Abstract
Postoperative acute respiratory distress syndrome (ARDS), characterized by bilateral infiltrates and severe hypoxemia, is a critical complication following thoracic surgery, particularly lung resection. Perioperative transfusions and mechanical ventilation strategies are potential contributors to ARDS development, yet the exact mechanisms driving this association remain incompletely understood. Despite advancements in surgical and perioperative care, ARDS remains a severe complication with a high mortality rate. This study explores the occurrence of ARDS in two patients with nonsmall-cell lung cancer postlung resection, focusing on the association of intraoperative blood transfusion and mechanical ventilation parameters. Both patients received intraoperative blood transfusions (>2 U red blood cells) and exhibited elevated peak plateau pressure (30–31 cmH₂O) with tidal volumes of 6 to 8 mL/kg. Postoperatively, both patients developed acute pulmonary edema and hypoxemia, requiring intensive care management, including mechanical ventilation, lung-protective ventilation strategies, restrictive fluid management, and pharmacological therapy. One patient recovered and was discharged, whereas the other died. ARDS after lung resection arises from synergistic insults, including transfusion-induced inflammation, mechanical ventilation stressors, and reduced pulmonary reserve. Strict adherence to lung-protective ventilation, restrictive transfusion volumes, and multidisciplinary perioperative care are essential to improve outcomes. Future research should prioritize multicenter studies to validate causal mechanisms and refine evidence-based protocols, addressing the urgent need for targeted therapies to reduce ARDS-related mortality.
Keywords
Introduction
Acute respiratory distress syndrome (ARDS) is a severe respiratory condition characterized by bilateral infiltrates on chest imaging and severe hypoxemia primarily due to noncardiogenic pulmonary edema. 1 The underlying pathological mechanisms of ARDS include the accumulation of protein-rich pulmonary edema in the alveoli and interstitium, accompanied by varying degrees of epithelial damage, endothelial barrier disruption, leukocyte infiltration, and impaired fluid and ion clearance. 2 Multiple mechanisms contribute to the development of ARDS, and their relative impacts may vary significantly among individuals. 3
Postoperative ARDS is one of the most severe pulmonary complications associated with thoracic surgery, particularly following lung resection. 4 Postoperative pulmonary complications are the most common adverse events after lung operations, with the incidence of ARDS reported to be 2%–5% following lobectomy or pneumonectomy. 5 Despite significant advances in surgical techniques and perioperative care, pulmonary complications, such as acute lung injury and ARDS, remain severe, albeit rare. Specifically, the incidence of ARDS ranges from 1.5% to 3.1%. 6 These complications substantially increase the rates of intensive care unit (ICU) admission and mortality. 7
Multiple risk factors exist for developing ARDS after lung resection, and the likelihood of ARDS increases with the coexistence of multiple risk factors.8,9 Moreover, the interaction between perioperative transfusions and intraoperative mechanical ventilation may play a key role in the development of postoperative ARDS. 10 Postoperative ARDS remains relatively uncommon but clinically significant, with reported incidence rates ranging from 0.2% to 3.0% after major surgery and up to 7%–10% following thoracic or lung resection procedures. Despite advances in perioperative care, mortality rates remain high, between 30% and 45%. 11 The present case report describes two patients who developed ARDS after lung resection for non-small-cell lung cancer (NSCLC), both of whom received perioperative transfusions and plateau pressure during mechanical ventilation. This report highlights the importance of optimizing transfusion protocols and ventilation strategies during lung resection to reduce the risk of ARDS. Further research is essential to identify preventive interventions and optimize perioperative management to improve the prognosis of patients undergoing surgery for lung cancer. Written informed consent was obtained from each individual for the publication of this article, and the final treatment outcome was recognized by the patient’s family.
