Abstract
Borrelia hermsii causes tick-borne relapsing fever (TBRF) in the Pacific Northwest. There are few reports of TBRF-associated acute respiratory distress syndrome. One such unusually severe case is described. Literature is reviewed including diagnosis, treatment, and prevention.
Keywords
Introduction
Tick-borne infections in the United States include diseases caused by bacteria, viruses, rickettsiae, and protozoa. Different ticks transmit different disease-causing agents, and some ticks can transmit multiple organisms. There are 2 families of ticks: Argasidae (soft ticks) and Ixodidae (hard ticks). There are many tick-borne diseases common in the United States and worldwide. Therefore, a doctor must obtain a detailed travel history when tick-borne disease is suspected. In the Inland Northwest, 2 tick-borne diseases are commonly seen: relapsing fever and tick paralysis. 1 The spectrum of illness in relapsing fever ranges from a mild, self-limiting illness to more severe cases. However, respiratory failure from acute respiratory distress syndrome (ARDS) is rare. This article provides a case report of a patient who survived ARDS associated with confirmed, unusually severe tick-borne relapsing fever (TBRF) transmitted under unusual circumstances. Neuroborrelosis resulted in hearing loss in this patient.
Case report
A hematologist admitted P.D., a 59-year-old healthy female, for the second time. She had been hospitalized several weeks previously for what appeared to be a virally induced thrombocytopenia. Her platelets were 19 000/mm3, her white count 4500/mm3, and her hemoglobin 12.6 g·dL−1. At that time her symptoms included a headache without fever or rash. She had a negative magnetic resonance imaging and magnetic resonance angiogram of the brain. Her bone marrow was consistent with idiopathic thrombocytopenic purpura. She was treated with prednisone and her platelet count rose to her pre-illness baseline of 245 000/mm3. At her follow-up hematology visit 2 weeks later, her temperature was 39° C, she had intense thirst and mild confusion and was unable to ambulate as a result of muscle pain, and she had pancytopenia (white count 1300/mm3, hemoglobin 8.9 g·dL−1, and a platelet count of 11 000/mm3). She had an elevated alkaline phosphatase (178 u·L−1), mild hypertransaminasemia (aspartate transaminase 128 u·L−1, alanine transaminase 156 u·L−1), and an elevated aldolase (45.6 u·L−1). Other labs were normal. The hematologist repeated a bone marrow biopsy prior to transfer to the hospital, and I was consulted at that time.
The patient had no history of camping, sleeping in rustic cabins, or tick exposure. She did comment about recent problems with increases in the chipmunk population in her upscale, forested neighborhood and had been clearing knapweed near her home. Another member of her household had had a brief febrile illness at about the time of her original hospitalization but had recovered without sequelae. Shortly after returning to Spokane, WA, from a vacation in Nevada, her former neighbor was hospitalized with fever and a severe respiratory illness requiring mechanical ventilation and antibiotics, but no infecting organism was found. Relapsing fever had not been considered in that case. The patient had traveled to the Galapagos 3 months prior to her illness, but no one else on that trip had become ill. She had also traveled to Africa 3 years before but had been healthy until this illness.
Physical exam was normal except for a temperature of 39°C and the presence of mild confusion and mild splenomegaly. A number of diagnostic tests, including peripheral smear for Borrelia, were done. The smear was packed with Borrelia, as was the bone marrow, which had been done immediately prior to admission (Figures 1 and 2). Re-review of labs and marrow from her previous admission showed no spirochetes. No evidence of malaria was seen. The patient was started on doxycycline and ceftriaxone, and she did well, with no Jarisch-Herxheimer reaction (JHR). Steroids were discontinued. Temperature, liver functions, and blood counts improved. The Spokane County Health Department and the Centers for Disease Control and Prevention (CDC) performed serologies that were consistent with at least several weeks of antibody response to B hermsii, as evidenced by enzyme immunoassay testing and by a very strong immunoglobulin M profile and mounting immunoglobulin G profile in Western immunoblots. Polymerase chain reaction amplification of TBRF targets from DNA extracted from the blood smear was successful and is currently being sequenced for genetic characterization and species identification by the CDC. Admission blood cultures and serologies for Rocky Mountain spotted fever, ehrlichiosis, cytomegalovirus, Epstein-Barr virus, parvovirus, and human immunodeficiency virus were all negative.
