Abstract
The magnetic ferrites were synthesized at ambient temperature through the precipitation method in aqueous solution at varying pH values and were used as novel adsorbents for heavy metal-containing wastewater treatment. The magnetic ferrites were applied for the removal of Cd(II) ion from wastewater. The synthesized magnetic ferrites were characterized by settling velocity, X-ray diffraction, scanning electron microscopy, laser particles size analyzer, and vibrating sample magnetometer. The effects of pH value and contact time on the adsorption process were investigated. The magnetic ferrites had a saturation magnetization value of 82.30 emu/g and a settling velocity of 2%, indicating easy separation from aqueous solution under magnetic field. The adsorption of Cd(II) onto the magnetic ferrites followed the pseudo-second-order kinetics and the Langmuir isotherm model. The most suitable pH condition for the synthesis of magnetic ferrite with optimal Cd(II) adsorption capacity was 9.0, and a maximum adsorption capacity of 160.91 mg/g for Cd(II) ions can be achieved. Based on the cost analysis, the magnetic ferrite was a cost-effective adsorbent for Cd-containing wastewater treatment.
Introduction
Cadmium-containing wastewater is generated in industrial operations such as mining and electroplating industries as well as the manufacture of fertilizers, batteries, paints, and pigments (Hu et al., 2011; Jian et al., 2009; Yan et al., 2017). Cadmium has been considered as one of the most harmful metals due to their high toxicity, low degradability, and high stability. Moreover, cadmium in aqueous environments tends to be accumulated and can cause several diseases and health disorders in humans and other living organisms (Hu et al., 2011; Ozay et al., 2009; Wang et al., 2011). The USA Environmental Protection Agency suggested an allowed amount of 0.005 mg/l for cadmium in drinking water (Wang et al., 2011). Thus, the treatment of wastewater contaminated by Cd(II) has always been a serious environmental issue.
Various physical and chemical strategies have been developed to treat Cd-containing wastewater, such as precipitation, ion exchange, and adsorption (Chen et al., 2013). Precipitation and ion exchange methods are expensive and may cause secondary pollution, while adsorption has been recognized as an effective and economic method (Chai et al., 2013). Since the adsorption efficiency mainly depends on the adsorbent properties, the selection of suitable adsorbent is essential. Currently, there are many researches focusing on the adsorbents for cadmium removal (Chen et al., 2011), including zeolite (Hao, 2012), red mud (Vaclavikova et al., 2005), chitosan (Zhang et al., 2011), resin (Liu et al., 2009), and activated carbon (Rao et al., 2006). However, these materials have some inherent shortcomings in their applications, such as low adsorption capacity, poor adsorption efficiency, or separation problems.
In recent years, there is an increasing interest in magnetic ferrite-based adsorbents due to their high adsorption capacities and unique magnetic properties for easy separation (Chen et al., 2009; Ren et al., 2017). The applications of magnetic ferrites have been expanded to permanent magnets, wastewater treatment, catalysts, and magnetic fluids (Berkovsky et al., 1993; Goldman, 1990; Kitamoto et al., 1999; Socoliuc et al., 2003). Magnetic ferrites can be produced directly from aqueous solutions as well as by solid-state reactions at high temperature. Conventional preparation of magnetic ferrite in aqueous system by coprecipitation methods requires the heating of the mixture up to 90°C (Hasegawa and Sato, 1967; Sato, 1970; Sugimoto and Matijević, 1980) or autoclave temperatures (Kunda and Rudyk, 2013). Moreover, these products must be annealed at temperatures between 500 and 1100°C to be crystallized (Gotic et al., 1998; Li et al., 1996; López et al., 1998). In the formation of magnetic ferrite for heavy metal removal from wastewater, the operation temperature has to be above 60°C (Okuda et al., 1975; Tamaura et al., 1991).
Currently, most researches only focused on the formation of various metal-bearing ferrites in aqueous system at high temperature or ambient temperature (25°C) (Perales Perez and Umetsu, 2000; Perales Perez and Umetsu, 2002; Perales Perez et al., 1998), while very few literature reported the optimization of other conditions for the synthesis of magnetic ferrites, i.e. the pH value and the oxygen supply. Specifically, solution pH value has a great impact on the synthesis of magnetic ferrites at ambient temperature, and thus affect the performance on the treatment of cadmium-containing wastewater.
