Abstract
Objective
To evaluate the effect of
Methods
Six-week–endurance-trained male Sprague-Dawley rats (n = 24) were randomly divided into 2 groups: control and experimental; the latter group was supplemented with
Results
There was a significant improvement in endurance exercise, as indicated by an increase in run to exhaustion following
Conclusion
Keywords
Introduction
Prolonged low-intensity exercise has been characterized by increased utilization of fatty acids, which eventually become the major energy source for muscles.
High-altitude exposure, both acute and chronic, is often characterized by hypophagia, body weight loss, and decrement in exercise capacity.
3
Exposure to high altitude elicits several physiological and metabolic adjustments, including an alteration in substrate metabolism.
4
While most studies have shown carbohydrates to be the preferred fuel in acute hypoxic conditions, some studies also indicate that fat may also be an important fuel at high altitude. Young et al
5
have reported higher resting free fatty acids after acute hypobaric/hypoxic exposure (2 hours at 4300 m) in lowlanders. Elevated levels of glycerol, a marker of lipolysis, have also been observed.
6
To aid in increased fat utilization, increased levels of carnitine could offer an advantage in hypoxic conditions.
Material and methods
Experimental Animals
Adult male albino Sprague-Dawley rats, 14 to 16 weeks of age (n = 24), were obtained from the animal house at the Defence Institute of Physiology and Allied Sciences (DIPAS). Rats were bred and maintained in a well-aerated room with a 12:12-hour light:dark cycle at the animal house facility of the DIPAS in Delhi, India. All experimental procedures were carried out in accordance with the guidelines of the DIPAS Ethics Committee.
Diet
Animal diet consisted of food pellets (Amrut Laboratory Animal Feeds, Pranav Agro Industries Ltd, Delhi, India) with 21% protein, 53% carbohydrate (wheat flour, roasted Bengal pulse, groundnut flour), 4% crude fiber, calcium, phosphorus, and refined oil enriched with stable vitamins A, D3, E, K, B1, B2, B5, B6, B12, and C. The energy density of the feed was 3.41 kcal·g−1. Food and water were provided ad libitum. The food pellets were provided in a metal cup (120-g capacity) fitted with an antiscatter rim to prevent spillage. The water was provided in 50-mL graduated glass bottles.
