Abstract
Objective
This study aimed to determine the efficacy, tolerability, and practicality of acetazolamide for the prevention of acute mountain sickness (AMS) in Nepali trekking porters early in the trekking season.
Methods
This study was a randomized, double-blind controlled trial with 400 male Nepali porters in the Mount Everest region of Nepal, trekking from Namche Bazaar (3440 m) to Lobuche (4930 m), the study endpoint. Participants were randomized to receive 250 mg acetazolamide daily or placebo, and AMS symptom scores (Lake Louise) were compared in highlanders vs lowlanders.
Results
Only 109 (27.2%) of the 400 porters completed the trial (28 highlanders, 81 lowlanders). The rest either dropped out (275/400 porters, 68.8%) or were excluded (16/400 porters, 4%). Acute mountain sickness occurred in 13 (11.9%) of 109 porters; all were lowlanders; 7 were taking acetazolamide, 6 taking placebo. Birthplace, acclimatization in the week before the trial, ascent rate, and rest days were the most important variables affecting the incidence of AMS. No highlanders, but 13 (16.1%) of 81 lowlanders had AMS (P = .016). Acclimatization in the pretrial week reduced AMS incidence (P = .013), as did a slower ascent rate (P = .0126), but rest days were the most potent prophylactic variable (P = .0001). Side effects were more frequent in porters taking acetazolamide than in the placebo group (P = .0001), but there were no serious side effects.
Conclusions
Acetazolamide was tolerable, but impractical for the routine prevention of AMS in Nepali porters. A good trekking schedule and adequate acclimatization remain the most effective preventive measures. This study identified lowland porters as a high-risk group for developing AMS.
Introduction
The trekking and mountaineering industry is a major source of income for the Nepali economy and attracts many lowland Nepalis to work as mountain porters. They carry heavy loads, and are often poorly equipped and inadequately informed of the risks of high altitude.
Acute mountain sickness (AMS) in trekkers and mountaineers ascending to high altitude remains a common problem1,2 and much research has been directed to its prevention. Nepali porters are as susceptible to AMS as trekkers 3 and a high incidence of AMS (37%) has been reported in non-Sherpa Nepali porters. 4
Acetazolamide, a carbonic anhydrase inhibitor, is effective and tolerable in preventing AMS.5,6 Clinical observations of acetazolamide prophylaxis (250 mg daily) in Nepali porters gave encouraging results, 7 suggesting that they could benefit from acetazolamide, especially early in the trekking season, when they are first exposed to high altitude. We were unaware of any controlled trials of the drug in these porters, who are small in stature, slightly built, and carry heavy loads (exercise increases AMS at simulated high altitude 8 ). Therefore, we undertook a double-blind controlled trial of 250 mg acetazolamide daily vs placebo to test the efficacy, tolerability, and practicality of acetazolamide for the prevention of AMS early in the trekking season.
Methods
Design
The Nepal Health Research Council approved the study protocol for a prospective randomized, double-blind controlled trial of 250 mg acetazolamide (Diamox, Wyeth Laboratories, Maidenhead, UK) daily vs placebo capsules. Trial medication (encapsulation and randomization of acetazolamide and placebo) was organized by DHP Ltd, Abergavenny, Monmouthshire, UK. The randomization code was sent directly by DHP to one of the authors of this study (A.P.), who was not directly involved in performing the clinical trial. He prepared the sealed envelopes containing the trial codes.
Participants
Each day from October 6, 2001 to November 4, 2001, Nepali doctors in Namche Bazaar (3440 m altitude), working in pairs, explained the study to small groups of porters and obtained informed consent from 403 adult (≥18 years old) male Nepali trekking porters heading for the Everest trek via Lobuche. The majority (291 porters) were TibetoBurmans, the rest were Aryans (106 porters) or Newars (6 porters). The 8 Nepali doctors were able to communicate with all of the porters in the Nepali language.
