Abstract
Breast cancer is the most common form of cancer and the second leading cause of cancer deaths among women in the USA. A quarter of all newly diagnosed cases are characterized by overexpression of HER-2 which is associated with poor prognosis. In March 2007, lapatinib, an oral dual-tyrosine kinase inhibitor was approved in combination with capecitabine for metastatic HER-2-positive breast cancer that has progressed on prior trastuzumab therapy, and in combination with letrozole for postmenopausal women with HER-2 and hormone receptor-positive advanced breast cancer. In the future, lapatinib may play an important role in dual HER-2 blockade with trastuzumab and other targeted agents for women with HER-2-overexpressing breast cancer.
Breast cancer is the most common form of cancer in women worldwide. In the USA, breast cancer accounts for approximately 200,000 new cases every year and is the second leading cause of cancer deaths among women. It is responsible for over 40,000 estimated deaths in 2013 [1]. About a third of all women with early stage cancer will eventually relapse with distant metastases [2,3].
An increased understanding in molecular signaling pathways has facilitated the development of novel targeted therapies. Based on tumor biology, breast cancer can be further classified in several distinct subtypes. A quarter of all newly diagnosed cases are characterized by overexpression of HER-2 on the surface of tumor cells [4,5]. HER-2 belongs to the ERBB family of receptor tyrosine kinases and has been shown to promote growth and survival of various epithelial malignancies. HER-2 overexpression in breast cancer has been associated with poor prognosis and overall survival (OS) [4]. As per recent 2013 recommendation from the American Society of Clinical Oncology and College of American Pathologists, a tumor is identified as overexpressing HER-2 either based on circumferential membrane staining that is complete, intense by immunohistochemical staining (scored as 3+ cell surface protein expression) or in situ hybridization (ISH) positive based on single probe average HER-2 copy number of >6.0 signals/cell or dual-probe HER-2/CEP17 ratio ≥2.0 with an average HER-2 copy number ≥4.0 signals/cell or a dual-probe HER-2/CEP17 ratio ≥2.0 with an average HER-2 copy number <4.0 signals/cell or dual-probe HER-2/CEP17 ratio <2.0 with an average HER-2 copy number ≥6.0 signals/cell. Immunohistochemical for HER-2 scored as 2+ requires further validation by ISH [6,7].
Trastuzumab (Herceptin, Genentech. Inc., CA, USA), a humanized monoclonal antibody directed against the extracellular domain of HER-2, was initially approved by the US FDA for HER-2-positive metastatic breast cancer in September 1998. The Phase III randomized trial that led to this approval enrolled 469 patients and showed that the addition of trastuzumab to chemotherapy improved objective response rates (ORRs) compared with chemotherapy alone (50 vs 32%, respectively; p < 0.001); time to progression (TTP) (median: 7.4 vs 4.6 months respectively; p < 0.001); duration of response (median, 9.1 vs 6.1 months, respectively; p < 0.001); and OS (median, 25.1 vs 20.3 months, respectively; p = 0.01) [8]. Since then, trastuzumab has altered the natural history of HER-2-positive disease [8,9], leading to significantly improved long-term survival. It is the standard of care for adjuvant, metastatic and neo-adjuvant HER-2-positive breast cancer [10,11].
The role of trastuzumab in primary breast cancer cannot be understated; however, it is associated with some limitations. The incidence of symptomatic heart failure in several trials evaluating single agent trastuzumab in metastatic setting is reported to be between two and 7% [12,13]. This risk is further increased with concurrent or sequential use of anthracycline in the adjuvant setting [13].
Trastuzumab is FDA approved for iv. (intravenous) administration; however, a fixed subcutaneous dosing schedule has been shown to be noninferior and acceptable alternative in HER-2-positive early breast cancer in a recent Phase III trial [14].
Moreover, trastuzumab therapy is complicated by acquired and denovo resistance [15]. Almost half of metastatic HER-2-positive disease will progress within 1 year of initiating treatment [16]. Trastuzumab has been associated with an increase in risk of CNS metastases as a site of first tumor recurrence [17,18]. Although it is a large monoclonal antibody with limited ability to cross the blood brain barrier, current evidence suggests that it improves OS and prognosis in HER-2-positive patients with brain metastases as a result of better control of systemic disease [19].
In March 2007, the US FDA approved labeling for Lapatinib (Tykerb, GlaxoSmithKline), an oral small molecule dual tyrosine kinase inhibitor with promising activity in HER-2-positive breast cancer.
Pharmacology
The ERBB protein family consists of four homologous members, in other words, ERBB 1/EGFR, ERBB 2/HER-2, ERBB 3/HER-3 and ERBB 4/HER-4. These receptors are composed of an extracellular domain that binds to ligands such as EGF, TGF-α, amphiregulin and neuregulin among others; a transmembrane region and a cytoplasmic tyrosine kinase domain. All ERBB isoforms exist in a predimerized state. HER-2 has no known endogenous ligands; however, it plays a vital role in forming heterodimers with ERBB 1, ERBB 3 and ERBB 4. Ligand binding induces a conformational change, protein re-arrangements and subsequent dimerisation of ERBB isomers. This results in activation of tyrosine kinases, autophosphorylation and recruitment of downstream signal transduction via MAPK and PI3-AKT cascades [4–5,20–21].
Lapatinib (GW572016) is a reversible, potent, small molecule tyrosine kinase inhibitor against the cytoplasmic tyrosine kinase domains of ERBB 1 and HER-2. Lapatinib, unlike trastuzumab, is active against the intracellular ATP binding site, thereby inactivating the PI3-AKT and MAPK pathways leading to apoptosis and cell death [22,23]. Figure 1 illustrates mechanism of action of lapatinib via ERBB signaling pathway.
