Abstract
This study, which is the first in a three-part series, deals with the encapsulation of photochromic dyes by spray drying. An aqueous ethyl cellulose dispersion and a spirooxazine-based photochromic dye were used as a shell and core material, respectively. The effects of main encapsulation parameters, such as solvent type, inlet temperature, feed rate, solid content, and aspirator rate were investigated. The encapsulation results were evaluated by scanning electron microscopy (SEM) images, particle size measurements, thermogravimetric analysis (TGA) and X-ray diffraction (XRD). The microcapsules obtained from a water-ethanol mixture exhibited photochromic properties. For microcapsule production, the optimum feed rate, total solid content, and aspirator rate were determined. Capsule formation improved with increased inlet air temperature. Spray drying to produce photochromic microcapsules could be a practical method for production of photochromic smart textiles.
Introduction
Photochromic materials change color reversibly under UV light exposure (sunlight or artificial UV light). This results from the absorption spectrum change of a molecule or crystal (reversibly) when the sample is irradiated by UV light. 1 There are different types of photochromic materials, such as inorganic and organic. Organic molecules can provide a pho-tochromic effect through various reactions, such as pericyclic reactions, cis-trans isomerization, and intramolecular trans-fers. 2 Among these molecules, one of the most widely used are molecules that can undergo pericyclic reactions. These dyes, which show photochromism as a result of ring opening/ closing reactions, are divided into five groups: spiropyrans, spirooxazines, naphthopyrans, diarylethenes, and fulgides.3,4 The first commercial applications of this photochromic dye class were made with easily-synthesized spiropyran dyes. However, with the discovery of spirooxazine dyes having greater photostability, and thus greater fatigue resistance, interest in spiropyran dyes gradually declined. 3 Within the scope of this study, it was decided to work with a spirooxa-zine dye, which is widely used and is relatively photostable. Spirooxazines change their colorless structure to a colored merocyanine form by ring opening reaction under UV irradiation and revert thermally to the colorless ring-closed form when UV light is removed (Fig. 1).4-7
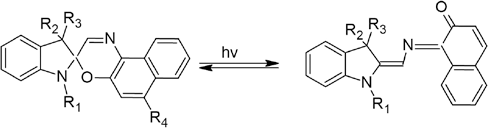
Structure change of a photochromic compound (spirooxazine). 7
Photochromic dyes have found use in various areas, such as the optical, plastics (e.g., drink bottles and toys), cosmetics (e.g., photochromic nail polishes), and textile industries due to their color changing ability by UV light.1,8 Commercial applications of photochromic textiles are mainly fashion-oriented designs. Studies on the use of photochromic dyes in the development of technical and smart textiles have also gained attention, especially in recent years. In this context, technical products, such as textile based sensors9,10 or UV protective textile materials, 1 can be developed by the application of photochromic dyes.
Photochromic textiles can be produced either by the incorporation of photochromic dyes into the polymer matrix during the production of synthetic fibres, 11 or through conventional finishing processes, such as impregnation, printing, or coating.1,5,12-16 Moreover, sol-gel applications of photochromic dyes were also reported.17-19
Photochromic dyes are small- to medium - size neutral molecules similar to disperse dyes. However, photochromic dyes are non-planar in their ring-closed form due to their two ring systems being connected through a spiro linkage. 14 In addition to this, unlike conventional textile dyes, photochromic dyes have low water solubility, low fiber affinity, and more sensitivity to environmental conditions, and thus, direct application of these dyes have drawbacks in the textile field. Aldib and Christie reported the formation of ground color after exhaust dyeing of polyester by photochromic dyes, possibly due to the fixing of the ring-opened colored form of the dye within the polyester or due to the thermal instability of the dyes under dyeing conditions. 5 Aldib and Christie, in a continuation of their previous study, indicated that the exhaustion of photochromic dye into the fiber in aqueous media was inadequate due to low water solubility, and they suggested the use of organic solvents to increase the dye yield. 13 Lee et al. applied exhaust dyeing on polyamide fibers and concluded similar results in terms of color yield due to the weak affinity of photochromic dyes to textile materials, and the poor diffusion ability into highly-crystalline synthetic fiber structures. 20 They stated that the exhaustion yield increased at higher temperatures. However, photochromic dyes are known to be sensitive to higher temperatures. For instance, Billah et al. found that photochromic dyes were susceptible to thermal decomposition in water at higher temperatures.14,16 In this context, encapsulation of photochromic dyes is considered as an alternative route for eliminating these disadvantages.
