Abstract
In this study, the second part of a three-part series, exergy analysis of microencapsulation of photochromic dyes by the spray drying method was investigated. Since the spray dryers are thermal systems, energetic examination of the system components is an important issue for sustainable production with high efficiency. Exergy analysis is an important tool used in recent years for the analysis, design, and performance evaluation of thermal systems. The spray drying process was investigated considering two subsystems: the heater and the dryer sections. An exergy model for the process of encapsulation of spirooxazine-based photochromic dye with ethyl cellulose by the spray drying method was proposed. The highest exergy efficiency was achieved at a drying temperature of 120 °C and using an aqueous system as the solvent.
Introduction
One of the main dimensions of sustainability is the efficient use of natural resources. Today, however, fossil fuels account for approximately 85% of primary energy use. 1 Due to the rapid depletion of fossil fuels, and more importantly, the emission of greenhouse gases, the use of fossil fuels should be limited to the maximum extent possible. For this reason, it is of great importance to analyze the energy consumption in all kinds of thermal processes and to determine the production methods with the most efficient energy use without compromising the product quality.
Textile production processes, and especially finishing processes, have high energy consumption. In addition, it is expected that the product’s specific energy consumption will be even higher, especially when it comes to the production of functional and technical textiles as the required processes become more complex.
An interesting area of technical and smart textiles are photochromic textiles. However, direct application of photochromic dyes onto textile materials is problematic due to the limited solubility in water, low temperature resistance, and low fastness. To overcome these problems, encapsulation of photochromic dyes can be used. Although various encapsulation methods are available, spray drying seems to be more suitable for industrial applications.
The spray drying method is a thermal process. Drying is one of the most energy-intensive basic processes due to the high latent heat of evaporation of liquids (especially water), and the use of inefficient hot air as the drying medium in many dryers. 2 Among drying methods, the second most energy-consuming method, subsequent to freeze drying, was reported to be spray drying.3–5 Therefore, to produce energy efficient photochromic microcapsules, the thermodynamic analysis of spray drying for the encapsulation of a photochromic dye should be investigated. However, conventional energy analyses indicate the quantity of the energy; they do not take into account the availability of energy. Energy equations often do not provide information about internal losses and are inadequate in many cases. 6
Recently, exergy analysis has gained importance as a prominent tool for the analysis, design, and optimization of thermal systems. Exergy is defined as “the maximum amount of useful work that can be obtained as the system is brought to equilibrium with the environment.”6–9 Energy is always conserved in a process. Exergy, on the contrary, is destroyed in actual processes. Exergy destruction throughout a process is proportional to the entropy arising due to process-related irreversibilities. Exergy analysis methodology is used to analyze the performance of a thermal system based on the second law of thermodynamics, which measures the quality of the energy forms interacting with the system. In this way, it displays the inefficiencies of the system or each of its sub-components;7,10,11 it can be used for a clearer identification of the causes, locations, and magnitudes of the inefficiencies. 7
Considering spray drying, studies on exergy analysis were quite limited. Aghbashlo et al.12–16 investigated the energy and exergy analyses and optimization of fish oil microencapsulation by spray drying in a series of studies. The authors examined the parameters such as drying air temperature, aspirator ratio, feed solution mass flow rate, and spraying airflow rate; a high improvement potential rate was reported, although the exergy efficiency of the system was low. Erbay and Koca17,18 proposed an exergy and exergoeconomic analysis methodology for white-cheese slurry drying with a pilot-scale spray dryer. The effects of atomization pressure and inlet–outlet air temperature on exergetic parameters were investigated. Jin and Chen 19 conducted the second-law analysis of the industrial-scale spray drying process for milk powder production. Saygi et al. 20 analyzed the spray drying of fruit puree; the exergy efficiency of the system was reported to be between 0.7% and 4.1%. The only study in the literature for exergy analysis of photochromic microcapsule production by spray drying is the preliminary study 21 presented by the authors of the present work. In that study, the exergy destruction of the spray drying system was quite excessive, and the exergy destruction of the heater sub-section was found to be higher compared to the dryer. The sustainability index of the system was also very low, which demonstrated the importance of increasing the efficiency of the spray dryers. On this account, the effects of selected spray drying parameters at which regular photochromic microcapsule formation was obtained on the exergetic parameters were investigated for the first time in the present study. This was done to put forth the most efficient process conditions for photochromic microcapsule production.
System Description and Exergy Analysis
Photochromic microcapsules were obtained using ethyl cellulose (Aquacoat ECD, 27% w/w) as shell material and a spirooxazine-based photochromic dye as core material in a laboratory scale spray dryer (Buchi B-290). Detailed information on the encapsulation method and parameters was given in the previous study. 22 Briefly, in spray drying, the drying air was first heated up to the drying temperature with an electric heater. Then, the solution including ethyl cellulose and photochromic dye, which was sprayed with compressed air from the nozzle in the drying part, came into contact with the drying air. During drying, the solvent in the sprayed solution was evaporated to form the microcapsules. The microcapsules obtained were collected in the bottom chamber of the device, and damp drying air was exhausted with an aspirator. For exergy analysis, the spray dryer was separated into two sub-components, namely, the heater and the drying chamber, as shown in Figure 1.