Experimental Design
The motorized rodent treadmill (Columbus Instruments, Columbus, OH, USA) consisted of a controller unit, motor assembly, 2 running lanes, and 2 grids to deliver electric shocks. The speed and incline of the running belt were increased stepwise during the course of training and were set to a final speed of 27 m·min−1 and a 15% incline. 9
The rats were trained/accustomed to treadmill running for 6 days, for 30 minutes each day, prior to the initiation of supplementation. This duration was found to be sufficient to accustom the animals to treadmill running. After this, “run to exhaustion” time was measured weekly for 5 weeks, at 27 m·min−1 speed and a 15% incline. During the exercise, when the rats would slip from the running lane onto the grid as a result of exhaustion, they would be propelled to run again on delivery of an electric shock, which consisted of 200-ms pulses delivered at the rate of 4 pulses·s−1 and an intensity of 163 V. Run to exhaustion time was noted as the time from the beginning of exercise until the rats got completely exhausted and remained stationary on the treadmill for longer than 10 seconds. 9
The protocol for the experiment is represented schematically in Figure 1. All the baseline recordings (ie, run to exhaustion time and biochemical measurements) were carried out 7 days prior to supplementation (R1) in all of the animals. The rats were randomly divided into 2 groups. In the experimental group,
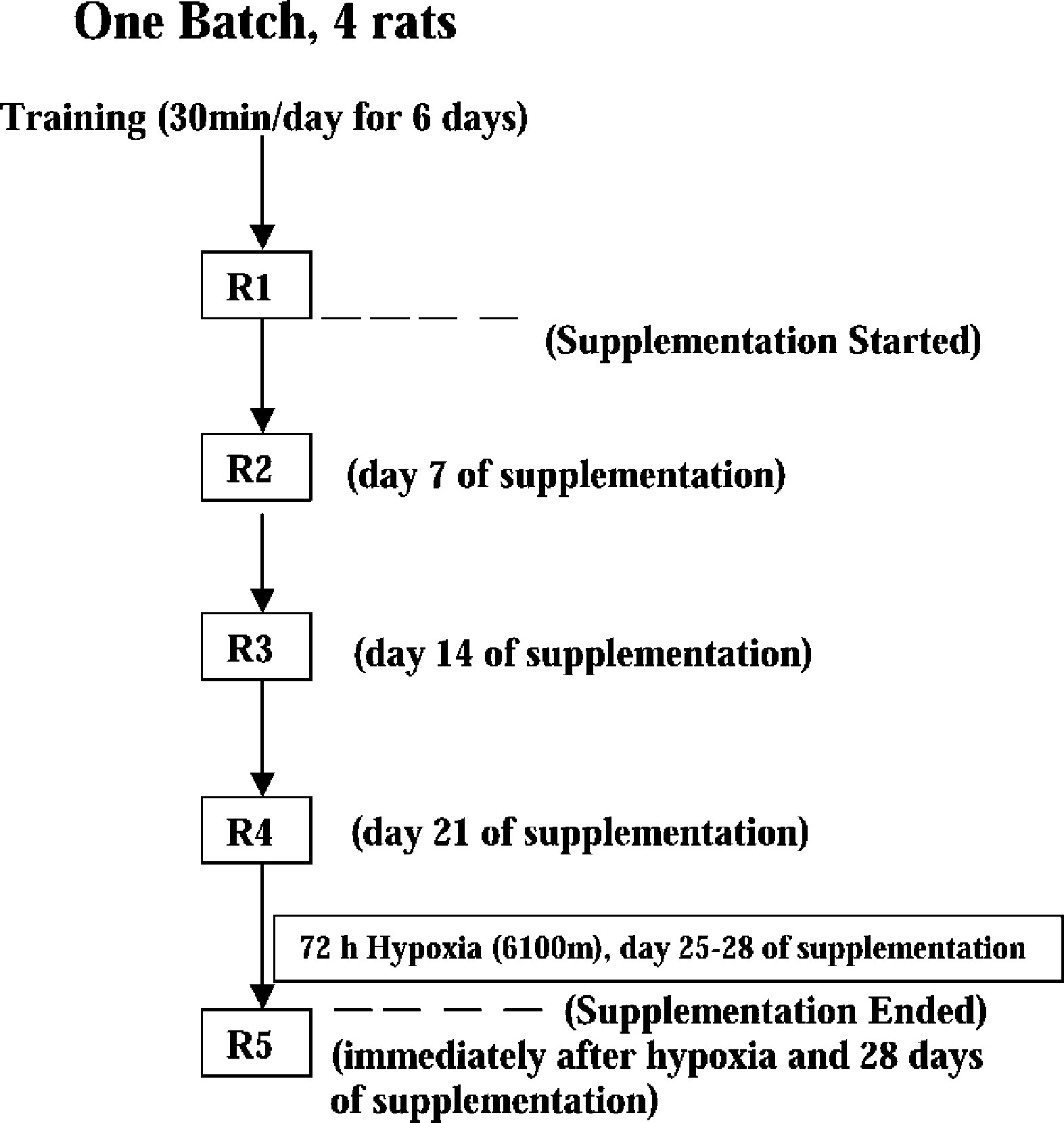
Schematic representation of the experimental protocol.
Hypobaric Hypoxic Exposure
The animals were exposed to hypobaric hypoxia equivalent to a 6100-m–high altitude using an animal decompression chamber (Seven Star Model 700, GDA, Delhi, India) at a temperature of 32 ± 0.5°C and a relative humidity of 50%, with fresh air flow at 5.5 L·min−1. Altitude was attained in 30 minutes with an ascent rate of 200 m·min−1. The level of decompression inside the chamber could be continuously monitored by a mercury manometer (349 mm Hg) and an altimeter, which directly gave the reading of the altitude above sea level. The animals were exposed continuously for 3 days at 23.5 h·d−1. They were brought to sea level between 0930 hours to 1000 hours, at which time their food and water were replenished, their food intake and body weight were recorded, and the supplement solutions were administered.