Case presentation
Case 1
The first patient was a 64-year-old man who presented with massive hemoptysis 2 weeks prior to presentation, with a total volume exceeding 400 mL. On admission, his vital signs were stable: temperature 37.3°C, heart rate 84 beats/min, respiratory rate 22 breaths/min, blood pressure 128/76 mmHg, and SpO₂95% on room air. Baseline laboratory results revealed white blood cells (WBC) 7.4 × 10⁹/L, hemoglobin 139 g/L, platelet count 64 × 10⁹/L, serum creatinine 70 µmol/L, C-reactive protein (CRP) 11 mg/L, and procalcitonin 0.08 ng/mL. Arterial blood gas analysis on room air showed pH 7.42, PaO₂ 84 mmHg, and PaCO₂39 mmHg. The patient underwent emergency bronchial artery embolization with satisfactory recovery. Chest computed tomography (CT; Supplemental Figure 1) revealed a 45 × 25 mm mass in the apical-posterior segment of the right upper lobe and mediastinal lymph node enlargement. Contrast-enhanced brain magnetic resonance imaging (MRI) and bone scintigraphy revealed no abnormalities. With a smoking history of over 30 years, pulmonary function tests indicated an expected forced expiratory volume in 1 s (FEV1) of 2.84 L and an actual measured value of 2.1 L, with an expected maximum voluntary ventilation (MVV) of 102.62 L/min and an actual value of 82.09 L/min. Echocardiography revealed an ejection fraction (EF) of 61%. Preoperative blood tests demonstrated a platelet count of 64 × 109/L. The patient received two therapeutic doses of leukocyte-depleted platelet apheresis, and subsequent testing suggested pseudothrombocytopenia. All standard preoperative assessments, including bronchoscopy, Positron Emission Tomography/Computed Tomography (PET/CT), and Diffusing Capacity of the Lung for Carbon Monoxide (DLCO), were performed with results supporting operability.
Intraoperatively, fresh blood oozed from the orifice of the middle lobe bronchus, necessitating a right upper lobectomy with middle lobe resection and standard mediastinal and hilar lymph node dissection. The estimated intraoperative blood loss was 700 mL, and 2 units of leukocyte-depleted red blood cell suspension were transfused. The surgical procedure, lasting 4 h and 27 min, employed volume-controlled ventilation (VCV) with tidal volumes maintained at 6–8 mL/kg predicted body weight. During single-lung ventilation phases, airway plateau pressure was strictly controlled at 30 cmH₂O. FiO₂ was maintained between 0.5 and 0.6, with positive end-expiratory pressure (PEEP) levels of 5–8 cmH₂O. Intraoperative transfusion of 2 units of leukocyte-depleted red blood cells was performed after approximately 2 h of surgery, following 600 mL blood loss. The postoperative diagnosis was a right upper-lobe squamous cell carcinoma (T2N1M0, stage IIB).
On postoperative day 3, the patient developed sudden respiratory distress. An urgent chest CT scan (Figure 1) revealed bilateral bronchial-alveolar opacities with ground-glass attenuation, suggesting pulmonary edema. Arterial blood gas analysis revealed a pH of 7.51, partial pressure of oxygen (PO2) of 49 mmHg, and partial pressure of carbon dioxide (PCO2) of 29 mmHg. Blood tests revealed a white blood cell count of 10.23 × 109/L (82.7%), a hemoglobin level of 82 g/L, a platelet count of 24 × 109/L, and procalcitonin of 0.29 ng/mL. The patient was transferred to the ICU for further treatment, intubated, and placed on mechanical ventilation for symptomatic management. Alternative causes such as cardiogenic pulmonary edema and infection were excluded based on normal cardiac function, negative cultures, and stable inflammatory markers. The diagnosis of ARDS was established according to the Berlin definition, based on the acute onset of bilateral pulmonary infiltrates, absence of cardiac failure, and severe hypoxemia.