Therefore, the objective of this work is to explore the optimal condition for the preparation of magnetic ferrite at ambient temperature for Cd(II) removal from wastewater. The magnetic ferrite was synthesized through precipitation method at four different pH conditions and were characterized by X-ray diffraction (XRD), scanning electron microscope (SEM), laser particle size analyzer (LPSA), and vibrating sample magnetometer (VSM). Their adsorption capacities toward Cd(II) were compared, and the effects of solution pH, adsorbent dosage, and contact time on the adsorption process of Cd(II) ions were also explored. In addition, the kinetics and isotherm studies on Cd(II) adsorption by the magnetic ferrites were also investigated to study the adsorption mechanisms.
Materials and methods
Materials
FeSO4·7H2O, NaOH, HCl, CdSO4, EDTA, zinc powder, sulfosalicylic acid, hexamethylenetetramine, and xylenol orange indicator were purchased from Hunan Chemical Reagent Company (Hunan, China). All the reagents were of analytical grade and were used without further purification. Double-distilled deionized water was used throughout the experiments.
Synthesis of the magnetic ferrite
At room temperature 25°C, four equivalent amounts of distilled water (1.5 l) were added into 2 l flat-bottom flasks. The flasks were purged with nitrogen for 10 min to remove dissolved oxygen from the water and then plugged with the rubber toppers to maintain a low level of oxygen. Ferrous sulfate weighing 7.5 g was dissolved in the distilled water with stirring. Then the solution pH values were adjusted by 1 mol/l NaOH and HCl to 9.0, 10.0, 11.0, and 12.0, and the magnetic ferrite products were labeled as F9, F10, F11, and F12. After the reaction was completed, the black precipitates were filtered and collected. The precipitates were washed repeatedly with ethanol and water and then dried in a vacuum oven at 60°C for the subsequent analyses.
Analysis of magnetic ferrites
The content of Cd(II) ions in the supernatant was analyzed by a flame atomic adsorption spectrophotometry. The crystal structures of the magnetic ferrites were examined by a MSAL-XD2 X-ray diffractometer, using Cu Kα radiation (λ = 0.1541 nm) in the range of 10–80° (2θ) at 30 kV and 30 mA. The morphology and size of the magnetic ferrite particles were characterized by a SEM microscopy (JSM-6360LV) at an electron acceleration voltage of 20 kV and LPSA, respectively. The magnetic intensity was measured by a VSM. The settling ability was determined by the settling velocity (SV) and the Fe2+/Fe3+ ratio was measured by the complexometric method.
Adsorption procedure
The adsorption of Cd(II) ions by the magnetic ferrites was performed in aqueous solution at 25°C and pH 8.0. The stock solution of Cd(II) ions (1000 mg/l) was prepared by dissolving an appropriate quantity of CdSO4 in double-distilled water, and a series of artificial wastewater with various initial Cd(II) concentrations ranging from 25 to 150 mg/l were obtained by diluting the stock solution into different extents. A fixed quantity of magnetic ferrite and artificial wastewater with varying concentrations of Cd(II) ions was added into 250 ml flasks and stirred throughout the experiment. The concentration of Cd(II) ions in the supernatant was measured using a inductively coupled plasma-optical emission spectroscopy to provide the residual concentration in solution. The removal rate (η) and the adsorption capacity (Qe) were calculated by the following equations


Results and discussions
Characterization of the magnetic ferrites
The SV is one of the most important indexes to reflect the sedimentation property of the precipitate in an aqueous environment. Here, SV was used to evaluate the settling capacity of the magnetic ferrites in water and to provide information about the compaction degree of the magnetic ferrites. Lower SV means that the magnetic ferrites were formed better. The SV was measured every 5 min during 30 min. The results are given in Figure 1.

The SV of the four ferrites.
