study by Di Leo comparing lapatinib plus paclitaxel with placebo plus paclitaxel as first-line therapy for MBC [29]. Patients were randomized to receive paclitaxel 175 mg/m2 every 3 weeks plus lapatinib 1500 mg/day or placebo. The incidence of diarrhea was significantly different between treatment arms (24 patients [8%] in the paclitaxel-lapatinib arm versus two patients [<1%] in the paclitaxel–placebo arm; p < 0.001). Crown et al., in a meta-analyses of more than 2000 patients with MBC from 11 clinical trials (Phase I, II or III) analyzed diarrhea events in patients treated with lapatinib [30]. Lapatinib was administered at doses ranging from 1000 to 1500 mg/day as monotherapy (n = 926) or in combination with capecitabine (n = 198) or taxanes (n = 687). Grade III diarrhea occurred in 9% of lapatinib-treated patients compared with 4% of patients not receiving lapatinib. Grade 4 events occurred in less than 1% of patients in both treatment group, and the mean number of diarrhea events per patient was similar in both groups (two events). Lapatinib-induced diarrhea was an early event (within 6 days) and was managed with loperamide and lomotil. The median duration of diarrhea with lapatinib was 5 days, with 85% of patients requiring no dose adjustment or interruption and only 2% discontinuing therapy owing to diarrhea. Finally, this side effect did not occur with a lapatinib-containing regimen when proactive monitoring and appropriate intervention were also employed.
Other reported side effects of lapatinib include rash (hand–foot syndrome and mouth syndrome) nausea and fatigue. In the large Phase III trial [15], grade 2/3 skin rash occurred in 29% of patients in the combination arm versus 14% in the capecitabine-alone arm. The majority of rash cases were mild-to-moderate with only 2% of grade 3 rash in the combination arm and 1% in the capecitabine-alone arm. This rash has been seen as a class effect of drugs that target the HER1 receptor. It's distribution predominates on sites with pilosebaceous units such as the scalp, face, neck and upper trunk. Lacouture et al. performed an analysis of dermatologic events (DEs) in patients with cancer treated with lapatinib [31]. DEs included hand–foot syndrome, rash, hair disorder, dry skin, pruritus/urticaria, skin disorder, skin infection and nail disorder. Among patients receiving lapatinib monotherapy, 55% experienced grade ½ DEs, 3% had grade 3 DEs and no grade 4 DEs were observed. Rash was the most common DE (43%, all grades) and developed between days 1 and 14 of starting treatment, with a median duration of 29 days. There were only 4 grade 3 rashes. Of the DEs, 3% led to a lapatinib dose reduction, 7% resulted in dose interruption, and 1% led to drug discontinuation.
Cardiac toxicity was an unexpected finding in the pivotal trial of trastuzumab [5] associated to HER2 targeting. Because lapatinib also targets HER2, cardiac toxicity was carefully monitored in the studies. However, in the pivotal Phase III study, EGF100151 [27], there were no reported differences in mean left ventricular ejection fraction (LVEF) between the two arms. In the combination arm, five women (one Printzmetal's angina/four asymptomatic) experienced cardiac events that were considered to be treatment related and fully recovered, whereas two patients in the capecitabine monotherapy group experienced a cardiac event unrelated to treatment; one remained unresolved. No symptomatic cardiac events occurred and there was no withdrawal from treatment owing to declines in LVEF, no cases of congestive heart failure and no decrease in the mean values in the group receiving lapatinib. Perez et al. analyzed the data from 44 studies involving 3689 patients enrolled in Phase I–III trials of lapatinib monotherapy and combination therapy in breast cancer and other solid tumors [32]. They reported cardiac events defined as symptomatic (grade 3 or 4 left ventricular systolic dysfunction according to the National Cancer Institute Common Terminology Criteria for Adverse Events) or asymptomatic (LVEF decreases > 20% relative to baseline and below the institution's lower limit of normal). Cardiac events were reported in 60 patients (1.6%) previously treated with anthracyclines (n = 12), trastuzumab (n = 14) or neither (n = 34). The event was not preceded by symptoms in 53 patients (83%) and the decrease in LVEF improved or resolved in 35 of these patients (88%) without any medication for the asymptomatic patients. The mean time of the onset of LVEF decrease was 13 weeks and the decrease was rarely severe. Thus, data from Perez support the notion that lapatinib-associated declines in LVEF, although potentially serious, are uncommon, usually asymptomatic and generally reversible. Of note, most patients treated with lapatinib in the meta-analysis had no prior exposure to cardiotoxic drugs such as anthracycline and/or trastuzumab, and thus represent a selected population.