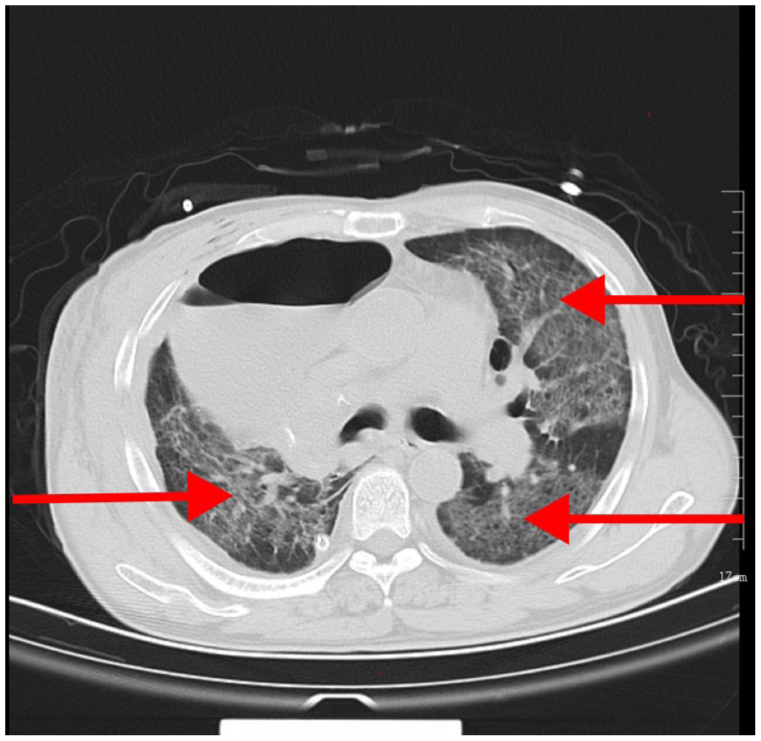
Case 1. Chest CT following the development of ARDS.
Extensive testing was conducted in the ICU to determine the cause of the sudden postoperative pulmonary edema. Laboratory results showed a B-type natriuretic peptide level of 4299 pg/mL and a D-dimer concentration of 4373 ng/mL. Due to limited clinical resources and urgent clinical deterioration, leukocyte antibody testing and cytokine profiling (e.g. IL-6, TNF-α) were not performed. Autoimmune marker analyses, including antinuclear antibodies, perinuclear antineutrophil cytoplasmic antibodies (P-ANCAs), cytoplasmic ANCA (C-ANCA), anticyclic citrullinated peptide, rheumatoid factor, antiextractable nuclear antigen (anti-ENA), C3, and C4, were all within the normal ranges. Given the possibility of infection, empirical antibiotics were initiated, along with anticoagulation therapy and low-dose corticosteroids. Over the next week, chest imaging showed resolving lesions. The patient received noninvasive positive pressure ventilation (NIPPV) to maintain oxygenation, along with glucocorticoids, diuretics, and prophylactic antibiotics. As clinical status improved, respiratory support was transitioned from NIPPV to high-flow nasal cannula and eventually standard nasal cannula.
On day 11 in the ICU, the patient was extubated and transferred to the general ward without further complications. The patient was discharged 11 days later.
Case 2
The second patient was a 74-year-old man with a smoking history exceeding 30 years, a 3-year history of type 2 diabetes, and a more than 40-year history of chronic hepatitis B (HBsAg-positive, anti-HBe-positive). The patient presented to the hospital after a routine physical examination that revealed a mass in the right lower lobe of the lung. On admission, his vital signs were stable: temperature 36.9°C, heart rate 80 beats/min, respiratory rate 20 breaths/min, blood pressure 126/78 mmHg, and SpO₂ 96% on room air. Baseline laboratory results were as follows: WBC 6.8 × 10⁹/L, hemoglobin 138 g/L, platelet count 210 × 10⁹/L, serum creatinine 68 µmol/L, CRP 10 mg/L, and procalcitonin 0.06 ng/mL. Enhanced chest CT (Supplemental Figure 2(a) and (b)) revealed an irregular nodule measuring 22 × 11 mm in the right lower lobe and a 6–7 mm nodule in the apical segment of the right upper lobe. Contrast-enhanced brain MRI and bone scintigraphy revealed no abnormalities. Pulmonary function tests reveal Aedan’s expected FEV1 of 2.54 L and an actual measured value of 1.8 L, with an expected MVV of 88.34 L/min and an actual value of 79.37 L/min. Echocardiography revealed a 60% EF. All standard preoperative assessments, including bronchoscopy, PET/CT, and DLCO, were performed with results supporting operability. The estimated intraoperative blood loss was 1000 mL, and 4 units of leukocyte-depleted red blood cell suspension were transfused. The patient underwent right upper lobe partial resection, complete right lower lobectomy, and systematic mediastinal lymph node dissection The 3 h 24 min procedure utilized VCV with tidal volumes of 6–8 mL/kg predicted body weight, maintaining plateau pressure at 31 cmH₂O throughout single-lung ventilation phases. FiO₂ ranged from 0.5 to 0.7, with PEEP maintained at 5–7 cmH₂O. Intraoperative transfusion of 4 units of leukocyte-depleted red blood cells was initiated after 90 min, synchronized with intraoperative bleeding control.