As shown in Figure 1, the settling velocities of the four magnetic ferrites all decreased with time. A rapid decrease was observed in the SV during the first 15 min, and then the SV decreased slower and eventually reached equilibrium. The final values were 2, 31, 10, 17% for F9, F10, F11, and F12, respectively. Within 30 min, the SV of F9 was lower than that of the others, indicating that the sedimentation property of F9 was the best, and the formation of F9 was better than the other three magnetic ferrites.
The morphology, crystal structure, and size of the as-synthesized magnetic ferrites were characterized by SEM, XRD, and LPSA analyses, respectively. The results are shown in Figure 2.

(a–d) SEM, (e) XRD, and (f–i) LPSA analyses of magnetic ferrites.
Figure 2(a) to (d) shows the SEM images of the four magnetic ferrites, and the particles of the magnetic ferrites were in the nanometer range, which was beneficial for equal aggregation in an aqueous environment. The XRD pattern of the magnetic ferrites (Figure 2(e)) shows 10 relatively strong reflection peaks at (111), (220), (311), (222), (400), (422), (511), (440), (620), and (553), all of which could be well indexed to the cubic inverse spinel structure for the magnetic ferrites (Su et al., 2010). The reflection peak positions and relative intensities for the ferrite suggested that the synthesized nanoparticles were Fe3O4 (Barakat and Schmidt, 2010; Maity and Agrawal, 2007). Furthermore, as shown in Figure 2(f) to (i), the particle size range distributions of the magnetic ferrites were 25–275 nm for F9, 50–550 nm for F10, 50–350 nm for F11, and 150–650 nm for F12, respectively. This also suggested that the particle sizes of the magnetic ferrites were uniformly distributed in the nanometer range, and F9 was more granular than the others.
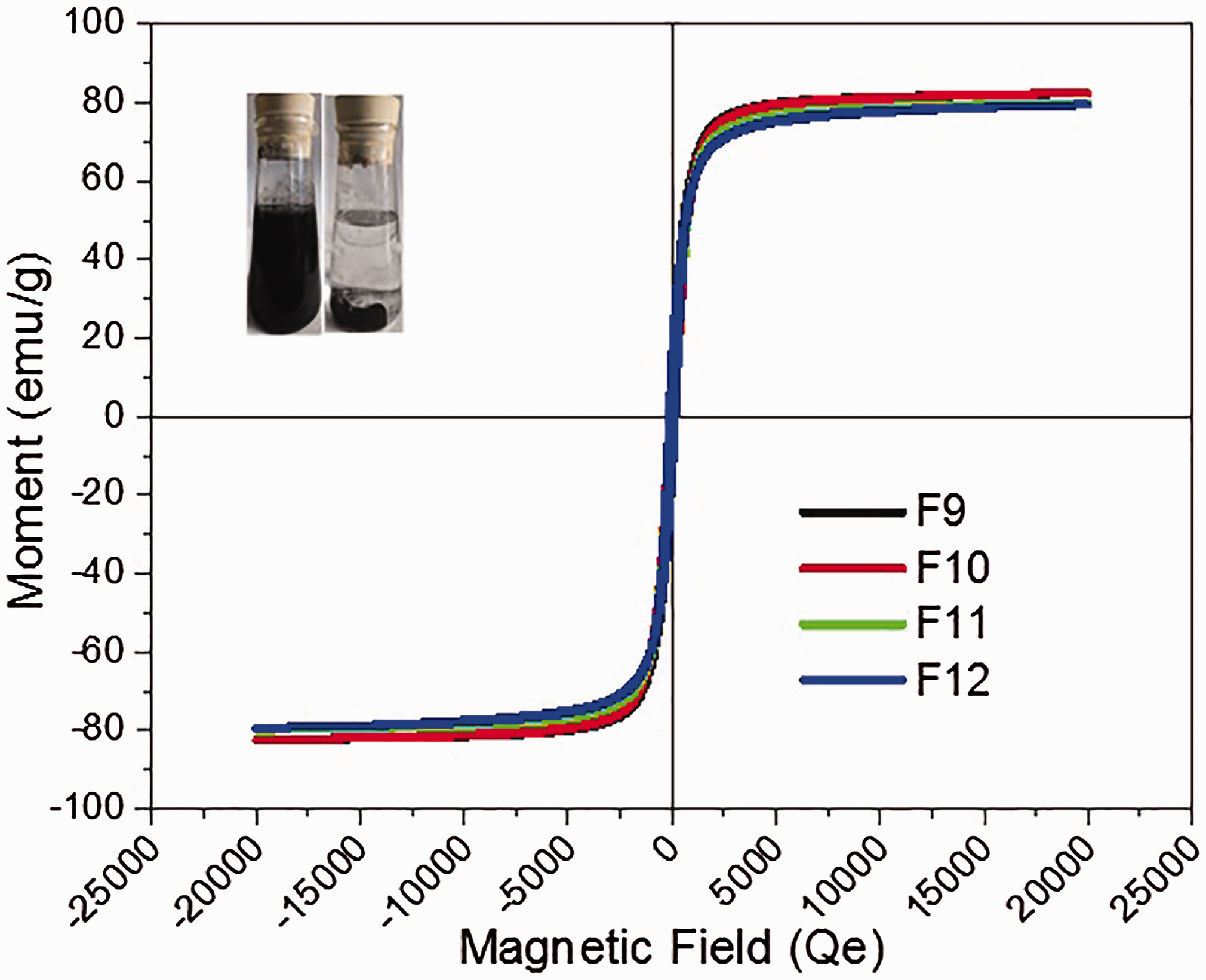
Magnetization curve of the four as-synthesized ferrites.
Solid–liquid separation is usually an arduous and costly issue in the treatment of complex wastewater containing heavy metal, and magnetic separation may offer a rapid and effective method to solve this problem. Magnetism is the unique feature of the magnetic ferrite. The magnetic property of the ferrites was measured by VSM at room temperature and the results are shown in Figure 3.