Liquid droplets, solid particles, or gases are coated with a continuous film or polymeric material by means of encapsulation; thereby, the encapsulated material is protected from external influences.21-23 Although there are many different methods for encapsulation, interfacial polymerization, coacervation, in-situ polymerization, and emulsification-solvent evaporation appear in the literature to be the most used methods for encapsulating photochromic dyes.1,24-29 However, spray drying is an encapsulation method that has many advantages, such as one-step processing, rapidity, industrial applicability, low dependence on the solubility properties of core and shell materials, low processing cost, and easy scale-up. 30
The aim of this study is to investigate the applicability of spray drying for photochromic dye microencapsulation. Ethyl cellulose and a spirooxazine-based commercial photochromic dye were selected as shell and core materials, respectively. The main purpose is the determination of the most suitable spray drying process parameters for capsule formation without disturbing its photochromic capability. The effects of solvent system, shell/core ratio, drying temperature, solution feed rate, and aspirator rate on resultant photochromic micro-capsule features, such as morphology, mean diameter, and thermal properties were conducted. To the best of the authors’ knowledge, there is no previously published study relating to the encapsulation of a photochromic dye by a spray drying method. Therefore, the process parameters determined are believed to be useful for the improvement of textile applications using photochromic dyes.
Experimental
Materials
The materials used in the encapsulation by spray drying are shown in Table I.
Materials used in the Encapsulation by Spray Drying
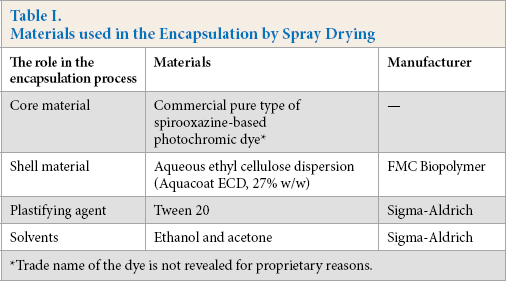
Trade name of the dye is not revealed for proprietary reasons.
Microencapsulation by Spray Drying
In encapsulation by spray drying, the core is dispersed in the solution of the shell material until the desired oil droplet size is obtained. This emulsion is fed into the heated compartment of the spray dryer through a nozzle and fed into droplets. The capsules are obtained at the end of drying using hot air. Although the capsule sizes formed in this method depend on the diameter and type of nozzle used in the spray dryer, parameters such as the feed rate applied during spraying, the viscosity of the feed solution, and the drying temperature also change the capsule sizes formed.30-32
In this study, a laboratory scale spray dryer (Büchi, B-290) was used for microencapsulation of a photochromic dye. The schematic illustration of the spray dryer is shown in Fig. 2. Microencapsulation solutions were pumped to the atomizer by a peristaltic pump (up to 30 mL/h) and sprayed into the drying chamber by compressed air (maximum of 8 bar). Simultaneously, drying air (heated by an electric heater up to 220 °C) was fed to the drying chamber (maximum air flow rate is 35 m3/h). Microcapsules were formed by the rapid evaporation of solvent during the concurrent flow of drying air and atomized droplets in the drying chamber. The resultant microcapsules were collected in a product collection vessel and the outlet air was exhausted to the environment by an aspirator after passing through a filter medium. The solution feed rate, drying air flow rate (aspirator rate), drying air temperature, and compressed air flow rate were adjustable.
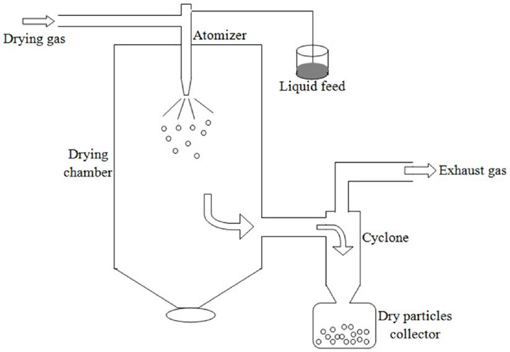
Schematic illustration of the spray drying system.