Perspective
Lapatinib & prevention of CNS metastases
Brain metastases develop in a third of patients with advanced HER2-postive breast cancer who are treated with trastuzumab and represent a challenge in the management of these patients [33–37]. This high incidence of CNS disease could result from a higher tropism for the CNS of HER2-positive disease or inability of trastuzumab to cross the blood–brain barrier [38,39].
Unlike trastuzumab, lapatinib is a small molecule that could cross the blood–brain barrier. Evidence of the efficacy of lapatinib for HER2-positive brain metastases has emerged from the Phase III study of Geyer [27]. In this study, fewer women in the combination therapy group than in the monotherapy group had new or progressive CNS metastases (4 vs 11), although this difference did not reach statistical significance (p = 0.10). Its safety and efficacy was evaluated in patients with HER2-positive brain metastases in an open-label, Phase II study conducted by Lin and colleagues [40]. Single-agent efficacy, although modest, led to further investigation in an international Phase II trial (EGF105084) [41]. A total of 242 patients who had HER2-positive breast cancer and progressive brain metastases in spite of trastuzumab and cranial radiotherapy were included. The primary end point was CNS objective response evaluated by volumetric reduction of CNS lesions in the absence of increasing steroid use, progressive neurologic signs and symptoms, or progressive extra-CNS disease. Brain magnetic resonance images were obtained every 8 weeks and were evaluated by central independent review. Two cohorts were defined based on Eastern cooperative oncology group performance status (0 or 1 vs 2) and prior treatment with a trastuzumab-based regimen (1–2 vs >2). CNS objective response rate was 6% in both cohorts. A total of 15 PRs (6%) were observed and 88 patients (37%) had stable disease for 8 weeks or more. After release of the results of the Phase III capecitabine plus lapatinib trial, this study was amended to allow patients with progressive disease with lapatinib alone to receive the combination lapatinib plus capecitabine at the FDA-approved combination dose. A total of 50 patients entered the extension phase and 20% experienced a CNS objective response while 40% had stable disease (volumetric reduction of > 20% in the CNS), confirming the antitumor activity of lapatinib in the CNS. Brain metastases are a significant cause of morbidity and mortality in patients with HER2-positive disease. Although progress has been made in the treatment of this complication, new therapeutic options are needed and the evaluation of lapatinib in this setting must continue in combination with other cytotoxic agents. In patients with both early-stage and HER2-overexpressing breast cancer, evaluation of prophylactic strategies with HER2-inhibiting small molecules that are able to cross the blood–brain barrier should also be investigated.
Lapatinib in first-line therapy
Many investigations of lapatinib were conducted in highly refractory MBC and demonstrated promising antitumoral activity. These data supported investigation of lapatinib as first-line therapy in MBC. In the EGF20009 Phase II study, Gomez et al. assessed the efficacy and tolerability of two schedules of lapatinib monotherapy (1500 mg daily or 500 mg twice-daily) in 138 trastuzumab-naive patients with HER2-positive advanced disease [42]. An independent review confirmed 33 partial responses among the 138 patients. The majority of responses occurred by week 8 and the median duration of response was 28.4 weeks. With an overall response rate of 24%, these data were comparable to historical data for trastuzumab in the first-line setting for HER2-positive MBC [4]. No differences were observed between the two doses of lapatinib. A recent Phase III, randomized, double-blind study compared lapatinib (1500 mg daily) and paclitaxel (175 mg/m2 every 3 weeks) versus paclitaxel (175 mg/m2 every 3 weeks) plus placebo in 579 trastuzumab-naive patients with MBC [29]. The originality of this study was that patients were retrospectively tested in blind for HER2 status. There was no significant difference in TTP and OS between the two arms in the overall population. A preplanned, blinded subset analysis was conducted in the 86 centrally defined HER2-positive patients (15%) tested on archived samples (n = 49, paclitaxel–lapatinib arm; n = 37 paclitaxel–placebo arm). TTP was significantly longer in lapatinib-treated patients versus patients receiving placebo (median: 36.4 versus 25.1 weeks) with an unadjusted HR of 0.53 (95% CI: 0.31–0.89; p = 0.005) as well as objective response rate (ORR; 63.3 vs 37.8%; p = 0.004), clinical benefit rate (CBR; 69.4 vs 40.5%; p = 0.011) and event-free survival (EFS; 35.1 vs 21.9 weeks; HR: 0.52; 95% CI: 0.31–0.86; p = 0.023). Even though the OS rates were not mature at time of publication (only 43% of events had occurred at the time of data lock), in those 43%, median OS was longer in patients receiving lapatinib, but this was not statistically significant. Interestingly, in the Slamon study comparing standard chemotherapy versus standard chemotherapy with trastuzumab in women with MBC overexpressing HER2, 96 patients received paclitaxel (175 mg/m2 every 3 weeks) and 92 paclitaxel (175 mg/m2 every 3 weeks) with trastuzumab (2 mg/kg once a week) after a loading charge [5]. OS was 38% in the association group versus 16% in the paclitaxel monotherapy arm, TTP was 6.9 versus 3 months, respectively, and OS was 22.1 versus 18.4 months, respectively.
