Abstract
There is a relative paucity of evidence to guide the adjuvant therapy of breast cancer in older adults. Herein, we review data from relevant clinical trials and retrospective analyses that assess systemic therapies in older adults. Chemotherapy, endocrine therapy, HER2-directed agents and bisphosponates in the adjuvant setting are discussed. Meta-analyses, such as the Oxford overview, suggest a decreasing benefit of chemotherapy with age. However, prospective trials do support a benefit from the addition of chemotherapy to endocrine therapy in older adults. In contrast to chemotherapy, the value of endocrine therapy appears to increase with age. In addition, available subset analyses suggest that the benefit from aromatase inhibitors relative to tamoxifen is independent of age. The use of HER2-directed therapy in older adults is challenged by the relatively low enrolment of this subset in prospective clinical trials. Nonetheless, the extent of enrolment may be proportional to the frequency of HER2 overexpression in this population. Finally, there are little data to support the use of adjuvant bisphosphonate therapy in older adults – ongoing prospective trials may address this issue. Each of the aforementioned therapeutic strategies will be augmented by efforts to personalize therapy for older adults. Clinical tools such as the geriatric assessment and biologic assays, such as the 21-gene recurrence score, may ultimately play a role in treatment algorithms for this unique demographic.
Keywords
Among an estimated 184,450 US patients diagnosed with breast cancer in 2008, the median age at the time of diagnosis was 61 years [1]. Age represents the most important risk factor for the development of breast cancer [101]; however, paradoxically, older adults (aged 65 years and older) are poorly represented in clinical trials [2]. In addition, a relatively paucity of data are available to specifically guide the treatment of older adults with variations in medical comorbidity and generalized health status. As a consequence of these and other factors, recent studies have suggested a widening gap in survivorship between older- and middle-aged adults with breast cancer [3]. Such data should provide impetus for further study of breast cancer therapy in an aging population. To date, several studies have addressed the role of surgery and radiation in the adjuvant treatment of breast cancer in older patients [4–7]. Herein, we discuss systemic adjuvant therapies in older adults, providing a framework for future studies in this unique demographic. Although a universal definition of ‘older adult’ is lacking, an age-cutoff of greater than 65–70 years is generally applied and was used to select references presented in this review.
Chemotherapy
Retrospective datasets
Data from the Early Breast Cancer Trialist' Collaborative Group (EBCTCG) has informed the design of numerous contemporary guidelines for nonmetastatic breast cancer management. The most recent update of these data suggests that there is an age-related decrease in the benefit of chemotherapy with respect to both disease-free survival (DFS) and overall survival (OS) (

Ratio of benefit from polychemotherapy versus not from the Early Breast Cancer Trialist' Collaborative Group overview analysis.
Survival, epidemiology & end result database analyses
Two survival, epidemiology and end result (SEER) database analyses provide further insight into the potential benefit of chemotherapy in older adults with breast cancer [10,11]. In the first study, women aged 66 years or older with hormone-receptor negative, nonmetastatic disease were identified. Associated SEER-Medicare claims were utilized to ascertain chemotherapy use or nonuse [10]. Of 5081 patients assessed, 1711 (34%) received adjuvant chemotherapy. Adjuvant chemotherapy was associated with a 15% reduction in mortality, with the greatest benefit observed in lymph-node positive patients and those lymph-node negative patients most likely to receive chemotherapy. The second SEER registry analysis assessed patients aged 65 years or older with stage I through III breast cancer diagnosed between 1991 and 1999 [11]. Inclusion in this study was not limited by hormone-receptor status, and, presumably for this reason, a smaller proportion of patients received chemotherapy (10.9%). The study suggested that women with lymph-node positive, estrogen receptor (ER)-negative breast cancer derived a survival benefit from chemotherapy, similar to the previous study. Interestingly, patients with ER-positive breast cancer did not benefit similarly from treatment.
