Abstract
The aim of this experimental study was to obtain disposable antimicrobial cotton fabrics through applications of tea tree oil/ ethyl cellulose (TTO/EC) microcapsules. For this purpose, the TTO/EC microcapsules were prepared and characterized via scanning electron microscopy (SEM) and Fourier transform infrared-attenuated total reflectance (FTIR-ATR) analyses. The characterized microcapsules were then applied to 100% cotton fabric samples via the padding method and were investigated by FTIR-ATR and SEM analyses. In addition, the antibacterial activities of TTO, TTO/EC microcapsules, and fabric samples were also assessed against
Introduction
Recently, essential oils have been given increased attention in the development of natural health care products due to their performance as antimicrobial, antifungal, antiviral, moisturizing, and regenerating agents, as well as their aromatic properties.1–4 The essential oil, tea tree oil (TTO), from the leaves of
In microencapsulation technology, an active substance, or core material, is covered by a suitable polymer wall on a very small scale.12,13 This technology has been used in various fields, including textiles, agriculture, pharmaceuticals, food, and cosmetics. Various core materials (e.g., antimicrobial agents, drugs, dyes, fragrances, fire retardants, oils, vitamins, insect repellents, and phase change materials) have been encapsulated for use in the textile industry.13–16 A great variety of microencapsulation techniques are available (e.g., emulsion polymerization, interfacial polymerization, in situ polymerization, and coacervation).13,16–19 Coacervation, which is divided into simple and complex coacervation, is based on the phase separation of one or many hydrocolloids.13,15 In this method, gelatin, maltodextrin, chitosan, sodium alginate, gum arabic, ethyl cellulose, and similar materials are generally preferred as the wall materials.13,20
The objective of the present study was to obtain disposable antimicrobial textile surfaces treated with TTO/EC microcapsules. TTO/EC microcapsules were used due to the difficulties of direct oil application on textile surfaces. First, the antibacterial activity of TTO was researched against
Materials and Methods
Materials
TTO (Huile Essentielle bio organic essential oil
In the application processes, 100% cotton woven fabric (46 thread/cm warp, 22 thread/cm weft, 270 g/m2Textile.) and a laboratory scale fulard were used. The fabric was desized, scoured, and bleached by the supplier Matesa Textile.
Method
TTO/EC microcapsules were prepared using the simple coacervation method as follows. Two phases (organic and aqueous) were prepared separately. For the organic phase, 2 g of ethyl cellulose was slowly added to 10 mL of a 10% (w/v) solution of TTO in ethyl acetate and mixed with a magnetic stirrer. Stirring proceeded until the ethyl cellulose completely dissolved. For the aqueous phase, 4% (w/v) Tween 20 was added to a 10% (w/v) solution of ethyl acetate in water stirred at 10,000 rpm with a high shear mixer, and stirring was continued for 10 min. The organic phase was then added dropwise into the aqueous phase and stirring was continued at 10,000 rpm for 60 min. Finally, 50 mL of cold water (10 °C) was added to the emulsion and it was stirred for 15 min to complete phase separation. The microcapsules were then filtered, washed with ethanol followed by water, and dried at room temperature (RT).
An application solution containing 50 g/L of microcapsules was applied to the fabric sample (20 × 30 cm) on the laboratory type fulard by padding. The sample was squeezed up to 90% pickup and dried at RT. Because the produced fabric samples were considered disposable, a crosslinking agent was not used in the application.
Microcapsule Characterization
Optical Microscope
The presence and the surface morphology of the microcapsules in solution were studied via optical microscope (Novex).
FTIR-ATR
FTIR-ATR analyses of the microcapsules, and treated and untreated fabric samples, as well as the raw TTO, were carried out by the transmission method using a Perkin–Elmer spectrophotometer (Spectrum 400) between 400-4000 cm–1. Resolution for the infrared spectra was 4 cm–1, and there were four scans for each spectrum.
SEM
The TTO/EC microcapsules and the treated samples were investigated by SEM (Zeiss Evo LS10). Before the analysis, the samples were rendered conductive with gold.