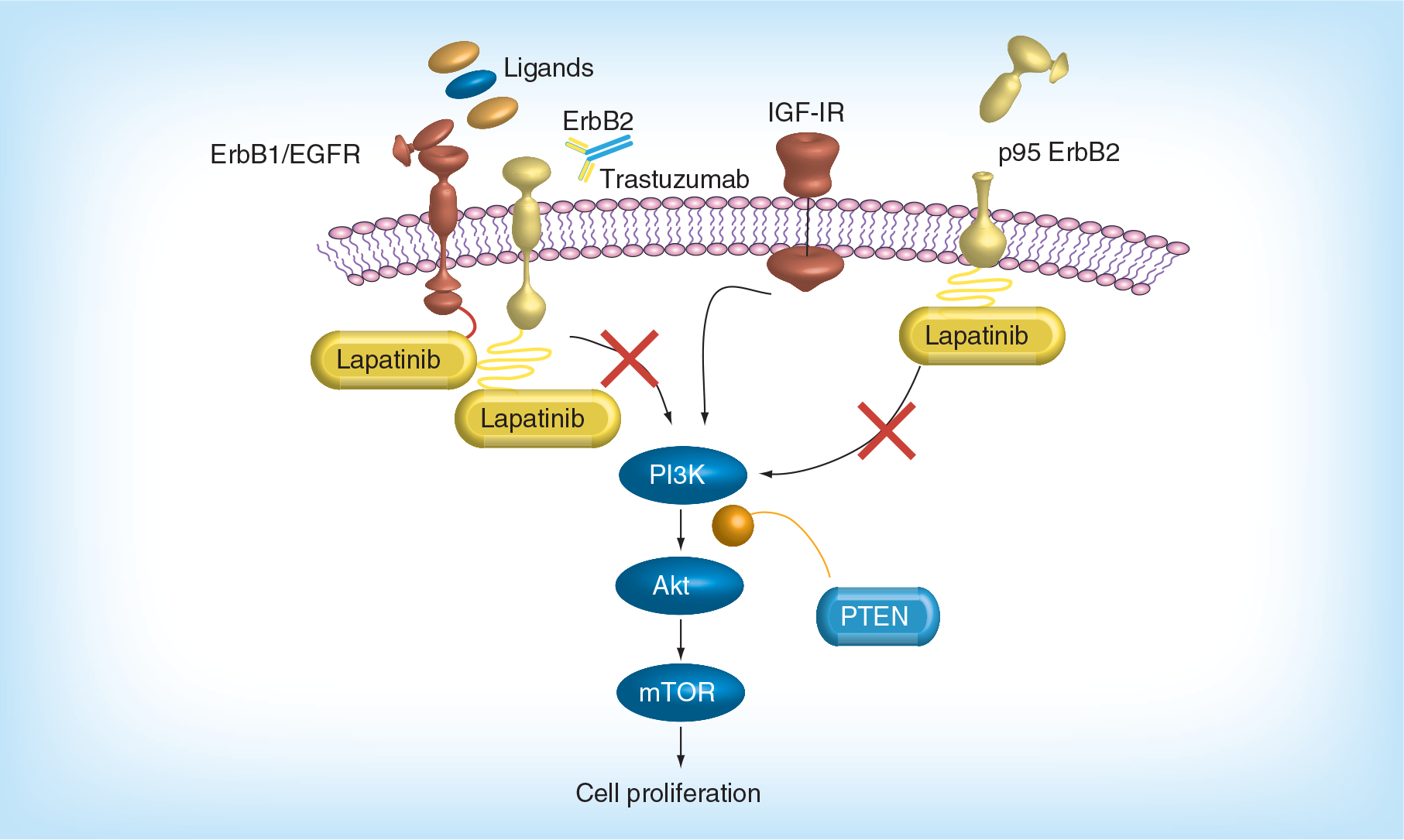
Preclinical laboratory studies suggest that lapatinib retains significant antitumor activity in HER-2-overexpressed, trastuzumab conditioned cell lines. In one study, lapatinib was shown to inhibit IGF-1 signaling [24]. O'Brien et al. evaluated the response of lapatinib and trastuzumab in 18 HER-2-overexpressed cell lines and showed that increased activation of PI3-AKT pathway was a potential mechanism for trastuzumab resistance that could be overcome by lapatinib [25].
Approximately 30% of HER-2-positive breast cancer lines express an amino terminally truncated carboxyl terminal fragment of HER-2, p95HER 2, which lacks the extracellular binding domain and are resistant to trastuzumab. These tumors have been associated with increased nodal metastases and shorter disease-free survival when compared with patients that overexpress the full-length HER-2 receptor [26,27]. Scaltriti et al. demonstrated that lapatinib retained antitumor activity in trastuzumab resistant truncated p95HER 2 isoforms as well [28]. These results were confirmed in a clinical sample combining patients from two large lapatinib studies (EGF 20009 and EGF 100151) with advanced HER-2-positive breast cancer. Tumors expressing p95HER 2 were sensitive to lapatinib. In fact, progression free survival and clinical benefit rate in both studies were not statistically different in the p95HER 2 positive and p95HER 2 negative groups [29]. Taking in consideration that p95HER 2 tumors are resistant to trastuzumab, these results suggest that lapatinib, as well as other TKIs, may be a preferred therapeutic option for these tumors.