Prospective studies
The aforementioned retrospective data provide intriguing signals for a benefit from adjuvant chemotherapy in certain subsets of older adults. However, prospective analyses are necessary for a more meaningful validation of these findings. To date, only two randomized, Phase III trials have been reported in populations of older adults [12,13]. The French Adjuvant Study Group (FASG)-08 trial addresses the benefit of chemotherapy in conjunction with endocrine therapy. In this trial, 338 patients aged 65 years or older with node-positive, operable breast cancer were randomly assigned to receive either tamoxifen alone (at 30 mg oral daily for 3 years) or tamoxifen in combination with six cycles of epirubicin (30 mg on days 1, 8 and 15 of a 28-day cycle). With a median follow-up of 72 months, an increased relative risk (RR) of relapse was observed in patients receiving endocrine therapy alone as compared with combination therapy (RR: 1.93; 95% CI: 1.70–2.17) (
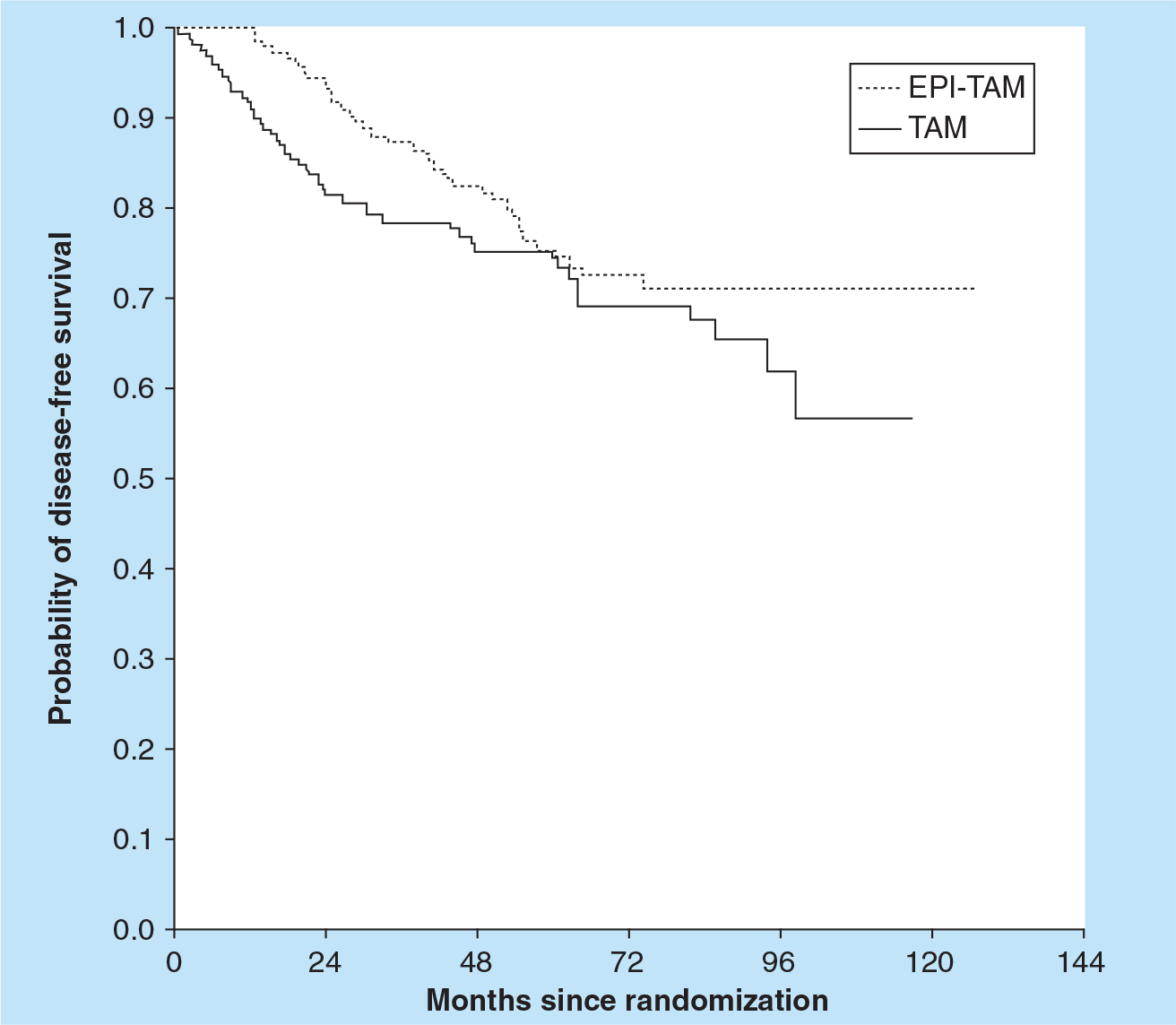
Kaplan–Meier curve representing disease-free survival from the French Adjuvant Study Group 08.