Antibacterial Activity
The antibacterial activities of the fabric samples, as well as TTO, were investigated against Gram-negative (
The antibacterial activity of TTO and TTO/EC microcapsules were evaluated qualitatively using AATCC TM147 (agar diffusion test). This test is based on the assessment of bacterial growth around and under the samples that were laid into the particular bacterial strain inoculated agar. Bacterial suspensions were prepared in a nutrient broth and diluted with buffer solution at pH 7. The standardized concentration of about 1.5–3 × 105 CFU/mL was applied to agar plates, they were inoculated at 37 °C for 24 h, and then test samples were placed on it. After incubating, the evaluation was carried out by the assessment of bacterial growth under the sample and by the size of the inhibition zone around the sample.13,21
Antibacterial activity of the treated and untreated fabric samples were evaluated quantitatively using AATCC TM100 (shake flask method). In this test, an inoculum with 100 mL of nutrient broth (NB) and incubated for 24 h at 37 ± 1 °C was used. The bacteria concentration was adjusted with NB to 2 × 105 cells/mL. Then, 1 ± 0.1 mL of the diluted inoculum was placed in each sample. The samples were incubated for 24 h at 37 ± 1 °C. After the incubation period, 100 mL of physiological buffer solution (8.0 g of NaCl, 0.2 g of KCl, 1.15 g of Na2HPO4, and 0.2 g of KH2PO4 in 1 L of distilled water) was added and the samples were mixed with a vortexer. To determine the number of living bacteria, the serial dilution plate count method was used. After incubation for 24 h, the percent reduction (R) of bacteria was calculated using the following formula.21,22
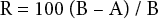
A is the number of bacteria recovered from the inoculated treated test sample in the jar incubated for 24 h, and B is the number of bacteria recovered from the inoculated treated test sample at zero contact time. All measurements were completed in a microbiology laboratory environment at ∼24 °C and 55% relative humidity (RH) and repeated four times.
Results and Discussion
TTO Characterization
The FTIR-ATR spectrum of TTO is given in Fig. 1. The major components of TTO, such as terpinen-4-ol, α-terpineol, γ-terpinene, and α-terpinene have aromatic structures. 10 TTO has many vibrations at 650–1000 cm–1due to the C-O-C aromatic rings. In addition, it has characteristic vibrations at 1000 (C–O–C stretching), 1100 (C-O stretching), 1400 (C=C stretching), 1700 (C=O stretching), 2900 (C-H stretching), and 3400 cm–1 (O-H stretching). 3,10
The FTIR-ATR spectrum of TTO.
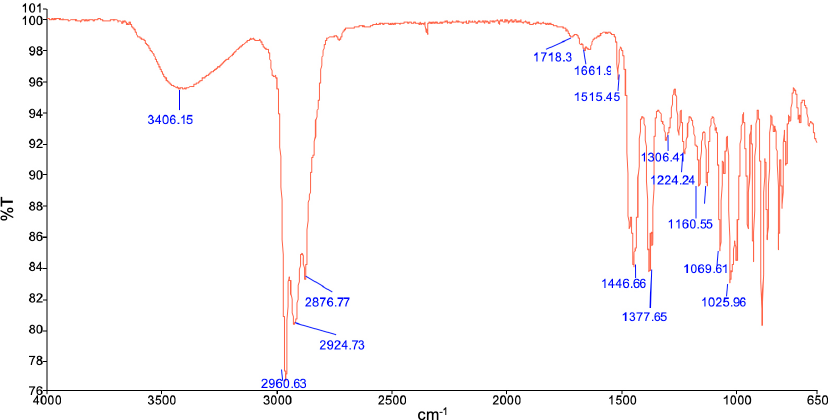
The antibacterial activity results of TTO using AATCC TM147 are given in Fig. 2. The inhibition zones formed by TTO against
The antibacterial activity of TTO against (a)
TTO/EC Microcapsule Characterization
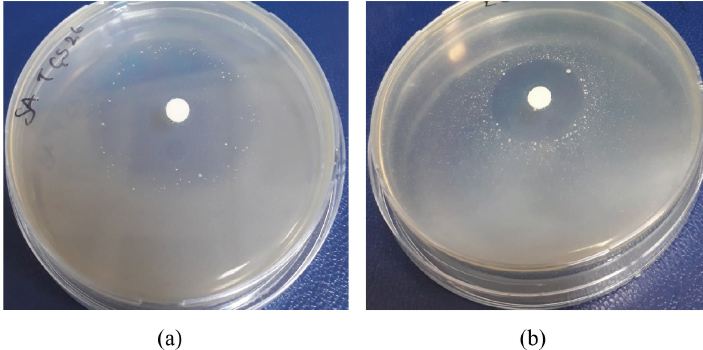
The antibacterial activity results of TTO/EC microcapsules according to AATCC TM147 are given in Fig. 3. The inhibition zones formed by TTO/EC microcapsules against
The antibacterial activity of TTO/EC microcapsules against (a)
The images from the optical microscope, which were taken of the microcapsule solution before filtering, and the SEM of the TTO/EC microcapsules can be seen in Fig. 4. TTO was encapsulated into EC, with 1–12 μm particle sizes. In addition, the microcapsules had spherical shapes and a smooth contour.

Images from the (a) optical microscope and (b) SEM of the TTO/EC microcapsules.