As calculated from Figure 3, the saturation magnetization values of the four ferrites were 82.30, 82.25, 79.79, and 79.44 emu/g for F9, F10, F11, and F12, respectively. This result demonstrated that the magnetic ferrites were essentially superparamagnetic. Therefore, the nanoparticles dispersed in water can be easily collected by an external magnetic field within several minutes and then can be readily redispersed upon shaking slightly. These results demonstrated that the particles exhibited good magnetic response and redisperse property, suggesting that the magnetic ferrite nanoparticles could be used as a potential adsorbent.
Cd(II) adsorption
Effect of pH
In the adsorption process, solution pH plays a key role in determining the charge on the surface of the adsorbent and the speciation of the metals in solution (Cornell and Schertmann, 1991). Thus, 0.2 g of magnetic ferrites was added into 200 ml of Cd(II) ion solution (100 mg/l) at different pH values and stirred for 1 h at 25°C. Since the point of zero charge of Cd(II) is 6.5–7.8 in solution (Mohapatra and Anand, 2007), the pH range of the solution for Cd adsorption was chosen as 6.0–9.0 according to the Eh–Ph property of cadmium and the pH range of natural water. The influence of pH condition on the adsorption of Cd(II) ion to the magnetic ferrites is shown in Figure 4. The adsorption capacities of the magnetic ferrites increased with increasing solution pH values. The adsorption capacities of F9, F10, and F11 increased slowly in solution pH range from 6.0 to 8.0, and a rapid increase occurred with pH range from 8.0 to 9.0. The highest adsorption capacities were 164.18, 58.47, and 76.21 mg/g for F9, F10, and F11, respectively. In contrast, the adsorption capacity of F12 increased slowly with elevated pH values and reached 36.96 mg/g at pH 9.0. The results showed that the adsorption capacity of F9 was the highest. At lower pH values, Cd(II) ions competed with H+ for the adsorption sites on the surface of the magnetic ferrite. As the pH was higher than 8.0, the Cd(II) ions easily formed hydroxide with hydroxyl ions, which might be attributed to the higher adsorption capacity. The adsorption capacities of the four magnetic ferrites reached the highest at pH 9.0, which can be attributed to hydrolysis and precipitation of cadmium species in the solution when the solution pH value was higher than the critical pH value of Cd(II) ion (approximately 8.0) (Wang, 1993). However, the products generated by the chemical precipitation method were difficult to be settled and separated in water. Therefore, 8.0 was selected as the solution pH for the adsorption of Cd(II) ion.