A second prospective trial in the elderly addresses the issue of polychemotherapy versus single agent chemotherapy. In CALGB 49907, patients aged 65 years or older with nonmetastatic breast cancer were randomized to standard multi-agent regimes of adriamycin and cyclophosphamide (AC) or cyclophosphamide, methotrexate and 5-fluorouracil (CMF), or single agent therapy with capecitabine [13]. The dose of capecitabine (2000 mg/m2 oral daily) was supported by a separate Phase II trial in older adults, in which a higher dose (2500 mg/m2 oral daily) produced excessive toxicity [14]. At a planned interim analysis, accrual was terminated at 633 patients, and data was recently presented with a median follow-up of 2 years. Compliance with oral therapy was good, exceeding that with infusional therapy with CMF (notably, compliance was highest with four cycles of AC chemotherapy, suggesting a relationship with duration of therapy). Efficacy data supported standard multi-agent chemotherapy, with patients on single agent capecitabine being 2.4-times (95% CI: 1.5–3.8) more likely to experience a relapse-free survival event and 2.1-times (95% CI: 1.2–3.7) more likely to die. With respect to toxicity, myelosuppression was more common with multiagent chemotherapy, whereas hand–foot syndrome was more common with capecitabine. In addition, two treatment-related deaths occurred with capecitabine therapy. Thus, the prospective experience to date supports the role of adjuvant chemotherapy in prolonging survival with standard infusional regimes as opposed to oral capecitabine for adjuvant therapy. Data from the ongoing ELDA study (comparing adjuvant weekly docetaxel to CMF) are eagerly awaited to determine if the type of infusional adjuvant therapy can be optimized [102]. Exploration of an adjuvant taxane regimen may be particularly relevant, given data from the recent US Oncology 9735 trial demonstrating an equivalent benefit from four cycles docetaxel and cyclophosphamide (TC) as compared with AC – recently presented subset analyses from this trial suggest a similar benefit from TC in older adults [15].
Toxicity considerations with chemotherapy
Several retrospective analyses contribute further insight into toxicity associated with chemotherapy in older adults. A meta-analyses of three CALGB trials (CALGB 8541: cyclophosphamide, doxorubicin and fluorouracil [CAF] in three dosing schedules; CALGB 9344: AC with or without paclitaxel; and CALGB 9741: CAF with paclitaxel every 2 vs 3 weeks) provides toxicity data for 6641 patients within age-based subsets [16]. As with the previously reported CALGB meta-analysis, older adults were poorly represented in the collective experience, with only 3% of patients aged 70 years or older. Across increasing age groups, there was a concomitant increase in the incidence of treatment-related deaths, ranging from 0.19% in patients under 50 years to 1.5% in patients over the age of 65 years. In addition, multivariate analysis revealed that older patients were more likely to have grade 4 hematologic toxicity or die of acute myeloid leukemia (AML) or myelodysplastic syndrome (MDS). The observation of an increased incidence of secondary hematologic malignancies is supported by a separate SEER-Medicare registry analysis [17].
A review of the International Breast Cancer Study Group (IBCSG) experience (similar to the CALGB experience) suggests an increase in treatment-related deaths with increasing age [18]. In a meta-analysis of IBCSG trials I through IX (all utilizing CMF-based chemotherapy), the incidence of treatment-related mortality was 1.28% in patients over 65 years, as compared 0.08% amongst patients 50 years or less. In this study, the majority of deaths attributable to chemotherapy in older adults were secondary to sepsis or other toxic effects of therapy (unspecified). Notably, recent American Society of Clinical Oncology guidelines do suggest the use of prophylaxis for febrile neutropenia with white blood cell growth factors in patients at high risk, including those of advanced age [19].