Since spray drying is a thermal process, the solvent used is one of the most important parameters affecting the properties of the resultant product. Therefore, in the first stage, an experimental plan was carried out for the determination of the most appropriate solvent system for the production of photochromic microcapsules. For this purpose, water, and two types of organic solvents that can dissolve the photochromic dye (ethanol and acetone) were examined. Table II shows the solution and process parameters used in solvent system determination. For single solvent systems, 3% w/v photochromic dye was first dissolved in the solvent, and then 3% w/v ethyl cellulose was added. A plastifying agent at a concentration of 1% w/v was then added to the solution, followed by stirring for 2 h. Along with single solvent systems, water/organic solvent mixtures were also investigated. For the systems containing water and organic solvent, photochromic dye (3% w/v) was first dissolved in an organic solvent and then added to the aqueous ethyl cellulose solution (3% w/v). In water/organic solvent mixtures, as a first approach, the ratio of organic solvent to ethyl cellulose solution was adjusted to 1:0.4, and the organic solvent was fully evaporated by stirring for 24 h at room temperature (RT) before spray drying. Thereby, an aqueous spray drying solution, in which the photochromic dye was thoroughly dissolved, was obtained. As a second approach, water/organic solvent mixtures at a ratio of 0.8:0.2 and 0.6:0.4 were also prepared, and these solutions were fed to the spray dryer without pre-solvent evaporation. A plastifying agent at a concentration of 1% w/v was used also in the water/organic solvent mixture trials. Each solution was spray dried at a 150 °C inlet air temperature, 3 mL/h feed rate, and 95% aspirator rate.
Parameters for Solvent System Investigation
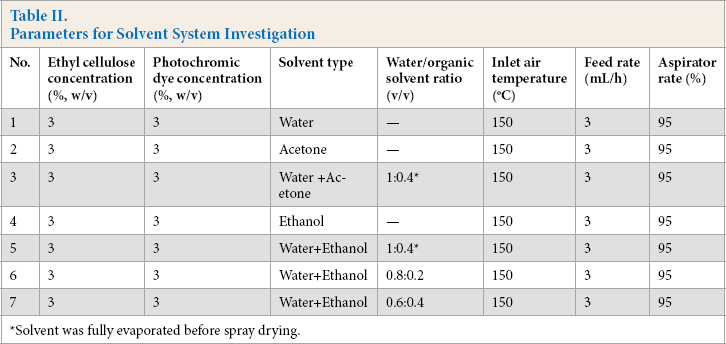
Solvent was fully evaporated before spray drying.
After determination of suitable solvent systems, in the second stage, a parametric study of spray drying was carried out. The ethanol/water solvent system was chosen for the parametric studies. At this stage, the effects of solid content (the total amount of shell polymer and core material), photochromic dye to ethyl cellulose ratio, inlet air temperature, feed rate of the solution, and aspirator rate of spray drying on the characteristics of the resulting photochromic microcapsules were investigated. The solution and process parameters used for the parametric study are shown in Table III. After spray drying, the resulting solid products were weighed and then stored in sealed containers until further analysis.
Selected Parameters for Spray Drying
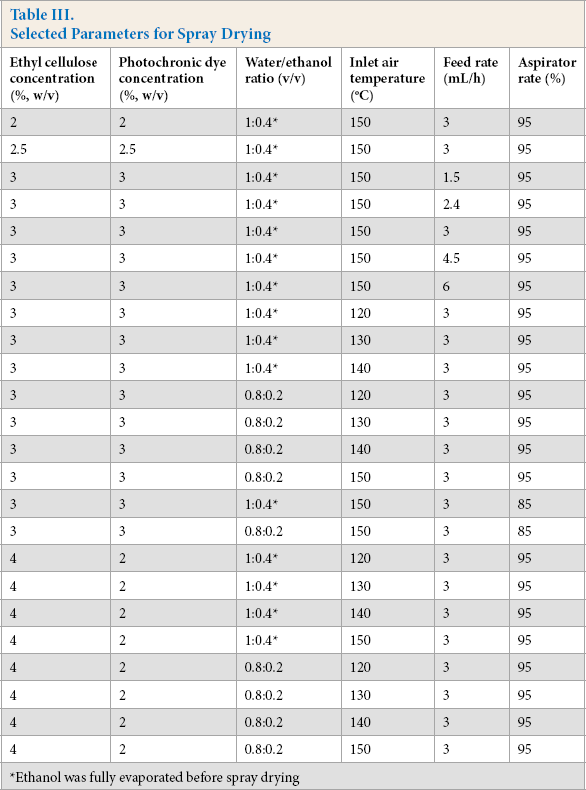
Ethanol was fully evaporated before spray drying
Characterization
The morphology of the resultant products of spray drying were visually evaluated using a scanning electron microscope (SEM, FEI Quanta250 FEG, FEI Inc., USA). All samples were sputtered with a thin layer of gold under vacuum using an Emitech K550X ion sputtering device before SEM measurements.