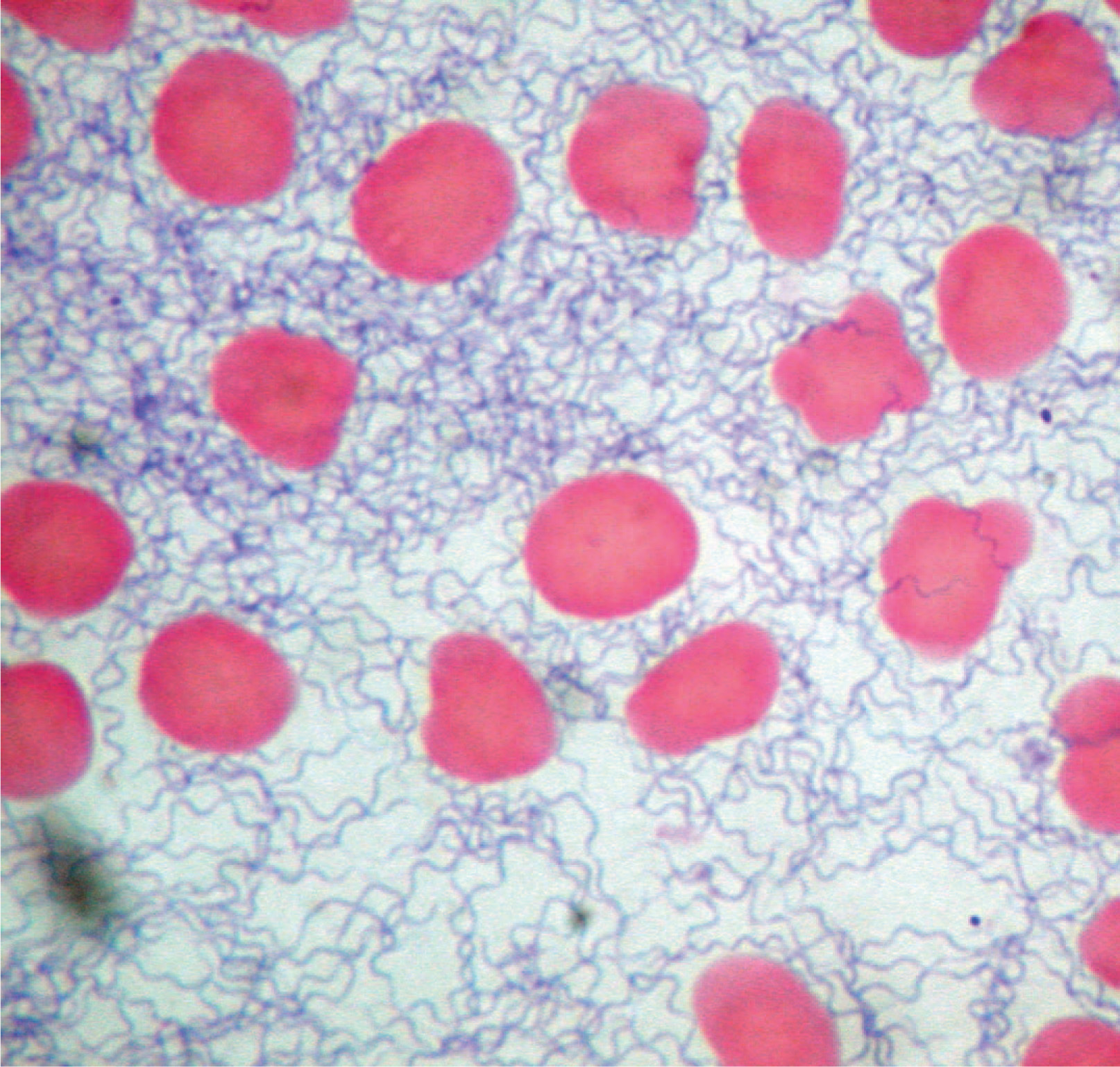
Peripheral smear done on admission to hospital revealing heavy spirochete load.
