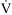Abstract
Introduction
AR36 is a pharmaceutical-grade plant extract used to support cardiovascular health in traditional Chinese medicine. Studies suggest that AR36 may prevent acute mountain sickness (AMS) during gradual ascent to high altitude. This randomized, placebo-controlled Phase 2 trial aimed to evaluate dosing regimens and assess efficacy and safety of AR36 for AMS prevention during rapid ascent.
Methods
Participants received placebo, low-dose AR36 (225 mg twice daily for 14 d prior and 5 d at altitude), or high-dose AR36 (12 d placebo, 300 mg twice daily for 2 d prior and 5 d at altitude). The primary efficacy outcome was 1993 Lake Louise Scoring System (LLSS) score on the morning after ascent. Safety was assessed through the proportion of treatment-emergent adverse events (TEAEs).
Results
One hundred thirty-two participants were randomized. Mean±SD age was 31.4±8.6 (range, 19–54) y. Baseline characteristics did not differ across groups. Lake Louise Scoring System scores on Day 16 in the placebo, low-dose, and high-dose groups were 4.03 (2.88), 4.42 (3.17), and 3.5 (2.31), respectively (placebo versus low-dose, P=0.462; placebo versus high-dose, P=0.574; n=110). The incidence of AMS on Day 16 was 66.7% in the placebo, 61.1% in the low-dose, and 55.3% in the high-dose group (P=0.66). The proportion of TEAEs in the placebo, low-dose, and high-dose groups was 38.4% (81), 28.4% (60), and 33.2% (70), respectively (P=0.205; n=127). There was no statistical difference between groups in LLSS, incidence of AMS, or TEAEs.
Conclusions
AR36 did not improve LLSS or AMS incidence using the current regimens. AR36 was well tolerated.
Introduction
Extended stay at high altitude has become increasingly common for recreation, work, and military purposes. Acute mountain sickness (AMS) describes a constellation of symptoms (including headache, gastrointestinal discomfort, fatigue, dizziness, and sleep disturbances) occurring after ascent to altitudes above 2500 m. 1 The pathophysiology of AMS has not been fully elucidated. However, research suggests that hypobaric hypoxia may lead to hyperventilation, fluid retention, altered gas exchange, increased intracranial pressure, and a systemic inflammatory state, 2 -4 all of which may underlie AMS symptoms. 5 In rare cases, AMS can progress to high-altitude cerebral edema or high-altitude pulmonary edema, which may be life-threatening. 1
Preacclimatization by gradual ascent may reduce the risk of developing AMS. 6 If gradual ascent is not possible, pharmacologic prophylaxis is recommended. 7 Acetazolamide, a carbonic anhydrase inhibitor, is first-line therapy for AMS prevention. 8 Although effective, acetazolamide can mimic AMS symptoms and may be associated with adverse effects that are poorly tolerated.8,9 The efficacy of acetazolamide during rapid ascent to high altitude varies across studies depending on the dose used and rate of ascent.8,10-16 Following rapid ascent to 3810 m, where the current study was conducted, the incidence of AMS after acetazolamide pretreatment was 39 to 55%.8,11-13 Rates in placebo at the same location have been reported as 63 to 69%.12,17 Effectiveness of acetazolamide may be reduced in individuals who are partly acclimatized. 18 Ibuprofen treatment, which is readily available and commonly used, led to AMS rates of 43 to 62%.13,17 These findings highlight the need for additional, alternative pharmacologic interventions that are both effective and well tolerated.
AR36, also known as T89, Dantonic, or Compound Danshen Dripping Pills, is a modernized preparation of a traditional Chinese medicine composed of pharmaceutical-grade extracts from the plants Danshen (Radix Salvia miltiorrhiza) and Sanqi (Radix Panax notoginseng).19,20 AR36 also contains Bing Pian (borneol) derived from the plant Dryobalanops aromatica, which enhances absorption of Danshen and Sanqi.19,20 AR36 has traditionally been used to treat myocardial ischemic diseases in China and is approved by the National Medical Products Administration for treatment of angina pectoris. 19 AR36 has been shown to increase oxygen saturation during high-altitude exposure 20 and reduce proinflammatory cytokines,21,22 which may reduce the inflammatory state that is thought to partly underlie AMS development. In addition, P notoginseng was shown to improve exercise tolerance in mice at simulated high altitude. 23
In a placebo-controlled pilot study conducted in 2014, pretreatment with AR36 during gradual ascent reduced the risk of developing AMS by 19% and increased exercise capacity. 20 The present prospective, double-blind, randomized, placebo-controlled Phase 2 trial aimed to assess the efficacy and safety of 2 AR36 dose regimens for prevention of AMS during rapid ascent to 3810 m to assess the feasibility of a trial at this location and to inform the design of a proposed pivotal Phase 3 trial in a US population.
Methods
Ethical Approval
Study protocols were approved by the University of California San Francisco Institutional Review Board (IRB) and conformed to the Declaration of Helsinki (IRB number: 17-23953; trial registration:
Study Participants
Participants were recruited via social media and local advertisements and were predominantly from the San Francisco Bay area but were accepted from farther away if they met study inclusion criteria and were able to attend all visits. Participants were prescreened to determine feasibility of participation in study procedures and to confirm lowland dwelling status. All participants were unacclimatized, healthy, nonsmoking adults aged 18 to 55 y who resided less than 1000 ft (approximately 305 m) above sea level. This age limit was selected to minimize the risk of occult comorbid cardiac conditions in older participants, because of planned daily exercise to exhaustion at high altitude. Participants were also required to pass a drug screen and physical examination and have normal baseline laboratory values (hematology, clinical biochemistry, and urinalysis) prior to enrollment. Major exclusion criteria included a medical history of cardiovascular, cerebrovascular, respiratory, renal, hepatic, or other significant comorbid conditions; blood oxygen saturation (SpO2) less than 95% at sea level; and surgery, blood donation, or treatment with any medications aside from oral contraceptives within 14 d of screening and during the study. The complete list of inclusion and exclusion criteria is provided in online Supplemental Table 1.