Size distribution of photochromic microcapsules was measured using a Malvern Nano ZS (Malvern Panalytical Ltd., USA) device. Before analysis, the aqueous solution of micro-capsules were stirred using an ultrasonic homogenizer. Ten the capsule solution was put into cuvettes for measurement.
Thermogravimetric analysis (TGA, TA Instruments SDT Q600, USA) was used to investigate the thermal behavior of the photochromic microcapsules. TGA thermograms was performed using nitrogen gas and a 10 °C/min heating rate between RT and 600 °C. TGA data were evaluated by comparing differential thermal analysis (DTA) results, such as endothermic peak temperatures, half-decay temperature, and residual mass rate.
Phase analysis of the photochromic capsules was performed using an X-ray diffractometer (Philips X'Pert Pro, Netherlands) between 2° and 40° (2θ) angles. XRD spectra were evaluated according to peak intensity.
Results and Discussion
A spirooxazine-based photochromic dye was encapsulated by ethyl cellulose via spray drying. The suitable solvent system for spray drying to obtain photochromic microcap-sules was first investigated. The dye to ethyl cellulose ratio and spray drying parameters were then examined in detail to improve the resultant microcapsule formation.
Solvent System Investigation
Fig. 3 illustrates the SEM images of the resultant products obtained by spray drying of photochromic dye microencapsulation solutions prepared by water, acetone, ethanol, and their mixtures. In the case of single solvent systems, microcapsule formation was observed only with the aqueous solution. When ethanol or acetone was used, capsule formation could not be obtained possibly due to the lower evaporation temperature of these solvents (boiling points for water, ethanol, and acetone are 100 °C, 78 °C, and 56 °C, respectively). 33 Gander et al. indicated that fast solvent removal from the polymer caused a rapid formation of solid particles. 34 They also stated that the diffusion of the solvent remaining in the core became difficult as a result of rapid shell formation, and thus the high vapor pressure formed in the core may cause the particles to burst. Paudel et al. explained the effect of solvent type on particle formation during spray drying in two ways: rush hour effect and tetris effect. 35 They stated that fast solvent evaporation causes rapid diffusion in the rush hour effect, and there is not enough time for recrystallization, while for the tetris effect, slow solvent evaporation provides sufficient time for reorganization. In the present study, similar to the studies in the literature, the microcapsule formation was obtained with water due to relatively slow evaporation instead of with solvents having low evaporation temperatures. On the other hand, although capsules can be obtained with the aqueous system, it was visually observed that these capsules do not change color under UV irradiation. This might be due to the inadequate dissolution of the photochromic dye in water. As Little and Christie also stated in their study, a photochromic effect from the undissolved photochromic dye was not expected due to its solid state. 36

SEM images of the products obtained after spray drying with different solvents.
To eliminate the solubility problem in water, organic solvent and water mixtures were further investigated. For this purpose, two approaches, (1) full evaporation of the solvent before spray drying (water to organic solvent ratio of 1:0.4 v/v) and (2) spray drying of the solutions including water/ organic solvent mixtures (water to organic solvent ratio of 0.6:0.4 and 0.8:0.2 v/v), were applied as shown in Table II. Precipitation of ethyl cellulose was observed for all water/acetone mixtures. Spray drying could not be carried out for the water/ ethanol ratio of 0.6:0.4, as a precipitate formed in the solution. On the other hand, stable ethyl cellulose and photochromic dye solutions could be obtained by water/ethanol mixtures at ratios of 1:0.4 and 0.8:0.2. In addition, it was difficult to dissolve the photochromic dye even at 0.8:0.2 water/ethanol ratio, so a 0.9:0.1 water/ethanol ratio was not studied. In this context, only two different water/ethanol ratios (1:0.4 and 0.8:0.2) could be studied, and successful capsule formation were observed after spray drying (Fig. 3). Moreover, color build up under UV irradiation was visually observed for the resultant microcapsules that categorically confirmed their photochromic behavior (Fig. 4). Accordingly, in the following parametric studies of spray drying, both aqueous systems prepared using ethanol, after the ethanol portion was fully evaporated before spray drying, were investigated.

