Abstract
Objective
Acclimatization at natural altitude effectively prevents acute mountain sickness (AMS). It is, however, unknown whether prevention of AMS is also possible by only sleeping in normobaric hypoxia.
Methods
In a placebo-controlled, double-blind study 76 healthy unacclimatized male subjects, aged 18 to 50 years, slept for 14 consecutive nights at either a fractional inspired oxygen (F
Results
Because of technical problems with the nitrogen generators, target altitude was not achieved in the tents and only 21 of 37 subjects slept at an average altitude considered sufficient for acclimatization (>2200 m; average, 2600 m). Therefore, in a subgroup analysis these subjects were compared with the 21 subjects of the control group with the lowest sleeping altitude. This analysis showed a significantly lower AMS-C score (0.38; 95% CI, 0.21 to 0.54) vs 1.10; 95% CI, 0.57 to 1.62; P = .04) and lower Lake Louise Score (3.1; 95% CI, 2.2 to 4.1 vs 5.1; 95% CI, 3.6 to 6.6; P = .07) for the treatment subgroup. The incidence of AMS defined as an AMS-C score greater than 0.70 was also significantly lower (14% vs 52%; P < .01).
Conclusions
Sleeping 14 consecutive nights in normobaric hypoxia (equivalent to 2600 m) reduced symptoms and incidence of AMS 4 days later on exposure to 4500 m.
Introduction
Ascent to altitudes above 2500 m frequently causes acute mountain sickness (AMS), a syndrome characterized by headache, nausea, dizziness, and insomnia, and ascent to higher altitudes can also occasionally cause potentially lethal high altitude pulmonary edema (HAPE) or high altitude cerebral edema (HACE). 1 The major determinants of the prevalence of these illnesses are altitude, individual susceptibility, rate of ascent, and degree of acclimatization caused by preceding exposures. 2 Slow ascent that would help to reduce severity and incidence of these illnesses is often not possible because of time constraints or financial reasons. Therefore, in a given setting of rapid ascent, preacclimatization or intake of drugs such as acetazolamide 3 or nifedipine 4 are often the only options to avoid or reduce the chance of experiencing AMS or HAPE.
There have been successful attempts to reduce AMS 5 and to improve the rate of ascent on climbing Mt Everest 6 by exposure to hypobaric hypoxia in the weeks preceding the altitude exposure. These studies, however, involved few subjects, were uncontrolled, and used procedures that interfere with regular daily activities. Furthermore, it was shown that staging ascent for 6 days at 2200 m reduces AMS severity at 4300 m by 44%, 7 but a placebo-controlled, double-blind study found only a minor, clinically irrelevant preventive effect on AMS at the same altitude after 8 days of sleeping 7.5 hours in normobaric hypoxia corresponding to an average altitude of 2600 m. 8
Exposure to hypoxia during sleep is an attractive possibility of acclimatization because it does not interfere with normal daily activities. It is likely that the short duration of total nights accounts for the mostly negative results in the study of Fulco et al.
