Abstract
Objective
The Lake Louise AMS Self-Report Score (LLSelf) is a commonly used, validated assessment of acute mountain sickness (AMS). We compared LLSelf and visual analog scales (VAS) to quantify AMS on Aconcagua (6962 m).
Methods
Prospective observational cohort study at Plaza de Mulas base camp (4365m), Aconcagua Provincial Park, Argentina. Volunteers climbing in January 2009 were enrolled at base camp and ascended at their own pace. They completed the LLSelf, an overall VAS [VAS(o)], and 5 individual VAS [VAS(i)] corresponding to the items of the LLSelf when symptoms were maximal. Composite VAS [VAS(c)] was calculated as the sum of the 5 VAS(i).
Results
A total of 127 volunteers consented to the study. Response rate was 52.0%. AMS occurred in 77.3% of volunteers, while 48.5% developed severe AMS. Median (interquartile range, IQR) LLSelf was 4 (3–7). Median (IQR) VAS(o) was 36mm (23–59). VAS(o) was linear and correlated with LLSelf: slope = 6.7 (95% CI: 4.4–9.0), intercept = 3.0 (95% CI: −10.0–16.1),
Conclusions
While both VAS(o) and VAS(c) for assessment of AMS appear to be linear with respect to LLSelf, the amount of scatter within the VAS is considerable. The LLSelf remains the gold standard for the diagnosis of AMS.
Keywords
Introduction
Acute mountain sickness (AMS) is a constellation of common symptoms including headache, nausea and vomiting, fatigue, dizziness or lightheadedness, and sleep disturbances in the setting of a recent ascent by an unacclimatized person. 1 The indistinctness and prevalence of these symptoms renders a standard, reproducible definition of AMS difficult. Because AMS may precede potentially fatal conditions such as high-altitude cerebral edema or high-altitude pulmonary edema, valid and reliable diagnosis of AMS is important. A tool that clinicians and researchers utilize to assess AMS is the Lake Louise AMS Symptom Score (LLS), which includes a self-reported questionnaire and a standardized clinical assessment.2,3 An abbreviated version of the LLS, the Lake Louise AMS Self-Report Score (LLSelf), has become the gold standard of assessing AMS by nonclinicians, including expedition leaders and commercial guides who often make decisions about a climber's ability to continue trekking. The LLSelf queries 5 symptoms: headache, nausea and vomiting, fatigue or weakness, dizziness or lightheadedness, and sleep. Each symptom is scored on a 4-grade ordinal scale from 0 (none or not present) to 3 (severe or incapacitating) for a combined minimal score of 0 and maximal score of 15.
The visual analog scale (VAS) has been suggested as a method for assessing AMS due to its use in other clinical settings. 4 The VAS is a 100-mm horizontal line that may be used to quantify symptoms in a continuous fashion from 0 to 100. It is easy to use and requires very little written language. There is a large body of literature on application of the VAS to medicine, and clinically significant measurements on the VAS are established for pain, nausea, fatigue, and sleep quality. 5 -8 These attributes suggest that the VAS may perform well for the evaluation of AMS.
Because of its simplicity, brevity, and lack of reliance on written language, a continuous measure of AMS such as the VAS may be easier to utilize and offer equivalent performance to a categorical measure such as the LLSelf. 4 The purpose of this study was to compare the VAS to the LLSelf on Aconcagua [6962 m (22,841 ft)], the highest mountain outside of Asia and one of the Seven summits. Our first objective was to determine if a single VAS for overall AMS [VAS(o)] correlates in a linear relationship with the LLSelf(o). Our second objective was to determine if the 5 individual VAS [VAS(i)], correlate in a linear relationship with the 5 items, LLSelf(i), that comprise the LLSelf. Our third objective was to determine if a composite VAS [VAS(c)], calculated as the sum of the five VAS(i), correlates in a linear relationship with the LLSelf(o). Our fourth objective was to determine if a single VAS(o) distance could accurately define severe AMS on Aconcagua.