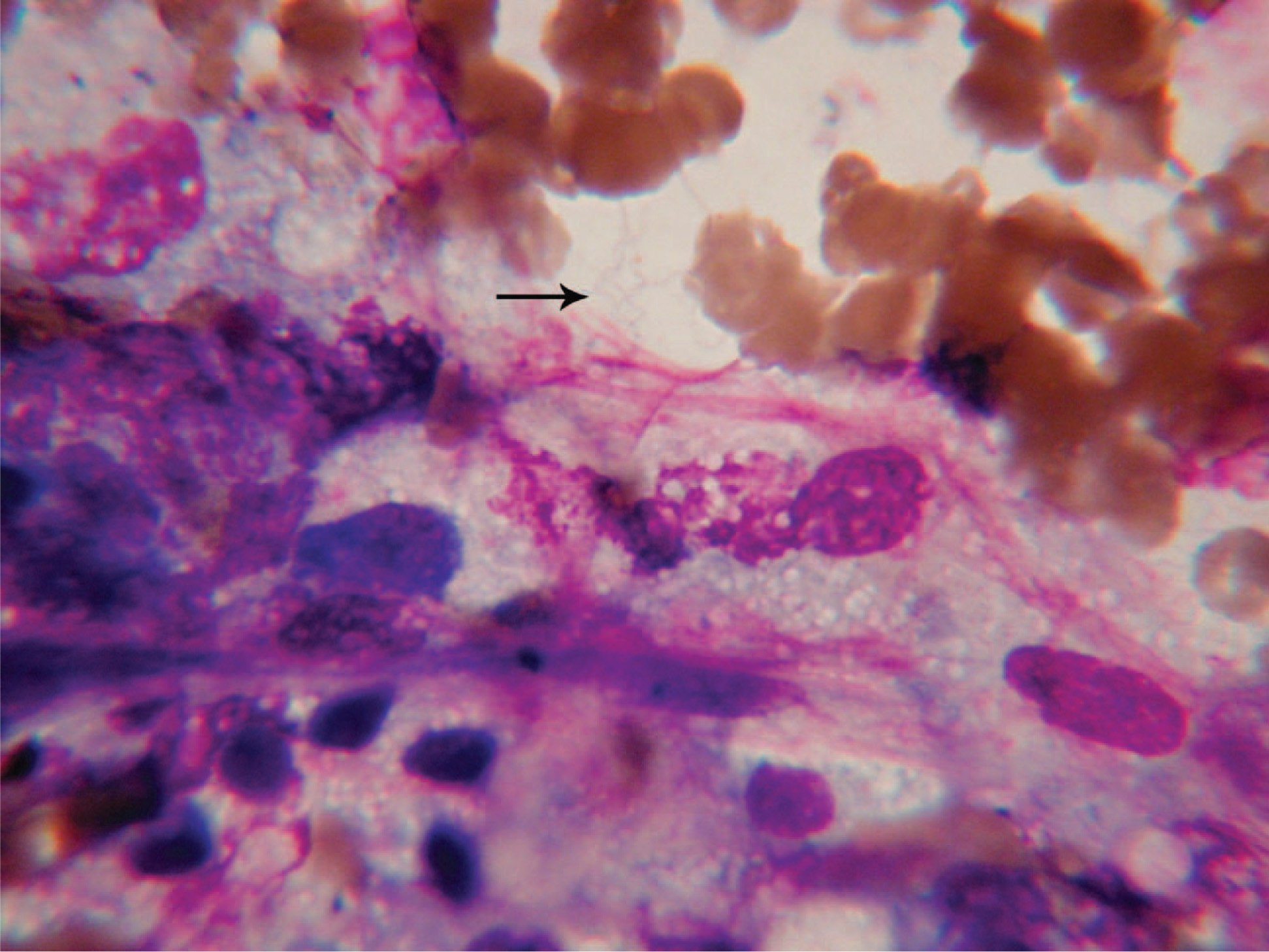
Bone marrow biopsy obtained prior to hospital admission, demonstrating multiple spirochetes shown by arrow.