Exclusion criteria were AMS, various medical conditions, (eg, gastroenteritis, diabetes mellitus, glaucoma, or epilepsy), sulphonamide allergy or any other previous drug reaction, or taking a different trek route that did not pass through the assessment stations. We enrolled 400 porters, having excluded 3 porters (gastroenteritis, AMS, and different trek route).
Protocol
Porters’ demography and pretrial altitudes were recorded at enrollment at the Namche Bazaar. It was usually possible to record the altitude at which porters stayed in the pretrial week, because the villages were usually on trekking routes and were recorded in guide books or on maps. The exact altitudes of porters’ birthplace villages were difficult to ascertain, therefore, porters were defined as highlanders or lowlanders depending on whether their village was above or within the rice-growing belt. This method was also used to define the altitude of sojourn during the monsoon. The upper limit of the rice growing belt is 3050 m of altitude.9,10
Each porter was given sufficient trial medication for 7 days, an identification card including the trial number, and a sealed envelope that was only to be opened in the event of illness or a Lake Louise AMS symptom score of at least 3, including headache plus at least one other symptom. 11 The envelope contained a rescue protocol in Nepali, including advice to consult a doctor, the trial number, and the identity of the trial medication (acetazolamide or placebo). Nepali doctors were stationed in pairs at Tengboche (3870 m), Somare (4040 m), and Lobuche (4930 m). Porters were asked to report to them and were assessed for AMS, using the Lake Louise AMS symptom score 11 translated into Nepali. The definition of AMS was a Lake Louise symptom score of at least 3 (headache plus at least one other AMS symptom). Porters were asked about side effects of medication and other symptoms, and compliance was checked by tablet counts. Porters were deemed to have completed the trial when they reported at all three stations, or if they had to exit because of AMS or side effects. Those who failed to report at one or more of the assessment stations were classed as dropouts. The only exceptions to this rule were the 5 porters who missed the second assessment station in the first 2 days of the trial because we had to move the station from Pheriche to Somare for logistical reasons.
The ascent rate was the altitude gain per time. Rest days were defined as days when the porter did not ascend.
Statistical Methods
Our previous experience 7 revealed a high incidence of AMS in porters without acetazolamide prophylaxis (75% suffered AMS at 3960 m). Therefore, we made a conservative estimate that the rate of AMS could be 40% in the untreated group, with a 50% reduction in the treated group. The sample size required to detect this difference with a statistical significance of 5% and a power of 80% is 91 porters in each group completing the study. We aimed to recruit 400 porters, assuming a dropout rate of greater than 50%, having sought advice from a local expert (B. Basnyat, oral communication, July 10, 2001).
The data were analyzed using GraphPad InStat, v3.00 for Windows 95, GraphPad Software, San Diego, CA. Nonparametric analysis was performed on the data obtained, because there is no previous experience within this field to assume that the data collected may be Normally distributed. Tests used to analyze the data were the Fisher exact probability test for comparison of proportions between groups and the Mann-Whitney test for comparison of Lake Louise scores between groups.
Results
Demographic details and the pretrial altitude location of the recruited porters are shown in Tables 1 and 2. Of the 400 porters recruited, only 109 porters (27.25%; 55 porters taking acetazolamide and 54 porters taking placebo) completed the trial, details of which appear in the flowchart in Figure 1.