The FTIR-ATR spectra of the TTO/EC microcapsules, comparatively with the core material (TTO) and the wall material (EC), can be seen in Fig. 5. The microcapsules had the important signals of both the core and wall materials. EC had crucial transmittance bands at 3300 (O-H stretching), 2900 cm-1 (C-H stretching), 1370 (C-H stretching), and 1100 cm–1 (C–O stretching) similar to those given in the literature. 23 The TTO/EC microcapsules had more transmittance (%) at 2900 (C-H stretching), 1370 (C-H stretching), and 1100 cm–1 (C-O stretching) due to TTO than did raw EC. Thus, the TTO was well microencapsulated into EC.
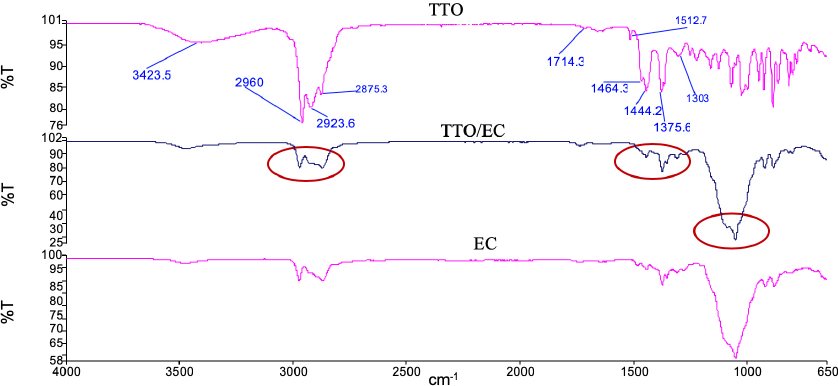
FTIR-ATR spectra of TTO, TTO/EC, and EC.
Characterization of TTO/EC Microcapsule Applied Fabrics
The SEM images of the TTO/EC microcapsule applied fabric sample are shown in Fig. 6, showing that the microcapsules were successfully applied to the textile surface by padding. In addition, the microcapsules penetrated into the fiber surface, with small particles homogenously dispersed.

SEM images of the TTO/EC microcapsule applied sample.
The FTIR-ATR spectra of the TTO/EC microcapsule applied sample compared with the untreated sample, can be seen in Fig. 7. The treated sample had vibration bands at 3300 (O-H stretching), 2900 (C-H stretching), 1600 (H-O-H stretching), 1100 (C-O stretching), and 1000 cm–1 (C-O-C stretching) from the cellulose chain. In addition, the FTIR-ATR spectrum of the treated sample was similar to the untreated one.
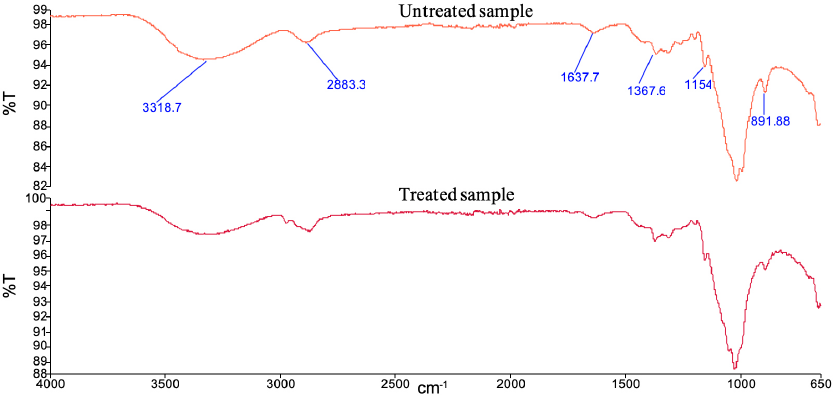
The FTIR-ATR spectra of the TTO/EC treated and untreated fabrics.
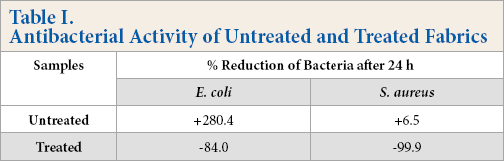
The antibacterial activity of the TTO/EC microcapsule applied sample, as well as the untreated sample, against
Antibacterial Activity of Untreated and Treated Fabrics
Conclusion
The objective of the present research was to produce disposable antimicrobial textiles including microcapsules of tea tree oil/ ethyl cellulose that could be used for medical/cosmetic purposes. For this purpose, TTO/EC microcapsules were prepared and characterized, after the oil was evaluated by FTIR-ATR and antibacterial activity testing. The microcapsules were characterized via optical microscope, SEM, and FTIR-ATR, and then applied to the 100% cotton woven fabrics by padding. The treated fabric samples were then examined by SEM, FTIR-ATR, and antibacterial activity testing.
The raw TTO had high antibacterial activity against