Seventy-six hours after initial antibiotic therapy the patient developed acute hypoxemic respiratory distress with pulmonary edema confirmed on chest radiograph (Figure 3). An echocardiogram showed an ejection fraction of less than 20% and moderate tricuspid regurgitation with a pulmonary artery pressure of 59 mm Hg. There were no electrocardiogram or enzyme changes to indicate infarct or myocarditis. The left atrial dimension was normal at 3.8 cm. Diastolic dysfunction could not be evaluated as a result of tachycardia. Pulmonary vein flow was not recorded. The patient remained slightly hypertensive, with a blood pressure of 140/90 mm Hg. Despite a rapid spontaneous return of ejection fraction to 35%, she developed worsening interstitial pulmonary edema, and pulmonary and cardiovascular services concurred that she had ARDS. She had Borrelia sepsis as a risk factor for developing ARDS, along with bilateral infiltrates on the chest radiograph. Arterial blood gases on 100% FIO2 showed a pH of 7.49, a PaO2 of 57 mm, and a partial pressure of arterial oxygen to fraction of inspired oxygen ratio of 57. A pulmonary artery catheter was not placed because of thrombocytopenia (platelets 15 000/mcL). She required mechanical ventilation with positive end expiratory pressure to correct hypoxemia and compliance. No vasopressors were needed. Ventilator support was no longer necessary after 4 days, and the patient was rapidly weaned off oxygen. She also developed mild transient disseminated intravascular coagulation, recurrent pancytopenia, and more liver function abnormalities (multisystem organ failure), despite normotension. She later also developed hyponatremia (falling from 141 to 117 mm·L−1 in 12 hours) with associated mental deterioration. A computed tomography scan of her brain was normal, and she was treated with normal saline and flurinef, and again she improved. Repeated smears for Borrelia were negative. Ceftriaxone was ultimately discontinued after 14 days. She was discharged home on doxycycline with a normal physical exam, normal chest radiograph, normal labs, an ejection fraction of 55%, and pulmonary artery pressure of 36 mm Hg. Pulmonary function testing was done at 5 months postdischarge. The forced vital capacity, forced expiratory volume in one second, forced expiratory volume in one second to forced vital capacity ratio, forced expiratory flow 25% to 75%, airway resistance, lung volumes, and diffusing capacity were normal. After discharge, she showed unilateral neurosensory hearing loss as her only sequela. Otorhinolaryngologists are following her, but they feel this loss is likely permanent.