On postoperative day 1, the patient developed a large amount of bright red hemorrhagic fluid (approximately 1700 mL within 18 h), suggesting active bleeding. An emergency second thoracotomy was performed for hemostasis, and 1000 mL of blood was found in the pleural cavity. Intraoperatively, the patient received 4 units of a leukocyte-depleted red blood cell suspension. The postoperative diagnoses were right upper-lobe squamous cell carcinoma (T1N0M0 IA1) and right lower-lobe squamous cell carcinoma (T1cN0M0 IA3).
On postoperative day 3, the patient developed sudden respiratory distress. An urgent chest CT scan (Figure 2(a)) revealed bilateral bronchial-alveolar opacities with ground-glass attenuation, indicating pulmonary edema. Arterial blood gas analysis revealed a pH of 7.47, PO2 of 43 mmHg, and PCO2 of 40 mmHg. Blood tests indicated a white blood cell count of 10.37 × 109/L (86.2%), a hemoglobin level of 93 g/L, and a procalcitonin level of 0.18 ng/mL. In addition to empirical antibiotics, high-flow oxygen therapy was administered at an oxygen concentration of 55% and a flow rate of 35–50 L/min. Diuretics and low-dose corticosteroids were also administered. Oxygen saturation was maintained at approximately 95% at rest. However, the patient’s condition worsened 7 days later, and he was transferred to the ICU for further treatment. The patient was intubated, and chest radiography revealed bilateral pulmonary edema with consolidation (Figure 2(b)). The diagnosis of ARDS was established according to the Berlin definition, based on the acute onset of bilateral pulmonary infiltrates, absence of cardiac failure, and severe hypoxemia. In the ICU, fiberoptic bronchoscopy was performed for sputum aspiration to promote lung re-expansion. Metagenomic next-generation sequencing of bronchoalveolar lavage fluid detected Haemophilus influenzae, Staphylococcus epidermidis, and Candida albicans, which are normal flora. No bacterial pathogens were isolated from blood cultures, and endotoxin levels were below the detection threshold (<5 pg/mL), suggesting that infection was unlikely to be the primary trigger of ARDS. Cardiogenic pulmonary edema was likewise considered unlikely given preserved left ventricular function and lack of response to diuresis.

Case 2. (a) Chest CT following the development of ARDS. (b) Chest X-ray following ARDS exacerbation.
The patient’s condition continued to deteriorate, and coagulation function tests indicated a plasma prothrombin time of 12.70 s, plasma D-dimer level of 36,821 ng/mL, and fibrinogen degradation product level >80,000 ng/mL. The patient was diagnosed with disseminated intravascular coagulation and subsequently developed multiple organ dysfunction syndrome. Despite rescue efforts, the patient ultimately died.
Based on the above tests and investigations, ARDS was considered the most likely cause of death, and acute pulmonary edema was identified as noncardiogenic. Intraoperative blood transfusion and surgical trauma are believed to be the causes of ARDS, as transfusion may induce direct cytotoxic effects or lung immune-mediated reactions, and surgery can trigger systemic inflammatory response syndrome.