Oncologists are often challenged by the decision to administer anthracycline chemotherapy in the older adult (particularly in those with comorbid cardiac disease) given the potential for anthracycline-induced cardiotoxicity [20]. A SEER-Medicare database review assessed 31,748 patients with stage I to III breast cancer diagnosed from 1992 to 1999 [21]. A total of 5575 patients (18%) had received adjuvant chemotherapy. Receipt of doxorubicin-based chemotherapy led to an increased rate of cardiomyopathy, CHF and heart disease. Notably, a separate SEER-Medicare database review including older adults with more advanced disease suggested that the use of doxorubicin led to an increase in cardiac toxicity across all strata of age [22].
An emerging body of evidence suggests an association between chemotherapy use and cognitive decline, a toxicity of particular concern in older adults. A series of 50 patients (aged 65 years or greater with stage I to III breast cancer treated with chemotherapy) were prospectively assessed at the Memorial Sloan-Kettering Cancer Center (MSKCC) [23]. In comparison with their perceived baseline memory, approximately 51% of study participants perceived a decline in their perceived memory 6 months after completion of chemotherapy. The most commonly cited defect in memory was the ability to learn new information. Cognition may be a predictor of survival amongst patients with breast cancer. Supporting this, a recent SEER-Medicare database analysis including 106,061 patients with breast, colon or prostate cancer suggested that survival was signifcantly worse in demented patients as compared with nondemented patients [24].
Endocrine therapy
Studies of tamoxifen in older adults
Available studies generally support the use of adjuvant endocrine therapy in older adults. The EBCTCG meta-analysis suggests that with increasing age, there is an increase in the benefit derived from tamoxifen therapy (specifically, a decreased annual event rate) (

Ratio of benefit from tamoxifen versus not from the Early Breast Cancer Trialist' Collaborative Group overview analysis.
Prospective validation of these findings is offered from a series of patients aged 65 years or greater with stage I to IIIA breast cancer (diagnosed from 1997 to 1999 in four separate geographic regions) [25]. Data from a total of 689 women was collected, 519 (76%) of whom received tamoxifen. At a median follow-up of 67 months, the 5-year OS was 81 and 70% with ever tamoxifen and no tamoxifen, respectively (HR: 0.53; 95% CI: 0.37–0.77). Notably, there were substantial differences in hormone receptor status in this study, with ER- or progesterone receptor (PR)-positive status in 84% of patients receiving tamoxifen as compared with 56% of patients not receiving tamoxifen (p < 0.0001). This difference was not accounted for in the multivariate analysis of survival – nonetheless, these data seem to largely support the benefit of tamoxifen therapy in older adults observed in the EBCTCG overview.
Once therapy with tamoxifen is initiated, maintenance of treatment emerges as a substantial issue in older adults. In a prospective analysis of 961 women (≥65 years) who initiated therapy for stage I to IIB breast cancer between 1990 and 1994, approximately 49% of patients discontinued use of tamoxifen prior to 5 years of therapy [26]. Patients who were over the age of 75 years had a substantially higher rate of discontinuation as compared with those patients between 65 and 75 years. Furthermore, patients with an increased incidence of medical comorbidity (as estimated by the Charlson Co-morbidity Index) had an increased risk of discontinuation. A second study provides further insight into the timing of and rationale for tamoxifen discontinuation. In this report, 516 women over the age of 65 years who were prescribed tamoxifen therapy for ER-positive, stage I to IIIA breast cancer were followed prospectively for 2 years [27]. Approximately 17% of patients discontinued tamoxifen during this 2-year period, with 68% of those patients discontinuing use within 1 year. Interestingly, women with neutral or negative beliefs about the value of tamoxifen had a much higher likelihood of discontinuing tamoxifen use. As a result of these findings, it is suggested that the degree of patient counseling and the attitude of the practitioner towards tamoxifen therapy presumably play a large role in the patient's decision to maintain therapy with tamoxifen. Questionnaire studies have, in fact, demonstrated that the perspective of the treating physician (as it pertains to tamoxifen therapy) is a strong predictor of tamoxifen utilization [28].