Images of the microcapsules powder obtained using water/ethanol mixture with the ratio of 1:0.4.
Parametric Study
The effects of solid content, shell/core ratio, solution feed rate, aspirator rate (i.e., drying air volume flow rate), and drying air inlet temperature on capsule formation were investigated.
In the studies examining the effect of total solid content on capsule formation, 4%, 5%, and 6% total solid content values were tested. The process parameters were applied as follows: 10% feed rate, shell/core ratio of 1:1, 1:0.4 water/ethanol ratio, 150 °C inlet air temperature, and 95% aspirator rate. The SEM images of the resultant products obtained for each solid content are shown at Fig. 5. Microcapsule formation was improved by the increase in total solid content and the best result was obtained when the total amount of solids was 6%, because higher solid content resulted in more viscous emulsions. As stated by Frascareli et al. and Tonon et al., viscosity enhancement decreased the particle precipitation rate, and thus, droplet coalescence was prevented by providing better emulsion stabilization.37,38 Therefore, an improvement in capsule formation was observed with increased solid content in the present study, however, concentrations above 6% could not be studied due to the problem of nozzle blockage.

SEM images of the products obtained at different solid content: 4%, 5%, and 6%.
The feed rate, which determines the duration the droplet remains in the dryer, is one of the most important parameters affecting droplet formation in spray drying. The droplet sprayed from the nozzle must be sufficiently dry before hitting the dryer wall, and therefore, the droplet should remain in the dryer for a sufficient time with an appropriate feed rate. 35 To investigate the effects of microencapsulation solution feed rate, spray drying with five different feed rates (1.5, 2.4, 3, 4.5, and 6 mL/h) were carried out. The spray drying process parameters used were 6% total solid content, shell/core ratio of 1:1, water/ethanol ratio of 1:0.4, 150 °C inlet temperature, and 95% aspirator rate. Fig. 6 shows the SEM images of the resultant products.

SEM images of the spray dried products obtained at different feed rates.
Microcapsule formation was improved with the increase in feed rate up to 3 mL/h. A further increase in the feed rate deteriorated microcapsule formation by not providing enough time for the particles to dry completely. 39 Based on these results, the optimum feed rate for the spray drying of photochromic microcapsules was determined to be 3 mL/h for this study.
Another parameter affecting the capsule properties during encapsulation with spray drying is the inlet temperature. As the inlet temperature increases, the moisture content in the product decreases, thereby increasing the rate of film formation on the surface of the droplet. 30 The effect of drying air temperature (inlet air temperature) of the spray dryer on microcapsule formation was investigated for air temperatures of 120 °C, 130 °C, 140 °C, and 150 °C. The spray drying process parameters were 6% total solid content, shell/core ratio of 1:1, water/ethanol ratio of 1:0.4 and 0.8:0.2, and 95% aspirator rate. The SEM images of the resultant microcapsules are shown in Fig. 7. Microcapsule formation could be obtained for all temperatures applied. However, improvement in microcapsule formation was visually observed with increased inlet temperature, due to the prevention of coalescence as a result of low moisture content and the tendency to form a crust faster. Similar findings were also reported by Alamilla-Beltrán et al. and Tonon et al.40,41

SEM images of the spray dried products obtained at different inlet temperatures.
The effect of the aspirator rate (drying air volumetric flow rate) was also investigated. Aspirator rates (85% and 95%) were compared for two solvent system. The spray drying process parameters were 6% total solid content, shell/core ratio of 1:1, and 150 °C inlet air temperature. Although microcapsule formation was observed using the 85% aspirator ratio, relatively better results were obtained using the 95% aspirator ratio (Fig. 8). Similar to the effect of the inlet temperature, faster crust formation occurs with the increase of the aspirator rate due to the increased drying speed and decreased moisture content.42,43

SEM image of the products at aspirator ratio of 85% and 95%.