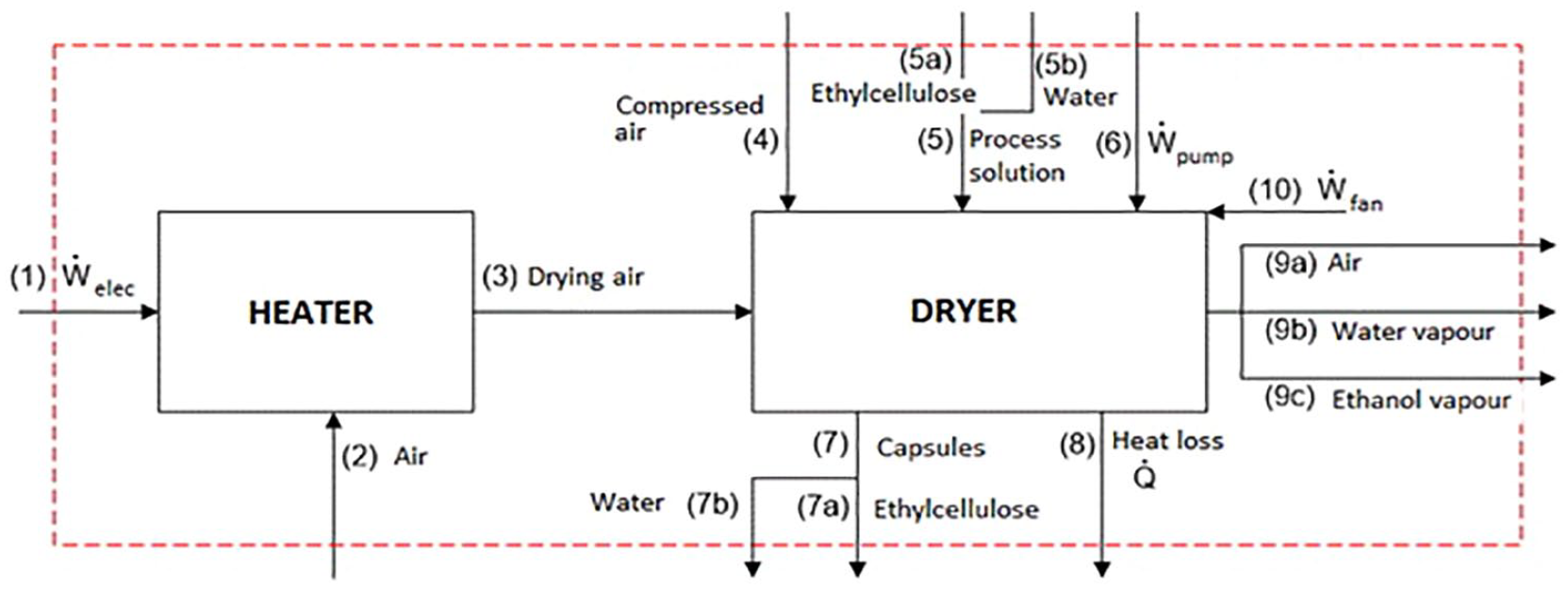
Control volume for spray dryer with coded streams used in equations.
The following assumptions were made for the analysis:
All processes are steady state and steady flow with negligible kinetic and potential energy effects.
Heat transfer to the system and work transfer from the system are positive.
Air is an ideal gas with a constant specific heat.
The temperature and pressure of the reference state are taken as the actual average ambient conditions (24 °C, 101.325 kPa) during drying experiments.
Chemical exergy changes are neglected.
Specific heats are assumed to be constant (
General mass, energy, and exergy balances of steady-state steady-flow systems were applied for exergy analysis.
The specific physical exergy is calculated in equation (1):

where
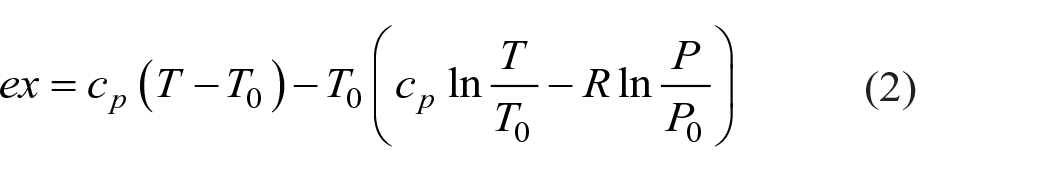
Specific enthalpy and entropy of water vapor and ethanol vapor were calculated by equations (3) and (4).
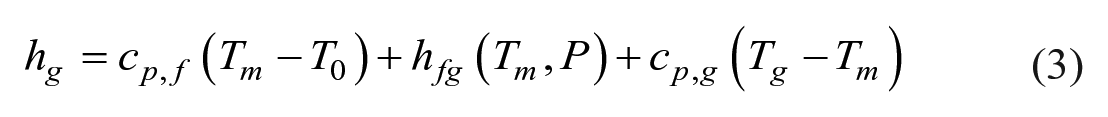
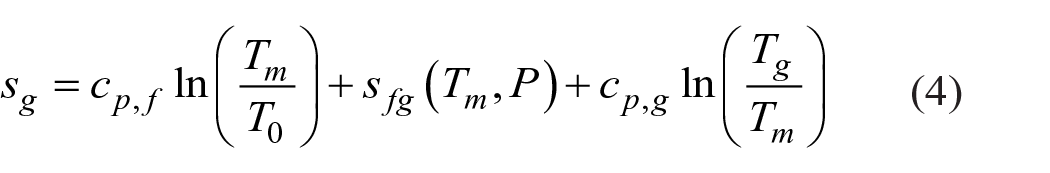
The total solid content in the encapsulation solution and the resultant microcapsules contains ethyl cellulose and photochromic dye. While the specific heat of ethyl cellulose is known, that of the photochromic dye is unknown. On the contrary, it is stated in the literature that the change in the solid content in the spray drying process had an insignificant effect on the exergy parameters of the system.15,20 Therefore, the specific exergy change of the photochromic dye during the process was neglected.