Methods
Study Setting and Study Volunteers
Enrollment was conducted at Plaza de Mulas base camp [4365 m (14,321 ft)] on the normal route to the summit of Aconcagua [6962 m (22,841 ft)] in Aconcagua Provincial Park, Mendoza, Argentina. Data collection occurred concurrently with a prospective study of the predictive value of postexercise SpO2 following completion of a 6-minute walk test for the outcome of successfully summiting Aconcagua. 9 Inclusion criteria were adult climbers (age 18 years and older) who hiked the normal route of Aconcagua, checked in with park rangers at base camp during the study period, and spoke English, Spanish, French, or German. Park authorities require climbers to check in with park rangers upon arrival to base camp on ascent and upon exiting the park upon descent. For climbers who met the inclusion criteria and consented to participate, there were no exclusion criteria. Enrollment occurred during 9 nonconsecutive days from January 15, 2009 to January 31, 2009.
Data Collection
Two investigators (JDV, MPL) obtained informed consent, collected basic demographic information, and distributed study postcards when climbers checked in with base camp physicians. Study materials were available in English, Spanish, French, and German. Volunteers proceeded towards the summit at their own pace with instructions to complete the postcard on the day of the expedition that they felt worst. The postcard contained a LLSelf and multiple VAS (Figures 1a and 1b for the English version). Six different standard 100-mm VAS lines with the word “none” at the left end of the line and “severe” at the right end of the line were utilized in this study. There were no other words, marks, or divisions on the line. For the VAS(o), volunteers were instructed to: “Please put a single slash mark on the line at the place that best represents the magnitude of your overall altitude sickness on the day you were feeling worst.” For each of the 5 VAS(i) corresponding to the 5 individual components that comprise the LLSelf, volunteers were instructed to: “Please put a single slash mark on the line at the place that best represents the magnitude of each of the following symptoms (headache, gastrointestinal symptoms, fatigue, lightheadedness, or difficulty sleeping) on the day you were feeling worst.”
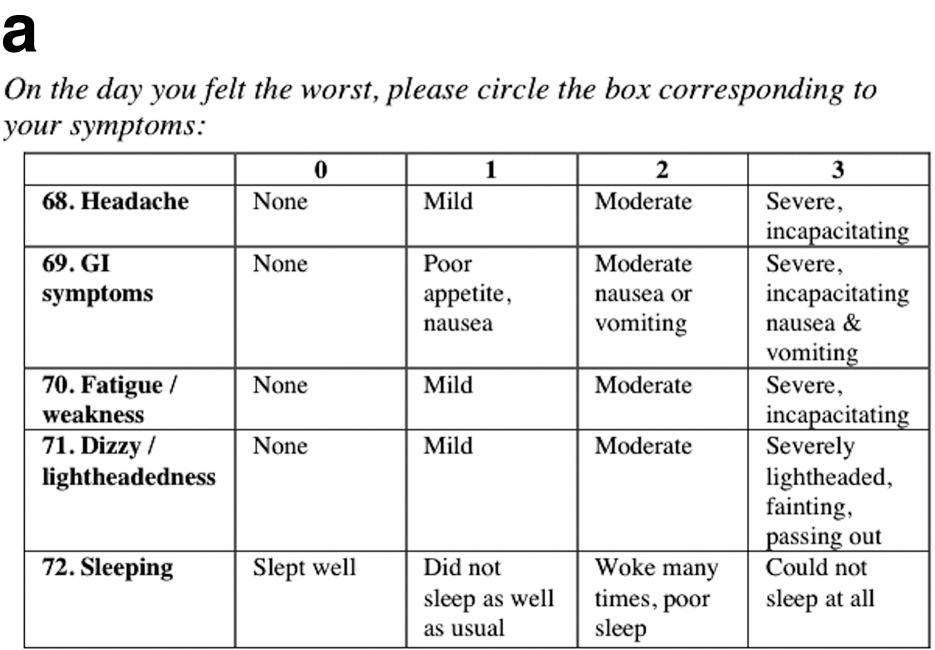
LLSelf component of the English version of the postcard used in the study.