Pharmacokinetics
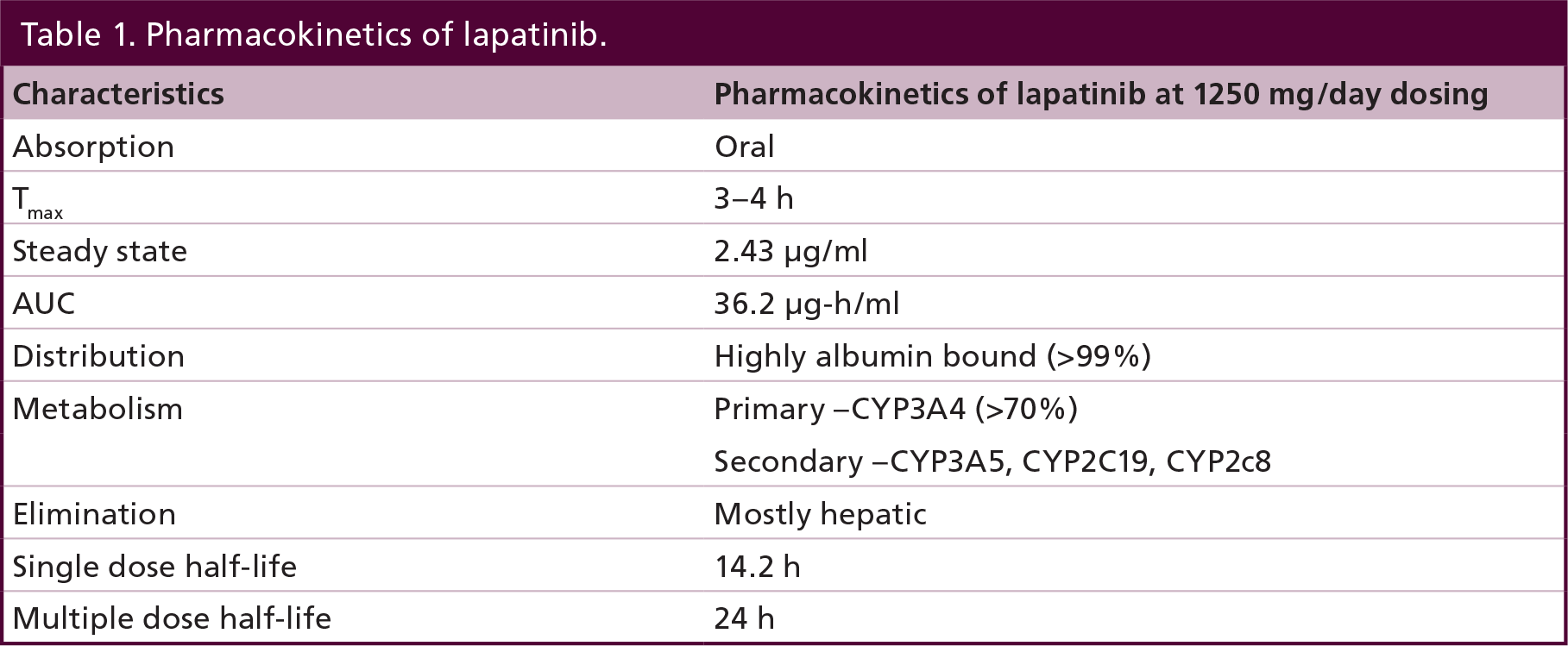
Lapatinib is orally administered as the monohydrated ditosylate derivative. In an initial Phase I study, Bence et al. evaluated safety and pharmacokinetics of lapatinib in single and multiple dose regimens from 10 to 1600 mg [30]. After oral administration, lapatinib reaches peak plasma concentration within approximately 4 h. Steady state levels can be achieved in 6–7 days, with an effective half-life of 24 h with daily dosing. Divided daily doses resulted in approximately twofold higher exposure at steady state AUC compared with the same total dose administered once daily. The area under serum concentration-time curve (AUC) for lapatinib increases threefold to fourfold when administered with food, hence it is preferred to take lapatinib on an empty stomach [31]. The difference in pharmacokinetics with multiple daily doses and the food effect should be kept in mind when designing trials, and more importantly, during patient instruction. Lapatinib is highly protein bound (>99%) to albumin and α 1 glycoprotein [31,32].
Elimination of the drug occurs mainly through fecal excretion (median fecal excretion 27%, ranging between 3 and 67%) and less than 2% is recovered in urine. Lapatinib has not been studied in patients with moderate and severe hepatic impairment (Child-Pugh B and C, respectively); however, a dose reduction to 750 mg once daily is recommended for patients with severe hepatic impairment. Lapatinib has not been studied in pediatric patients or patients with renal impairment [32]. Please refer to Table 1 for a summary of pharmacokinetics of lapatinib at a daily dose of 1250 mg/day.
Pharmacokinetics of lapatinib.
Lapatinib is extensively metabolized in the liver via cytochrome P450 (CYP3A4, CYP3A5, CYP2C19 and CYP2C8), primarily through CYP3A4. It is a substrate and inhibitor of CYP3A4. Smith et al. (2004) studied pharmacokinetic effects of CYP3A4 inhibition with concomitant ketoconazole in 22 patients receiving a single 100 mg daily dose of lapatinib [33]. There was a 3.6-fold increase in AUC of lapatinib and a 1.7-fold increase in elimination half-life compared with control. Likewise, Herendeen et al. (2004) showed that carbamazapine, a known CYP450 inducer, resulted in a 72% reduction in AUC [34]. Hence, concomitant administration of strong CYP3A4 inhibitors should be avoided in patients receiving lapatinib. In addition, antacids should be avoided for 1 hour before and after lapatinib administration, because of interference with absorption (please refer to Table 2 for a list major drug interactions with lapatinib).
Major drug interactions with lapatinib.