8
Therefore, we hypothesized that sleeping 14 instead of 8 consecutive nights in a well-tolerated hypoxic environment at home using nitrogen-enriched air would significantly reduce severity and incidence of AMS most likely by inducing ventilatory acclimatization. This hypothesis was tested during a 20-hour exposure in a normobaric hypoxia room at an ambient P
Methods
Subjects and Study Design
We recruited 84 healthy, nonsmoking male subjects who did not take any medication and who had not stayed above 2000 m during the last 2 months before the study. Seventy-six of them were found eligible, agreed to participate, and were randomly assigned to one of the study groups. Three dropped out during the study and 73 finished the study protocol (Figure 1). They had a mean age of 26.5 years (range, 18 to 48 years) and a mean body mass index of 23.6 kg/m2 (range, 19.1 to 35.2 kg/m2). History of AMS was not assessable in all but 3 subjects because of lack of appropriate altitude exposures. Subjects were randomly assigned in blocks of 6 to normoxic or hypoxic treatment, which consisted of sleeping for 14 consecutive nights at home under a tent that was ventilated by either normoxic (control group) or hypoxic (treatment group) air. A few days before the intervention they were exposed in a hypoxia room for 4 hours at a fractional inspired oxygen (F
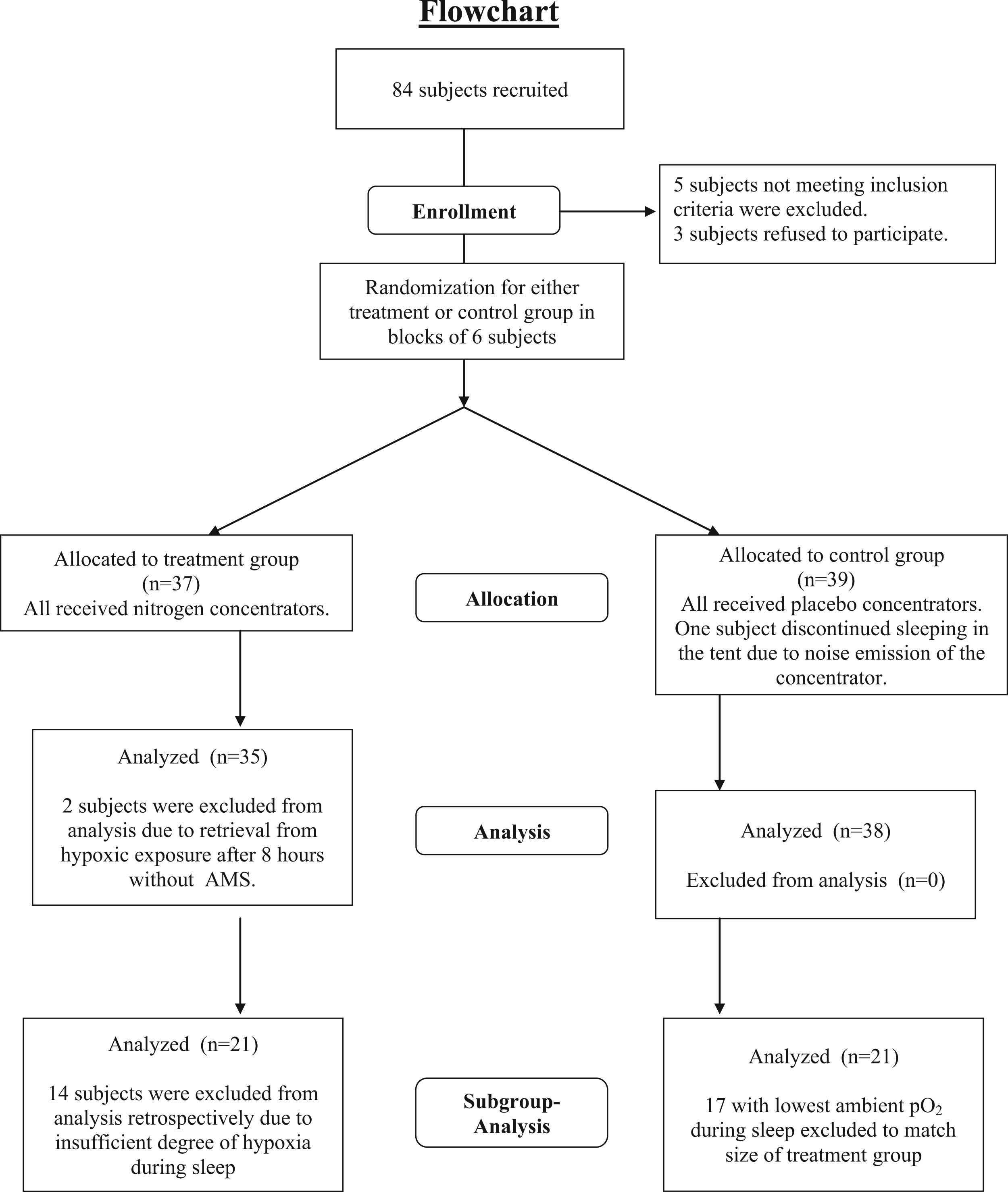
Cohort flow diagram. Flowchart of enrolment and randomization of the subjects and overview of data analysis.