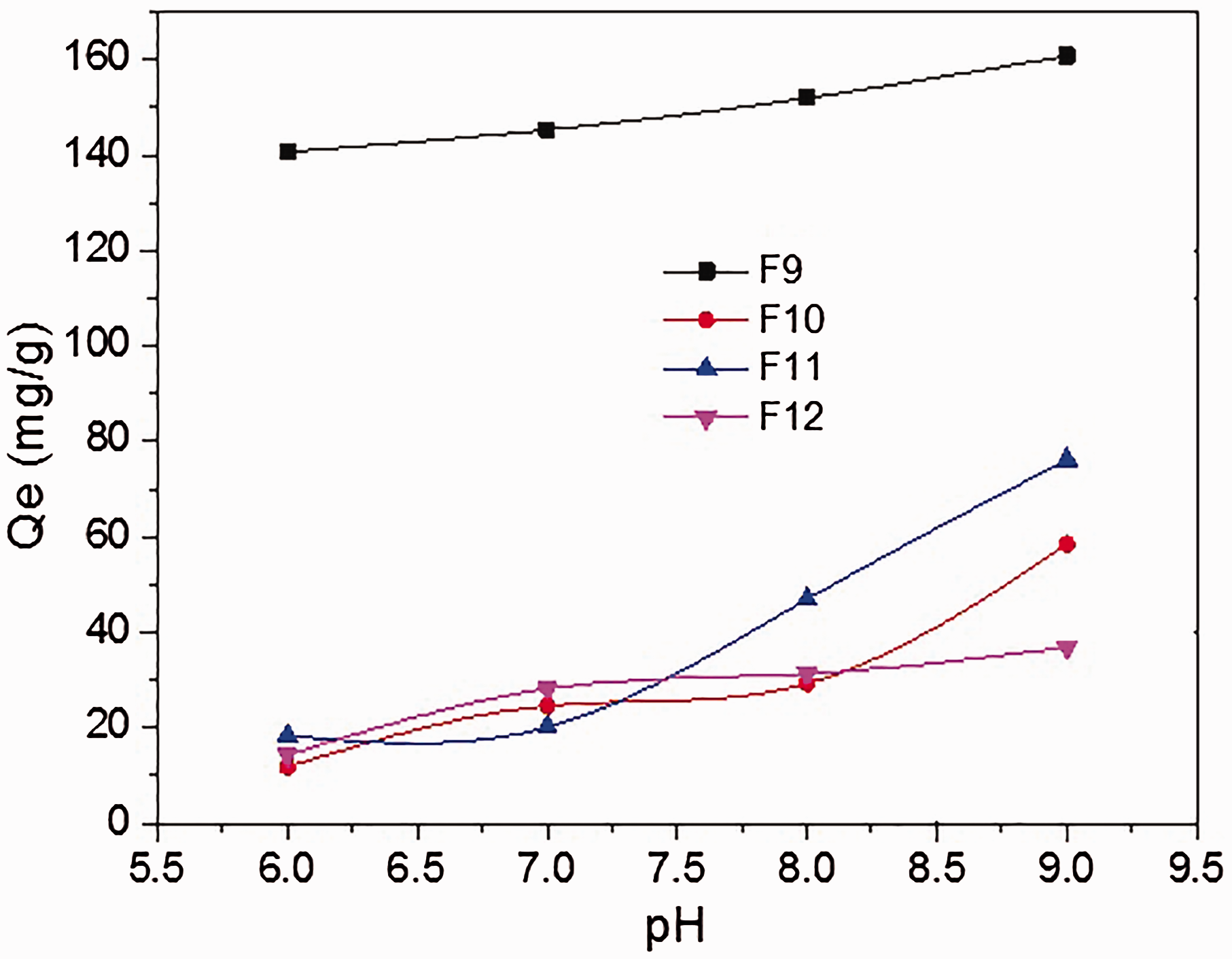
Effect of pH on the adsorption of Cd(II) ions by the four magnetic ferrites.