The exergy rate of each stream was calculated according to equation (5) and the exergy balance equations for the heater and dryer of the spray dryer were calculated as in equations (6) and (7), respectively, using the codes of each stream given in Figure 1.

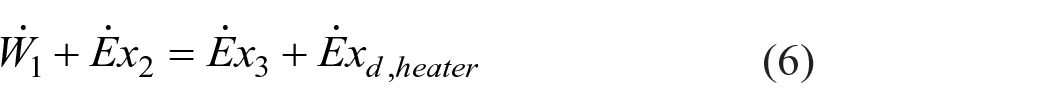
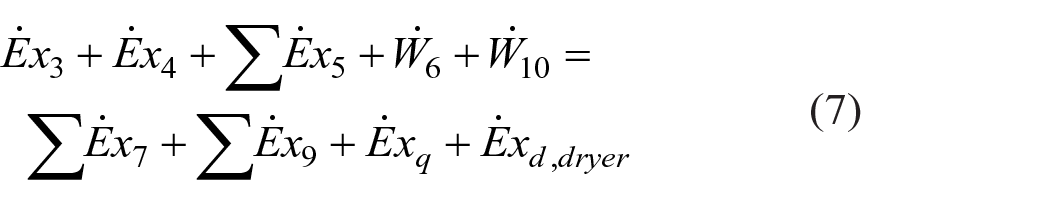

For the exergy efficiency of the spray dryer, both total system exergy efficiency and exergy efficiencies of each subsystem (heater and dryer) were calculated and discussed. The product (
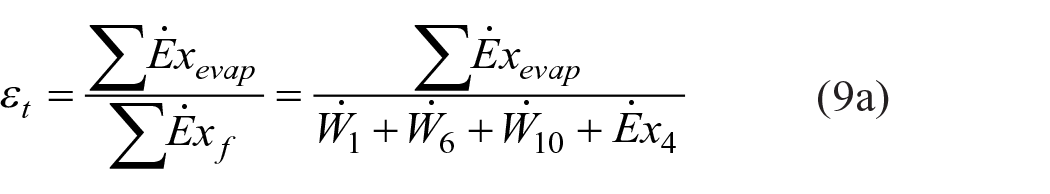


Exergy efficiencies of the heater and dryer were calculated according to equations (10) and (11), respectively:


Energy efficiency of the system was calculated by the ratio of the energy required for the evaporation of the solvent to the energy inlet to the system as in equation (12):
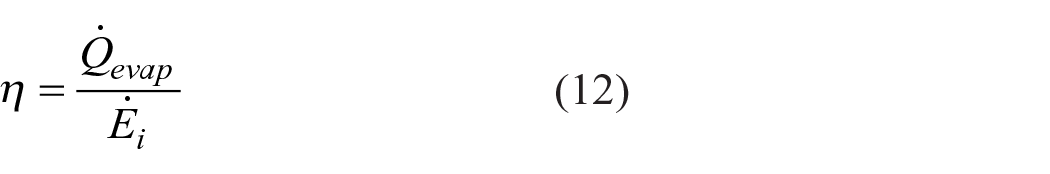
The aforementioned exergy analysis model was used to investigate the effects of spray drying parameters on the exergy destruction rate and exergy efficiency of both the total system and each subsystem. For this purpose, the main process parameters were selected to be the spray drying air temperature, aspirator rate (corresponds to drying air flow rate), microencapsulation solution mass flow rate, ethyl cellulose concentration, and water:ethanol ratio of the encapsulation solution. The selection values of these parameters were made according to the previous study, 22 considering the values at which a regular microcapsule formation was obtained. Parametric values for which capsule formation cannot be achieved were not taken into account. Accordingly, the parametric data used for photochromic microcapsule production in this study are listed in Table 1.
Spray drying parameters used for exergy analysis.

1.0/0.4 ratio of water–ethanol was used for solution preparation to completely dissolve the photochromic dye, and ethanol was fully evaporated before spray drying. 22
Flow rates of drying air, compressed air, and microencapsulation solution were obtained from the device catalog 24 considering the machine settings. The inlet and outlet air temperatures of the drying chamber were obtained directly from the screen of the device. Spray dryer surface temperatures and material temperatures were measured with an infrared (IR) thermometer (Proscan 520, TFA-Dostmann). The remaining data were calculated by the energy balance equations based on the actual operational data.