VAS of the English version of the postcard used in the study.
Upon descent, volunteers deposited the postcards in drop boxes at base camp or the park entrance from January 15, 2009 through February 20, 2009. Alternatively, volunteers returned the postcards to the investigators via US mail.
Statistical Analysis
General descriptive statistics are presented as means (95% CI) for variables in which normality could be assured and medians (interquartile ranges, IQRs) for variables in which normality could not be assured. Two investigators (JDV, MPL) measured the distance in millimeters from the left end of the line to the slash mark. The investigators were blinded to each other's measurements, and interrater reliability was calculated using the intraclass correlation coefficient (ICC). A composite VAS, VAS(c), was created by summing the 5 VAS(i) for each volunteer. The scale of the VAS(c) is 0 to 500 mm. For ease of comparison with VAS(o), the VAS(c) data were normalized and are represented as percentages on a 0 to 100 scale. Upon analyzing the LLSelf data, normality could not be assured. Spearman's and Kendall's rank correlation tests (
Results
Volunteer Characteristics
One hundred twenty-seven volunteers consented to the study, of whom 66 (52.0%) returned postcards with outcome data. Not all postcards were completed entirely, so the number of VAS available for each comparison with LLSelf ranges from 45 to 56. Table 1 shows the baseline characteristics of the volunteers, who were predominantly male. LLSelf and VAS data are shown in Table 2. Median (IQR) LLSelf was 4 (3–7). Fifty-one volunteers (77.3%) developed AMS (LLSelf ≥ 3 and headache) while 32 volunteers (48.5%) developed severe AMS (LLSelf ≥ 5).
Baseline characteristics of the volunteers
LLSelf and VAS data
LLSelf, Lake Louise AMS Self-report Score; VAS, visual analog scale; VAS(o), VAS overall; VAS(c), VAS composite; VAS(i), VAS individual; IQR, interquartile range; ICC, intraclass correlation coefficient;
The number of available datapoints ranges from 45 to 66 because some postcards were only partially completed.
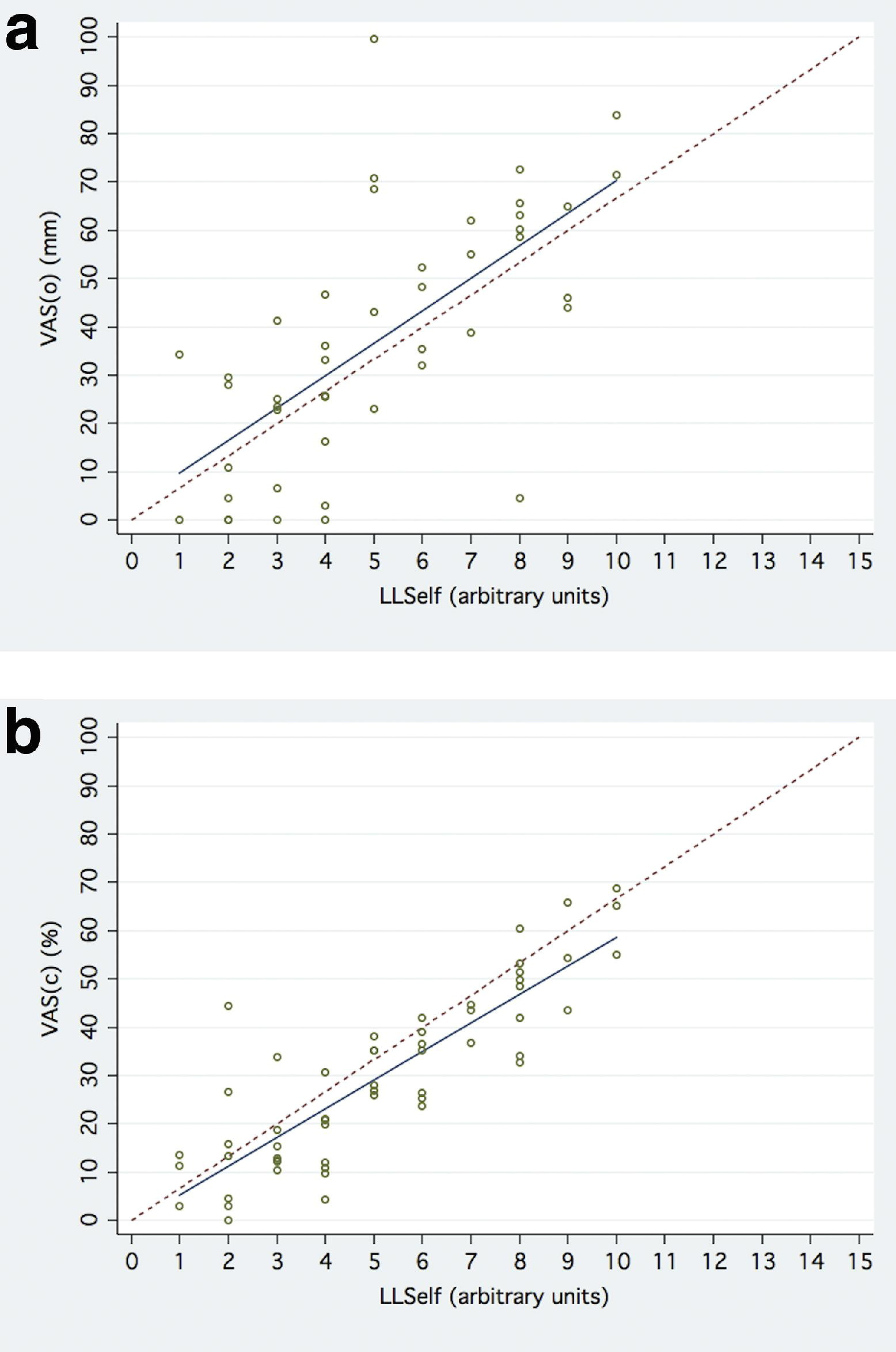
Relationship Between VAS(o), VAS(c), and LLSelf(o)
The relationships between VAS(o), VAS(c), and LLSelf(o) are depicted in Table 2 and graphically in Figures 2a and 2b. Median (IQR) VAS(o) was 36 mm (23–59). VAS(o) was significantly correlated to LLSelf(o):
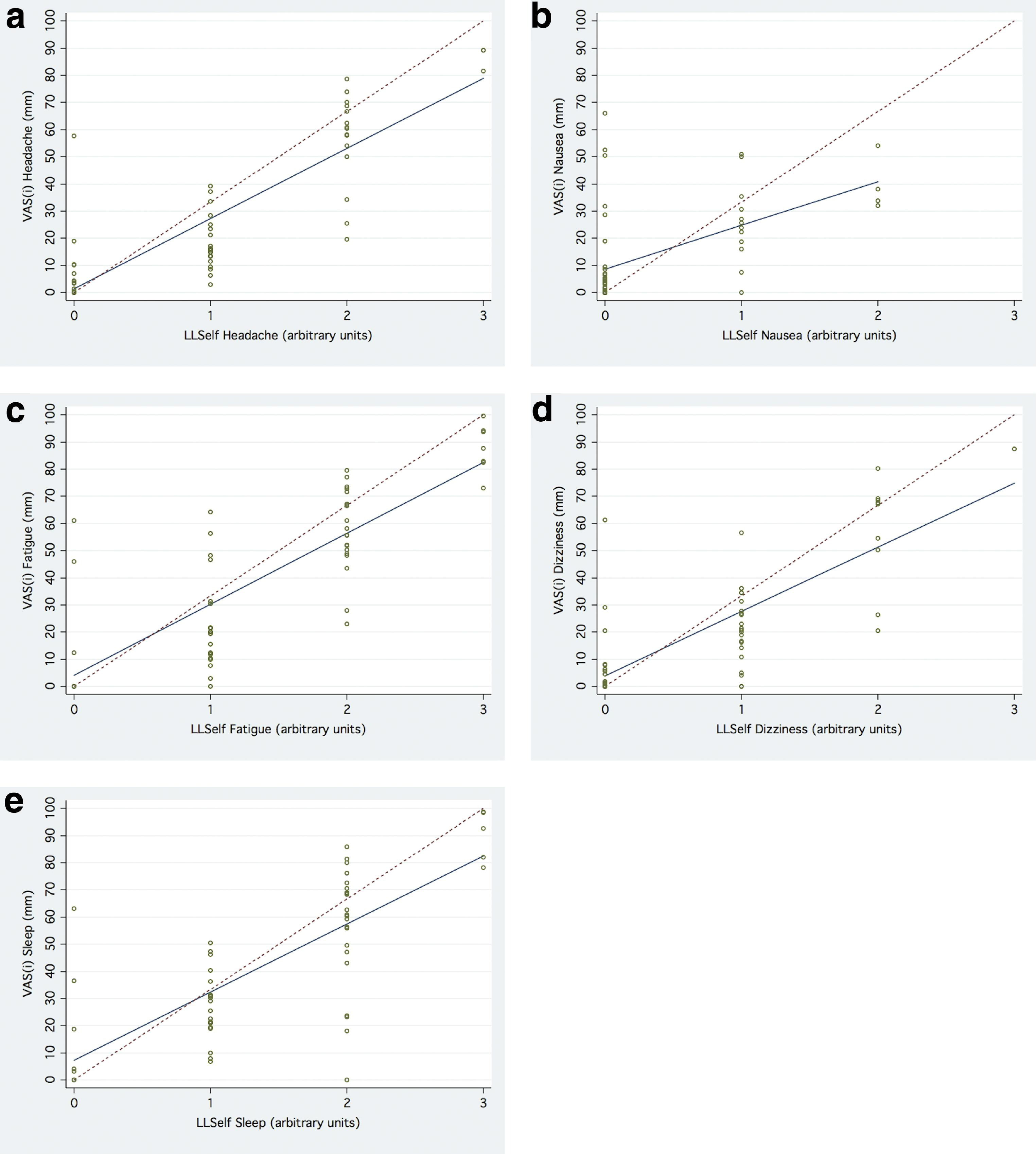
Relationship Between VAS(i) and LLSelf(i)