Effect of contact time on the adsorption of Cd(II) ions
To explore the effect of contact time on the adsorption of Cd(II) ion onto the magnetic ferrites, 0.2 g of the magnetic ferrite was added into 200 ml of 200 mg/l Cd(II) solution at 25°C and at pH 8.0. The adsorbed Cd(II) ion at different time intervals was measured, and the results are shown in Figure 5. From Figure 5(a), the adsorption capacities of the four ferrites increased with time extension and reached an equilibrium after 150 min. Specifically, the adsorption capacity of F9 increased most significantly and reached equilibrium to a value of 160.91 mg/g after 150 min. The final adsorption capacities of F10, F11, and F12 were 115.88, 110.90, and 105.82 mg/g, respectively.

(a) Effect of contact time on the adsorption of Cd(II) ions at pH 8.0. (b, c) The fitting of pseudo-first-order (b) and pseudo-second-order (c) kinetic models for Cd(II) uptake by the magnetic ferrites.
The adsorption capacity reached equilibrium due to the absence of internal diffusion resistance (Ahmaruzzaman and Gayatri, 2011; Langmuir, 2015). Fast adsorption of Cd(II) ions was observed initially, after which the Cd(II) ion adsorption capacity underwent a trend to equilibrium because the adsorption sites of the adsorbent were occupied with the adsorbate. The results confirmed that the adsorption capacity of F9 was the highest among the four adsorbents, suggesting that F9 was prepared successfully and was superior over other magnetic ferrites.
The adsorption kinetics can reflect the rate of adsorption. The pseudo-first-order (Chen et al., 2017) and pseudo-second-order (Chen et al., 2017) kinetic models were used to investigate the adsorption of Cd(II) ions by the magnetic ferrites, and their Lagergren forms were expressed in equations (3) and (4), respectively, as follows


Parameters calculated from the pseudo-first- and second-order kinetics models during Cd(II) adsorption by the four ferrites.

From Table 1, the correlation coefficient (R2) values of pseudo-first order were lower than 0.95 for all four magnetic ferrites, indicating that the fitting was not very well. The Qe values of the magnetic ferrites calculated from the first-order kinetics were lower than the experimental data (Table 1). This result was consistent with previous study (Badruddoza et al., 2010).
For the second-order kinetics, there were high values of R2 (higher than 0.99), showing good fitting results. Therefore, the adsorption of Cd(II) ions followed the pseudo-second order kinetics, which was also reported by previous studies of the adsorption of Cd(II) ion from aqueous solution by other adsorbents (Benguella and Benaissa, 2002; Pérezmarín et al., 2007). Thus, the adsorption of Cd(II) ion onto the magnetic ferrites was a multistep process involving both sorption on the external surface and diffusion into the interior of the adsorbent (Gupta et al., 2013; Liu et al., 2013). Furthermore, the process was possibly a chemisorption through valence force by sharing or by exchange of electrons between the magnetic ferrites and Cd(II) ions.
Adsorption isotherms
Adsorption isotherms can illustrate the relationship between the amount of adsorbate on the adsorbent (Qe) and the equilibrium concentration in bulk solution (Ce) at constant temperature. To further investigate the adsorption process, experiments were carried out at 25°C at pH 8.0, and 0.2 g of the magnetic ferrites was added into 200 ml of solution with various Cd(II) concentrations, i.e. 25, 50, 75, 100, 125, and 150 mg/l. The adsorption of Cd(II) ion onto the magnetic ferrites was analyzed using the Langmuir (Wang et al., 2015) and Freundlich (Chai et al., 2013) isotherm models. The Langmuir isotherm model is based on the assumption of homogeneity on the surface of adsorbent with a finite number of identical sites and a constant adsorption potential. The Freundlich isotherm is an empirical equation based on a highly heterogeneous surface adsorption. These two models are expressed in equations (5) and (6), respectively, as follows
Langmuir equation

Freundlich equation


(a) Langmuir adsorption isotherms and (b) Freundlich adsorption isotherms of the four ferrites.
The parameters of adsorption isotherms of Cd(II) ions.