Results and Discussion
Effects of System Parameters on the Exergy Destruction Rate
Figure 2 illustrates the effect of drying air temperature on the exergy destruction rate (at an aspirator ratio of 95% and ethyl cellulose concentration of 3%). As the drying air temperature increased, exergy destruction rate increased linearly. In general, it was found that the exergy destruction rate in the heater was higher than that of the dryer subsystem. When the system sub-components were examined, it was seen that the main subsystem affected by the increase in drying air temperature in terms of the exergy destruction rate was the heater, due to the conversion of electrical energy to low-grade thermal energy. On the contrary, although the exergy destruction rate of the dryer increased depending on the drying air temperature, this increase was not significant, since the process solution flow rate in the drying section was fixed. In normal drying processes, drying accelerates as the drying air temperature increases, therefore energy efficiency can be increased by working at higher material flow rates. However, in the case of photochromic microcapsules, it was observed that microcapsule formation could only be obtained at a fixed solution flow rate. 22 Therefore, the flow rate of the microencapsulation solution was constant since there was a restriction of capsule formation. Since the drying air outlet temperature also varied proportionally to the air inlet temperature, the increase in the drying air temperature in the dryer subsystem did not affect the exergy destruction rate significantly. In addition, it was observed that the use of water or water–ethanol mixture for the photochromic dye solution did not have a significant effect on the exergy destruction rate. The exergy destruction rate of the heater for both solvents was the same since the same mass flow rate of air was heated up to drying temperature.
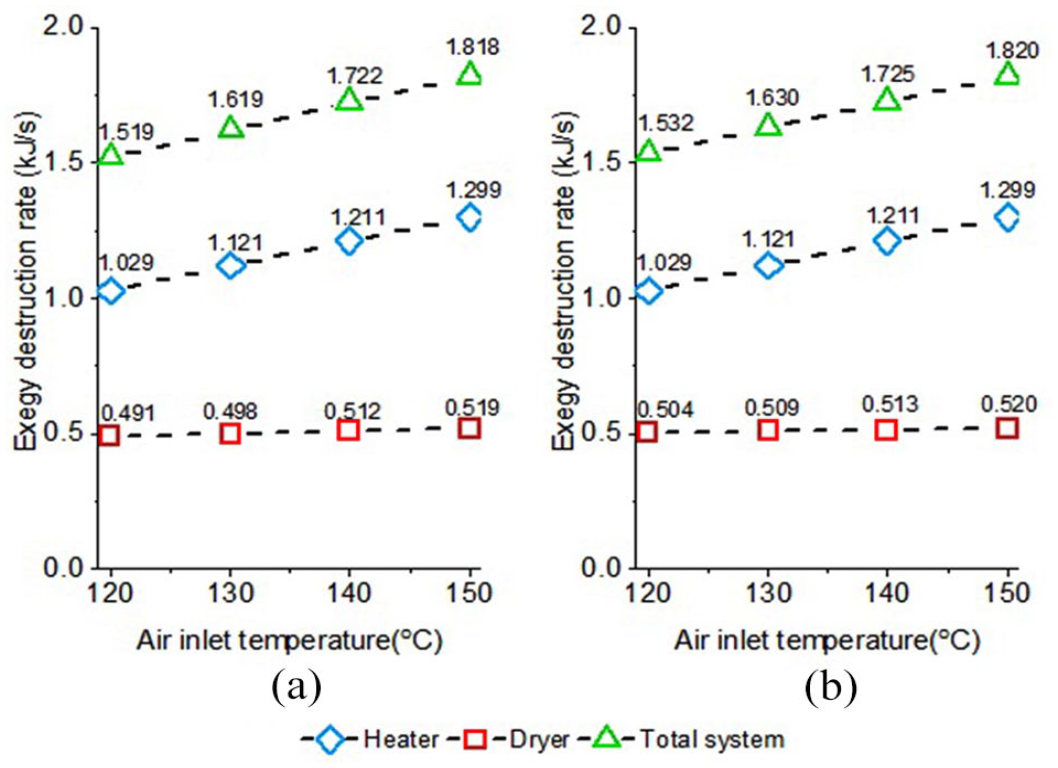
Effects of drying air temperature on exergy destruction rate: (a) photochromic dye water–ethanol solution and (b) photochromic dye aqueous solution.
Figure 3 shows the effects of aspirator ratio on the exergy destruction rate (at 150 °C drying air temperature, 3% ethyl cellulose concentration, and a water:ethanol ratio of 0.8:0.2). The drying air was sucked or blown through the device by the aspirator motor. The amount of drying air can be increased or decreased by regulating the aspirator speed. 25 Therefore, the aspirator ratio is directly related to the airflow rate that leaves the system, and therefore enters the system. As the aspirator ratio decreases, the mass flow rate of the air inlet decreases. In this case, less electrical power will be used in the heating subsystem to heat this lesser amount of air required for drying. As this reduces the exergy rate input to the system, the exergy destruction rate decreases. On the contrary, as in the drying air temperature investigation, the aspirator ratio was an insignificant factor on the dryer subsystem.