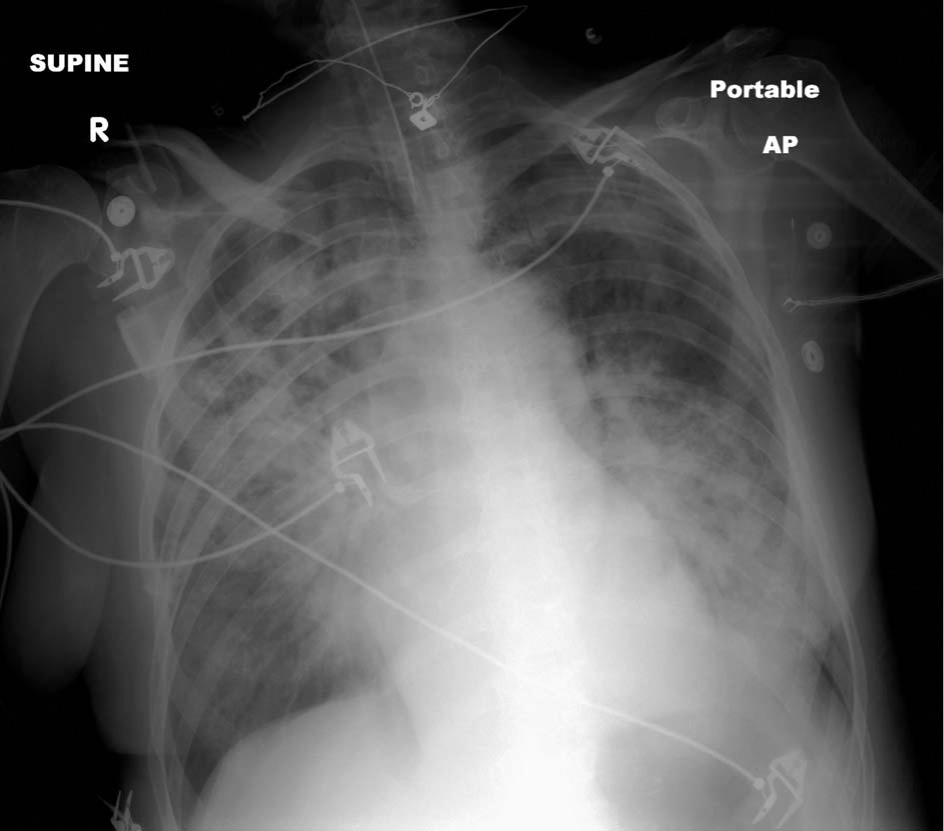
Chest radiograph with bilateral pulmonary infiltrates, done after intubation.
Discussion
There are 2 main forms of relapsing fevers: epidemic or louse-borne relapsing fever and endemic or TBRF. The body louse transmits louse-borne relapsing fever from human to human. Tick-borne relapsing fever is transmitted to humans through the bite of infected soft ticks from rodents and other animals. 2 Tick-borne relapsing fever is found throughout most of the world, although endemic areas include Colorado, California, the Pacific Northwest, southern British Columbia, 3 –8 plateau regions of Central and South America, central Asia, Mediterranean countries, and most of Africa. An outbreak of TBRF caused by B hermsii, and near Browne's Mountain in Spokane, WA, was considered the largest outbreak of TBRF reported in the western hemisphere. 9 Nearby chipmunk and vole nests in rotten logs and stumps contained ticks harboring the disease. Studies have shown that pine squirrels, chipmunks, and meadow voles experience prolonged spirochetemia. 10 This patient had been complaining about increased chipmunk populations following construction near her home. Ornithodoros (soft) ticks are obligate blood feeders; they maintain continuous Borrelia infections in salivary gland tissue and transmit spirochetes when feeding. 11 These ticks may survive for 20 years without feeding and may remain infectious for up to 10 years following infection. Infected ticks pass the spirochetes to their offspring. Unlike hard ticks, which may remain attached for several days, soft ticks remain attached for only 5 to 20 minutes while feeding, frequently at night, and they lack a painful bite. Most people are unaware of soft tick bites. Infection may result from the bite or from contamination of the wound or skin by tick secretions. 12 Tick-borne relapsing fever is more common in men because of their more frequent exposure. Most recent cases and outbreaks have occurred in rustic cabin or vacation home settings at elevations greater than 1200 m in coniferous forests in the western United States. 3 –7 Tick-borne relapsing fever normally occurs in summer months. It can, however, occur in winter, particularly when people go into rodent-infested cabins and start fires that warm the cabin and produce the carbon dioxide and heat that attract the ticks. The true incidence of TBRF in the United States is unknown because it is not a nationally reportable disease. Each year about 25 cases are reported to the CDC. Fifty-eight cases were documented in Washington State between 1980 and 1993. 8 Since then, there have been about 5 to 8 cases a year in Washington. In 2007 there were 4 cases, including this one. 1
Relapsing fever Borrelia are gram-negative helical bacteria that normally measure 0.2 to 0.5 μm in width and 5 to 20 μm in length. They are visible with light microscopy and have the corkscrew shape typical of all spirochetes. They have a unique process of DNA rearrangement in their linear DNA. Each time the DNA is read a different antigenic marker, also known as a variable major protein, is created. This allows the organism to evade the immune system and therefore cause recurrent patterns of fever and other symptoms. 13 It is for this reason that, although there is limited information on the immunity of TBRF, there have been patients who developed the disease more than once. 14
After a tick bite, infected victims may develop a pruritic eschar at the site, but the lesion is usually resolved by the onset of clinical symptoms. A rash, ranging from a macular eruption to petechiae and erythema multiforme, develops in 25% to 30% of patients. 15 The characteristic clinical feature of TBRF is abrupt onset of fever lasting about 3 days, but ranging from 12 hours to 17 days; an afebrile period of variable duration; and then relapses, with return of fever and other clinical manifestations. Untreated patients have had up to 13 febrile episodes. 8