Clinical trials
Lapatinib's efficacy in breast cancer has been evaluated in several studies in advanced, metastatic and recently in neo-adjuvant trials. Lapatinib has been tested alone or in combination with hormonal, biologic or chemotherapeutic agents. (Please see Tables 3–5 for a summary of major clinical trials with lapatinib and common adverse events reported).
Summary of major Phase III trials of lapatinib combination therapy in metastatic breast cancer.

Summary of clinical trials of lapatinib in HER-2-positive early breast cancer.

List of Grade 3 or 4 adverse events with lapatinib combination therapy.

LVEF: Left ventricular ejection fraction.
Lapatinib in metastatic setting Monotherapy
Burris et al. (2005) reported a Phase I dose escalation trial with lapatinib in 67 heavily pretreated metastatic solid tumors overexpressing ERBB 1 and/or ERBB 2. This study noted four partial responses; all four were breast cancer patients. Overall, lapatinib was well tolerated in the study at doses up to 1600 mg daily with clinical activity observed at doses ranging from 650 to 1600 mg/day. The most frequent side effects observed were diarrhea (42%) and skin rash (31%). No grade 4 toxicities were observed. This study did not report any cardiotoxicity or interstitial pneumonitis seen with other ERBB inhibitors [31].
Phase II trials evaluating safety and efficacy of lapatinib in HER-2-positive breast cancer patients previously treated with trastuzumab have shown ORRs between 20 and 30% [35]. Gomez et al. reported results from a Phase II randomized study of lapatinib as first-line monotherapy in HER-2-positive locally advanced or metastatic breast cancer [36]. A total of 138 patients were treated with 1500 mg daily or 500 mg twice daily dosing for a median of 17.6 weeks. Seventy-six percent had hormone receptor negative disease. Observed overall response rate was 24% and the progression-free survival (PFS) at 6 months was 43%. No significant difference was observed in outcomes between the two dosing groups. Although the authors concluded that lapatinib was effective as monotherapy in first-line setting, this did not appear to offer significant clinical benefit over single agent trastuzumab. A randomized trial comparing the two in first-line setting has not been reported.
Combination therapy Lapatinib with chemotherapy
In the Phase III randomized trial that led to the FDA approval, the efficacy and safety of lapatinib was evaluated in combination with capecitabine in 399 patients with advanced or metastatic HER-2 overexpressing breast cancer previously treated with an anthracycline, a taxane and trastuzumab. This trial compared lapatinib at a daily dose of 1250 mg in combination with capecitabine at 2000 mg/m2 on days 1–14 of a 21-day cycle to capecitabine alone at 2500 mg/m2 on days 1–14 every 21 days. The median TTP was 27.1 versus 18.6 weeks (HR: 0.57; p = 0.00013) for the lapatinib combination and capecitabine alone arms, respectively. The response rates were 23.7 versus 13.9% for the lapatinib combination and the capecitabine alone arms, respectively. There was no statistical difference in OS between the two groups; however, early termination and crossover of control arm patients to the lapatinib combination at disease progression in the study design made it difficult to interpret OS [37].
Although the toxicities observed with the lapatinib combination were similar to those in the capecitabine alone arm, an increased incidence of diarrhea and rash was noted with the combination. The most frequent adverse reactions during treatment with combination were diarrhea (65%), palmar-plantar erythrodysesthesia (PPE; 53%), nausea (44%), rash (28%), vomiting (26%) and fatigue (23%). Grade 3 or 4 adverse events that occurred with a frequency greater than 5% in patients on the combination arm were diarrhea (13%) and PPE (12%). A 2% incidence of generally reversible decreased left ventricular function in the combination arm was noted [37].
Another randomized placebo controlled double-blind Phase III study evaluated lapatinib in combination with paclitaxel in the first-line setting in 579 women with advanced metastatic breast cancer. Eighty-six patients (15%) tested positive for HER-2 overexpression. In this particular subset of patients, treatment with paclitaxel-lapatinib resulted in statistically significant improvements in PFS (35.4 vs 25.1 weeks, respectively; p = 0.005), ORR (53.3 vs 37.8%, respectively; p = 0.023) and clinical benefit rate (CBR) (69.4 vs 40.5%, respectively; p = 0.011) compared with paclitaxel-placebo. No differences between treatment groups were observed for any end point in HER-2-negative patients. The rate of cardiac events was low, and no difference in cardiac toxicity was observed between treatment arms [38].
Lapatinib with endocrine therapy
On 29 January 2010, the FDA granted accelerated approval to lapatinib in combination with letrozole for the treatment of postmenopausal women with hormone receptor-positive metastatic breast cancer that overexpresses the HER-2 receptor.
The Phase III trial that led to this FDA approval was a multinational randomized placebo-controlled trial of lapatinib plus letrozole versus placebo plus letrozole in 1286 patients with hormone receptor-positive metastatic breast cancer who had not received prior therapy for metastatic disease. Patients were randomly assigned to receive lapatinib (1500 mg once daily) plus letrozole (2.5 mg once daily) or placebo plus letrozole (2.5 mg once daily). Among 219 patients (17%) who were HER-2-positive, the lapatinib plus letrozole combination had a median PFS of 35.4 weeks, compared with 13.0 weeks for the placebo plus letrozole arm (HR: 0.71; p = 0.019); ORR increased from 15 to 28%(OR: 0.4; 95% CI: 0.2–0.9; p = 0.021). The difference in OS was not statistically significant [39].
The safety profile of lapatinib in the study population was consistent with previously reported clinical data. The most common adverse reactions (at least 10%) in the lapatinib plus letrozole arm were diarrhea, rash, nausea and fatigue. The frequency of grade 3 or 4 decreases in left ventricular ejection fraction was less than 1% [39].