The relationships between VAS(i) and LLSelf(i) are depicted in Table 2 and Figures 3a–3e. All 5 VAS(i) were significantly correlated with their respective LLSelf(i). However, there was significantly more scatter observed with the VAS(i) than the VAS(o) and VAS(c). Additionally, the degree of correlation and linear scaling characteristics of the VAS(i) varied. For VAS(i)headache, VAS(i)fatigue, VAS(i)dizziness, and VAS(i)sleep, the 95% CI of the intercept included 0, and the upper limit of the 95% CI of the slope (29.3–32.0) approached the slope of the line of identity (33.3). VAS(i)nausea was least significantly correlated with LLSelf(i) and most distorted from the line of identity. Again there was substantial noise in the data, most pronounced in the range of VAS(i)sleep of 0 to 86 mm for the 24 volunteers with LLSelf(i)sleep = 2 (Figure 3e).
Determination of a VAS(o) Distance That Correlates to Presence of Severe AMS on Aconcagua
We were interested in establishing a VAS distance that corresponds to a particular LLSelf score. An ROC curve utilizing LLSelf ≥ 5 as the reference variable and VAS(o) distance as the classification variable was constructed. The area under the curve was 0.92 (95% CI: 0.79, 0.98). An overall VAS distance between 35 and 43 mm correctly classified 86.7% of climbers with a LLSelf ≥ 5, yielding a sensitivity of 78.3 to 87.0%, a specificity of 86.4 to 95.5%, a positive likelihood ratio of 6.38 to 17.3, and a negative likelihood ratio of 0.15 to 0.23. This is depicted graphically in Figure 4.