The initial febrile period terminates with rapid defervescence or a “crisis,” accompanied by drenching sweats and intense thirst. 15 Febrile periods in TBRF are associated with bacteremia. Resolution of fever occurs when the host develops an adequate antibody response to the spirochete. During afebrile periods, the spirochete remains “hidden” in organ tissue and undergoes antigenic conversion to a new serotype. Relapse occurs when the new serotype causes bacteremia. 15 Shaking chills, severe headache, myalgias, arthralgias, upper abdominal pain, photophobia, cough, nausea, and vomiting frequently accompany the fever. The temperature is usually greater than 39°C (102.2°F), and patients may manifest extreme muscular weakness and lethargy. Splenomegaly develops in approximately 40% and hepatomegaly in 18% of TBRF patients. 15
Acute respiratory distress syndrome associated with TBRF previously appeared to be quite rare. Davis et al 16 reported a case occurring in a parturient woman in Utah. However, personal communications from Paul Mead, MD, and the October 19, 2007, issue of Morbidity and Mortality Weekly Report provide evidence that ARDS may actually be a more common complication of TBRF than previously appreciated. 17 Including the California and Nevada cases described in the CDC report, 65 cases reported victims living in or visiting the Lake Tahoe area during the usual incubation period of 2 to 18 days before illness, and of these patients, 46% required hospitalization and 5% developed ARDS. Including the single case described in the CDC report (thought to be from exposure in Chelan County, WA), 46 cases of TBRF were reported in Washington between 1996 and 2005, of which 80% required hospitalization, and 6% had ARDS. All 3 ARDS cases occurred after 2001. 17 The patient in Spokane had not visited the Tahoe area, although the neighbor who was hospitalized in Nevada had. Titers run by the CDC on serum from the patient's son and the neighbor (at my request) were negative for TBRF. All ARDS cases the CDC recently reviewed occurred after 2001, but further surveillance is needed to determine whether the risk for developing ARDS is increasing. 17 Like my patient, the 3 cases the CDC reviewed were in women who had received antimicrobial therapy before onset of ARDS.
From 10% to 40% of TBRF patients have neurological involvement. 15 The most common neurological complications are meningismus and facial nerve palsy. Facial palsy, when present, typically occurs after the second febrile episode and usually resolves within 2 to 9 weeks. Other reported neurological complications include neuropsychiatric disturbances, encephalitis, peripheral neuropathy, myelitis, pathologic reflexes, iritis, or iridocyclitis. 15 This patient developed a unilateral hearing loss several weeks after discharge and has not improved in 5 months. This complication is known to occur in TBRF.18,19
Thrombocytopenia is a common manifestation of all tick-borne disease and is thought to result from decreased production, hypersplenism, consumption due to widespread endothelial intravascular coagulation, and immune-mediated platelet destruction. 20 In retrospect, there is a question regarding whether this patient's thrombocytopenia improved as a result of steroid treatment or because her TBRF was in remission. Also, a question remains regarding whether the steroids made her second and third crises more severe by decreasing her immune response to the spirochetemia.