Study Design
This Phase 2 study was designed to perform an exploratory analysis comparing efficacy and safety of 2 different AR36 dosing regimens in a healthy US population in order to optimize trial logistics and plan future Phase 3 pivotal trials. The targeted enrollment of 132 participants (44 per group) was based upon a previous trial of gradual ascent in 141 participants 20 as well as the anticipated rates of AMS at this elevation.
This study investigated 2 different AR36 dosages and regimens. We hypothesized that a lower dose with a longer pretreatment period could gradually increase plasma drug levels and have a similar efficacy profile and a better safety profile compared with a shorter, higher-dose regimen. Participants were randomized 1:1:1 by block randomization to 3 treatment groups: placebo, low-dose AR36 (14 d treatment before ascent continued 5d at altitude), and high-dose AR36 (12 d placebo, then 2 d treatment before ascent and continued 5d at altitude). The low-dose AR36 group received a constant dose of 225 mg twice daily for 14 d at sea level and for 5 d after ascent. The high-dose AR36 group received placebo twice daily for the first 12 d at sea level, followed by 300 mg twice daily for 2 d at sea level and for 5 d after ascent. The placebo group received placebo treatment twice daily for 14 d at sea level and for 5 d after ascent (Figure 1).
Study drug and ascent schedule for each treatment group. n=110.
Investigators and participants remained blinded to group assignments until trial completion. To maintain blinding, all investigational product capsules provided by the manufacturer were identical. Compliance with treatment regimens was self-reported by participants via a drug and symptom log from Days 1 to 14 and was ensured by direct observation on Days 15 to 19. On Day 15, participants ascended from sea level to 3810 m (Nello Pace Laboratory at Barcroft Station, White Mountain Research Center) by automobile over 8 to 10 h and remained there until completion of the study on Day 19. Study data were collected over 2 operational seasons (June–October 2018 and June–October 2019).
Outcomes
The primary efficacy outcome was mean 1993 Lake Louise Scoring System (LLSS) score on Day 16 (the morning after ascent). Secondary efficacy outcomes included mean SpO2, exercise tolerance, systolic and diastolic blood pressure (BP), heart rate, and area under the curve (AUC) of the 1993 LLSS–time curve during rapid ascent and stay at high altitude (Days 15–19). Area under the curve was used as a novel method for assessing a composite of severity and duration of AMS. Relative risk for the development of AMS over the entire study period and the total incidence of AMS the morning after arrival were also calculated. As acute acclimatization is generally thought to occur within 3 to 4 d at this altitude, data from Days 15 to 18 were used for analysis of SpO2, BP, and heart rate. The primary safety outcome was the proportion of treatment-emergent adverse events (TEAEs) across treatment groups.
Acute Mountain Sickness Assessment
Acute mountain sickness symptoms were assessed using the 1993 LLSS at each prealtitude visit and twice daily while at altitude. 24 This version of the LLSS was used because the revised score 25 only became available in 2018, after IRB and US Food and Drug Administration approval and after study initiation. The 1993 LLSS contains 3 domains: a self-administered questionnaire for headache, gastrointestinal symptoms, fatigue/weakness, dizziness/lightheadedness, and difficulty sleeping; a functional assessment of the degree to which AMS symptoms caused a reduction in activity; and a clinical assessment of mental status, ataxia, and peripheral edema. All clinical assessments were performed by study investigators. Each symptom was scored as 0, 1, 2, or 3, indicating no, mild, moderate, or severe symptoms, respectively. Pain from headache was assessed using a visual analog scale. 26 -28 Acute mountain sickness was defined as a mean combined 1993 LLSS score of ≥3 in the presence of a headache and at least 1 other symptom. 24 A post hoc calculation of LLSS score using the 2018 definition was also performed.
Laboratory and Physiologic Assessments
Physiologic measurements, including SpO2, BP, and heart rate, were obtained after at least 5 min of rest in a seated position at the following time points: enrollment (before initiation of drug), on the day before ascent (day 14), on the day of ascent (day 15), and on each day after arrival (Days 16–19). Blood oxygen saturation and heart rate were monitored using a Radical-7 Pulse CO-Oximeter (Masimo Co, Irvine, CA) applied to participants’ index fingers. For each sea level visit, a single measurement was taken. At altitude, these data were collected 4 times per day: before breakfast, before lunch, before dinner, and before sleep. Laboratory assessments (hematology, clinical biochemistry, and urinalysis) and electrocardiograms were also obtained at sea level and at altitude.
Exercise Test
At sea level, participants completed physiologic and AMS assessments followed by an exercise tolerance test consisting of cycling to exhaustion using a ramp protocol. After a 3-min unloaded warm-up at 30 W, resistance was increased to a starting power corresponding to participant weight; exercise load started at 110 W for participants weighing ≥60 kg and at 90 W for those weighing <60 kg. Regardless of starting weight, the load was increased by 5 W every 20 s until participants reached exhaustion, which was marked by cadence decreasing below 40 revolutions/min. At altitude, this test was performed in the afternoon on the day of arrival (Day 15) following physiologic and AMS assessments and every morning for subsequent days (Days 16–19) following physiologic and AMS assessments. The exercise protocol was designed to capture maximal oxygen consumption (
Efficacy Analysis
Descriptive statistics, including mean, standard deviation, standard error of the mean, median, interquartile range, and range, were reported for continuous variables. Categorical variables were summarized using counts and percentages. Statistical analyses for detecting significant differences from baseline (before ascent) or compared with placebo included the Kruskal-Wallis test, Mann-Whitney U test, χ2 test, Tukey’s 2-sided test, and analysis of variance. Alpha was set to 0.05. Analyses were performed using Prism 9.0 (GraphPad Software, San Diego, CA) and R 3.1.1 (open source;
Safety Analysis
Safety analyses were performed in all participants treated with at least 1 dose of the study drug or placebo. At sea level, AMS symptoms were included as adverse events because of the lack of confounding factors. At altitude, however, AMS symptoms were not included as adverse events unless they were severe and affected study participation since they were already captured by the LLSS. As such, AMS symptoms resulting in study withdrawal were recorded as severe adverse events because of their rarity at this altitude. The Medical Dictionary for Regulatory Activities was used to classify all adverse events according to organ system and preferred term. Severity of TEAEs was assessed using Common Terminology Criteria for Adverse Events, version 5.5.