The statistical power analysis yielded a sample size of 40 subjects. When analyzing the data of the first 40 subjects, we discovered that many subjects had not been exposed to the intended degree of hypoxia because of technical problems discussed in the section describing the devices. Avoiding the identified causes for failure to reach sufficient hypoxia, the study was repeated in another group of 40 subjects.
Measurements in the Hypoxia Room
Exposure to hypoxia took place in a laboratory unit located at 100 m above sea level equipped with 3 beds, a shower, and a toilet. Ambient P
AMS was assessed by the Lake Louise Score 9 and the AMS-C subscore of the Environmental Symptom Questionnaire. 10 Subjects were considered to have AMS when the AMS-C score was ≥0.7 and the Lake Louise Score (questionnaire and clinical examination) was ≥5. This criterion for AMS had been applied in a previous study, 11 the results of which were used for the power calculation of this investigation. When one score was borderline and the other fulfilled the criterion score, subjects were also classified as having AMS as shown in Table 1. The classification regarding questionable cases of AMS was performed before the examiners were unblinded. Sleep quality in the laboratory and at home was assessed by the question of the Lake Louise Score regarding sleep: 0 = slept as well as usual, 1 = did not sleep as well as usual, 2 = woke up many times, poor night’s sleep, 3 = could not sleep at all.
Individual data of all subjects
Individual data on acute mountain sickness (AMS) classification (0 = no AMS, 1 = AMS), Lake Louise (LL) score, AMS-C score, altitude equivalent of average hypoxic exposure (altitude), allocation to study group (Hx, hypoxia; Nx, normoxia), and group assignment for subgroup analysis.
Indicates termination of study after 16 hours.
Indicates termination of study after 8–10 hours.
“No record” means no or insufficient recording available.
Ventilation and end-tidal P
Exposure in Hypoxic Tents
Tents and oxygen generators (Integra TEN; Sequal, San Diego, CA) modified to produce nitrogen-enriched air were obtained from a company selling such devices to athletes for sleeping or training in hypoxia. Air and nitrogen flow, respectively, were regulated via O2 (KE 25; GS Yuasa Corporation, Kyoto, Japan) and CO2 sensors built in a control unit that was placed inside the tents. In 3 of 6 nitrogen concentrators (“placebo generators”), nitrogen supplementation was blocked and they always supplied room air independent of the selected altitude on the control panel. Subjects had no information