Hyponatremia, which occurred in this patient, secondary to increased antidiuretic hormone secretion in response to hypovolemia, is observed in 56% of patients. 15
Previous studies have shown that the heart suffers significant injury during experimental relapsing fever when the immune response is impaired. 21 The pathogenesis of Borrelia carditis is not understood. The overall prognosis is good, although delayed recovery can occur. In this particular case, the patient's ejection fraction fell to less than 20%, but without any enzyme elevation to indicate muscle necrosis or myocarditis. Ejection fraction improved in 24 hours and ultimately returned to normal (55%). Ventricular function is often decreased during sepsis, even with an increase in cardiac output. 22 This function may recover in days. It may be due to circulating depressant factors, including cytokine tumor necrosis factor alpha and IL-1B in addition to problems with preload and systemic vascular resistance, or other factors. 23 In this one case, it is unclear whether the cardiac dysfunction was primarily due to Borrelia sepsis or secondary to ARDS.
Relapsing fever in pregnancy results in a high incidence of spontaneous abortion, premature birth, and perinatal morbidity. Fetal death is probably caused by direct placental invasion by spirochetes, resulting in thrombocytopenia and retroplacental hemorrhage.15,24
The clinical diagnosis of TBRF requires knowledge of the epidemiology of the disease and a high index of suspicion. Tick-borne relapsing fever occurs only sporadically. A history of recent exposure to old cabins, caves, or any rodent-friendly environment suggests the diagnosis. Routine laboratory tests are of little value. The white blood cell count is usually normal but may be increased or decreased. A left shift is often present. Thrombocytopenia is common but nonspecific. The cerebral spinal fluid is often abnormal, with a lymphocytic pleocytosis (typically 10 to 2000 cells/mm3), usually with a normal glucose and elevated protein. The diagnosis is confirmed by demonstrating spirochetes on peripheral blood smears. A routine peripheral blood smear (Wright-Giemsa stain) from a febrile patient is initially positive in 70% of cases. 15 The diagnostic yield can be increased by examining thick smears and by staining with acridine orange using fluorescence microscopy. The visual yield of spirochetes in peripheral blood smears diminishes with each successive febrile sampling. B hermsii can be cultured in BSK-II medium, with the yield increasing in acutely febrile patients. 15 Paired acute and convalescent samples of antibody by enzyme immunoassay testing and Western immunoblots may confirm the diagnosis and differentiate it from other tick-borne diseases. Monoclonal antibodies (H9826) that bind to B hermsii are also being developed. 25 These type-specific assays may be helpful in characterizing the disease severity associated with specific species of Borrelia.
Tick-borne relapsing fever is probably underreported because not all states consider it a reportable disease. In 1987 it was removed from the list of nationally notifiable conditions. However, 11 states (Arizona, California, Colorado, Idaho, Nevada, New Mexico, Oregon, Texas, Utah, Washington, and Wyoming) still require TBRF to be reported to their State Health Departments. Montana may institute reporting in the future.