Results
Study Participants
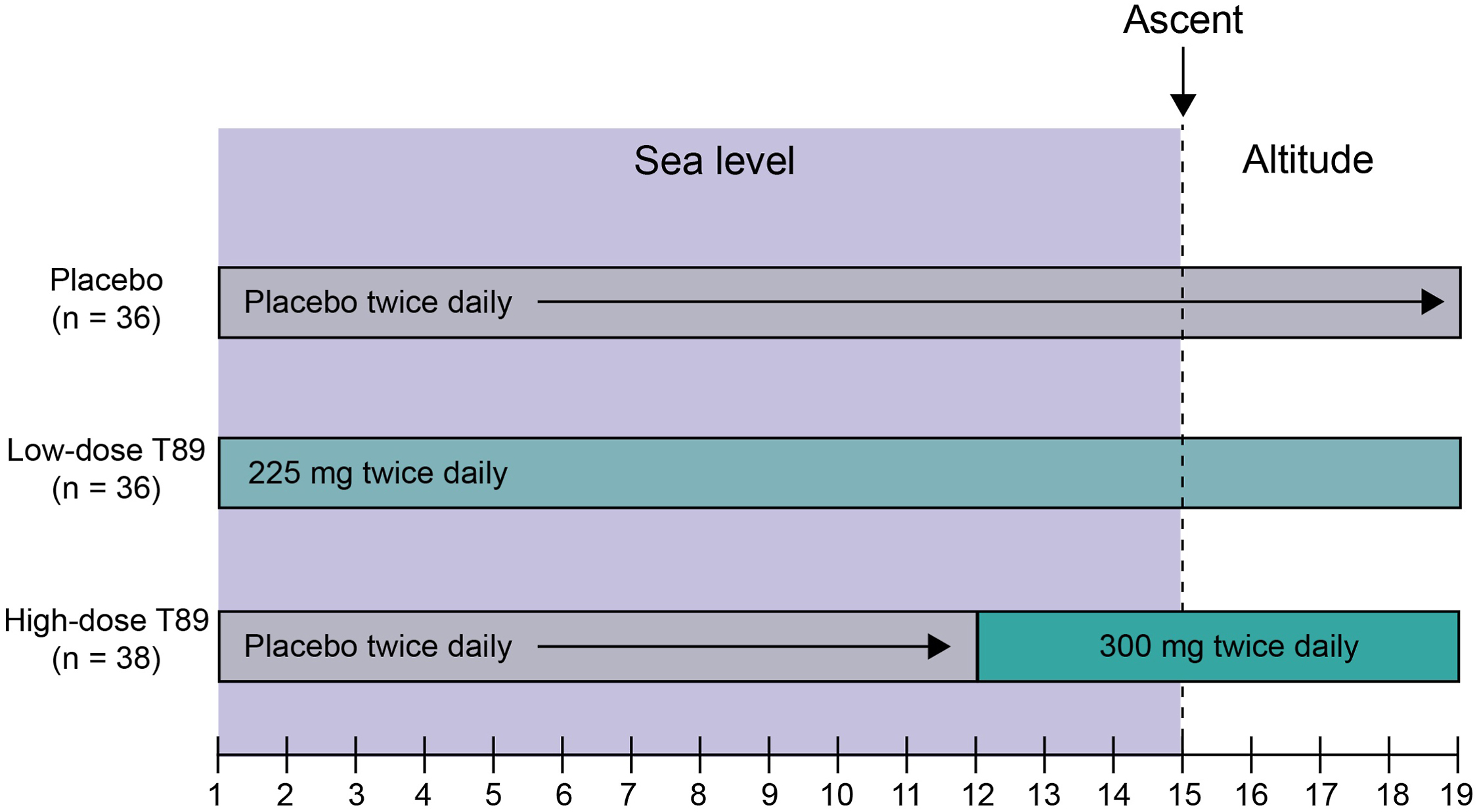
In total, 132 participants (44 placebo, 44 low-dose AR36, and 44 high-dose AR36) were randomized. Baseline characteristics were not statistically different across treatment groups (Table 1). Fifty-seven participants (43%) were female and 70 (53%) were White. Mean age was 31.4±8.6 (range, 19–54) y. Mean body mass index for the study population was 23.4±3.0 kg/m2. Overall, 22 participants withdrew from the study before ascent to altitude. Of these, 11 were withdrawn after inclement weather made Barcroft Station inaccessible, 2 were withdrawn because of consumption of prohibited concomitant medications, and 9 were lost to follow-up. Of the 110 participants who ascended, 4 withdrew from the study at altitude and 106 completed all study visits (Figure 2). Compliance was 100% in both AR36 groups on Days 15 to 19 as the drug was directly administered.
Baseline participant characteristics
BMI, body mass index.
Participant flow diagram. N=132.