Discussion
With advancements in surgical techniques, patient selection, anesthesia, and ICU management, complication, and mortality rates following lung cancer resection have significantly decreased.12–14 However, postoperative lung injury remains a major cause of morbidity and mortality. ARDS, the most severe form of postoperative lung injury, commonly occurs after pneumonectomy but can also occur after sublobar or wedge resection.15,16 The incidence of ARDS after lung resection is 2%–8%, with a higher incidence of 7%–12% following pneumonectomy. 17 In this case report, two patients with NSCLC developed ARDS after lung resection. Common features in both patients included intraoperative blood transfusion, elevated airway pressure, large tidal volume, and delayed onset of postoperative ARDS. These factors have also been reported as risk factors for ARDS.18–20
Synergistic effects of intraoperative blood transfusion and mechanical ventilation on ARDS
Perioperative blood transfusion constitutes a significant risk factor for postoperative ARDS, particularly transfusion-related acute lung injury (TRALI) characterized by acute hypoxemia with bilateral infiltrates in the absence of cardiac dysfunction. While classical TRALI pathogenesis involves donor-derived antibodies or bioactive lipids, our cases highlight alternative mechanisms: surgical stress combined with RBC-derived microparticles may activate pulmonary endothelial TLR4/NF-κB pathways, as evidenced in recent experimental models.21,22 Importantly, mechanical ventilation acts as a critical cofactor through two interrelated pathways: high tidal volumes directly exacerbate alveolar neutrophil infiltration and protein leakage, while ventilator-induced priming of circulating neutrophils and endothelial barrier dysfunction collectively lower the threshold for lung injury initiation. 23
Clinically, both patients underwent prolonged procedures with substantial blood loss requiring multiple transfusions, correlating with established dose-dependent associations between transfusion volume and ARDS risk. 24 This pathophysiological interplay underscores the necessity for integrated preventive strategies. Clinical management should prioritize restrictive transfusion protocols guided by dynamic hemodynamic monitoring, coupled with rigorous implementation of lung-protective ventilation (tidal volume < 6 mL/kg predicted body weight, plateau pressure <30 cmH2O). Postoperative surveillance must include serial arterial blood gas analysis and chest imaging in high-risk cohorts.
Impact of intraoperative mechanical ventilation and surgical extent on ARDS risk
Intraoperative mechanical ventilation parameters and the extent of surgical resection synergistically influence postoperative lung injury. Mechanical ventilation strategies directly modulate pulmonary stress, with elevated plateau pressure and excessive tidal volumes (>10 mL/kg) recognized as key drivers of ventilation-induced lung injury (VILI). These factors induce alveolar overdistension, disrupt the alveolar-capillary barrier, and amplify inflammatory cascades through cyclic recruitment-derecruitment injury.25,26 Experimental studies in porcine ARDS models demonstrate that lung-protective strategies combining low tidal volumes (VT < 6 mL/kg) with optimized PEEP reduce histological lung damage while maintaining hemodynamic stability. 27 In parallel, the extent of pulmonary resection critically determines postoperative functional reserve. Extensive resections (e.g. pneumonectomy or multilobectomy) impose disproportionate mechanical strain on residual lung tissue, exacerbating ventilation-perfusion mismatches and interstitial fluid overload. 28
In the presented cases, both mechanisms converged: elevated intraoperative peak pressures (exceeding 30 cmH2O) compounded the physiological burden of multilobar resections. This dual insult likely precipitated alveolar epithelial injury through volutrauma and surgical parenchymal loss, creating a permissive environment for ARDS development. Current evidence advocates strict adherence to lung-protective ventilation protocols (VT < 6 mL/kg predicted body weight, plateau pressure <30 cmH2O) during major thoracic procedures, particularly when extended resections are anticipated. 10 Proactive hemodynamic monitoring and judicious fluid management further mitigate cumulative injury in this high-risk cohort.