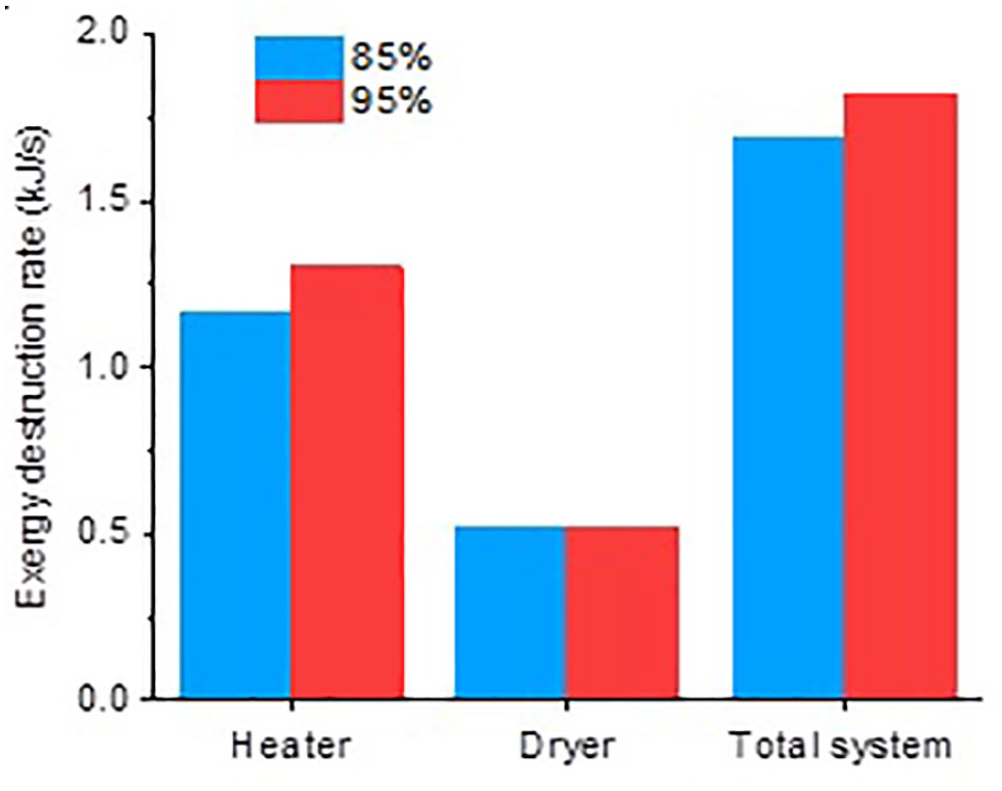
Effects of aspirator ratio on exergy destruction rate.
As shown in Table 1, the flow rate of the microencapsulation solution was kept constant. Also, although the concentration of ethyl cellulose changed, the total amount of solids in solution (ethyl cellulose, photochromic dye, and auxiliary agents) remained constant. Therefore, the amount of water (and ethanol) in each solution did not change. As a characteristic of the drying processes, except heat losses, almost all of the thermal energy was used to evaporate the solvent (here water and ethanol). Hence, in the present range of process parameters, there was no significant change in the inlet and outlet energy/exergy rates of the system. In consequence, the change in the concentration of ethyl cellulose in the microencapsulation solution had no significant effect on exergy destruction.
Effects of System Parameters on Energy and Exergy Efficiency
Exergy efficiency was found to be lower than energy efficiency as expected. Depending on the process parameters, the energy and exergy efficiencies of the total system were 5.28%–7.81% and 3.40%–4.37%, respectively. This indicated that the exergy efficiency of photochromic dye microencapsulation by spray drying was quite low. The high latent heat of vaporization of liquids, and for convective dryers such as spray drying, the inefficient air-to-material heat transfer and exhaust air loss 26 make the drying processes energy-intensive. Moreover, for spray drying, the use of high drying temperature 12 and the need for evaporation of a large amount of water in the solution 27 led to a high-energy/exergy input, which caused low exergy efficiency. The present exergy efficiency values were found to be compatible with the results of the studies on spray drying in the literature.12,13,18,20 Given the low efficiency, any exergetic improvement to be performed was very important. In this context, the first action to be taken is to examine and optimize the process parameters, which is also the aim of this study. Other potential measures include exhaust gas heat recovery and drying air recirculation.27,28
Figure 4(a) shows the effect of drying air temperature and solvent system on total system energy and exergy efficiency (using an aspirator ratio of 95% and an ethyl cellulose concentration of 3%). In addition, a subsystem exergy efficiency investigation depending on the drying air temperature is shown in Figure 4(b). First, when the system sub-components were examined, it was observed that the exergy efficiency of the heating part increased and the exergy efficiency of the drying part decreased as the drying air temperature increased. Drying air with higher temperature gives a higher exergy content. Therefore, at higher drying temperatures, electrical power was converted to a higher-grade thermal energy compared to low temperature. For this reason, the exergy efficiency of the heating section was increased by the drying air temperature. On the contrary, since the encapsulation solution feeding rate was constant, as the drying air temperature increased, the same amount of water (and ethanol) was evaporated with higher-grade energy, therefore the exergy efficiency of the drying subsystem decreased with increasing temperature.