Efficacy Analysis
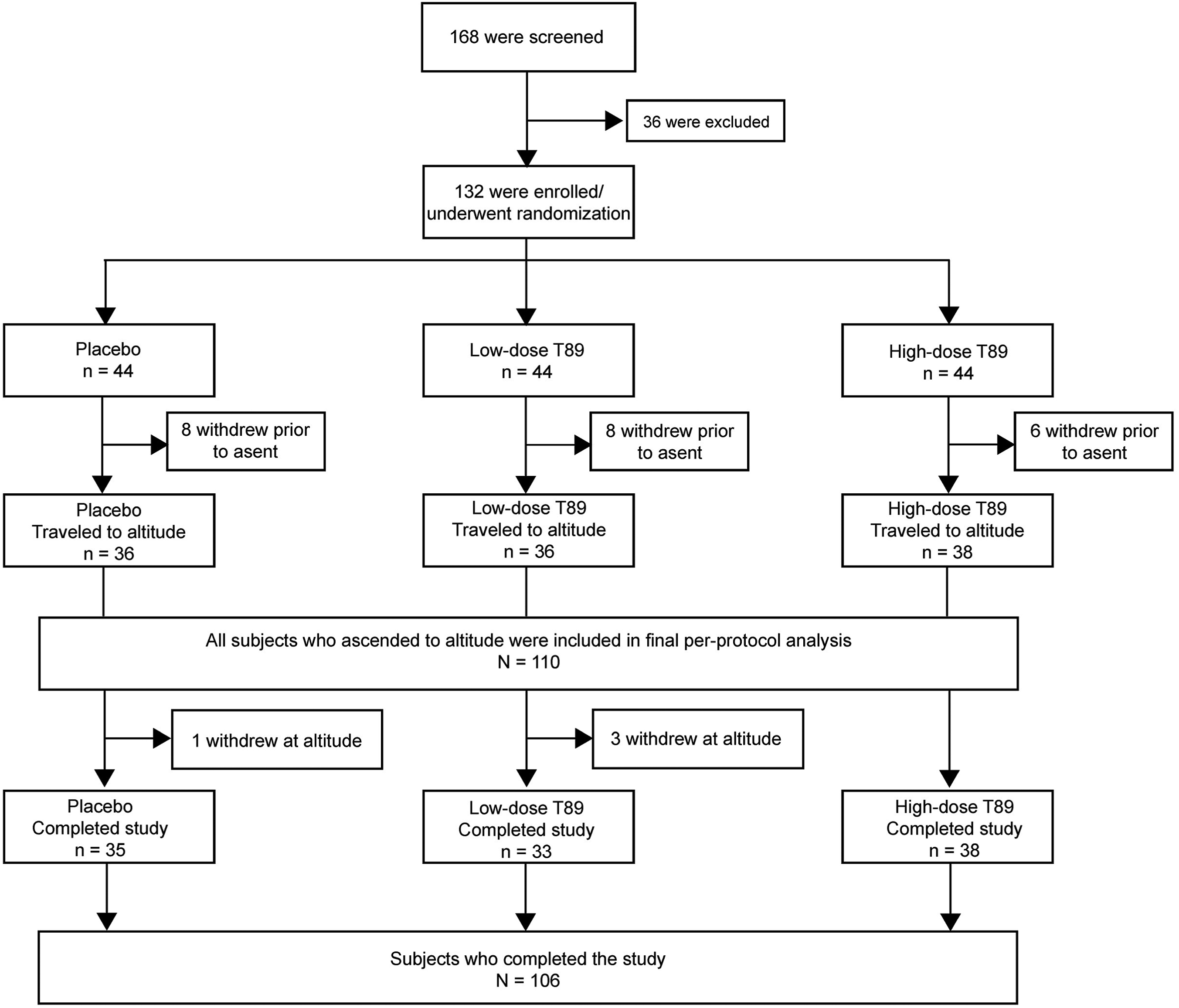
A total of 110 participants (36 placebo, 36 low-dose AR36, and 38 high-dose AR36) reached the planned altitude and were included in the efficacy analysis. Using the 1993 consensus definition, LLSS scores on the morning after ascent (Day 16) in the placebo, low-dose, and high-dose AR36 groups were 4.03 (2.88), 4.42 (3.17), and 3.5 (2.31), respectively (placebo versus low-dose, P=0.462; placebo versus high-dose, P=0.574) (Table 2). There were no significant differences in 1993 LLSS scores on any of the days spent at altitude (Days 15–19) (Figure 3, Table 2). Post hoc analysis using the 2018 LLSS consensus definition showed a trend toward lower LLSS score in the AR36 groups compared to the placebo group, but this was not significant (see online Supplemental Figure 1). Separate analyses were performed for headache scores using the 1993 LLSS consensus definition (see online Supplemental Table 2). Headache scores in the placebo, low-dose, and high-dose AR36 groups were 1.06 (0.92), 1.33 (1.04), and 1.00 (0.84), respectively (placebo versus low-dose, P=0.34; placebo versus high-dose, P=0.95).
Mean 1993 Lake Louise Scoring System score at altitude
LLSS, Lake Louise Scoring System.