Tamoxifen versus aromatase inhibitors
Large datasets comparing various schedules of tamoxifen and aromatase inhibitors have emerged in recent years. The Arimidex, Tamoxifen, Alone or in Combination (ATAC) trial represents the longest experience to date, with data reported at a median follow-up of 100 months [29]. The trial compares 5 years of anastrazole therapy with 5 years of tamoxifen therapy (data from a combination therapy arm was omitted after an initial analysis owing to a lack of efficacy or tolerability over tamoxifen alone). Overall, with 46,202 women-years follow-up, the study demonstrated an overall benefit in DFS with anastrazole therapy (HR: 0.85; 95% CI: 0.76–0.94), with no effect on OS (HR: 0.97; p = 0.70). Although data in older adults was not separately reported, subjects aged 65 years or greater seemed to derive a DFS benefit nearly identical to those patients under 65 years (HR: 0.77 and 0.76, respectively). The Breast International Group (BIG) 1–98 trial provides a similar comparison, with 5 years of tamoxifen therapy compared with 5 years of letrozole (comparison to two unique treatment arms employing endocrine switching has been reported separately) [30]. In 4922 patients (at a median follow-up of 71 months), a nonsignificant increase in OS was observed with letrozole alone as compared with tamoxifen alone (HR: 0.87; 95% CI: 0.75–1.02). At the most recent update, the trial demonstrated no substantial differences in survival end points with endocrine switching. A separately reported subset analysis specifically assessed 1795 patients over the age of 65 years [31]. In comparison with younger cohorts, older patients were less likely to complete treatment, but nonetheless had significantly improved DFS as compared with tamoxifen.
Recent efforts have looked at extended durations of endocrine therapy (i.e., beyond 5 years of tamoxifen). A report from the National Surgical Adjuvant Breast and Bowel Project (NSABP) B-14 trial suggested that 5 years of tamoxifen therapy may be superior to longer durations of endocrine therapy [32]. However, preliminary recent results from the larger, multinational ‘Adjuvant Tamoxifen – To Offer More?’ (aTToM) and ‘Adjuvant Tamoxifen, Longer Against Shorter’ (ATLAS) suggest a benefit in DFS with greater than 5 years of tamoxifen therapy [33,34]. Age-related differences in benefit have not yet been elicited from these trials at this point. Beyond the strategy of extended tamoxifen therapy, the National Cancer Institute of Canada-Clinical Trials Group (NCIC-CTG) MA.17 trial assessed treatment with letrozole beyond 5 years of tamoxifen. As compared with placebo, a significant benefit in DFS and OS was noted with extended letrozole therapy in patients with ER- and PR-positive disease [35]. A separately reported analysis of patients aged 70 years or greater suggested the effect of letrozole was similar among age groups [36]. Furthermore, toxicity and quality of life were similar in patients aged 70 years or older receiving letrozole or placebo.
Toxicities of endocrine therapy
A long-term safety analysis from the ATAC trial provides insight into cumulative toxicities from these tamoxifen and aromatase inhibitors [37]. While aromatase inhibitors lead to decreases in bone mineral density (BMD), tamoxifen is thought to have a protective effect on the bone in postmenopausal women [38,39]. In the ATAC trial, more fractures were recorded in the group receiving anastrazole. However, hip fractures (associated with greater morbidity than all other fractures combined) were similar in both arms, and the incidence of fractures overall equalized beyond the 5 years of therapy. Nonetheless, concern for decreasing BMD and resultant complications with aromatase inhibitors have prompted independent guidelines for use of bisphosphonates with these agents [40].
In contrast to the aromatase inhibitors, tamoxifen leads to a substantial risk for vascular events, with a large meta-analysis suggesting a statistically significant increase in the risk of stroke (HR: 1.49; 95% CI: 1.16–1.90) and endometrial cancer (HR: 2.70; 95% CI: 1.94–3.75) [41]. Such considerations may weigh heavily in the decision to administer aromatase inhibitors in the elderly patient, given the oft presence of multiple comorbidities.