Recordings
The rats were tested for endurance exercise immediately postexposure (within 5 minutes). The food intake was recorded to the second decimal place; recording occurred daily at the same time in the morning. The food intakes were then subsequently expressed as kcal·100 g−1 body weight of the animals. The body weight was recorded every day on a weighing balance with a sensitivity of 0.1 g.
Blood Sampling and Biochemical Estimation
The animals were anesthetized using anesthetic ether. Postexercise blood samples were drawn from the ophthalmic orbital plexus. The drawing of blood samples coincided with the recording of run to exhaustion time. The samples were collected in heparinized tubes and subsequently centrifuged at 1000g for 10 minutes at 4°C. The aliquots of plasma were stored at −70°C to assay TC and HDL cholesterol. Glucose was assayed immediately after separation of plasma to assess the status of carbohydrate utilization under hypoxic conditions. Plasma glucose estimations were carried out using commercially available kits (Boehringer Mannheim GmBH Diagnostica, Penzberg, Germany) based on the glucose oxidase-peroxidase method. Total cholesterol and HDL cholesterol were estimated in the plasma by an enzymatic method using commercially available kits (Source Diagnostics, Shimla, India).
Statistical Analysis
Comparisons between control and experimental groups were made using repeated-measures analysis of variance (2 × 4 mixed model), and further multiple comparisons tests were completed using Student Newman Keul's multiple range tests. The preexposure (R4) and postexposure (R5) comparisons for the same experimental animals were analyzed using paired t tests. The level of significance was P < .05.
Results
Effects of l -Carnitine Treatment Under Normobaric/Normoxic Conditions
Run to exhaustion time was significantly increased by ∼36% in the experimental group on day 7 (R2) both in comparison to basal value as well as in comparison