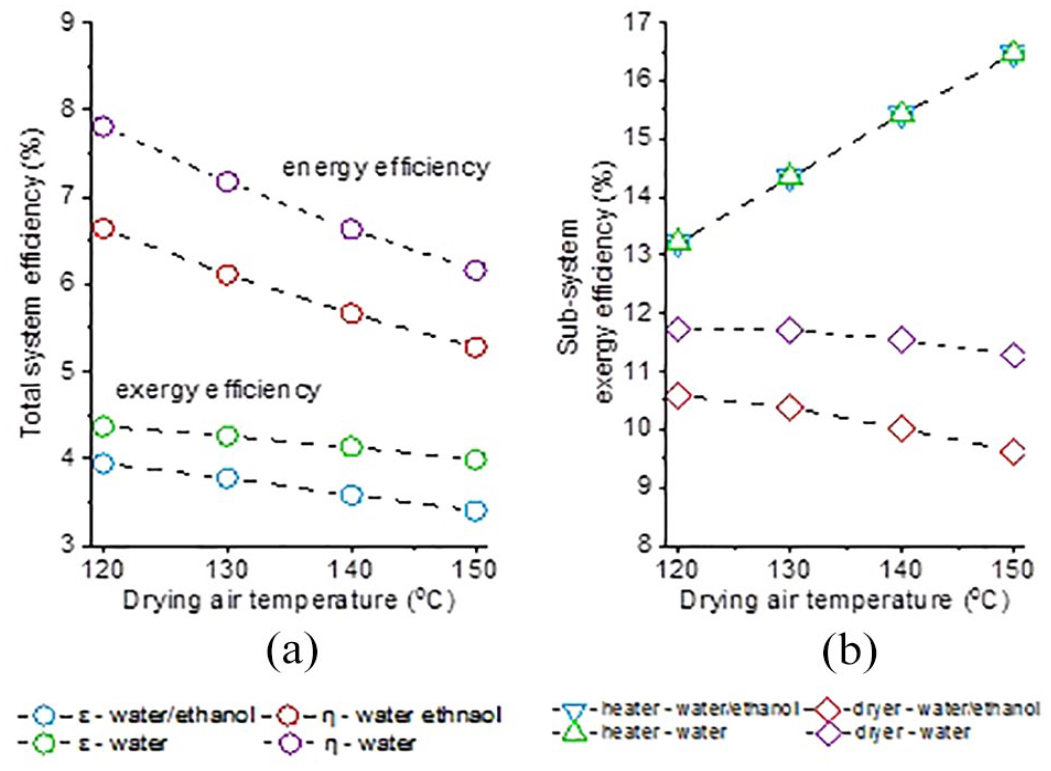
Effects of drying air temperature and solvent system on energy and exergy efficiency: (a) total system energy and exergy efficiency and (b) subsystem exergy efficiency.
Total system energy and exergy efficiency decreased as the drying air temperature increased, as in the dryer subsystem. In the literature, the effect of drying temperature differed for different drying systems. In the studies of Erbay and Koca 18 and Saygi et al., 20 the increase in the drying temperature caused an increase in exergy efficiency, which was associated with the increase in the drying rate. On the contrary, as in the studies of Aghbashlo et al.,12,13 when the feed rate of the spray solution was kept constant, higher drying air temperatures resulted in a decrease in exergy efficiency. This was consistent with the results of the present study. Exergy efficiency, in this study, is the ratio of exergy required to evaporate water (and ethanol) to exergy input. Therefore, exergy efficiency is inversely proportional to the drying air exergy rate. 29 In the analyzed system, as the drying air temperature increased, more exergy input was used to evaporate the same amount of water (and ethanol)—the amount of solvent is constant as the solution flow rate cannot be changed—which means a higher exergy destruction and also a nonessential use of exergy, most of which will be lost with the exhaust air. Therefore, it has been demonstrated that in such cases where the flow rate of the process solution must be kept constant, higher drying temperatures will be an inefficient application for spray drying. On the contrary, it should be noted that the resultant microcapsule diameter is closely related to the drying air temperature and for photochromic dye microencapsulation a decrease in the spray drying air temperature led to an increase in the mean diameter of microcapsules. 22
Although the change in exergy efficiency dependent on the drying air temperature seemed to be quite low numerically, it should be noted that this change was significant given the very low exergy efficiencies of the drying process. 30 For example, when the drying temperature decreased from 150 °C to 120 °C, the exergy efficiency increased by about 16% for encapsulating with a process solution containing water–ethanol. This increase was considered to be important for drying systems.
Another result drawn from Figure 4 is that when aqueous ethyl cellulose solution was used, total energy and exergy efficiency was higher compared to water–ethanol mixture. Latent heat of evaporation of ethanol is much lower than that of water, therefore less energy would be adequate to evaporate ethanol. Since the exergy input was the same in both cases, evaporation of the water–ethanol mixture will be more inefficient. Moreover, it was found that the effects of the change in drying air temperature was found to be more pronounced for photochromic dye water–ethanol solutions. For example, when the drying air temperature was reduced from 150 °C to 120 °C, the exergy efficiency of the aqueous system increased by 9.5% while that of the water–ethanol system increased by 16%. Therefore, lower drying temperatures should be selected for water–ethanol mixture. When subsystems were examined, the same tendency was observed for the drying subsystem. However, as expected, the exergy efficiency of the heater was not affected by solution solvent type (water or water–ethanol mixture), since the air flow rate and temperature were constant for each comparison.