regarding the level of hypoxia because the display on the control unit showing the ambient O2 concentration was hidden while the display of the altitude remained visible for selection of the altitude. Furthermore, a flowmeter on the generator showing the nitrogen flow to the tents was also covered by a metal plate. Subjects were asked to sleep for 8 hours each night, starting with an altitude of 2500 m (15.4% O2) and increasing the altitude every night by about 100 m (decrease O2 by 0.2%) until 3300 m (14% O2) was reached. This altitude was kept constant for the last 7 days, resulting in an overall average exposure of 3043 m per night. The O2 and CO2 signals were continuously registered on notebook computers and were used after the study to calculate the O2 and CO2 concentration in the tents throughout each night. In addition, oxygen saturation (Sp
Analysis of CO2 production in the tents revealed a good compliance regarding the time spent in the tents (7.4 ± 0.8 h/night), although the target F
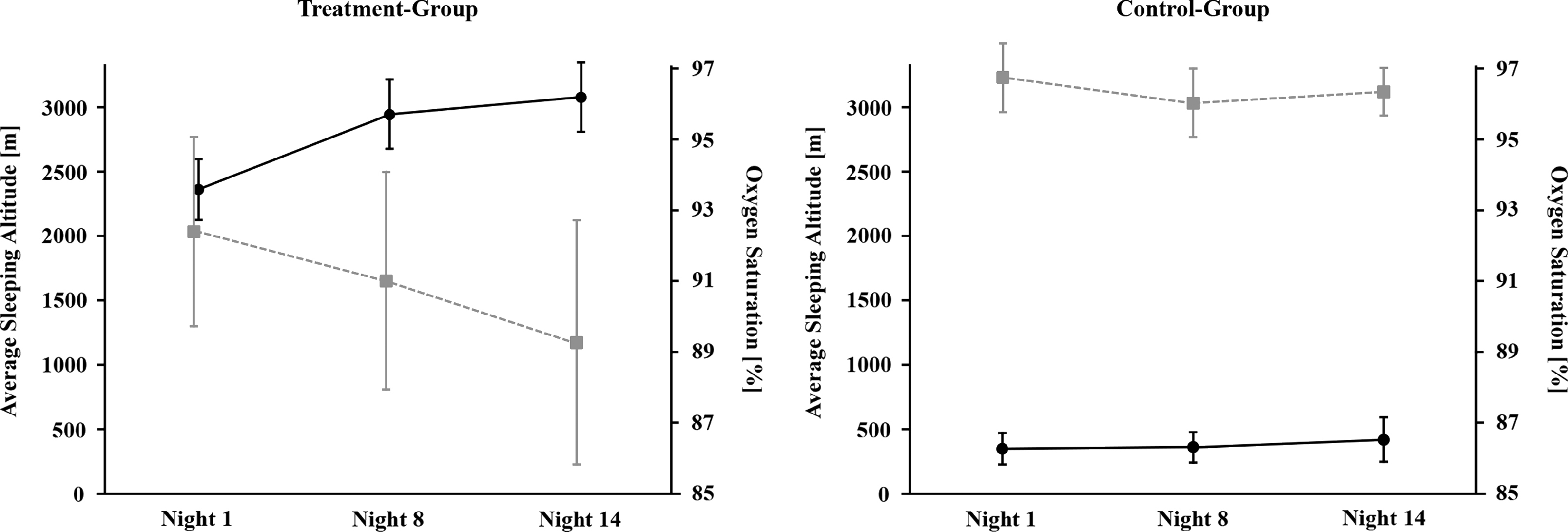
Average sleeping altitude ± SD (black circles) and average oxygen saturation ± SD (gray squares) during exposure in the tent for the treatment group (left) and the control group (right). Oxygen saturation decreases with increasing sleeping altitude in the treatment group, whereas in the control group it remains within normal range throughout the whole study.
Statistical Analysis
The calculation of the sample size was based on an estimated AMS prevalence of 50% found in a comparable study. 11 We calculated a sample size of 20 subjects per group for detecting a reduction of the incidence of AMS from 50% to 20% with a power of 80% (α = 0.05). The frequency of AMS between treatment groups is compared by χ2 test. Values between the groups with and without AMS are compared by Mann-Whitney U tests, and 2-sided probability values are reported. Values are reported as mean values and 95% CI, unless otherwise stated. Correlation analysis was performed by linear regression analysis (Pearson correlation). The level of statistical significance was set at P < .05 (2-sided) for all tests.
The aim of the study was to compare subjects who acclimatized while sleeping at approximately 3000 m for 2 weeks with subjects who slept close to sea level. Therefore, we also performed a subgroup analysis of those subjects who slept at an altitude that might induce acclimatization during a repeated exposure of 8 hours. A recent study demonstrated that first the symptoms attributable to hypoxia start occurring between 2134 m and 2438 m within 9 hours.