Comorbidities, prognostic implications, and study limitations
Both patients exhibited significant comorbidities that amplified ARDS susceptibility. In case 2, chronic hepatitis B and diabetes likely impaired innate immunity, exacerbating transfusion-related inflammatory cascades. The extensive resections—right upper lobectomy with middle lobe resection (case 1) and bilobectomy (case 2)—reduced functional lung capacity, lowering the threshold for volutrauma-induced hypoxemia. While surgical trauma independently activates systemic inflammation, the convergence of transfusion-related injury (e.g. TRALI), mechanical ventilation stressors (e.g. VILI from high tidal volumes), and comorbidities aligns with a multiple-hit model of ARDS pathogenesis. This mechanism reflects a “second-hit” process, in which perioperative transfusion acts as an additional inflammatory trigger superimposed on surgical and ventilatory stress. The combined insults may synergistically promote endothelial damage, capillary leak, and alveolar flooding, leading to delayed-onset postoperative ARDS.
Postoperative ARDS carries a mortality rate of 40%–60%, with outcomes contingent on early intervention. 29 In these cases, divergent outcomes emerged: case 1 recovered through aggressive ICU management, while case 2 succumbed to disseminated intravascular coagulation and multiorgan dysfunction syndrome, underscoring ARDS-associated lethality. Current therapies remain supportive (e.g. lung-protective ventilation, restrictive fluid strategies), highlighting the unmet need for targeted interventions.
This study’s findings, while illustrative, are constrained by its retrospective design and the inherent limitations of a case report format. Although the analysis reinforces associations between intraoperative blood transfusion, ventilation parameters, resection extent, and ARDS, prospective multicenter studies are required to validate causality and refine preventive protocols (e.g. transfusion thresholds, optimal PEEP titration). From a clinical perspective, early recognition and prevention of postoperative ARDS are crucial. Preventive strategies include careful intraoperative fluid and transfusion management, lung-protective ventilation, and close postoperative monitoring of oxygenation, inflammatory markers, and imaging to enable prompt detection and intervention. Future research should prioritize mechanistic investigations into TRALI-VILI interactions and clinical trials evaluating protocolized interventions in high-risk cohorts.
Conclusion
Postoperative ARDS following lung cancer resection stems from multifactorial interactions, including intraoperative blood transfusion, high tidal volumes, and extensive surgical resection, which synergistically exacerbate alveolar injury and inflammation. Restrictive transfusion protocols and strict lung-protective ventilation (tidal volume < 6 mL/kg, plateau pressure <30 cmH2O) are pivotal to mitigating these risks, while comorbidities and reduced pulmonary reserve further heighten susceptibility, necessitating tailored perioperative management and vigilant monitoring. Despite advancements, ARDS mortality remains alarmingly high (40%–60%), underscoring the urgent need for targeted therapies beyond supportive care. Future efforts should focus on multicenter studies to validate causal mechanisms, refine evidence-based protocols, and elucidate molecular pathways underlying transfusion-ventilation injury interactions, complemented by multidisciplinary collaboration to optimize outcomes in high-risk patients.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X251400426 – Supplemental material for Case report: Causes of acute respiratory distress syndrome complications after lung resection
Supplemental material, sj-docx-1-sco-10.1177_2050313X251400426 for Case report: Causes of acute respiratory distress syndrome complications after lung resection by Liu Chang, Li Zhengjun, Zhang Yumeng, Wang Tao, Zhang Xiangchao, Xie Liwei, Song Shiyuan and Ren Yi in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
The authors thank the patients who participated in this study.
Author contributions
C.L.: Writing—original draft, writing—review, and editing. Z.L.: Conceptualization, Data curation, project administration, writing, review, and editing. Y.Z. and T.W.: Formal analysis, Software, Supervision. X.Z., L.W., and S.S.: Writing of the original draft. Y.R.: Conceptualization, Validation, Visualization, and Writing of the original draft.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Shenyang Science and Technology Planning Project (No. 22-321-33-70).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The original contributions presented in this study are included in the article/supplemental material. Further inquiries can be directed to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