A separate and unique consideration in older adults is that of polypharmacy. In a survey of 3005 community-residing individuals between the ages of 57 and 85 years, approximately 4% of individuals were potentially at risk of having a major drug–drug interaction [42]. Tamoxifen is metabolized to the potent antiestrogen endoxifen by the enzyme cytochrome P450 2D6 (CYP2D6), and polymorphisms in the encoding gene may result in variations in tamoxifen metabolism [43]. Beyond its role in tamoxifen conversion, CYP2D6 is responsible for the metabolism of multiple other agents, including several commonly used antihypertensives and antidepressants [44,45]. The use of these agents concurrently with tamoxifen may result in altered serum levels of endoxifen, as demonstrated prospectively with the combination of paroxetine and tamoxifen [46]. Therefore, practitioners must exercise caution in administering tamoxifen to older adults who are subject to polypharmacy.
HER2-directed therapy
Approximately 14% of invasive breast cancer in patients over the age of 75 years is HER2-overexpressing, in contrast to an estimated 25–30% in the overall breast cancer population [47,48]. Several multinational trials have substantiated the benefit of the HER2-directed monoclonal antibody, trastuzumab, for use as an adjuvant therapy in HER2-overexpressing breast cancer [49–51]. These trials include a relatively small number of older adults, albeit somewhat in proportion to the number of older adults who are HER2-overexpressing. A combined analysis of the National Surgical Adjuvant Breast and Bowel Project (NSABP) B-31 and North Central Cancer Treatment Group (NCCTG) trials suggested that the addition of trastuzumab to standard chemotherapy resulted in a 50% reduction in the risk of recurrence and a 33% reduction in the risk of death at 2-year median follow-up [50]. A total of 3351 patients were included in this combined analysis, of whom 535 patients (16%) were aged 60 years or older. The Herceptin Adjuvant (HERA) trial employed a somewhat similar randomization, and included an equal percentage of patients over the age of 60 years (16%) [50]. Results from this study were largely similar to those of the NCCTG/NSABP B-31 combined analysis.
Of particular concern with the use of trastuzumab is the associated risk of cardiac disease, particularly in combination with anthracycline chemotherapy. A multivariate analysis of data from the NSABP B-31 trial suggested that age was a significant predictor of CHF with trastuzumab and chemotherapy [52]. Furthermore, increasing age was associated with an increased risk of cardiac events in the NCCTG N9831 study [53]. A potential strategy to minimize the risk of cardiac toxicity is the use of nonanthracycline chemotherapy with trastuzumab. The Breast Cancer International Research Group (BCIRG 006) trial compares one such regime (a combination of docetaxel, carboplatin and trastuzumab, or TCH) to standard anthracycline-based regimes [51]. At an interim analysis, similar efficacy was noted amongst the treatment arms. In addition, lower rates of cardiotoxicity were observed with TCH. Limited data with respect to age-related differences in efficacy or toxicity of this regime are available at this time; these findings are eagerly anticipated. A separate way in which to reduce cardiac toxicity could be shortening the duration of trastuzumab therapy. A strategy of 9 weeks of trastuzumab therapy was shown to result in a statistically significant benefit in recurrence-free survival (HR: 0.42; 95% CI: 0.21–0.83) with no associated decrease in left ventricular ejection fraction or cardiac failure [54]. A study of 6 versus 12 months of trastuzumab in combination with taxane-based chemotherapy is ongoing [102].
Bisphosphonates
The relationship between BMD and breast cancer incidence remains elusive. One prospective analysis of 1504 women aged 75 years or greater compared BMD at three separate sites, and correlated these findings with breast cancer incidence with 7 years of clinical follow-up [55]. Interestingly, patients in the highest tertile of BMD had a two- to three-fold increased risk of breast cancer as compared with those women in the lowest tertile. A combined
Given these data, the use of bisphosphonates as adjuvant breast cancer therapy in older adults remains the subject of debate. Recently, data has emerged to support an antitumor effect of zoledronic acid in younger, premenopausal patients when combined with adjuvant endocrine therapy and ovarian suppression [58]. Several anticipated datasets may yield further information regarding the benefit of bisphosphonate therapy in an enlarged demographic. The Ibandronate With or Without Capecitabine in Elderly Patients With Early Breast Cancer (ICE) trial represents one prospective evaluation of bisphosphonates in older adults (

Schema for the Ongoing Ibandronate With or Without Capecitabine in Elderly Patients with Early Breast Cancer trial.