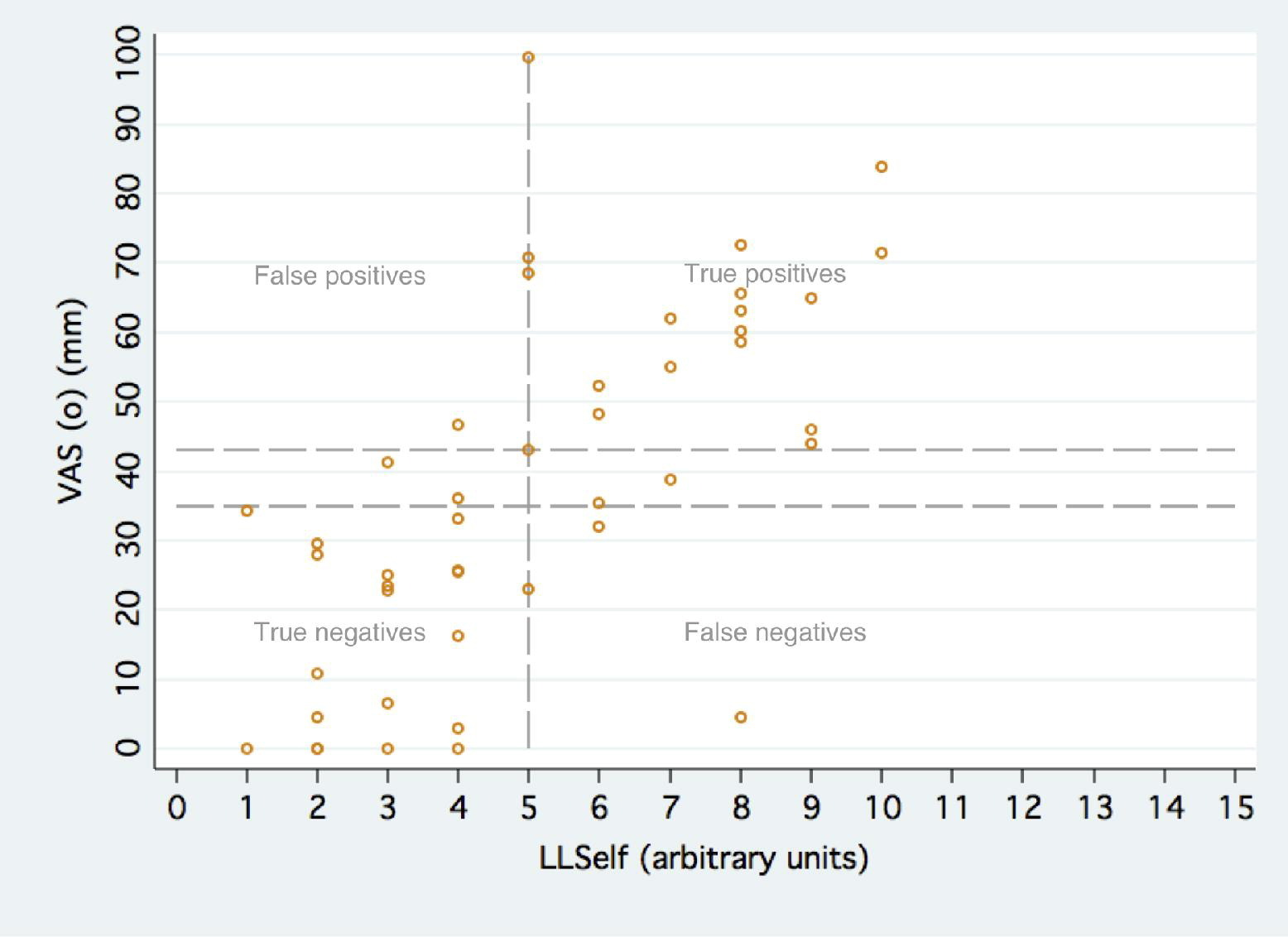
Graph demonstrating the accuracy of a single VAS(o) distance of 35 to 43 mm for the classification of climbers with severe AMS. Vertical dashed line is through LLS = 5, indicating the presence of severe AMS. Horizontal dashed lines at VAS = 35 and VAS = 43 represent the values that correctly classified 86.7% of volunteers with severe AMS.