to the control group. However, there was no further improvement on days 14 (R3) and 21 (R4) of supplementation. Run to exhaustion time was also significantly increased at all time periods (R2–R4) in the experimental group and did not increase significantly in the control group. As shown in Figure 2, there was no significant alteration in food intake recorded before hypobaric hypoxic exposure in both groups. Comparison of body weight between the 2 groups showed a significant decrease in body weight in the experimental group as compared to the control group after days 12, 15, 18, and 21 of supplementation (Figure 3). Table 1 depicts plasma glucose, TC, and HDL cholesterol values.
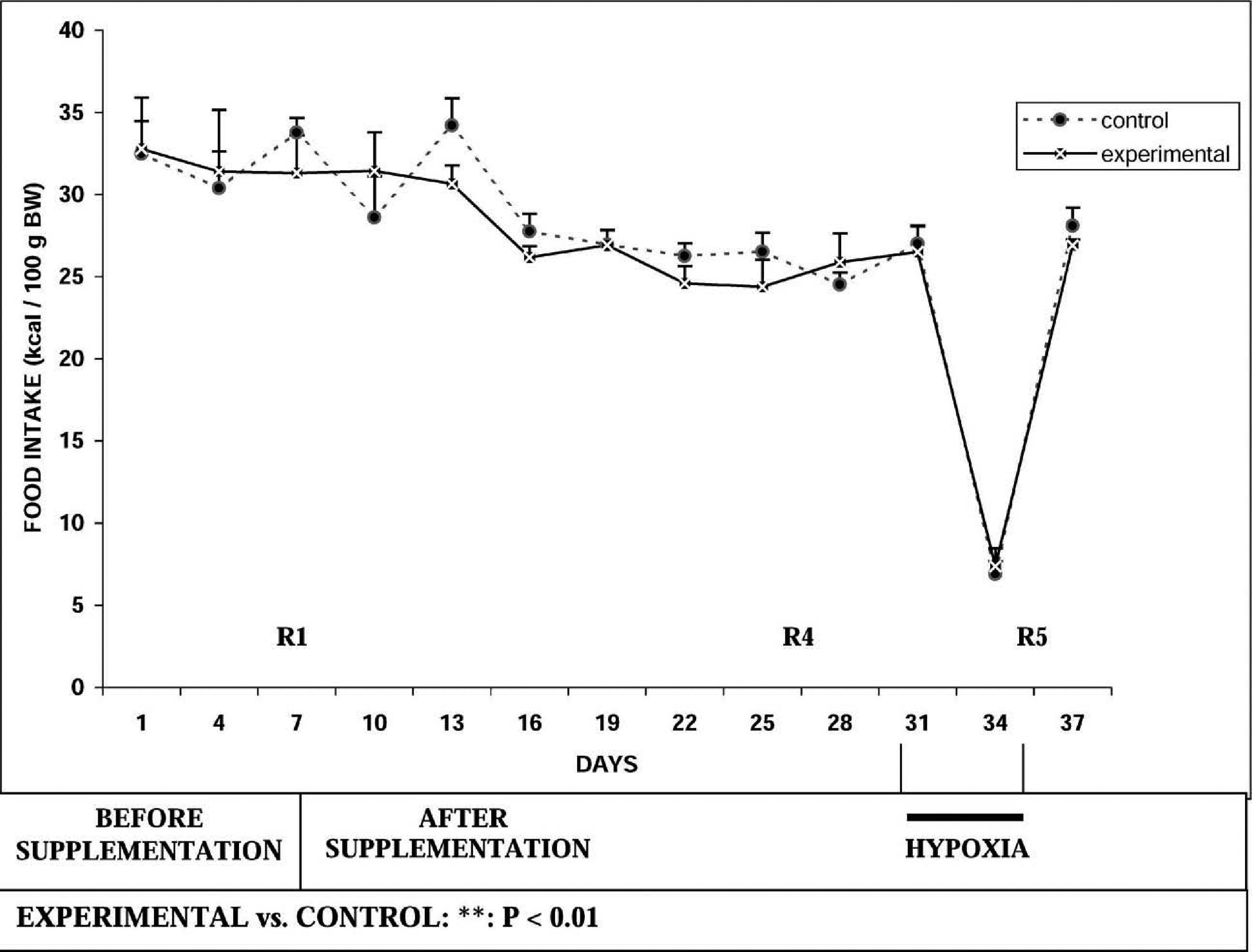
Effect of
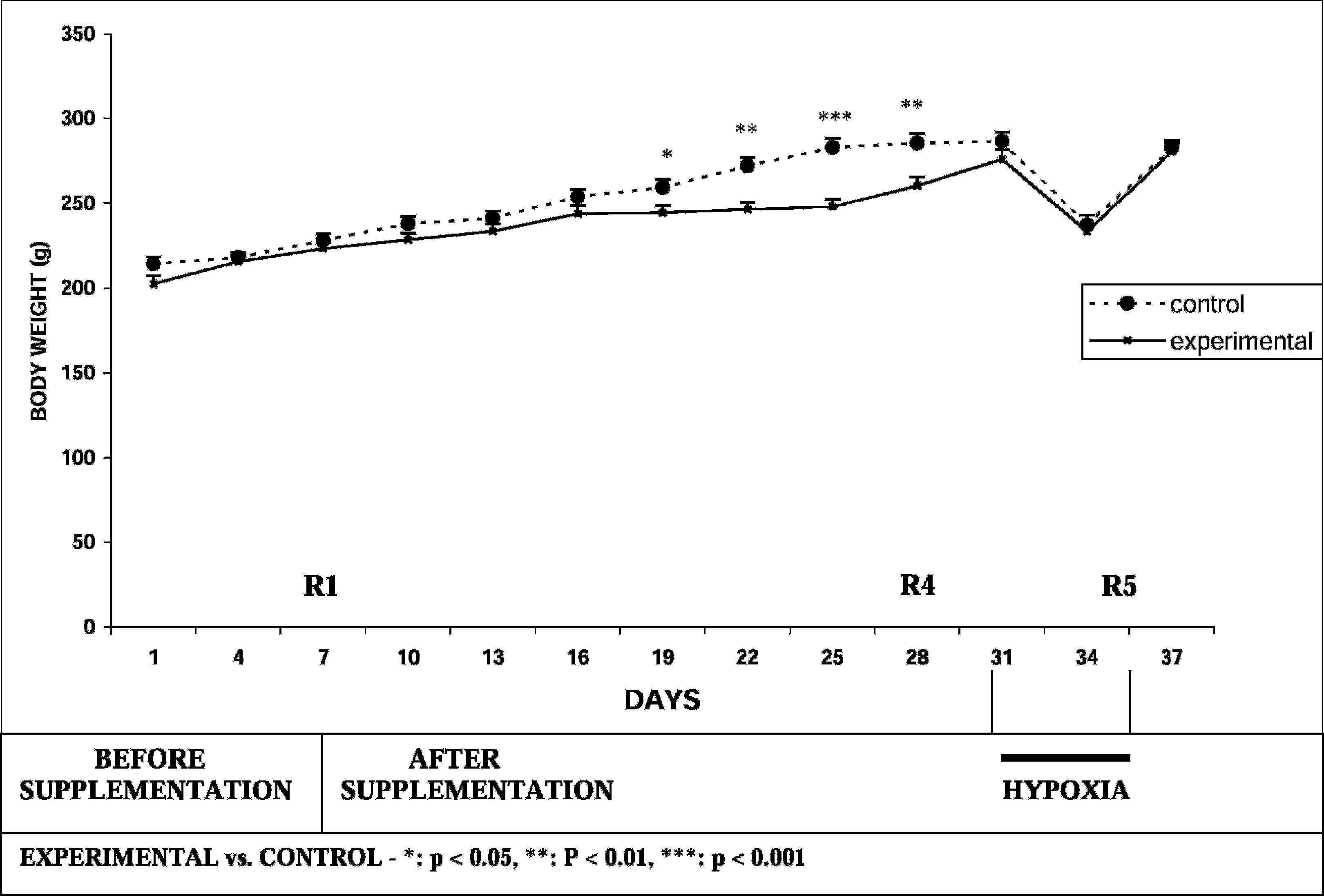
Effect of
Effect of
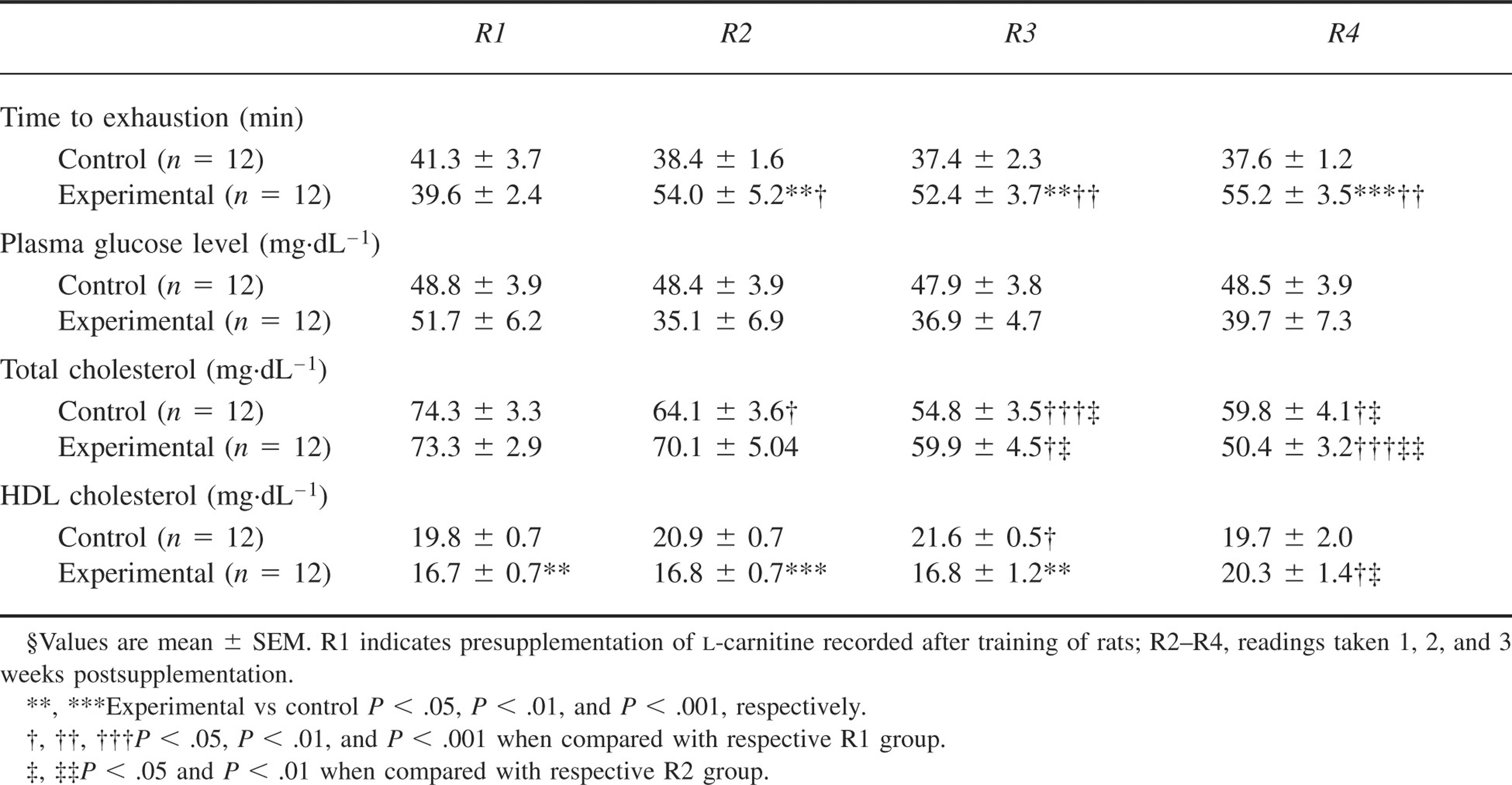
Comparison of plasma glucose between the experimental and control groups showed no significant differences. There was also no effect of supplementation of
Effect of l -Carnitine Supplementation Under Hypobaric Hypoxia
The run to exhaustion time was higher in the experimental group than in the control group following exposure to hypobaric hypoxia (P < .05). There was no significant intragroup change in time to exhaustion following hypobaric hypoxic exposure (R5) in the experimental group. There was an increase in the control group when compared to R4. There was a significant decrease of food intake in both the experimental and control groups (P < .001) upon exposure to hypobaric hypoxia (Figure 2). There was a reduction in body weight in both the experimental and control groups after hypoxic exposure (P < .001) (Figure 3). As depicted in Table 2, plasma glucose and TC levels did not change significantly after hypobaric hypoxic exposure in both groups. There was a significant increase in HDL cholesterol following hypobaric hypoxic exposure in both the control group (P < .05) and the experimental group (P < .01).