Determining the benefits & risks of adjuvant therapy
In comparison with younger women, older women are more frequently diagnosed with invasive breast cancers that are of, hormone receptor positive, HER2-negative, lymph node negative and are of low-grade histology [47]. The determination of possible benefit from adding chemotherapy to endocrine therapy in this population is aided by the use of biologic assays such as the 21-gene recurrence score (OncotypeDx; Genomic Health, San Francisco, CA, USA). This assay incorporates a reverse transcription-PCR-based quantitation of genes that encode for hormone receptors, HER2 and other proliferative factors. A mathematical algorithm summarizes this data in a numerical ‘recurrence score’. This test was validated on paraffin-embedded specimens obtained from NSABP B-14, an adjuvant trial comparing tamoxifen alone or in combination with chemotherapy [59]. In this retrospective analysis, patients with a recurrence score less than 18 did not benefit from the addition of chemotherapy. By contrast, patients with scores greater than or equal to 31 had an improved OS when chemotherapy was added to tamoxifen. As yet undetermined is the benefit of adding chemotherapy when the recurrence score is between 19 and 31. This assay is useful in guiding treatment recommendations for node negative, hormone receptor positive, HER2-negative breast cancers and has been incorporated into breast cancer treatment guidelines. The 21-gene recurrence score has also been shown to predict outcomes in patients with node positive disease; however, further validation in this subset of patients is necessary [60].
In older women, comorbidities may be more life limiting than early-stage breast cancer and the benefits of chemotherapy (even in women with a high recurrence score or positive lymph nodes) may be outweighed by associated toxicities. Computer-based programs such as Adjuvant! On-Line provide the patient and practitioner with a reasonable estimate of therapeutic benefit with various treatment modalities, derived from the patient's age, tumor size, estrogen receptor status and comorbidities [103]. Recent data indicate that Adjuvant! On-Line is highly sensitive to inputs related to patient comorbidity (to an even greater extent than biologic characteristics of the tumor) [61]. Although the program is subject to a great deal of interpreter variability via difficulties in characterization of comorbid conditions, it is of considerable benefit in helping the patient consider treatment options.
Key elements of the geriatric assessment
Functional status
– Activities of daily living
– Instrumental activities of daily living
Comorbidity
Nutritional status
Cognitive function
Psychosocial function
Polypharmacy
Data taken from [68].
Conclusion
Despite tremendous progress in the evolution of adjuvant therapy for breast cancer patients at large, limited evidence exists to specifically define appropriate treatment of the older adult. To date, only two prospective, randomized trials in older adults have been reported [12,13], and virtually all data pertaining to endocrine and HER2-directed therapy in this group are derived from subset analyses [29,31,49,50,62]. Furthermore, variations in health status and increasing comorbidity with age complicate therapeutic decision-making in older adults. In order to aid practitioners in the clinical management of this unique demographic, several guidelines have been established. The Senior Adult Oncology Guidelines set forth by the National Comprehensive Cancer Network (NCCN) provide a detailed framework in which to assess the overall health status of the patient, incorporating patient-specific data related to life expectancy and functional status [104]. Separately published Breast Cancer Treatment Guidelines (also from the NCCN), suggest that the use of adjuvant chemotherapy in patients aged 60 years or older should be individualized, with consideration of the potential incremental benefit of chemotherapy with increasing age (as observed in the EBCTCG overview analysis).
Complementing the NCCN guidelines, the International Society of Geriatric Oncology (SIOG) has generated a consensus statement pertaining to older adults with breast cancer [63]. These recommendations were derived from a task force of experts challenged to assess all published data and international meeting abstracts related to this domain. The data presented encompasses not only systemic adjuvant therapy, but data related to radiation therapy, surgery and treatment of metastatic disease. This concise manuscript provides the practitioner with a useful context in which to recommend and discuss treatment options with the older adult.