12
We selected 2134 m as a cutoff level for the treatment group, assuming that an altitude that causes symptoms attributable to hypoxia will also induce some altitude acclimatization during a similar duration exposure. Therefore, the results of the 21 subjects who slept on average above 2134 m (range, 2211 to 3291 m; mean, 2599 m) were compared with those of an equal-sized group who had slept at the highest inspiratory P
Results
Analysis of All Subjects
There was no significant difference in age, body mass index, and history of AMS between treatment groups. Individual data on exposure and AMS scores of all subjects are shown in Table 1. The overall incidence of AMS was 42%, without significant differences between treatment and control groups regarding the criterion score and the symptom scores. There was a trend to less AMS in those assigned to sleep in hypoxia, with 34% AMS (12 of 35 subjects) vs 50% (19 of 38 subjects) in the control group (P = .25). The respective symptom scores were 3.6 (95% CI, 2.8 to 4.5) and 4.9 (95% CI, 3.9 to 6.0; P = .13) for the Lake Louis Score and 0.50 (95% CI, 0.32 to 0.68) and 0.99 (95% CI, 0.62 to 1.36; P = .17) for the AMS-C score. Linear regression analysis between mean “altitude” of the hypoxic exposure and Lake Louise or AMS-C scores is statistically significant with P = .04 and P = .01, respectively, but yields only r2 values of 0.06 and 0.09, respectively, indicating that only 6% and 9% of the variability of the respective scores can be explained by differences in normobaric hypoxia during sleep. Exponential regression models did not result in better correlation coefficients.
It is well documented that residency above 900 m has some preventive effect on AMS. 13 The lack of a good correlation between the degree of hypoxia and AMS scores may be explained by a lack of acclimatization during the short exposure time (14 nights of 8 hours each) at intermediate altitudes between 1000 and 2000 m. Therefore, we also performed a subgroup analysis based on criteria that came close to the initial intention.