Lapatinib with trastuzumab (dual HER-2 blockade)
Preclinical data indicate lapatinib has a synergistic effect in combination with trastuzumab in HER-2-positive breast cancer cell lines [40]. Scaltriti et al. were able to show enhanced antiproliferative and increased apoptosis of this combination in vivo in HER-2-positive tumor xenografts [41]. Lapatinib reduces HER-2 ubiquination, prevents HER-2 degradation and induces the formation of inactive dimers at the cell surface that in turn provides an increase in trastuzumab binding and a greater trastuzumab-mediated immune response.
An initial Phase I study to evaluate safety and dosing of lapatinib (750–1500 mg once daily dosing) in combination with trastuzumab in 54 patients with HER-2 overexpressing metastatic breast cancer revealed the optimal tolerated regimen to be 1000 mg daily of lapatinib with standard weekly trastuzumab. The most frequent drug-related grade 3 toxicities included diarrhea (17%), fatigue (11%) and rash (6%). There was no increase in cardiotoxicity with the combination [42]. It was noted that there was significant fatigue in the 24 h following the trastuzumab infusions; for this reason three-weekly dosing of trastuzumab was not attempted.
The Phase I trial demonstrated a significant clinical activity of dual HER-2 blockade, even in patients that had been heavily pretreated. This prompted the development of a randomized Phase III trial which demonstrated that lapatinib plus trastuzumab improved PFS (12.0 vs 8.1 weeks; HR: 0.73; 85% CI: 0.57–0.93; p = 0.008) and OS (14 vs 9.5 months; HR: 0.74; 95% CI: 57–0.97; p = 0.026) compared with lapatinib monotherapy in a study population of 291 patients with HER-2-positive metastatic breast cancer who experienced disease progression during prior trastuzumab therapy. Improvements in absolute OS rates were 10% at 6 months and 15% at 12 months in the combination arm. This survival benefit was more significant in the 163 patient who had HER-2-positive and ER-negative disease. Of note, 77 patients in this trial crossed over from control arm to combination therapy at disease progression [43].
Grade 3 and 4 adverse events were experienced in 26% patients versus 16% in combination and lapatinib monotherapy arms, respectively. The incidence of grade ≥3 diarrhea was similar between the two treatment arms (seven vs 7%). Rash was noted at a higher incidence in patients receiving monotherapy compared with patients receiving combination therapy (29 vs 23%, respectively).
Ten (4.4%) out of 223 patients who received combination treatment experienced grade 3 or worse cardiac events, which is consistent with the incidence of heart failure noted with trastuzumab monotherapy in previous trials. There did not appear to be an increased risk of decline in left ventricular (LV) function with this combination. This study suggests that dual HER-2 blockade represents an effective and safe nonchemotherapeutic approach in patients with HER-2-positive advanced metastatic breast cancer that have progressed on prior trastuzumab containing regimens [43].
Lapatinib & other nonchemotherapeutic agents
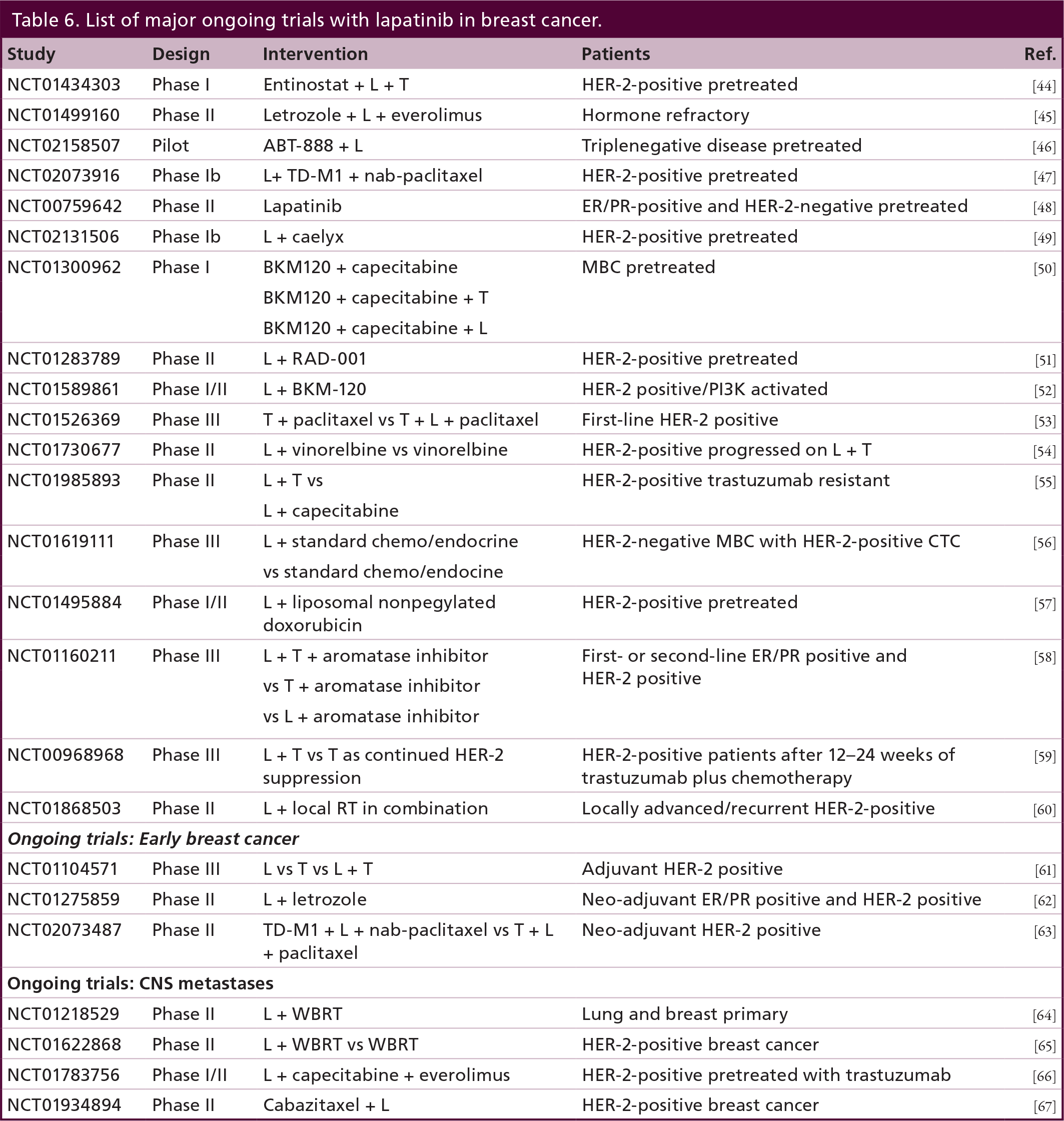
Please refer to Table 6 for a summary of major ongoing clinical trials with lapatinib in HER-2-positive breast cancer.