While guidelines for management of breast cancer in the older adult remain in their infancy, adherence to generalized guidelines appears to be rather poor. A review of 1568 patients (≥55 years) treated at the University of Texas MD Anderson Cancer Center, USA between 1997 and 2002 for stage I to IIIa breast cancer suggested that older women were less likely to receive adjuvant chemotherapy and endocrine therapy in concordance with accepted guidelines (p < 0.001 for both) [64]. Emerging data suggests that the adherence to guideline-based therapy may reduce a widening gap in breast cancer mortality with increasing age [65].
A separate strategy for improving outcomes in older adults with breast cancer might include the broader incorporation of novel patient assessments. Tools such as the self-administered cancer-specific geriatric assessment might provide more personalized data, incorporating co-morbidities and overall health status, and may therefore offer a more accurate assessment of life expectancy and resultant adjuvant therapy benefit [66]. Attempts to utilize the geriatric assessment (Box 1) in clinical practice thus far appear to be challenged by a continued reliance on standard clinicopathologic variables [67]; more recent efforts to incorporate the geriatric assessment into prospective clinical trials may yield valuable results. It is foreseeable that these useful clinical tools will be integrated with relevant biologic and computational assessments (i.e., the 21-gene recurrence score and Adjuvant! On-Line, respectively) in order to more precisely characterize the older adult.
Future perspective
The studies presented herein underscore the paucity of clinical data available to define the role of systemic adjuvant therapy in the older adult. Prospective analyses limited to older adults (such as CALGB 49907 and FASG 08) represent truly unique efforts, and provide perhaps the most relevant insights into management of this demographic. Admittedly, such trials are challenging to conduct. As an alternative, upcoming clinical trials of systemic adjuvant therapy may instead focus on proportional accrual of older adults with sufficient numbers to draw meaningful conclusions within this subset. Furthermore, incorporation of novel assessment tools in clinical trial design will allow for prospective validation of these modalities. These tools include both clinical examinations (such as the geriatric assessment) and biologic assays (such as the 21-gene recurrence score). Finally, the use of therapeutic agents with enhanced tolerability and decreased toxicity (i.e., liposomal anthracyclines or albumin-bound paclitaxel) may present a useful alternative in this subgroup of patients – age-specific studies of these agents are ongoing [102].
Executive summary
Age represents the most important risk factor for the development of breast cancer – despite this, older adults are largely underrepresented in clinical trials.
Only two randomized, prospective studies related to systemic adjuvant therapy in older adults have been reported to date: the French Adjuvant Study Group (FASG) 08 trial; and Cancer and Cancer and Leukemia Group B (CALGB) 49907 trial.
FASG 08, comparing endocrine therapy alone to chemotherapy with endocrine therapy, suggested that the latter strategy may lead to an improvement in disease-free survival (DFS).
CALGB 49907, comparing standard multiagent infusional regimens to single-agent capecitabine, suggested that the latter regimen is inferior in older patients, with respect to both overall survival and DFS.
Prospective data suggest that older women with breast cancer derive a survival benefit from adjuvant tamoxifen therapy (as compared with no tamoxifen), although compliance with therapy remains a substantial issue.
In the comparison of tamoxifen versus aromatase inhibitors as adjuvant endocrine therapy, subset analyses from large, randomized trials indicate that older adults derive a similar relative benefit from aromatase inhibitors as compared with the population at large.
Studies of trastuzumab have enrolled a relatively small proportion of older adults, perhaps proportional to the degree of HER2-overexpression in this population. Age may be a predictor of cardiotoxicity related to the combination of trastuzumab and chemotherapy.
Insufficient evidence exists at this time to guide the use of bisphosphonates as adjuvant breast cancer therapy in older adults.
Novel tools to assess the older adult are needed to inform decisions regarding therapeutic assignment. Clinical tools, such as the geriatric assessment, allow for individualized characterization of health status and life expectancy. Biologic tools such as the 21-gene recurrence score predict outcome independently of age, and may thusly be of great use in this population.
Efforts must be made in clinical trial enrolment to include a more representative proportion of older adults. In addition, age-specific trials in older adults may more precisely address clinical dilemmas in this population.