More recently, a study of combination of lapatinib with letrozole for front-line postmenopausal hormone-receptor (HR)-positive MBC was presented in San Antonio, TX, USA. A total of 1286 patients (219 HER2-positive) with HR-positive untreated MBC were randomized to once-daily treatment with letrozole 2.5 mg and lapatinib 1500 mg or letrozole and placebo. Patients were stratified according to presence of visceral versus bone only disease and time since completion of prior adjuvant endocrine tamoxifen therapy. Median PFS in the HER2-positive population was increased from 3 months in the letrozole group to 8.2 months in the lapatinib/letrozole group (HR: 0.71; 95% CI: 0.53–50.96; p = 0.019), showing that lapatinib significantly improved the clinical efficacy of letrozole for HR-positive and HER2-positive MBC [43].
Association of lapatinib with trastuzumab
Preclinical data showed that there is a potential synergy with no crossresistance in HER2-expressing cell lines [18,44]. Because of their different mechanism of action, it is necessary to assess the association of lapatinib and trastuzumab. Storniolo et al. reported the combination of escalating doses of lapatinib (750 mg/day and 500 mg/day) in combination with trastuzumab at 4 mg/kg as loading charge followed by 2 mg/kg/week in 54 patients with MBC [45]. Eight responses were observed among the first 50 evaluable patients, one CR and seven PRs. Toxicity was mostly diarrhea, nausea and fatigue and no patient experienced a symptomatic decline in LVEF. The recommended dose of lapatinib in this Phase I trial was 1000 mg/day in combination with a conventional dose of trastuzumab.
Cardiac toxicity is a common toxicity of these two treatments. Therefore, monitoring of cardiac function is very important. Storniolo analyzed data from four trials involving 238 patients who received the combination of lapatinib and trastuzumab [46]. A total of 203 patients received the combination of lapatinib plus trastuzumab and 35 received the combination with a taxane. With cardiac monitoring every 8 weeks, no patient had a symptomatic cardiac event, four patients (1.7%) had a single asymptomatic decrease in LVEF and one patient (0.4%) had two asymptomatic decreases. Two patients with a decrease in LVEF had received prior regimen with anthracyclines and two patients had received previous anthracycline and trastuzumab. Thus, this combination seems to be feasible and safe from a cardiac point of view. A randomized Phase III trial comparing lapatinib plus trastuzumab to lapatinib monotherapy in patients with MBC was reported in ASCO 2008. Subjects were randomized to receive either combination treatment: oral lapatinib 1000 mg daily plus trastuzumab 4mg/kg administered as an intravenous loading dose, followed by 2 mg/kg intravenous weekly or monotherapy treatment: oral lapatinib 1500 mg daily. In these 296 heavily pretreated patients, there was a significant improvement in clinical benefit rate and PFS for patients in the association arm [47].
Predictive factor of response
Breast cancer treatment is increasingly based on molecular profiling of the tumor rather than histology alone in order to administer the appropriate treatment to the corrcet patient, and thus to target the tumor and the putative responding patient. Identification of predictive factors of response to therapy is crucial.
Cameron reported an updated analysis of the pivotal trial with results of a biomarker analysis [28]. Tissue samples from 320 patients (80% of patients included) were tested for several biomarkers including EGFR by immunohistochemistry (IHC), and HER2 by fluorescence in situ hybridization (FISH) and IHC, and thus 241 patients were centrally confirmed as HER2-positive. The PFS was correlated with the HER2-positive status in an exploratory analysis. They looked for the performance of FISH and IHC predictors for benefit from the addition of lapatinib. In the 265 patients with HER2 amplification by FISH, irrespective of IHC results, addition of lapatinib to capecitibine improves PFS (HR: 0.471; 95% CI: 0.332–330.368; p < 0.0001) and in the 211 patients with 3 + staining on IHC, irrespective of FISH, results are similar (HR: 0.472; 95% CI: 0.318–310.701; p = 0.0002). However, the effect was uncertain in patients with HER2 IHC 2+ or HER2 IHC 1+ tumors. No association was identified between EGFR expression level and PFS. The baseline level of HER2 extracellular domain (ECD) was exmained in 367 (92%) patients. Previous studies suggest that elevated serum HER2 ECD levels correlate with poor response to chemotherapy [48]. These results confirm a clear correlation between high baseline HER2 ECD levels and shorter PFS in the capecitabine group, but not in the combination group.