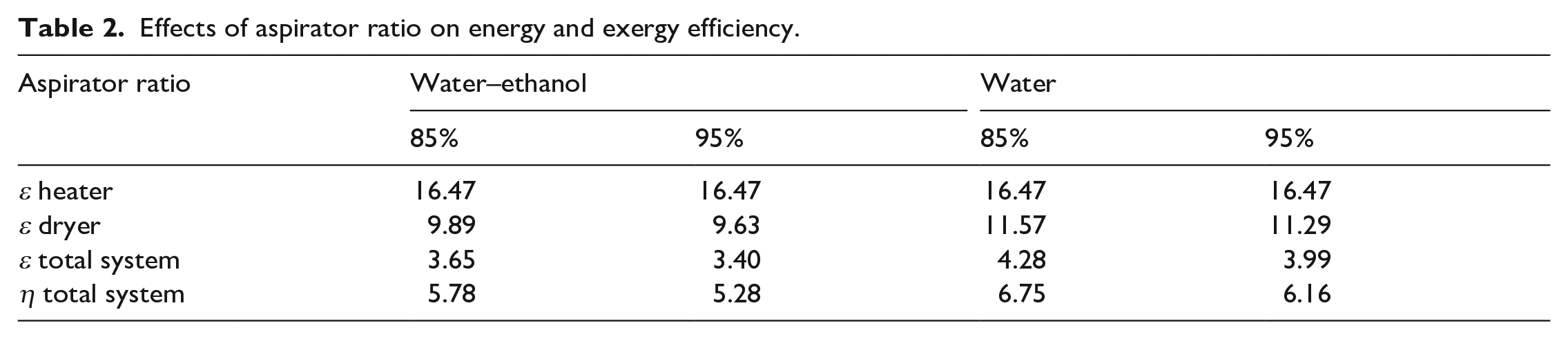
The effect of the aspirator ratio on energy and exergy efficiency is shown in Table 2 (150 °C drying air temperature, 3% ethyl cellulose concentration). No effect was observed in the heater subsystem by the change in the aspirator ratio since the drying temperature was kept constant, whereas, for the dryer subsystem, the exergy efficiency increased by the decrease in aspirator ratio, since the same amount of water (and ethanol) was evaporated with less air. This also applied to total system energy and exergy efficiency. The rate of exergy efficiency increase according to the aspirator ratio did not differ for cases where a water or water–ethanol mixture was used as solvent.
Effects of aspirator ratio on energy and exergy efficiency.
The effect of ethyl cellulose concentration on energy and exergy efficiency was insignificant. For example, for ethyl cellulose concentrations of 3% and 4%, the energy efficiency was found to be 5.28% and exergy efficiency was 3.40% and 3.41%, respectively (at 150 °C drying temperature, 95% aspirator ratio, and water–ethanol mixture).
Effects of System Parameters on Exergy Loss Rate
In thermal systems, besides exergy destruction rate and exergy efficiency parameters, exergy losses are also important. Exergy losses are the waste exergy rate streams via material streams or heat transfer to the surroundings. In the analyzed system, exergy loss rate streams arose by exhaust air containing water (and ethanol) vapor discharged from the chimney and heat transfer from the device surface. Dincer 31 reported that the exergy rate lost by heat transfer can be up to 25% of total exergy input for industrial spray dryers.
Figure 5 illustrates the change in exergy loss rate and exergy loss ratio (percentage of losses in exergy input) depending on the drying air temperature (aspirator ratio 95%, ethyl cellulose concentration 3%, and aqueous system). In the figures,
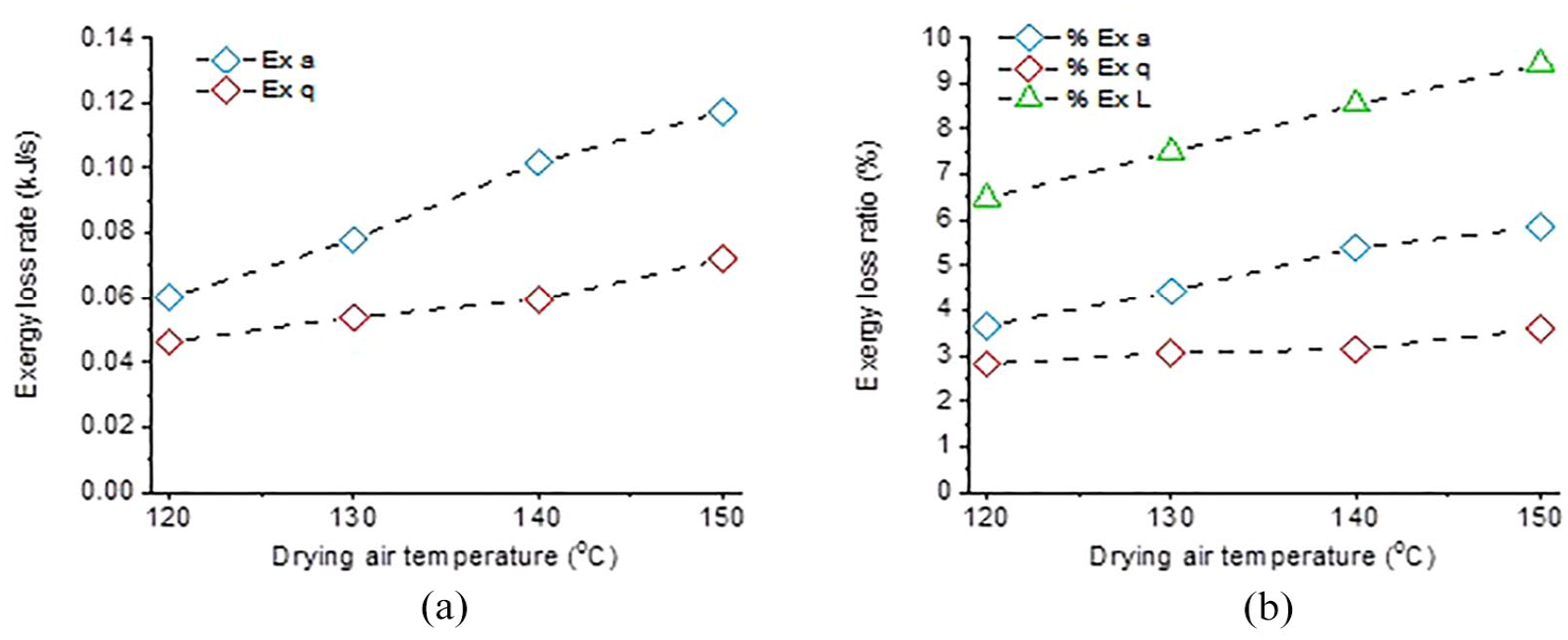
Effects of drying air temperature on (a) exergy loss rate and (b) exergy loss ratio.