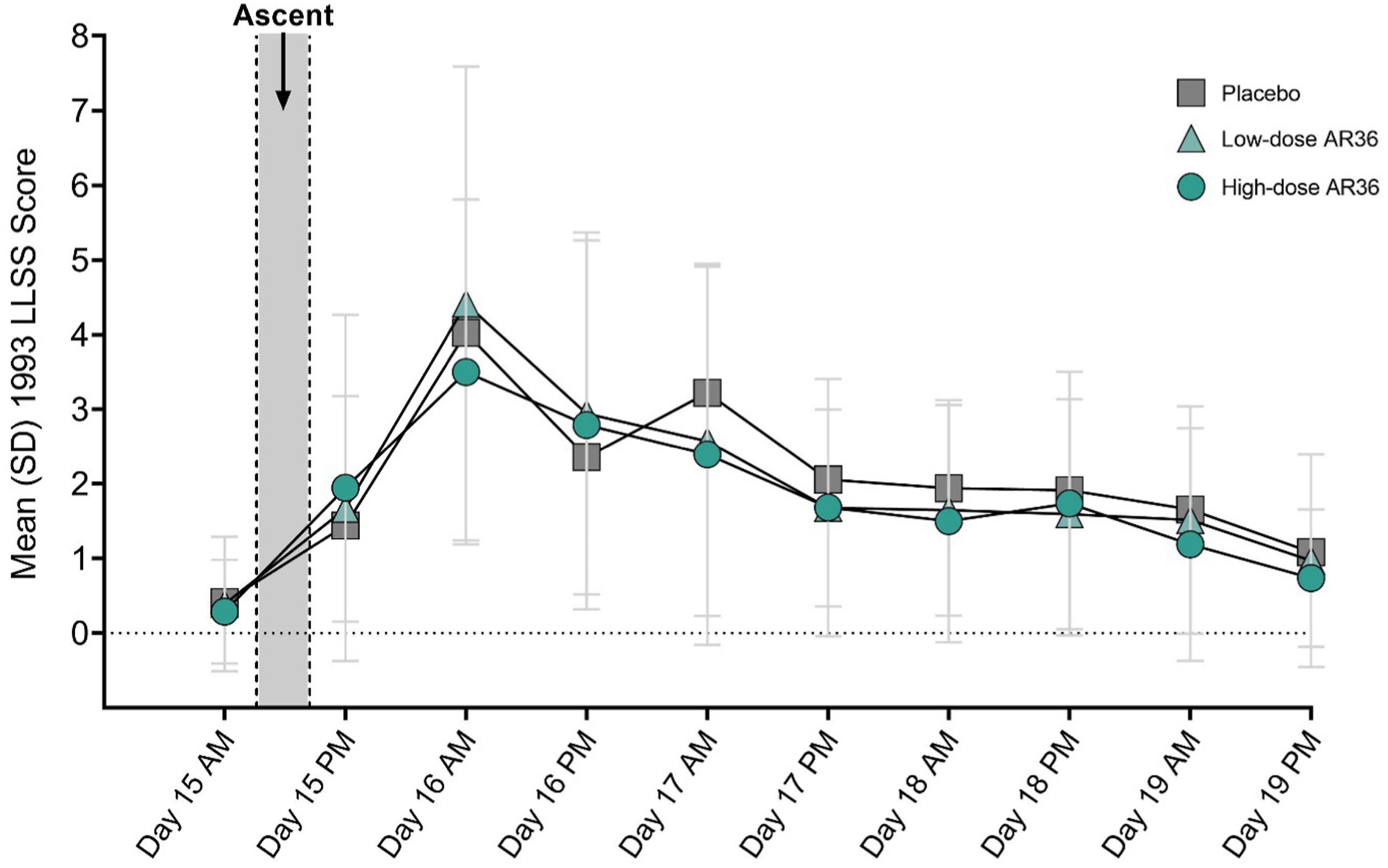
Mean±SD LLSS scores for the entire stay at altitude (Days 15–19) using the 1993 consensus definition. Day 15, n=110; Day 19, n=106. LLSS, Lake Louise Scoring System; SD, standard deviation.
There were no sex differences in 1993 LLSS score in either the placebo or low-dose AR36 groups. Interestingly, females in the high-dose AR36 group had higher LLSS scores compared to males, although post hoc analysis did not identify a difference at any specific time point (P<0.001) (see online Supplemental Table 3). Acute mountain sickness incidence on the morning of Day 16 was 66.7% in the placebo group, 61.1% in the low-dose AR36 group, and 55.3% in the high-dose AR36 group (P=0.66). The relative risk (95% CI) for AMS across the