Johnston et al. conducted a predictive biomarker analysis for response to advanced inflammatory breast cancer treated with lapatinib [49]. Baseline biomarkers (β-catenin, bcl-2, ER, heregulin, p53, pEGFR, pHER2, pHER3, PR, RhoC and TGF-α) were evaluated in 45 patients by IHC with the following scores 1+, 2+ or 3+. The remaining biomarkers, E-cadherin, IGF-1R and PTEN, were defined as positive when IHC values were 2+ or 3+. Patients were assigned to cohort A (HER2+; n = 30) and B (HER2-/EGFR+; n = 15). Only one patient in cohort B responded to lapatinib, demonstrating that HER2 overexpression but not EGFR is predictive of response to lapatinib. The baseline molecular profile of tumors that responded to lapatinib versus those that did not in cohort A showed that tumors that coexpressed pHER2 and pHER3 were more likely to respond than tumors that did not coexpress the phosphorylated receptors. In this study, 67% of clinical responders were PTEN deficient, suggesting that PTEN status does not preclude response to lapatinib. Press et al. recently performed a retrospective analysis of HER2 and EGFR status in available breast cancer tissue from EGF30001 (paclitaxel ± lapatinib in HER2-negative/unknown MBC; n = 579) and EGF100151 (capecitabine ± lapatinib in HER2-positive MBC; n = 399) [50]. HER2 gene amplification by FISH, HER2 mRNA by reverse transcription-PCR, HER2 protein expression by HercepTest IHC, EGFR mRNA level by RT-PCR, and EGFR protein by IHC were analyzed and compared with clinical outcome. He concluded that HER2 gene amplification/overexpression status, not EGFR status, correlates with lapatinib response, but lapatinib responsiveness is not related to the level of HER amplification among women with HER2-amplified breast cancer.
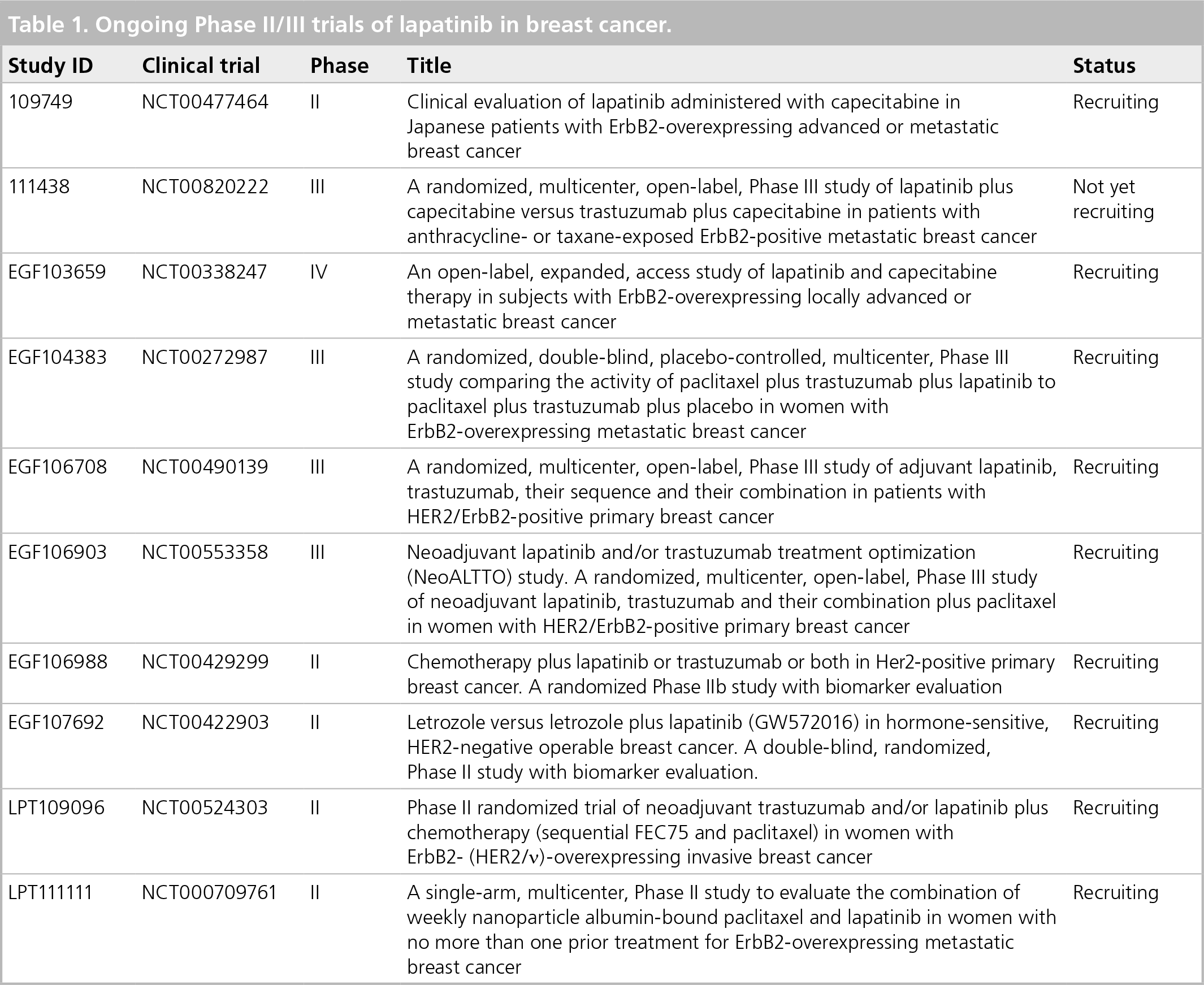
Ongoing trials
Numerous studies are in progress evaluating lapatinib as monotherapy or in combination with other therapies in a variety of cancers. Ongoing Phase II, III or IV trials of lapatinib in breast cancer are listed in Table 1 [101].
Ongoing Phase II/III trials of lapatinib in breast cancer.
Conclusion
Lapatinib is an oral, small-molecule, dual kinase inhibitor that targets reversibly both HER2 and EGFR. Lapatinib in association with capecitabine is now a standard of care for the treatment of patients with advanced or metastatic breast cancer whose tumors overexpress HER2 and who have received prior therapy including an anthracycline, a taxane and trastuzumab.