Characterization of Photochromic Microcapsules
All of the aforementioned experiments were carried out with a 1:1 shell/core ratio. To investigate the effects of the shell/core ratio for photochromic microcapsules, the experiments were repeated at a 2:1 shell/core ratio under the conditions where optimum microcapsule formation was achieved (feed rate: 3 mL/h; aspirator ratio: 95%; inlet temperature: 120 °C, 130 °C, 140 °C, and 150 °C; and water/ethanol ratio: 1:0.4 and 0.8:0.2). The SEM images of these experiments are shown in Fig. 9. As in the trials with 1:1 shell/core ratio, capsule formation was observed for 2:1 shell/core ratio and improved with increased inlet air temperature. Similar to the study of Lie et al., in the present study, more spherical capsules were obtained with the increased shell/core ratio. 44

SEM images of the products obtained with 2:1 shell/core ratio at different inlet air temperatures.
According to the SEM images, the achievable main spray drying parameters for microcapsule formation were visually determined. For this study, a total solid content of 6% w/v, solution feed rate of 3 mL/h, inlet air temperature of 120 °C to 150 °C, aspirator rate of 85% to 95%, and a shell/core ratio of 1:1 and 1:2 were found to be the optimal spray drying parameters for the production of photochromic microcapsules. To characterize the resultant products, particle size measurement, TGA, and XRD analysis were applied to selected photochromic microcapsules produced using several production parameters (Table IV).
Mean Diameter of the Capsules Obtained at Different Process Parameters
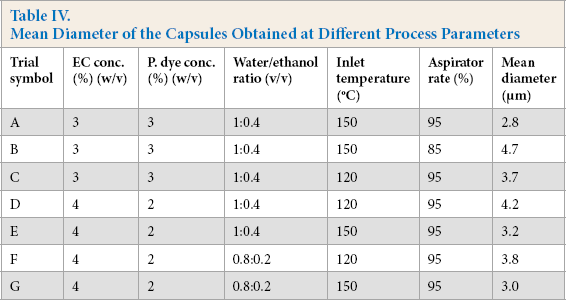
The mean diameter of the photochromic microcap-sules obtained from particle size measurements are given in Table IV. The average sizes of photochromic dye capsules were measured to be ∼3-5 µm. The aqueous system in which the organic solvent was evaporated before spray drying (water/organic solvent rate of 1:0.4) led to a greater mean microcapsule diameter. This might possibly be due to the lower drying rate of the totally aqueous system compared to the water/ethanol mixture during spray drying. It was observed that the increase in the inlet air temperature reduced the microcapsule diameter, which might be due to the rapid solidification as a consequence of the higher drying rate. Also, the increase in the ethyl cellulose concentration in the microencapsulation solution led to an increase in the mean diameter possibly due to the larger feed droplet size as a result of the higher viscosity of the solution. Decrease in the aspirator rate also led to a higher mean particle diameter, which might be due to the alleviation of the drying rate. According to the particle size distribution charts, in general the use of the water/ethanol mixture and a 2:1 shell/core ratio were observed to ensure more homogeneous microcapsule formation.
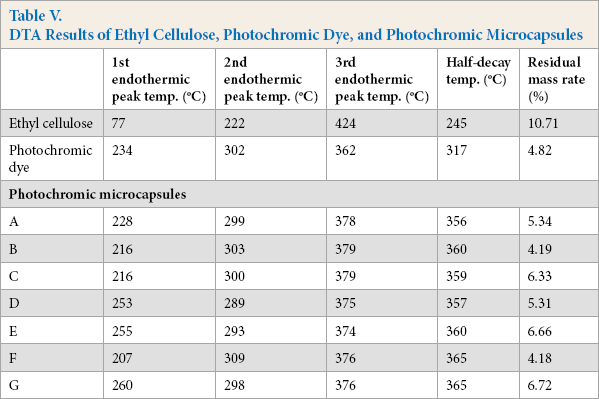
TGA curves of ethyl cellulose, photochromic dye, and photochromic dye capsules are shown in Fig. 10. In addition, DTA data for photochromic microcapsules are given in Table V. When the TGA and DTA results of photochromic microcapsules were examined, it was observed that the encapsulation process improved the thermal resistance of the dye. On the other hand, the TGA curves of microcapsules produced in different spray drying process parameters were found to be similar. It was thought that the encapsulation parameters had no significant effect on the thermal properties of the resultant microcapsules. DTA data showed that the microcapsules were degraded more slowly than the photochromic dye itself (Table V). For example, the photochromic dye lost half of its weight at around 317 °C, whereas for photochromic microcapsules, this temperature increased up to 355-365 °C. Similarly, the 3rd endothermic peak temperatures were increased by the encapsulation. In this context, the encapsulation process improved the thermal properties of the photochromic dye.