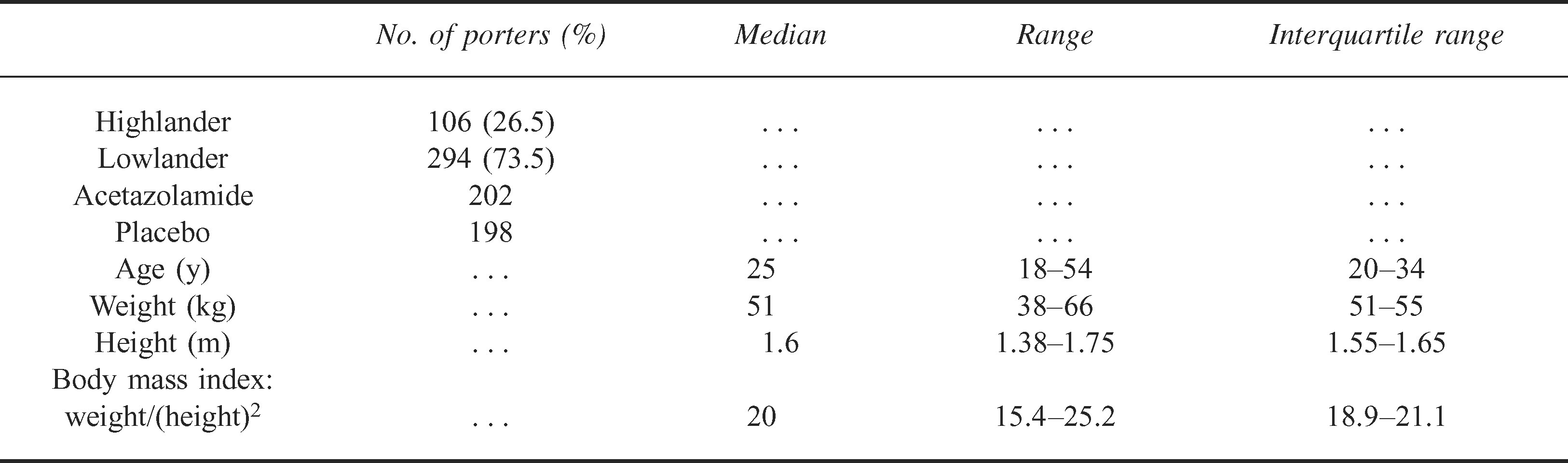
Porters’ demographic details
Location of porters during Monsoon and pretrial week

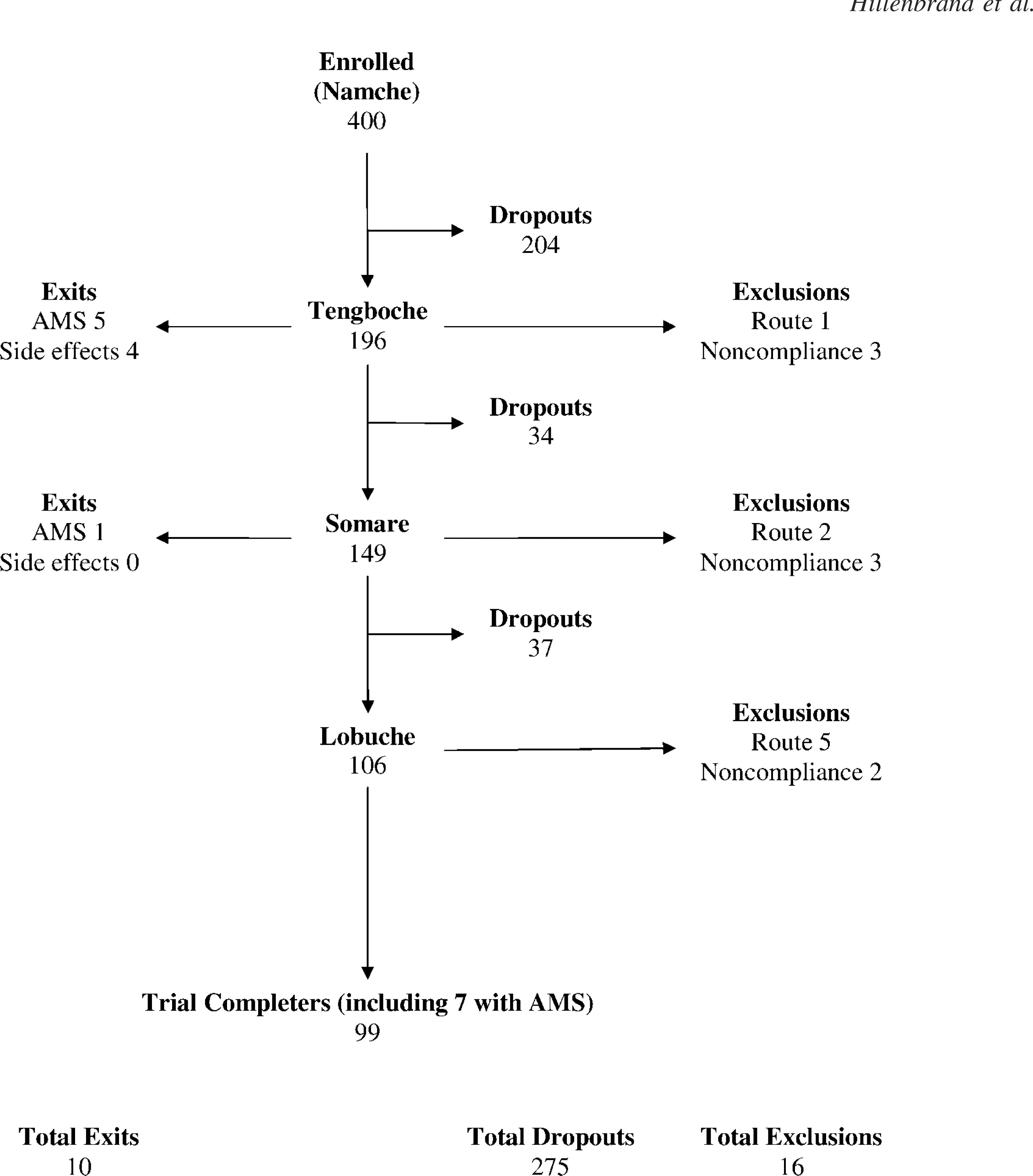
Flow chart of 400 porters enrolled in the trial.
Porters who Failed to Complete the Trial
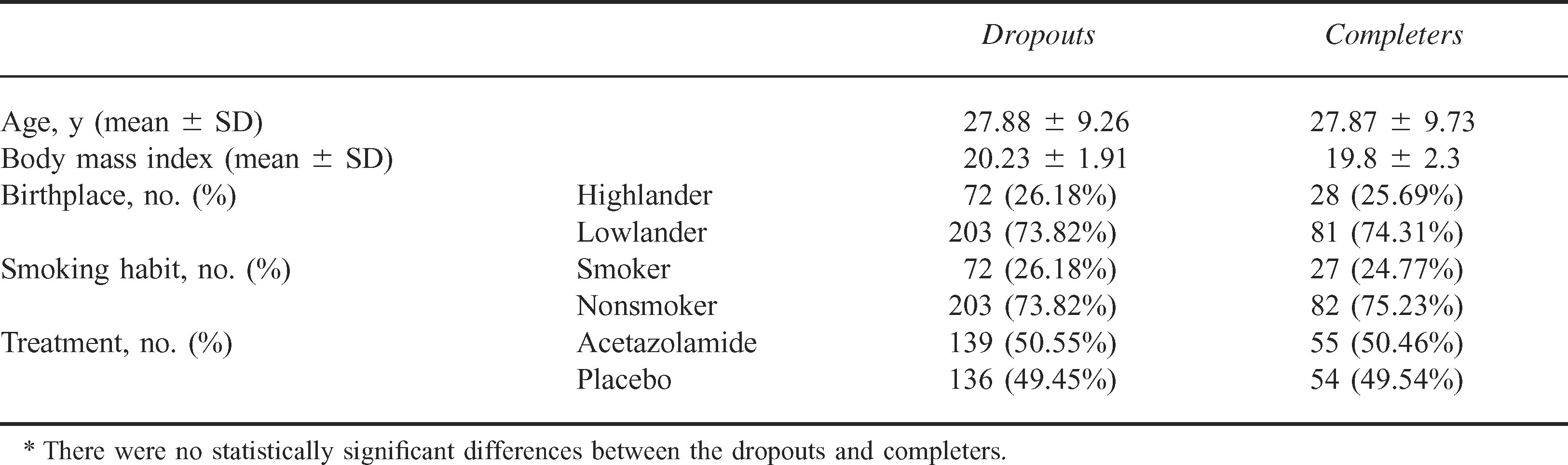
Most porters (275 porters; 68.75%) dropped out of the trial; 92 porters missed 1 station, 61 porters missed 2 stations, and 122 porters missed all 3 stations. Treatment allocation and demographic data were similar in porters who completed the trial and in those who dropped out (Table 3). Sixteen porters (4%) were excluded from the analysis, 8 porters for deviating from the standard trek route and 8 porters for noncompliance with medication. Three noncompliers accepted medication from a friend, 3 porters took acetazolamide, 1 porter received medicine from a trekker, and one porter simply failed to take his medication.