List of major ongoing trials with lapatinib in breast cancer.
Lapatinib has been studied in combination with other biologic agents in metastatic breast cancer. A Phase II study of lapatinib in combination with pazopanib (oral small molecule inhibitor of VEGFR, c-KIT and PDFGFR) versus lapatinib alone in treatment of HER-2-positive metastatic breast cancer yielded ORR rates of 44% and reduction in disease progression from 27 to 19% in prespecified interim analyses [68].
Lapatinib in early breast cancer
Lapatinib alone demonstrates fairly limited activity in the adjuvant setting. In the TEACH trial, 3161 HER-2-positive early breast cancer patients who had previously received adjuvant therapy but not trastuzumab, were randomized to receive daily lapatinib 1500 mg versus placebo for 12 months. Lapatinib failed to show significant benefit in PFS compared with placebo (HR: 0.83; 95% CI: 0.70–1.00; p = 0.053) [69].
Several neo-adjuvant trials have assessed the efficacy of the lapatinib and trastuzumab combination in HER-2-positive early breast cancer. The Phase III randomized NeoALTTO study enrolled 455 invasive operable HER-2-positive breast cancer patients. A total of 154 patients received lapatinib (1500 mg), 149 trastuzumab and 152 the combination of lapatinib (1000 mg) and trastuzumab. Anti-HER-2 therapy alone was given for the first 6 weeks; weekly paclitaxel was then added to the regimen for a further 12 weeks, before definitive surgery was undertaken. After surgery, patients received adjuvant chemotherapy followed by the same targeted therapy as in the neo-adjuvant phase to 52 weeks. Pathologic complete response (pCR) rate was significantly higher in the combination arm (51.3%; 95% CI: 43.1–59.5]) compared with trastuzumab alone (29.5%; 95% CI: 22.4–37.5; difference 21.1%; 95% CI: 9.1–34.2; p = 0.0001). However, there was no significant difference in pCR between the lapatinib (38 of 154 patients [24.7%; 18.1–32.3]) and the trastuzumab (difference −4.8%; −17.6–8.2; p = 0.34) groups. No major cardiac dysfunction occurred. Frequency of grade 3 diarrhea was higher with lapatinib alone (23.4%) and lapatinib plus trastuzumab (21.1%) than with trastuzumab monotherapy (2.0%). Similarly, grade 3 liver-enzyme alterations were more frequent with lapatinib (17.5%) and lapatinib plus trastuzumab (9.9%) than with trastuzumab alone (7.4%) [70].
The Phase III NSABP B-41 trial evaluated the efficacy and safety of neo-adjuvant combination chemotherapy (doxorubicin/cyclophosphamide) followed by paclitaxel plus lapatinib or trastuzumab or the combination of both in 529 women with HER-2-positive, operable breast cancer. The pCR rates were 53.2, 52.5 and 62% for the lapatinib, trastuzumab and trastuzumab/lapatinib arms, respectively. Grade >3 toxic effects included diarrhea (2, 20 and 27% with lapatinib, trastuzumab and trastuzumab/lapatinib arms, respectively) and symptomatic left ventricular systolic dysfunction (4, 4 and 2%, respectively) [71].
In another multicenter neo-adjuvant Phase II study by Rimawi et al. (2013), 64 women with stage II and III operable HER-2-positive disease were treated with dual blockade without chemotherapy. All eligible patients received trastuzumab weekly and daily lapatinib (1000 mg) for 12 weeks. Women with ER positive disease also received daily letrozole. Overall, in-breast pathologic complete response (pCR; ypT0-is) was 27% (ER positive: 21%; ER negative: 36%). The rate of low-volume residual disease (ypT1a-b) was 22% (ER positive: 33%; ER negative: 4%). This trial suggests that some select patients with HER-2-positive tumors may not need chemotherapy, and more complete blockade of HER-2 and ER receptors may represent an effective treatment option in these women [72].