Discussion
This is the first study to graphically report itemized VAS that correspond to each of the 5 LLSelf components and the first study to present the concept of a composite VAS. This study was performed on Aconcagua [6962 m ([22,841 ft)], 1 of the Seven summits and the highest mountain in the world outside of Asia. As such, the median LLS was 4, over three-quarters of the subjects developed AMS, and nearly half of the subjects developed severe AMS. Both overall VAS and composite VAS correlated with LLSelf in a linear manner.
The VAS has been used extensively in the medical literature as a measure for evaluating headache, nausea, fatigue, dizziness, and sleep, which comprise the 5 components of the LLSelf score.
5
-8 Indeed, studies have characterized clinically significant changes in VAS for 4 of the 5 symptom components of the LLSelf that have been studied.
6
,7,12,13 The VAS has only recently been applied to high-altitude medicine. Ours is the third field study evaluating VAS for assessing AMS, although experts in high-altitude medicine have encouraged consideration of its use in additional studies.
4
Wagner and colleagues performed the initial study describing the use of VAS for assessment of AMS.
14
In their study of 356 climbers on Mt. Whitney (4419 m), mean LLSelf was 2.8, mean overall VAS was 14.4 mm, and the two were reasonably correlated (
There are some similarities and multiple key differences among the present study and these prior studies. First, the correlation of VAS(o) to LLSelf has been moderate to high in all three studies, ranging from 0.65 14 to 0.84,14,15 despite differences in study settings and spoken languages. Second, there was at least one critical methodological difference among the studies. Both the present study and the Wagner study utilized a standard 100-mm VAS, whereas Kayser utilized an 85-mm scale for VAS(o) and a 40-mm scale for VAS(i). One might expect a 100-mm continuous scale to be more sensitive to small change than an ordinal scale of 0 to 15. The use of 40- and 85-mm scales complicates this comparison. Third, the sample size of the studies varies significantly. In our study and Wagner's, single measurements of VAS and LLSelf were obtained from 66 and 356 volunteers, respectively. In Kayser's study, 168 datapoints were obtained from a smaller sample of 14 volunteers who repeated the VAS(o), VAS(i), and LLSelf 12 times each over the course of 1 week. There may have been a learning bias to completing a VAS that the volunteers in our study and Wagner's did not experience. Fourth, Kayser noted that the linearity of the relationship between VAS and LLSelf in their dataset was distorted, such that there is a threshold effect for both the VAS(o) and VAS(i). They noted that at LLSelf < 5, the VAS tended to underreport AMS, whereas at LLSelf > 5, the VAS tended to overreport AMS. They concluded that the VAS(o) and VAS(i) for assessment of AMS is not a linear scale. Our data are mixed. VAS(o) and VAS(c) are clearly linear with respect to LLSelf. On the other hand, VAS(i)nausea is clearly nonlinear, exhibiting the same threshold effect described by Kayser. The relationship between VAS(i) headache, fatigue, dizziness, and sleep and their respective LLSelf(i) is less clear. The 95% CI of the slopes of their regressions lines approach the slope of the line of identity. Whether these VAS(i) truly exhibit the same threshold effect that Kayser identified or if our study was underpowered to detect the linearity of the relationship between VAS and LLSelf (which had been assumed and was first questioned after our study was concluded) remains to be seen.