Reduction of the aspirator ratio also reduced total exergy losses. However, the effect here is not as significant as the change in drying air temperature (exergy loss ratio for 95% and 85% aspirator ratios were 9.4% and 9.0%, respectively), due to the small difference in the selected aspirator ratio values.
The general results obtained as a result of exergy analysis for the encapsulation of photochromic dyes by spray drying are summarized in Table 3. The selection of lower drying temperature, lower aspirator ratio, and the use of an aqueous solvent system would be beneficial for the exergy efficient production of photochromic microcapsules by spray drying.
Summary of exergy analysis results.
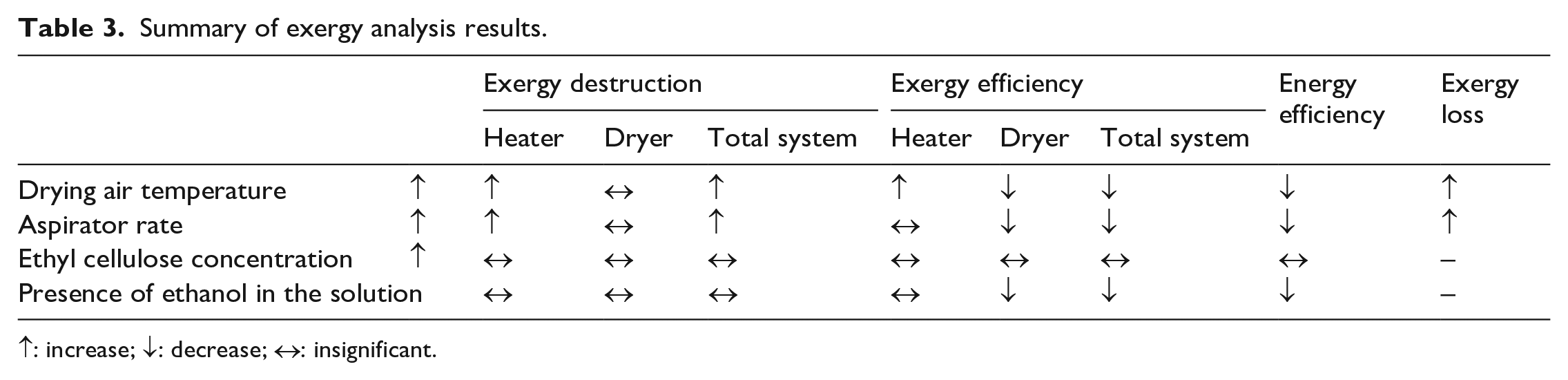
↑: increase; ↓: decrease; ↔: insignificant.
Conclusion
Exergy analysis of spray drying for the production of photochromic microcapsules with ethyl cellulose was presented. An exergy model considering two subsystems, the heater and dryer, was proposed. The effects of drying air temperature, aspirator ratio, ethyl cellulose concentration, and the type of solvent used in the encapsulation solution on the exergetic parameters, such as exergy destruction rate, exergy efficiency, and exergy loss rate, were investigated. Exergy destruction of the system was found to be affected by the drying air temperature and aspirator rate. The total system energy and exergy efficiency were found to be between 5.28% and 7.81%, and between 3.40% and 4.37%, respectively. The main parameters affecting the exergy efficiency of the system were found to be the drying air temperature, aspirator rate, and the type of solvent. Considering the investigated process parameters at which regular microcapsule formation was obtained, the selection of a drying air temperature of 120 °C, an aspirator ratio of 85%, and the use of aqueous photochromic dye solution resulted in the highest exergy efficiency. These results are thought to be a guide in the production of photochromic textiles to ensure a sustainable production with the efficient use of energy in addition to quality parameters.
Footnotes
Appendix 1
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to gratefully acknowledge The Scientific and Technical Research Council of Turkey (TUBITAK) through Project No. 214M428 for financial support of this research project.