Subgroup Analysis
AMS scores of those 21 subjects who on average slept at an ambient P
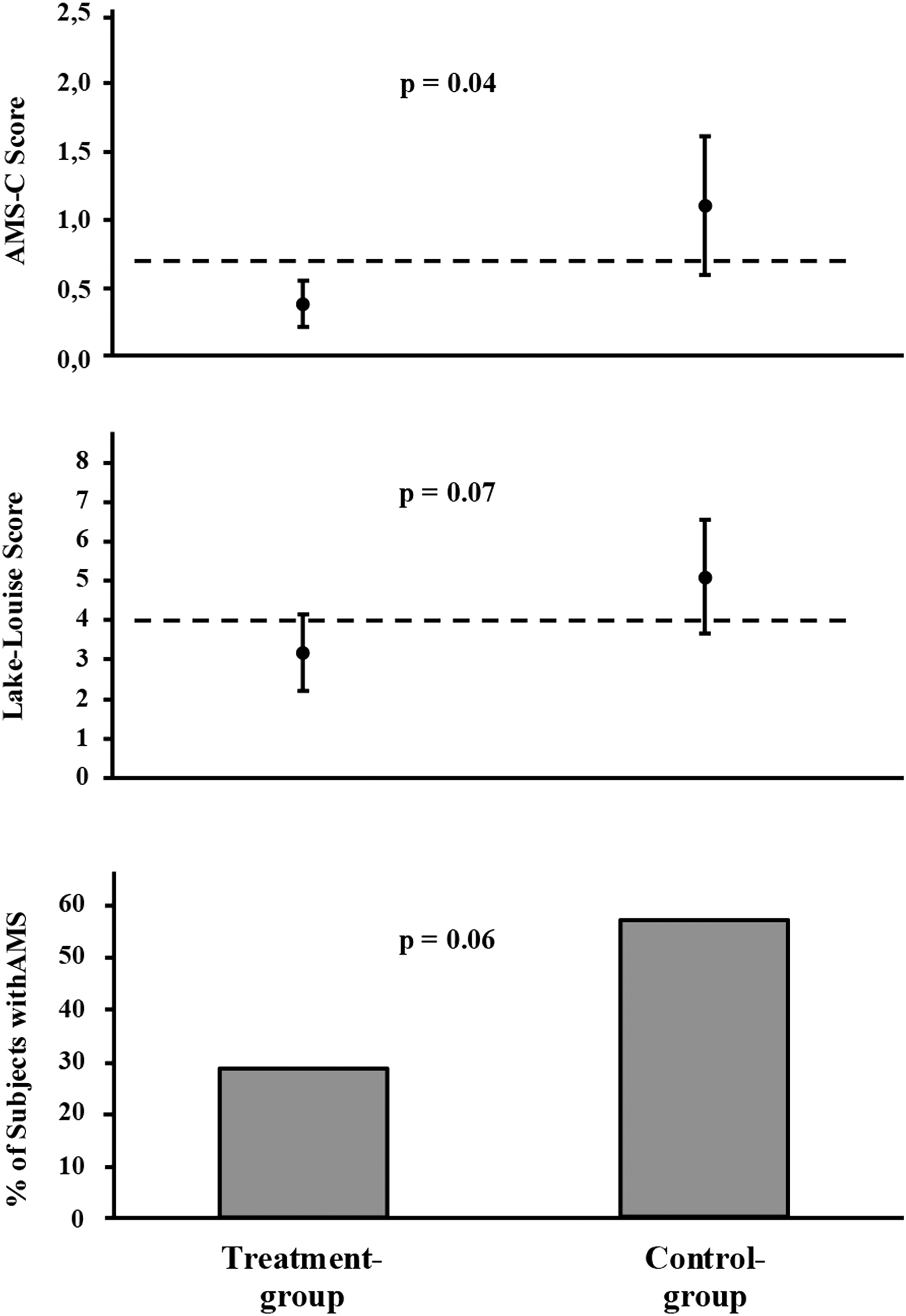
Mean values and 95% CI of AMS-C score (top) and Lake Louise score (middle) as well as incidence of acute mountain sickness (AMS; bottom) in 21 subjects of the treatment group and 21 subjects of the control group; dashed lines indicate cutoff level for diagnosis of acute mountain sickness with respective score.
To test whether sleeping in moderate hypoxia had an effect on ventilation and blood gases, we compared the data measured after 4 hours of exposure to 12% O2 before and after treatment. The results are shown in Table 2. There was no difference in ventilatory acclimatization between treatment groups. We only observed insignificant trends to higher Pa
Effect of treatment on blood gases and ventilation
Mean values and 95% CI of capillary blood gas analysis, pulse oximetry (Sp
Treatment group (n = 21), control group (n =21).
Blood gases and ventilation in relation to acute mountain sickness
Mean values ± 95% CI of capillary blood gas analysis, pulse oximetry (Sp
We further analyzed the sleep quality of the 42 subjects involved in the subgroup analysis. After the first 3 nights, sleep quality in the tent expressed as an average sleep score for days 4 to 14 was significantly impaired compared with a control night before the intervention in both groups. Mean scores in the treatment group were 0.43 (95% CI, 0.27 to 0.59) during vs 0.05 (95% CI, 0.00 to 0.14) before the treatment and 0.45 (95% CI, 0.22 to 0.67) vs 0.10 (95% CI, 0.00 to 0.22), respectively, in the control group. The data demonstrate that there was only a mild impairment of sleep that was the same in both groups during treatment. Furthermore, sleep score was considerably less than when sleeping at 12% O2, which resulted in scores of 1.35 (95% CI, 0.86 to 1.84) for the treatment group and 1.77 (95% CI, 1.39 to 2.15) for the control group.