The role of VAS in the assessment of AMS remains unclear at this point. The theoretical benefits are a continuous scale that is less reliant on written language with established clinically significant differences for most of the symptoms that comprise the LLSelf. Results from this study and the three additional studies of VAS in high altitude medicine are mixed. Careful analysis of the data from all studies illustrates the significant scatter of VAS distances for a particular LLSelf score. Some degree of scatter is to be expected when comparing a 4-grade categorical scale with a 100-mm continuous scale. One would expect unidirectional scatter at the endpoints 0 and 3, and bidirectional scatter at the internal points 1 and 2. The varying degree of scatter (accuracy), the number of points in each dataset (sample size), and the relative position of the points on the scale (prevalence of AMS) ultimately affect the linearity of the relationship between VAS and LLSelf. In Kayser's study, 15 a small sample size and lower prevalence of AMS resulted in a nonlinear relationship. Wagner 14 had a large sample size and low prevalence of AMS and did not comment on linearity. Our study is of moderate sample size and higher prevalence of AMS. An appropriately-powered study on a mountain with a significant prevalence of AMS that analyzes both VAS(o) and VAS(i) is needed. Ideally, this study would be conducted in multiple languages to determine if linearity exists or varies among languages or literacy rate.17,18 For external validity, we recommend all future studies of VAS for assessment of AMS utilize standard 100-mm scales. If proven linear, the utilization of serial VAS measurements to follow the course of AMS over time in the same manner that serial LLSelf assessments are completed seems reasonable. Nevertheless, at the present time the LLSelf remains the gold standard for the assessment of AMS.
Our study is not without certain limitations. First, our response rate was 53.2%, and a number of our responses were incomplete. Although less than ideal, this response rate is in line with other field studies of AMS. 17 ,19,20 Second, due to high prevalence of AMS on Aconcagua, our data may be not be representative on peaks at less extreme altitude where the prevalence of AMS is lower. Third, although we intended to directly compare VAS performed in multiple languages, our response rate precluded meaningful subgroup analysis. Fourth, utilization of the VAS is dependent upon human measurement. To control for this in our study, two investigators independently measured distances. The intraclass correlation was > 0.99 for all measurements, which is consistent with past studies. 14 Finally, at least one study suggests that utility of serial VAS measurements may be unreliable for absolute initial measurements of < 37.6 mm. 21
In summary, we have demonstrated that both overall and composite visual analog scales correlate with Lake Louise AMS Self-Report Score for assessment of acute mountain sickness on Aconcagua. We have also presented data for individual visual analog scales corresponding to the 5 symptoms that comprise the Lake Louise Score. While both overall and composite visual analog scales for assessment of acute mountain sickness appear to be linear with respect to LLSelf, the amount of scatter within the VAS is considerable. The LLSelf remains the gold standard for the diagnosis of AMS at present.
Competing financial interests
The authors have no conflicting financial interests to disclose.
Ethical approval
The Institutional Review Board of Northwestern University granted approval of human subjects research (IRB#2863-001), and all volunteers signed a document of informed consent. The Department of Natural Resources of the Mendoza Provincial Government granted the investigators permission to conduct the study (Resolución #836).
Footnotes
Presented at Wilderness Medical Society Medical Conference and Annual Meeting, Snowmass, CO, July 2009.
Acknowledgments
The authors wish to thank Heber Orona, Roel Quique Apolinario Villafán, Malena Giglio, and the staff of Aconcagua Trek for their expedition support; the base camp physicians and park rangers of Aconcagua Provincial Park for their time, equipment, and cooperation; Marisa Chumil and Silvia Palmerin for their Spanish translations; François Blumenfeld and Eliana Vagalau for their French translations; Kristina Ogilvie and Misha Kovacevic for their German translations; and James Harris of Orbital Technologies Corporation for constructing the drop-boxes.