In contrast to the high pCR observed with this combination in the neo-adjuvant setting, first results from the large Phase III randomized adjuvant dual HER-2 blockade ALTTO trial recently discussed at American Society of Clinical Oncology June 2014 annual meeting were disappointing [73]. From June 2007 to July 2011, 8381 nonmetastatic invasive HER-2-positive patients were randomized to one of four arms – either lapatinib (L) and trastuzumab (T), trastuzumab followed by lapatinib, lapatinib alone or trastuzumab alone. Anti-HER-2 therapy was initiated after completing all chemotherapy (n = 4613), concurrently with a taxane following anthracycline (n = 3337), or concurrently with a nonanthracycline, platinum-containing regimen (n = 431). The lapatinib monotherapy arm was closed in Aug 2011 for futility and was not presented. The primary endpoint for this trial was invasive disease-free survival (DFS). L + T versus T arms were tested for superiority and T→L versus T were tested for non inferiority at 1.11 margin. A value of p < 0.025 was required for statistical significance. At a median follow up of 4.5 years there was no statistical difference observed in DFS in either arms. HR for DFS was 0.84 (97.5% CI: 0.70–1.02; p = 0.048; 4-year DFS% = 88 vs 86%) for L + T versus T and 0.93 (97.5% CI: 0.76–1.13; noninferiority p = 0.044; 4-year DFS% = 87 vs 86%) for T→L versus T. Diarrhea (75 vs 20%), rash (55 vs 20%) and hepato-biliary (23 vs 16%) adverse events were more frequent in L + T versus T. Cardiac safety was promising (<1% primary cardiac events) considering >97% study population had received an anthracycline-based regimen in addition to anti-HER-2 therapy.
The failure of the ALTTO to confirm results with neo-adjuvant NeoALTTO study has profound implications in drug development in early breast cancer. For over 2 years, pCR from smaller and more feasible neoadjuvant studies has been used as a surrogate marker to obtain accelerated drug approval. However, disparate results from the largest randomized adjuvant trial conducted in HER-2-positive patients raises questions to this approach.
Lapatinib in inflammatory breast cancer
Although inflammatory breast cancer is uncommon (<5% of all breast cancers) in the USA, it represents a highly aggresive form that predominantly affects the younger population. Johnston et al. (2008) conducted a Phase II study of lapatinib in 45 patients with recurrent or anthracycline refractory inflammatory breast cancer who had not received previous trastuzumab therapy. Fifty percent of HER-2-positive patients exhibited a clinical response [74]. Another Phase II trial reported by Spector et al. (2006), which evaluated lapatinib monotherapy for treatment-refractory inflammatory breast cancer (IBC), indicated that high ERBB2, phosphorylated-ERBB2 and IGF-IR co-expression are predictors for clinical response to lapatinib monotherapy in patients with treatment-resistant inflammatory breast cancer. ERBB2 overexpression, but not ERBB1 expression alone, indicates sensitivity to lapatinib in this subset of breast cancer. The study enrolled 34 relapsed/refractory IBC patients. However, clinical response data was available from 22 patients of which 17 had biopsies analyzed at a reference lab and were assigned to Cohorts A (n = 11) and B (n = 6). Cohort A was comprised of ERBB2- positive patients, irrespective of ERBB1 status, while cohort B consisted of patients with ERBB1-positive and ERBB2-negative tumors. Cohort A showed clinical responses in eight of the 11 patients, whereas none of the six patients in cohort B showed any clinical response [75].
Kaufman et al. reported on an open-label Phase II study of 1500 mg of lapatinib given once daily in 126 patients with relapsed or refractory ERBB2-positive inflammatory breast cancer. Although none of the patients had a complete response, 39% of the patients had a partial response. The findings included a median response of 19.4 months (95% CI: 12.8–26.4) and a median OS of 11.2 months (95% CI: 9.1–13.5) for patients who responded to lapatinib, compared with a median OS of 8.4 months (95% CI: 5.9–9.4) for nonresponders to lapatinib [76].
Lapatinib in CNS metastases
Brain metastases occur in 30–50% of patients with metastatic HER-2-positive breast cancer. Preclinical data with lapatinib in mice models have shown promising results [77].
In an open-label Phase II trial, 39 patients with HER-2-positive breast cancer patients with new or progressive brain metastases while on trastuzumab therapy were given lapatinib monotherapy at a starting dose of 750 mg twice a day in 4-week cycles. If patients had not received previous radiation therapy, the patients were required to be asymptomatic. The median time to tumor progression was 3 months (95% CI: 2.3–3.7). Seven patients were progression-free in both central nervous system and non-CNS sites at 16 weeks, and three patients had a decline in volume of CNS metastasis by 30% [78].
A Phase II trial compared lapatinib plus capecitabine with lapatinib plus topotecan in patients with progressive CNS metastases after trastuzumab and radiotherapy. The lapatinib plus topotecan arm was terminated after 22 patients were enrolled, as toxicity and a lack of efficacy were shown. The ORR in CNS for the lapatinib plus capecitabine arm was 38% (95% CI: 13.9–68.4) [79]. The more recent Phase II LANDSCAPE study, evaluated the activity of lapatinib plus capecitabine on CNS metastases in 45 patients with HER-2-positive MBC who had not received previous whole-brain radiation therapy. Twenty-four patients (57%) achieved an objective CNS response, 22 patients (52%) had a partial response (PR) and 2 (5%) had a complete CNS response (CR). Median TTP was 5.5 months and median OS was 17.0 months [80]. These results suggest a role for lapatinib-based therapy in the treatment of CNS metastases.