entire study period was 0.82 (0.59–1.11) in the low-dose AR36 group and 0.85 (0.62–1.13) in the high-dose AR36 group (Figure 4). The AUC of the 1993 LLSS–time curve, a measure of the total AMS experience over the period at high altitude, was 19.37 (3.92), 18.71 (4.1), and 17.25 (4.2) in the placebo, low-dose, and high-dose groups, respectively (P=0.931) (Figure 5).
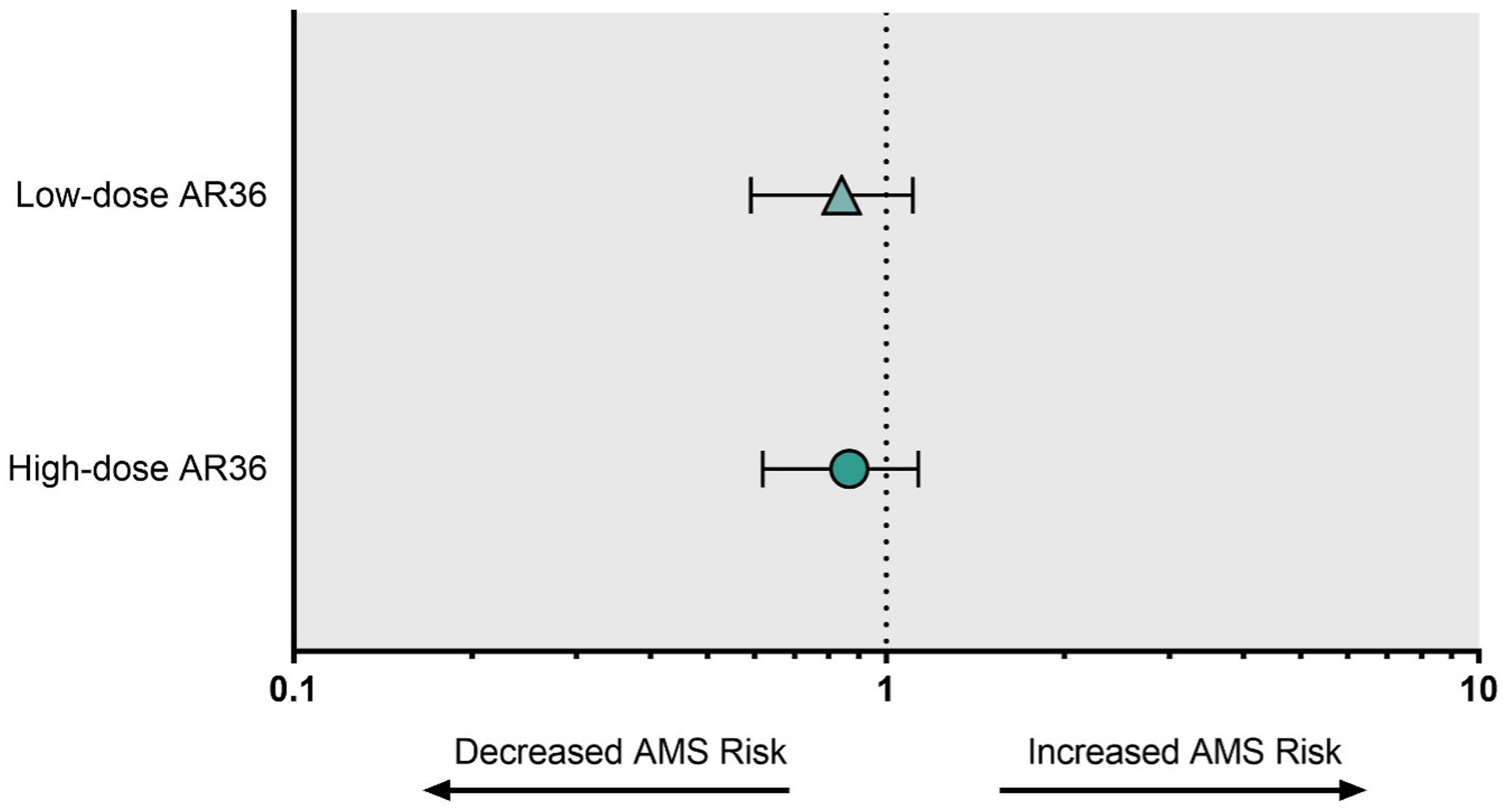
Relative risk for AMS in the low-dose AR36 and high-dose AR36 groups. n=110. AMS, acute mountain sickness.
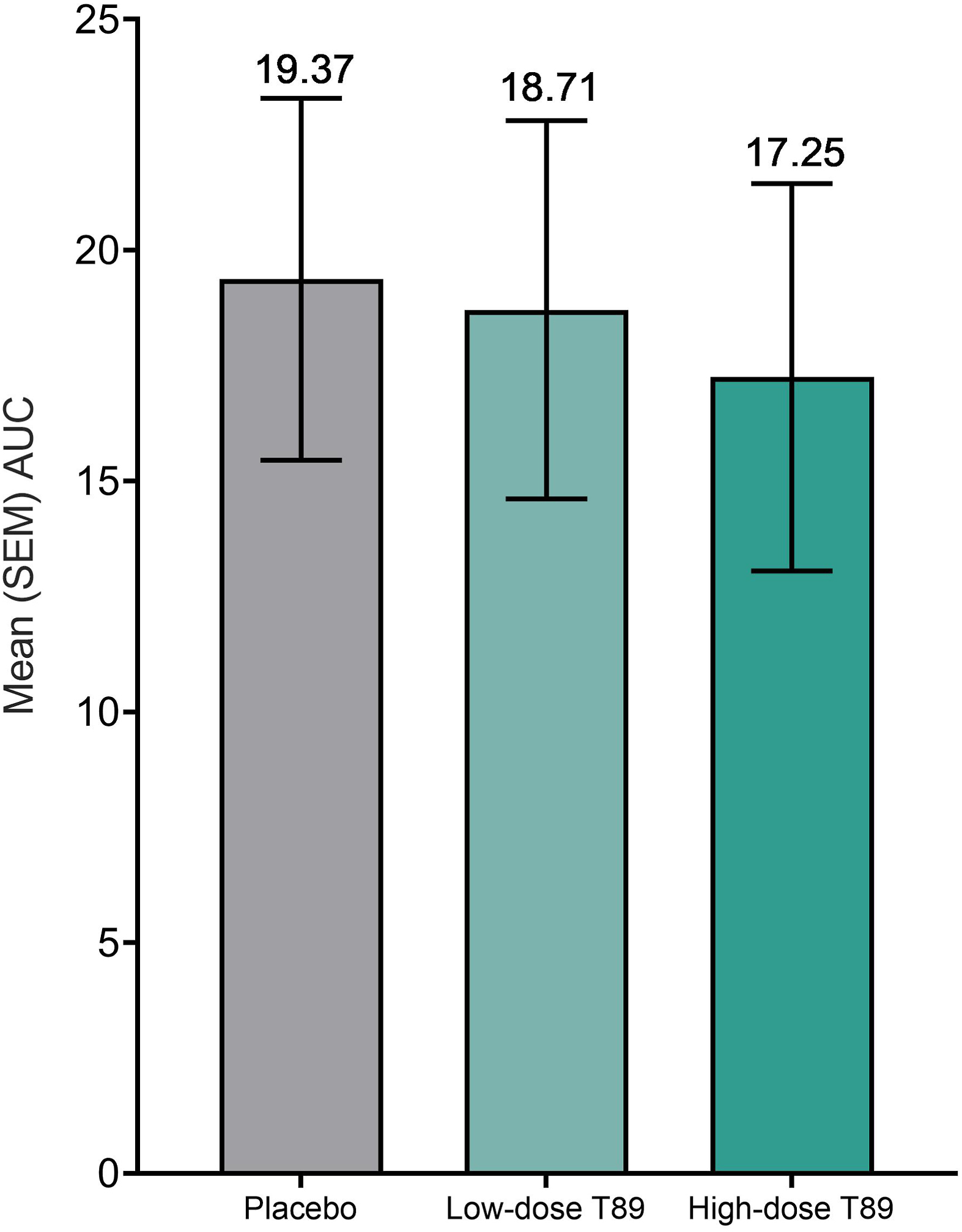
Mean±SEM AUC of the Lake Louise Scoring System–time curve for participants who completed all Lake Louise Scoring System assessments during the study. n=106. AUC, area under the curve; SEM, standard error of the mean.