DTA Results of Ethyl Cellulose, Photochromic Dye, and Photochromic Microcapsules
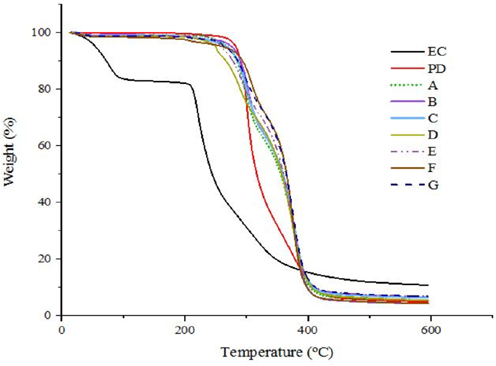
TGA curves of ethyl cellulose (EC), photochromic dye (PD), and photochromic microcapsules A-G.
XRD spectra of ethyl cellulose, photochromic dye and photochromic microcapsules are shown in Fig. 11. The XRD graph of the photochromic dye shows characteristic crystalline peaks, while the XRD graph of ethyl cellulose shows amorphous peaks. When XRD graphs of photochromic microcapsules were examined, it was observed that the intensity of sharp peaks belonging to the photochromic dye decreased and softer peaks were formed. In this context, XRD analysis revealed that the photochromic dye was encapsulated with ethyl cellulose.
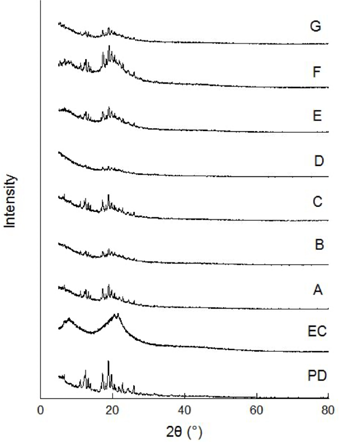
XRD spectra of ethyl cellulose (EC), photochromic dye (PD), and photochromic microcapsules A-G.
Conclusion
This study showed that spray drying is a suitable method for photochromic microcapsule production. Ethyl cellulose was used as the shell material in the encapsulation of the spirooxazine-based photochromic dye. It was determined that the solvent system chosen is critical for the preservation of photochromic properties. Accordingly, a water-ethanol mixture was found to be the most suitable solvent. Increasing solid content, shell/core ratio, drying temperature, and aspirator rate improved microcapsule formation. Furthermore, thermal resistance of the photochromic dye was improved by the encapsulation process. The average size of the microcapsules was measured to be ∼3-5 µm depending on the process parameters.
Spray drying is known to be a cost-effective, continuous, and scalable process for microencapsulation. Due to these advantages, obtaining photochromic microcapsules by this method is believed to have good potential for solving the quality problems arising from textile applications of neat pho-tochromic dyes, and the development and industrial uptake of photochromic smart textiles. In this context, it is important to examine the textile applications and photochromic performances of the resultant microcapsules. Also, since spray drying is a thermal process, it should also be examined in terms of energy use. For this purpose, in the following stages of this research, the energy/exergy analysis of the spray drying method and the textile applications of the resultant photo-chromic microcapsules will be investigated.
Footnotes
Acknowledgements
The authors would like to gratefully acknowledge The Scientific and Technical Research Council of Turkey (TUBITAK) through Project No. 214M428, and Ege University, scientific research projects, through Project No. 16-MUH-079 for financial support to this research project. The authors also would like to thank Sun Tekstil for allowing use of the spray drying device in this study.