Dropouts and completers: demography, smoking habits, and trial group*
AMS Incidence and Related Factors
Acute mountain sickness occurred in 13 (11.9%) of 109 porters who completed the trial, 7 porters taking acetazolamide and 6 porters taking placebo. The difference in AMS incidence between these 2 groups was not statistically significant, neither was the difference in AMS scores (acetazolamide: median, 1.0; range, 0–6; and interquartile range, 0–1.5 vs placebo: median, 1.0; range, 0–5; and interquartile range, 0–1.0).
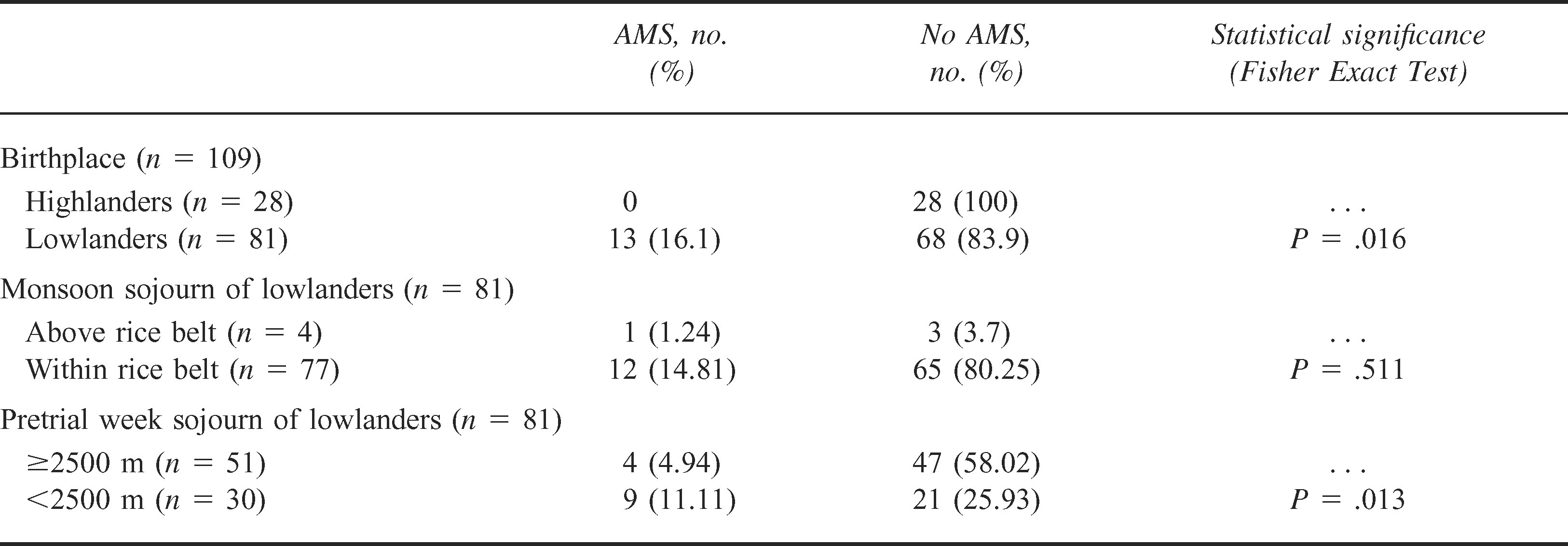
The most important factors affecting the risk of developing AMS were birthplace, location 1 week before the trial (Table 4), ascent rate, and, in particular, rest days (Table 5). Highlanders were minimally affected by altitude; AMS occurred only in lowlanders (13/81 lowlanders, 16.1%; P = .016, Fisher exact test). The median AMS score (1.0) was the same in both groups (range, 0–2; interquartile range, 0–1 in highlanders; and range, 0–6; interquartile range, 0–2 in lowlanders), although there was a marked difference in distribution. The data are represented in Figure 2.