Effect of
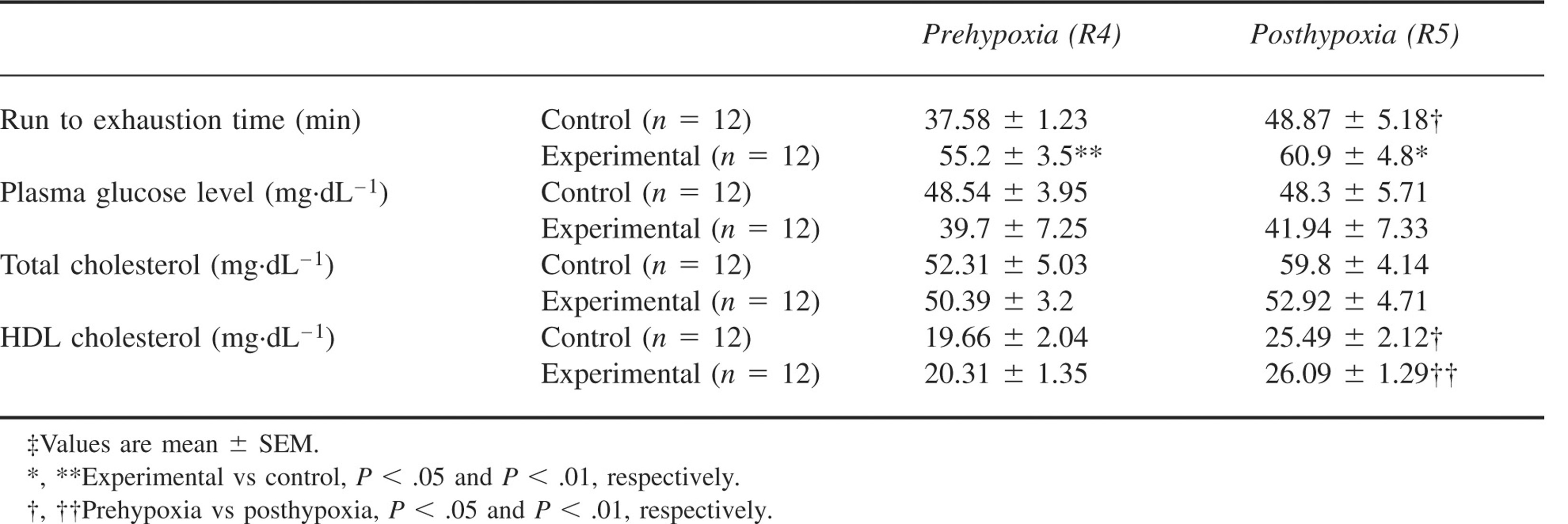
Discussion
The improved endurance exercise following
The effect of
In the present study, exercise training per se may have contributed to a lack of any detrimental effect of hypoxia on physical work capacity. It was reported
16
that training may attenuate myocardial beta-adrenoceptor downregulation; the latter contributes to limitation of V
In addition to endurance exercise, hypobaric hypoxia influences food intake and body weight.
19
In the present study, exposure to hypoxia reduced food intake and body weight. Supplementation with
Body weight loss is another manifestation of hypobaric hypoxia.18,19 In the present study, weight loss after 72 hours of hypobaric hypoxia was of a similar magnitude (ie, 19.8% and 17.3%) in the experimental and control groups, respectively. There was a decrease in body weight between day 19 (corresponding to 2 days before R3) and day 28 (R4) in the experimental group compared to the control group; it is possible that this decrease can be attributed to an improvement in endurance exercise following
In the present study, we were interested in assessing carbohydrate utilization by plasma glucose assay. However, neither
Total cholesterol and HDL cholesterol were also estimated. Endurance exercise reduced the levels of plasma TC in successive weekly recordings taken immediately following exercise performance in both the supplemented and unsupplemented groups of rats. Hypobaric hypoxic exposure (72 hours) had no effect on plasma TC levels.
The changes in plasma cholesterol elicited in the present study are well supported by the reports documenting the effects of exercise on plasma lipids. 8 The regulation of substrates during exercise is multifactorial and depends upon 1) dietary and nutritional status; 2) hormonal milieu; 3) exercise mode, intensity, and duration; and 4) training status.26,27 There is also a marked variability in the response (HDL cholesterol) to the same level of exercise. 28 The effects of exercise on HDL cholesterol also depend upon other baseline factors, such as level of initial HDL cholesterol and triglyceride levels.29,30
The present study had the following limitations: 1) the intensity of exercise could not be graded in terms of V
In conclusion,
Footnotes
Acknowledgments
The authors gratefully acknowledge the contribution of Dr Y. K. Sharma, Scientist ‘E’ for statistical analysis of the data.