Compared with the low-dose group, the high-dose AR36 group demonstrated higher SpO2 at bedtime on Day 15 (low-dose AR36, 81.91 [5.16]; high-dose AR36, 84.74 [4.67]; P=0.045) and at breakfast on Day 18 (low-dose AR36, 86.44 [5.37]; high-dose AR36, 89.92 [3.84]; P=0.008)] (Figure 6; see online Supplemental Table 4). No differences were observed in BP (see online Supplemental Figure 2 and Supplemental Table 5), heart rate (see online Supplemental Figure 3 and Supplemental Table 6), or exercise tolerance (Table 3) between placebo and AR36 groups on any days at altitude.
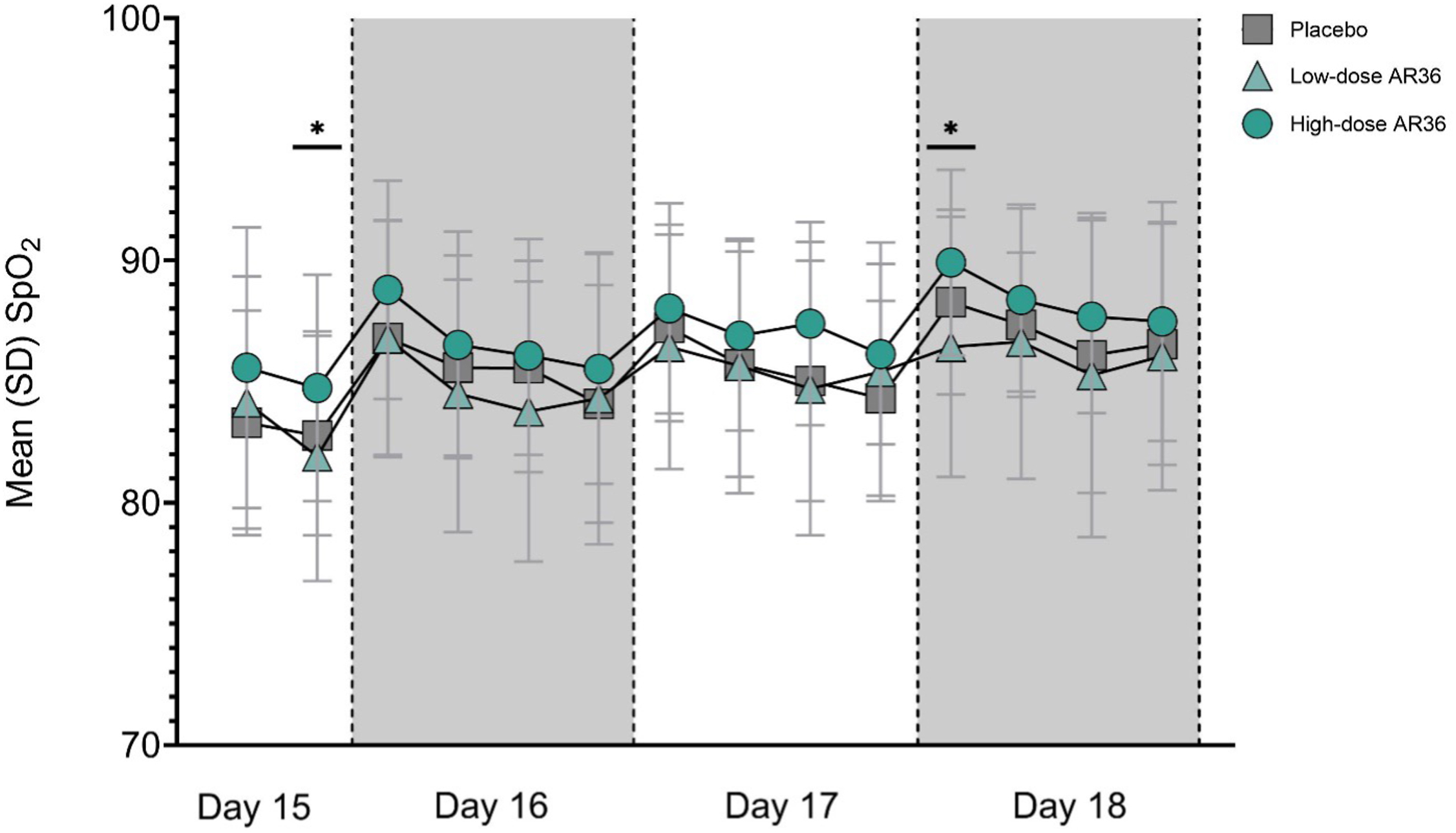
Mean±SD SpO2 values at high altitude (Days 15–18). Day 15, n=110; Day 18, n=106. SD, standard deviation; SpO2, blood oxygen saturation. ∗P<0.05
Mean maximum power output during exercise and change in power from baseline
Safety Analysis
A total of 127 participants received at least 1 dose of the study drug or placebo and were included in the safety analysis. Duration of exposure to the study drug was 0 d in the placebo group, 19 d in the low-dose AR36 group, and 7 d in the high-dose AR36 group. There was no significant difference in the proportion of TEAEs across treatment groups (placebo, 38.4% [81]; low-dose AR36, 28.4% [60]; high-dose AR36, 33.2% [70]; P=0.205). Overall, 211 TEAEs were reported in 80 participants (63%), and 94% of TEAEs were mild. The most frequently reported TEAE classes included gastrointestinal disorders (57 TEAEs in 39 participants; 31%), nervous system disorders (48 TEAEs in 37 participants; 29%), respiratory disorders (28 TEAEs in 20 participants; 16%), general and administration site conditions (18 TEAEs in 16 participants; 13%), and musculoskeletal disorders (19 TEAEs in 13 participants; 10%) (Table 4, see online Supplemental Table 7).