Acute mountain sickness (AMS) incidence and related factors: birthplace and acclimatization
Acute mountain sickness (AMS) incidence and related factors: ascent rate and rest days in lowlanders

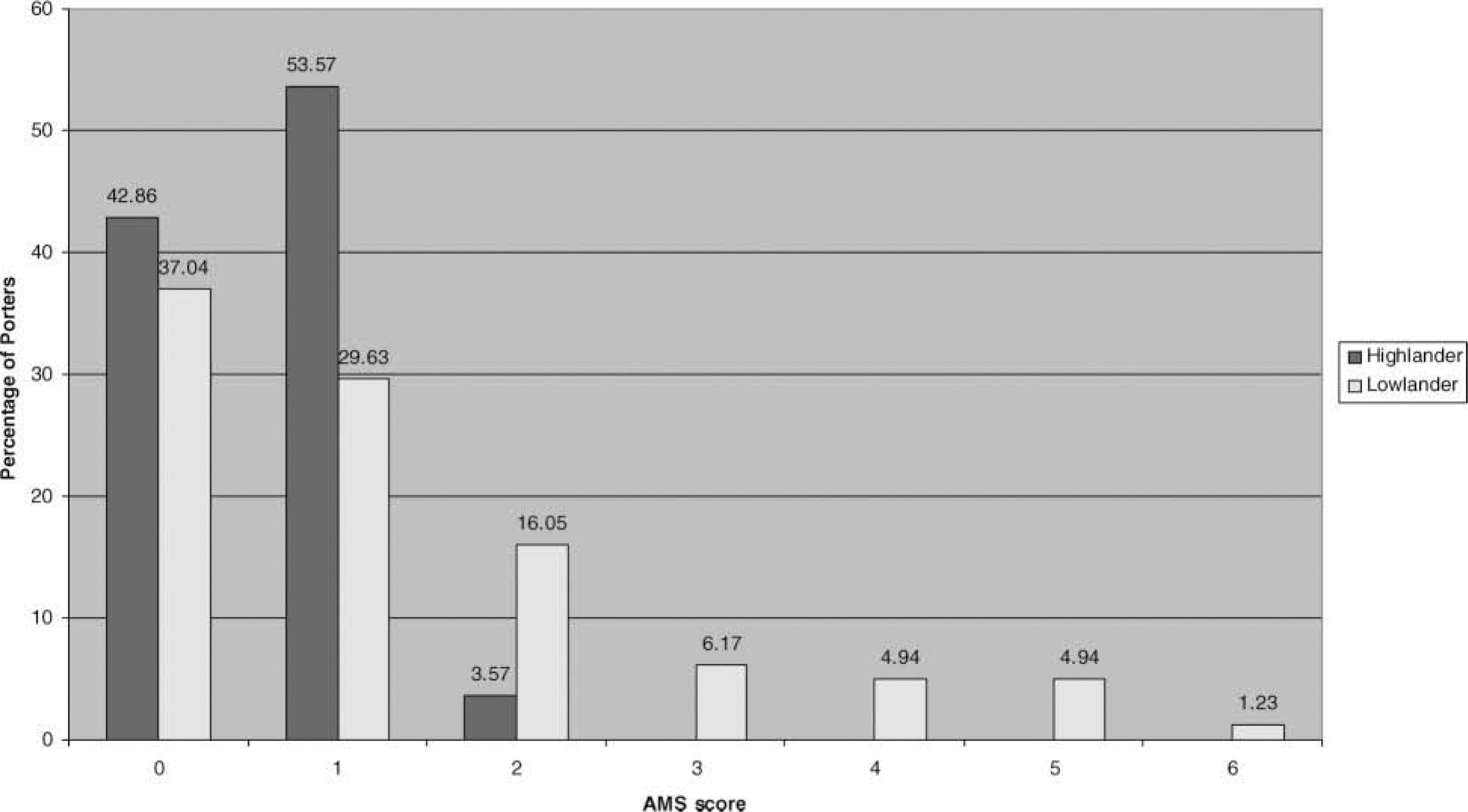
Acute mountain sickness (AMS) score in highlanders vs lowlanders.
Side Effects
Side effects, especially paresthesiae and numbness, were frequently reported (85.5% taking acetazolamide; 38.9% taking placebo). They caused exit from the trial in only 4 (3.7%) of 109 porters, who were all taking acetazolamide. Significantly more side effects occurred in porters taking acetazolamide than in those taking placebo (47 vs 21 porters; P = .0001, Fisher exact test). There were no serious side effects.
Discussion
We were unable to prove that acetazolamide prevents AMS in Nepali porters because the power of the study was seriously reduced both by the low incidence of AMS (13/109 porters, 11.9%) and the high dropout rate (275/400 porters, 68.8%). We chose a dosage of 250 mg acetazolamide daily, rather than the standard dosage of 500 to 750 mg,5,6 because Nepali porters are much lighter than Westerners. Indeed, a recent placebo-controlled trial showed that a dosage of 250 mg daily reduced the incidence of AMS by 50.6% in trekkers, 12 and a study reported in 2005 found that a dosage of 250 mg acetazolamide daily was as effective as 750 mg acetazolamide daily in preventing AMS in Western trekkers. 