Novel HER-2 agents
Although trastuzumab and lapatinib have proven clinical benefit in HER-2-positive advanced breast cancer, most patients will eventually progress within a year of therapy. New targeted therapies have been in development in hope of overcoming resistance. One such agent that has received the US FDA approval in metastatic and neo-adjuvant setting is pertuzumab, a humanized monoclonal antibody that binds at a different epitope of the HER-2 extracellular domain (subdomain II) compared with trastuzumab (subdomain iv.). Pertuzumab prevents HER-2 from dimerizing with other ligand-activated HER receptors, most notably HER-3. Pertuzumab was approved for front line metastatic HER-2-overexpressing breast cancer patients based on Phase III study of 808 patients with HER-2-positive metastatic breast cancer randomized to placebo plus trastuzumab plus docetaxel (control group) or pertuzumab plus trastuzumab plus docetaxel (pertuzumab group) as first-line treatment until the time of disease progression or the development of toxic effects that could not be effectively managed. The primary end point was independently assessed PFS. The median PFS was 12.4 months in the control group, as compared with 18.5 months in the pertuzumab group (hazard ratio for progression or death, 0.62; 95% CI: 0.51–0.75; p < 0.001). The safety profile was generally similar in the two groups, with no increase in left ventricular systolic dysfunction [81]. Final long-term OS results after a median follow up of 50 months show an unprecedented prolongation in survival of 15.7 months with pertuzumab combination as compared with the control group [82] (median OS – 56.5 vs 40.8 months; HR: 0.68; p = 0.0002).
The development of antibody–drug conjugates is yet another novel approach in targeting HER-2-overexpression. Ado-trastuzumab-DM1 (T-DM1) combines the HER-2-targeted antitumor properties of trastuzumab with the antimicrotubule agent DM1 (derived from maytansine) allowing preferential intracellular drug delivery to the HER-2-positive tumor cells. Unlike lapatinib; trastuzumab, pertuzumab and T-DM1 can all induce antibody-dependent cellular cytotoxicity.
In the Phase III EMILIA trial, 991 patients with HER-2-positive metastatic breast cancer who had previously been treated with trastuzumab and a taxane, were randomly assigned to T-DM1 or lapatinib/capecitabine combination [83]. The T-DM1 group showed significant improvement in the median PFS (9.6 vs 6.4 months; 95% CI: 0.55–0.77; p < 0.001), median OS (30.9 months vs 25.1 months, HR for death from any cause: 0.68; 95% CI: 0.55–0.85; p < 0.001) and ORR (43.6 vs 30.8%; p < 0.001) with a lower incidence of grade 3 toxicity (57 vs 41%). Based on the results of the EMILIA trial, the FDA approved T-DM1 as a second-line agent in HER-2-positive metastatic breast cancer.
Other small molecule TKI's
Afatinib is an orally active irreversible dual inhibitor of EGFR and HER-2 receptors. In a Phase II study, afatinib monotherapy in heavily pretreated HER-2-positive metastatic breast cancer demonstrated partial response (PR) in four patients (10% of 41) and stable disease in 11 patients (37% of 41) [84].
Neratinib is an orally active irreversible inhibitor of EGFR, HER-2 and HER-4 receptors. In Phase II open-label clinical trial, 240 mg of oral neratinib was administered to trastuzumab pretreated (n = 66) and a trastuzumab naive cohort (n = 70). The ORR was 24 and 56%, respectively, and the most common grade 3 toxicity was diarrhea [85].
Conclusion
Numerous targeted therapies have evolved in the past 5 years that show promising activity in HER-2-positive breast cancer. Lapatinib is one such orally available drug with a favorable safety profile showing sustained activity in trastuzumab resistant tumors. Lapatinib is currently FDA approved in combination with capecitabine for metastatic HER-2-positive breast cancer that has progressed on prior trastuzumab therapy, and in combination with letrozole for postmenopausal women with HER-2 and hormone receptor-positive advanced breast cancer. In addition lapatinib shows promising activity in HER-2-positive disease with CNS metastases.
Future perspective
There has been a recent plethora of promising new agents for the treatment of HER-2-positive breast cancer. Pertuzumab, another humanized monoclonal antibody and TD-M1, an antibody-drug conjugate have been approved by the FDA for the treatment of metastatic disease and are in ongoing Phase III clinical trials in the adjuvant setting. Afatinib and neratinib are just two of several other small molecule tyrosine kinase inhibitors in development.
In the future, the challenge will be to optimize and individualize sequence and combinations of these agents as we seek safe and effective treatment for HER-2-positive disease – in some instances without conventional chemotherapy.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Executive summary
HER-2 amplification accounts for 20–25% of all newly diagnosed breast cancer cases.
Trastuzumab, a monoclonal antibody against the extracellular domain of HER-2 has changed the natural history of this disease. However, it does have some limitations including drug resistance and cardio-toxicity.
Lapatinib is an orally bioavailable, reversible dual small molecule tyrosine kinase inhibitor active against ERBB1 and ERBB2 by acting at the ATP binding site of the intracellular domain, thus inactivating phosphorylation of PI3K-AKT and RAS-RAF mechanisms leading to apoptosis and cell death.
Lapatinib is currently US FDA approved in combination with capecitabine for metastatic HER-2-positive breast cancer that has progressed on prior trastuzumab therapy, and in combination with letrozole for postmenopausal women with HER-2- and hormone receptor-positive advanced breast cancer.
Lapatinib is overall well tolerated in combination therapy for metastatic disease with minimal cardiac toxicity and diarrhea and skin rash being most common adverse events reported in major clinical trials.
Pertuzumab, another humanized monoclonal antibody and TD-M1, an antibody-drug conjugate have been approved by the FDA for the treatment of HER-2-positive metastatic disease and are in ongoing Phase III clinical trials in the adjuvant setting. Afatinib and neratinib are just two of several other small molecule tyrosine kinase inhibitors in development.
In the future, the challenge will be to optimize and individualize sequence and combinations of these agents for HER-2-positive disease – in some instances without conventional chemotherapy.