Summary of treatment-emergent adverse events by organ system
TEAEs, treatment-emergent adverse events.
Three of the 4 participants who withdrew from the study at altitude were from the low-dose AR36 group, and 1 was from the placebo group. Two participants (low-dose AR36 group and placebo group) voluntarily withdrew because of intolerable AMS symptoms. One participant from the low-dose AR36 group was evacuated from the altitude station and withdrawn for safety reasons after he developed high-altitude pulmonary edema on Day 18. Because of the clear temporal relationship between ascent and symptom resolution on descent, this event was deemed unrelated to drug administration. One participant from the low-dose AR36 group was withdrawn because of elevation in transaminases on Day 16. Both aspartate aminotransferase and alanine aminotransferase normalized within 1 wk when re-evaluated at sea level.
Discussion
This Phase 2 study was designed to explore the efficacy and safety of AR36 in healthy US participants during rapid ascent to altitude and to determine appropriate dosing regimens and operational study procedures. Compared with placebo, the 2 AR36 dosing regimens did not show a significant difference in the primary outcome of 1993 LLSS score on the morning after ascent (Day 16), nor did they show a difference in relative risk for the development of AMS over the course of the study. However, higher LLSS scores in females in the high-dose AR36 group suggest that there may be sex differences in the effects of AR36, which may warrant further investigation in a subsequent trial. Administration of AR36 appeared to be well tolerated, with 94% of all TEAEs being of mild severity, and with most adverse events recorded as mild gastrointestinal upset or headache prior to ascent (these symptoms were captured by the 1993 LLSS at altitude). The safety analysis did not show a higher proportion of TEAEs in the AR36 groups compared with placebo, even at the higher dose.
We aimed to determine whether a longer, low-dose regimen versus a shorter and more feasible high-dose regimen would yield similar treatment effects. There did not seem to be an advantage to a longer, lower dose regimen, as neither reached significance for AMS incidence or LLSS score. Most physiologic variables were unchanged, but high-dose AR36 was associated with higher SpO2 compared with that in the low-dose AR36 group. Although not significantly different, lower AMS incidence, lower LLSS AUC, and lower LLSS scores may have reached significance with a sample size of 150 to 200 subjects, which should be considered in future trials. A higher dose of AR36 over a shorter period may be more practical and may justify administration using this regimen in future studies. In addition, the AR36 dosages in this study may have been too low to produce a significant effect as measured by the LLSS. Notably, the AR36 dosages administered in this study were both lower and less frequent than an earlier randomized, placebo-controlled pilot study that showed lower AMS incidence with Compound Danshen Dripping Pills during gradual ascent to altitude. 20 Higher doses of AR36 than were used in this trial may be required for the compound to be effective.
Although AR36 did not result in a significant reduction in AMS incidence or a difference in exercise tolerance, high-dose AR36 did result in a modest but significantly higher SpO2 at altitude compared with low-dose AR36. This improvement in oxygenation may have resulted in increased oxygen delivery to tissues and a resultant decrease in tissue hypoxia. Several studies have shown a correlation between hypoxemia and development of AMS, 29 -35 and prior research has shown that acetazolamide use was associated with higher SpO2 compared with placebo. 36 However, other studies have failed to show that higher oxygen saturation was protective against AMS.37,38 Therefore, the modest increase in peripheral arterial oxygenation detected in this study following treatment with high-dose AR36 is of unclear clinical significance.
The exercise protocol used in our study was designed to capture VO2 max and did not demonstrate significant differences in exercise tolerance between treatment groups. As the protocol was designed to capture 
Study Limitations
This Phase 2 study was limited by the lack of a formal power analysis. The drug was previously tested in a study that had a 36-h ascent to altitude followed by a 1-d stay during which a higher dose and different dosing regimen were used compared with those used in this study. Due to the difference in rate of ascent and dosing regimen, a power analysis was not performed. This lack of power analysis resulted in a trial that was underpowered to detect a difference in efficacy measures based on the results obtained.
A second limitation is that the LLSS, the standard assessment for AMS, is inherently dependent on a participant’s subjective interpretation of AMS symptoms. This subjectivity likely resulted in a high degree of variability, which limited our ability to detect meaningful signals in the data with this small sample size. However, we believe that the key limitations of the current Phase 2 study can be addressed by a larger sample size and a simplified dosing regimen using a higher dose of AR36.
Conclusions
Under the current dosing regimens, AR36 did not significantly lower LLSS scores or reduce AMS incidence compared to placebo. AR36 was well tolerated at both dose levels. A larger study with a higher dose is needed to determine whether AR36 could be effective and safe for prevention of AMS.
Footnotes
Acknowledgments
We thank all participants involved in this study and all members of the University of California San Francisco (UCSF) AR36 working group (Helge Eilers, Philip Bickler, John Feiner, Andrea Olmos, Odmara Barreto Chang, and Koa Gudelunas, all of UCSF Anesthesia). Medical writing support was provided under the direction of the authors by Jennifer Shepherd, PhD, and Shervonne Poleon, PhD, from Porterhouse Medical Group.
Author Contributions: UCSF study staff designed the study in collaboration with Tasly Pharmaceuticals and were solely responsible for conduct of the study, collection of data, and analysis of the data obtained independent of Tasly Pharmaceuticals. AS and GC contributed equally to the development of this manuscript. All authors had access to the data and were involved in drafting and revising this manuscript. All authors approved the final manuscript.
Financial/Material Support: This study was funded by Tasly Pharmaceuticals. Development of this manuscript was supported by Azurity Pharmaceuticals, Inc, through Porterhouse Medical.
Disclosures: AS, MD, and YE have no disclosures. JS has received funding for unrelated projects from Pfizer, California Bureau of Cannabis Control, and the National Institute of General Medical Sciences. GC has received funding from the Foundation for Anesthesia Education and Research for unrelated research on brain development and anesthesia exposure.
Supplemental Materials
Supplementary material associated with this article can be found in the online version at