13 The low dosage used in our study seemed adequate, as shown by the systemic dose-dependent side effects of the drug (paresthesiae and diuresis). Side effects were frequent, but tolerable in the vast majority of porters. Only 3.7% of porters left the trial because of intolerable side effects, and there were no serious drug reactions. Although acetazolamide was tolerable, it does not seem to be a practical routine solution for preventing AMS in Nepali porters. Education of porters on the prevention and recognition of AMS, encouragement of employers and trek leaders to provide safer ascent schedules and adequate food, clothing, and shelter are all essential measures to protect this vulnerable group.
The low incidence and mildness of AMS surprised us, because our previous experience had led us to expect a higher incidence so early in the trekking season. 7 In trying to explain the unexpectedly low incidence of AMS, we found several contributory factors: porters’ birthplace, pretrial acclimatization, ascent rate, and rest days.
Although the porters’ origin determined their susceptibility to AMS (only lowlanders were affected), pretrial acclimatization was also important. Its benefit was most apparent in porters who had spent the week before the trial at at least 2500 m. When we arrived at Lukla (2820 m), it was clear that many porters were waiting there for days to get work; tourist numbers were reduced in 2001. No less than 31 of the 109 porters who completed the trial had been at Lukla in the week before the trial. Rest days and, to a lesser extent, a slower ascent rate, 14 made a major contribution to the low incidence of AMS.