Discussion
This randomized, placebo-controlled, double-blind study demonstrates that sleeping in moderate normobaric hypoxia equivalent to an average altitude of 2600 m for 14 consecutive nights significantly reduces the severity and the incidence of AMS assessed by AMS-C scores during a subsequent exposure to normobaric hypoxia (equivalent to 4500 m) that took place 4 days after the last night spent in hypoxia. Absence of AMS was associated with higher Sp
Although our conclusions are based on a subgroup analysis, we consider them valid because the selection of subjects for the subgroup analysis was based on criteria that are in accordance with the primary purpose of the study, which was to compare subjects who did and did not sleep at levels of hypoxia that have been shown to induce acclimatization and prevent AMS during a prolonged continuous stay at high altitude. Despite not reaching the intended average level of hypoxia (equivalent to an altitude of 3043 m), we demonstrated significant effects on AMS assessed by the AMS-C score. This score is used in many studies as the only parameter to assess AMS, 14 particularly in those looking at the effects of various forms of preexposure to hypoxia for prevention of AMS at Pikes Peak (4300 m). 15 The reduction of AMS assessed by the Lake Louise Score was of borderline significance. In a larger sample with greater statistical power, the differences in Lake Louise Scores would most likely become statistically significant because AMS-C and Lake Louise Score are closely related. 16
Our study provides further insight into earlier work on this issue and suggests that a longer duration of exposure might be necessary to induce a sufficient degree of acclimatization. An earlier double-blind, placebo-controlled study showed that sleeping 7 consecutive nights at normobaric hypoxia equivalent to 2220 to 3100 m had no significant overall effect on AMS assessed by AMS-C scores after rapid ascent to 4300 m despite improving Sp
Previous uncontrolled studies used more severe hypobaric hypoxia (4300 m and higher) that lasted 4 and more hours per day for effective acclimatization and prevention of AMS.5,6 Such a procedure is not compatible with a normal professional life and depends on the availability of hypobaric chambers. Our study shows that a preventive effect on AMS can be achieved by a degree and mode of application of normobaric hypoxia that does not interfere with daily activities and that is quite well tolerated. Interestingly, subjective judgment of sleep quality was similar between the intervention and control groups and indicated minimal disturbance, which can be attributed to increased humidity and temperature inside the tents as well as to the noise of the generator (about 30 dB). Subjects usually placed the generator in an adjacent room. One subject, however, dropped out of the study because he did not tolerate the noise.
We suspect that some subjects had not completely closed their tents because of the humidity and high temperatures and that the generators could not provide enough nitrogen to compensate for the leak. Thus, these subjects did not reach the intended simulated altitude and insufficient capacity of the generators may be a concern with this method, particularly with the use of bigger tents. Insufficient supply of nitrogen would, however, easily be detected outside a study setting because the display on the oxygen sensor inside the tent would be visible and indicate the oxygen concentration. Users should be told to let the generator run for 1 to 2 hours at the desired level of hypoxia before bedtime to check whether the desired altitude is reached inside the closed tent. Alternatively one could set up a continuous registration of P
The reduction in the incidence of AMS is comparable with the reduction reported from other studies using 500 to 750 mg of acetazolamide per day for prevention of AMS. 17 Because the effect of our intervention was tested in normobaric hypoxia, we cannot exclude that the outcome might be somewhat different with exposure to real high altitude, in which additional factors like hypobaria and exertion may play a role. We are confident with regard to hypobaria that its influence is minimal because we repeatedly 11 observed the same symptoms 18 with a comparable prevalence in normobaric hypoxia as we do at an altitude of 4559 m. 2 Exercise and exertion, which were not involved in our testing, may enhance AMS. 19 On the other hand, we exposed subjects within minutes to hypoxia equivalent to 4500 m, whereas mountaineers usually take a few days to reach this altitude, even on Kilimanjaro. Thus, the immediate exposure may compensate for the lack of exercise as an AMS risk factor in our setting.
Blood gas analysis and pulse oximetry shown in Table 3 demonstrates that subjects with AMS had a lower Sp
In summary, symptoms of AMS during acute exposure to hypoxia equivalent to an altitude of 4500 m are significantly reduced for at least 4 days after sleeping 14 consecutive nights in normobaric hypoxia equivalent to an altitude of 2600 m. Thus, acclimatizing at home during sleep may be an alternative for those who consider taking drugs for prevention of AMS when travelling to high altitude.
Footnotes
Acknowledgments
The authors would like to thank Mrs Ingrid Slater for assistance in preparing the manuscript and Mrs Martina Haselmayr, Mrs Xenia Goetzelmann, and Mrs Marion Becker for technical assistance.
☆
The study is registered on ClinicalTrial.gov with the title: “Prevention of Acute Mountain Sickness by Intermittent Hypoxia” (NCT 00559832).