The CDC has not developed specific treatment guidelines for TBRF. However, tetracycline and erythromycin are both effective in treating relapsing fever. A 7- to 10-day course of 500 mg orally 4 times a day is generally recommended in tick-borne disease. 26 Borrelia are also sensitive to penicillin and chloramphenicol, but treatment failures have been reported with penicillin. 15 If there are symptoms of meningitis or encephalitis without clinical or radiological evidence of elevated intracranial pressure, lumbar puncture showing increased cells or protein would call for the use of parenteral ceftriaxone for 14 days, as the brain may serve as a reservoir for Borrelia. 18
Jarisch-Herxheimer reaction occurs after the first dose of antibiotics in as many as 50% of patients. It is often severe and may be fatal. When initiating antibiotic therapy, a patient should be watched closely for a reaction for the first 4 hours after the antibiotic is given. 27 The reaction may be difficult to distinguish from a febrile crisis, with rigors and decreased blood pressure. Cooling blankets and appropriate use of antipyretic agents may be useful. The reaction begins with a rise in body temperature and exacerbation of existing signs and symptoms; vasodilation and a fall in blood pressure follow. This complex reaction is mediated in part by products of mononuclear leukocytes. The leukocytes are stimulated by increased contact with antibiotic-altered spirochetes. Neither endotoxin nor complement appears to be necessary in the pathogenesis of JHR, and JHR cannot be prevented by prior steroid treatment or by waiting until the patient is afebrile. Patients who are receiving the initial dose of antibiotics for relapsing fever should receive an intravenous infusion of isotonic saline in anticipation of a possible JHR. Patients may require up to 4 L within 24 hours. This amount is generally sufficient to counteract the hypotension. 15
Limiting exposure to ticks reduces the likelihood of TBRF infection. In persons exposed to tick-infested habitats, prompt, careful inspection and removal of crawling or attached ticks is an important method of preventing disease. Risks increase for individuals camping in mountain forests. When staying in a cabin, campers should check the sleeping areas for evidence of rodents; avoid sleeping on the floor or on a bed that touches the wall; and if the dwelling has been unoccupied, change and wash all bedding before use. A building does not have to be obviously rodent infested to pose a risk of TBRF. Although most TBRF cases occur in older, rustic cabins, this patient was infected in or near a clean, modern home. Insecticides containing permethrin can be sprayed on boots and clothing and will last for several days. Repellents containing deet (N, N-diethyl-m-toluamide) can be applied to the skin but will last only a few hours before reapplication is necessary.
People living in TBRF-prone areas should eliminate the rodents currently in a residence and take measures to prevent future reinfestation. They should thoroughly check the outside walls and foundation of the building for any openings through which rodents might enter; ensure that external doors and windows close with a tight seal; keep all food and garbage in tightly sealed containers; promptly clean up any leftover or spilled food; refrain from feeding squirrels, chipmunks, and other rodents; store firewood outside, away from walls; and stack wood raised a few inches off the ground to discourage rodents from using these areas as nesting sites. Direct control of ticks in an infested building is necessary for comprehensive prevention of TBRF. Eliminating rodents without tick control may actually increase the risk of people becoming infected, because hungry ticks may seek out humans upon which to feed if rodents are unavailable. 12
Conclusions
Acute respiratory distress syndrome may result from a variety of infections. Previously, this complication was not commonly associated with B hermsii. However, several cases of particularly severe disease associated with ARDS have recently been reported.16,17 The patient in my case had unequivocal evidence of B hermsii based on blood smears and serology. The extent and duration of her pulmonary dysfunction could be related to increased virulence of the organism, to her overwhelming infection (as evidenced on smears and bone marrow), to steroid suppression of the immune response to her spirochetal illness, or to a combination of these factors.
Tick-borne relapsing fever is endemic in the Inland Northwest. It should be considered in patients living in or visiting the area who present with a severe flu-like illness and/or ARDS. The diagnosis is best made by demonstration of spirochetes in the blood during the febrile period. Antibiotic treatment is effective. Prevention and control of the disease depend on limiting patient exposure and tick control.
Footnotes
Acknowledgments
The author would like to thank Felix Martinez, MD, and William A. Dittman, MD, for the high-quality images they contributed to this manuscript. I would also like to thank Paul Mead, MD, from the CDC, for assistance with additional testing and information regarding in press Morbidity and Mortality Weekly Report reports on ARDS cases; Martin Schreifer, MD, from the CDC, for additional serological testing; and Dorothy MacEachern of the Spokane County Public Health Department for help in coordinating CDC contacts.