It is possible that porters who thought they were at risk of AMS decided not to take part in the trial in case they lost work because of ill health; this factor could have reduced the incidence of AMS in the group studied. Conversely, it could be argued that the porters who did join the trial perceived the benefit of medical care during their trek, including the possibility of medication to prevent AMS. Another possible source of bias in the 109 porters completing the trial is that they were a compliant subgroup who stayed enrolled in the trial and might have been more likely to preacclimatize and to ascend more slowly. Some porters who dropped out may have done so because of AMS, and this might also help to explain the surprisingly low incidence of AMS in those who completed the trial.
We were disappointed by the high dropout rate (275/400 porters, 68.8%). Most of the porters who dropped out (153/400 porters, 40.75%) failed to report at one or two stations. The remaining porters (122/400 porters, 30.5%) missed all three assessment stations, therefore it was impossible to determine why they had dropped out. There was no difference in demography between porters who dropped out and those who completed the trial (Table 3). The doctors had communicated with all of the porters in the Nepali language and the porters had apparently understood the instructions that were given. The reasons for dropping out included trek route changes, difficulty in finding accommodation in villages, and active discouragement by sirdars (porter managers) from taking medication, despite our strenuous efforts to inform them of the trial at the outset. Perhaps some porters simply did not want to take the medication, despite having agreed to do so, and this may reflect a culture averse to taking medication. Some saw the possibility of commercial gain, because trial medication found its way into a local shop in Namche Bazaar and was on sale several months after the trial (S. Johnson, oral communication, July 27, 2002).
Conclusions
In conclusion, the high dropout rate and other irregularities indicate poor compliance. This suggests that routine acetazolamide prophylaxis in Nepali porters would be impractical even if it were desirable, although it may be helpful in some vulnerable individuals. However, we have identified lowland porters as a high-risk group, especially at the start of the trekking season. They should be targeted for nonpharmacological measures to prevent AMS, particularly a good trekking profile allowing adequate acclimatization.
Footnotes
Acknowledgments
We thank the Jerwood Foundation and the Sir Samuel Scott of Yews Trust for grants; and the Good Hope Hospital NHS Trust Charitable Fund, the Holy Trinity Parish Church, the Royal Sutton Fun Run, and many individuals for generous donations towards the funding of this study. We thank Wyeth Pharmaceutical Company, Maidenhead, Berks, UK, for supplying acetazolamide (Diamox) for the study; DH P Ltd, Abergavenny, Monmouthshire, UK for preparing the trial medication; and Dr Sarah Walters, Senior Clinical Lecturer in Public Health and Epidemiology and Honorary Consultant in Public Health Medicine, Birmingham University, for advice on statistics. We are grateful to Mr Doug Scott, CBE; Captain Tek; Mr Kinley Dendup; Mr Pukar Shrestha (via the Surgery in Nepal Association); Mr Pasang Temba Sherpa; and Mr Anu K. Lama of the Kathmandu Environmental Education Project for guidance when we planned the study. We are particularly indebted to Dr Buddha Basnyat, Medical Director, Himalayan Rescue Association and the Nepal International Clinic, Kathmandu, for his advice and support; and to Mr Pasang Tamang for helping to recruit porters participating in the study. We thank Angmaya Sherpa for allowing us to use Hotel Namche as a base for our study and we are grateful to the lodge owners at Tengboche, Somare, and Lobuche for their hospitality. P.H., Y.S., and Birmingham Medical Research Expeditionary Society members planned the study; A.P. recruited the Nepali doctors who participated in planning, acquired the data, and treated sick porters; and P.H. and Y.S. coordinated the study on the Everest trek. P.H., Y.S., D.S., and A.P. analyzed and interpreted the data and drafted the paper, with advice from Birmingham Medical Research Expeditionary Society members.
*
Reprints will not be available from the author.
1
The authors have no conflicting interests in this work.