Future perspective
Several questions justify ongoing evaluations. Brain metastases are a particular challenge for women with advanced HER2-positive breast cancer. First, the role of lapatinib must be still investigated as a treatment for, but also to prevent or delay metastases to the CNS. Second, the synergy demonstrated by lapatinib and trastuzumab in vitro warrants clinical evaluations: their non-crossresistant mechanisms of action and nonoverlapping toxicity profiles provide the opportunity to associate the two drugs in order to prevent the development of resistance and to enhance the efficacy. At last, given the results in the metastatic setting, lapatinib is moving forward in early breast cancer trials through two multicentric international studies: Adjuvant Lapatinib and/or Trastuzumab Treatment Optimization (ALTTO) and Neoadjuvant Lapatinib and/or Trastuzumab Treatment Optimization (NeoALTTO) studies.
Executive summary
Lapatinib is the first reversible, dual kinase inhibitor of the EGF receptor (EGFR) and HER2.
Lapatinib interrupts signal transduction from EGFR and HER2 by competing with ATP for the intracellular ATP-binding domain of these tyrosine kinase receptors.
Lapatinib is administered as an oral drug and its absorption is incomplete and variable.
Lapatinib is bound to albumin and α-1 acid glycoprotein and is extensively metabolized by cytochrome P450 (CYP) isoenzymes, CYP3A4 and 3A5.
Lapatinib is a substrate and inhibitor of the efflux transporters breast cancer resistance protein (ABCG2) and P-glycoprotein (ABCB1).
The bioavailability of lapatinib is susceptible to alteration when administered with food.
Lapatinib is approved in combination with capecitabine for the treatment of patients with advanced or metastatic breast cancer whose tumors overexpress HER2 and who have received prior therapy including an anthracycline, a taxane and trastuzumab.
The recommended dose of lapatinib is 1250 mg administered orally once-daily for 21 days in combination with capecitabine 2000 mg/m2/day (administered orally in two doses 12 h apart) on days 1–14 in a 21-day cycle.
Most adverse events are generally mild to moderate.
Diarrhea, palmar plantar erythrodysesthesia, rash, nausea and fatigue are mostly reported.
Cardiac toxicity occurs rarely and mostly as an asymptomatic and reversible decrease of left ventricular ejection fraction.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