From Figure 6 and Table 2, the Langmuir isotherms showed that the maximum adsorption capacities of the magnetic ferrites were 43.48, 50.00, 27.40, and 46.51 mg/g for F9, F10, F11, and F12, respectively. The corresponding correlation coefficients (R2) were 0.987, 0.985, 0.946, and 0.964, respectively. The correlation coefficients (R2) described by the Freundlich model were 0.949, 0.994, and 0.599 for F9, F10, and F11, respectively, while F12 could not be described by the Freundlich isotherm. The results indicated that both the Langmuir and Freundlich adsorption isotherms fitted well with the experimental data of F10. This may be attributed to the preparation and structure of the magnetic ferrite. The Langmuir isotherm model was optimal for the characterization of the adsorption of Cd(II) ions onto F9, F11, and F12, suggesting an uniform distribution of active sites for Cd(II) ion adsorption with monolayer coverage (Gupta et al., 2013; Shah et al., 2013). The n values for F9, F10, and F11 were in the range of 0–10, suggesting that these three magnetic ferrites were favorable for the adsorption of Cd(II) ions at pH 8.0 (Gong et al., 2011; Hameed et al., 2008). The higher value of Kf for F9 suggested a stronger adsorption capacity of Cd(II) ions by F9 than the other magnetic ferrites, in consistent with the above results.
Cost analysis of magnetic ferrite preparation
To evaluate the potential commercial application of magnetic ferrites for Cd-containing wastewater treatment, cost analysis of magnetic ferrite production is performed referring to previous literatures (Banerjee et al., 2016; Dhawane et al., 2016). The overall cost consists of the raw material sources (Mondal et al., 2016), preparation methodology, and subsequent treatment (Mondal et al., 2016). The cost estimation of synthesizing 1 kg of magnetic ferrite is calculated in China Yuan (RMB) as follows:
I. Cost of raw material (CRM) = cost of ferrous sulfate (FeSO4·7H2O, CFS) + cost of sodium hydroxide (NaOH, CSH) = units × per unit cost = 3.6 kg × 0.13 RMB/kg (CSF) + 1.04 kg × 2.5 RMB/kg (CSH) = 3.068 RMB.
II. Cost of water usage (CW) = units × per unit cost = 5 kg × 0.003 RMB/kg = 0.015 RMB.
III. Cost of electricity consumption (EC) = units × per unit cost = 0.4 kW h × 0.6 RMB/kW h = 0.24 RMB.
IV. Cost of cleaning magnetic ferrite (CCMF) = units × per unit cost = 3 kg × 0.006 RMB/kg = 0.018 RMB. The magnetic ferrite production was washed by the distilled water obtained from laboratory setup before experiment.
Therefore, the overall cost for magnetic ferrite production = CRM + CW + EC + CCMF = 3.068 + 0.015 + 0.24 + 0.018 = 3.341 RMB. The net cost of magnetic ferrite production was the overall cost.
Based on the above cost estimation, the total cost for preparation of 1 kg of magnetic ferrite was 3.341 RMB, which was quite lower than other commercial adsorbents (Banerjee et al., 2017). The results suggested that the synthesized magnetic ferrites can be used as a cost-effective adsorbent for the removal of heavy metal from aqueous solution. In addition, the spent magnetic ferrites particles can be easily collected and further used as ingredients for road construction or brick formation with additional modifications if required (Mukherjee et al., 2017).
Conclusion
In this study, the magnetic ferrites were prepared at various pH conditions and applied to remove Cd(II) ions from wastewater. The synthesized magnetic ferrites were uniformly distributed in the nanoscale and showed good magnetism, which was beneficial for solid–liquid separation. The optimal pH value for Cd(II) ion adsorption by the magnetic ferrites was 8.0, and the adsorption of Cd(II) ion onto the magnetic ferrites followed pseudo-second-order kinetics. Both Langmuir and Freundlich adsorption isotherms described well with the adsorption by F10, and the Langmuir isotherm model was optimal for the characterization of the adsorption of Cd(II) ions onto F9, F11, and F12, suggesting an uniform distribution of active sites for Cd(II) ion adsorption with monolayer coverage. To conclude, the most suitable pH value for the synthesis of magnetic ferrite is 9.0, and a maximum adsorption capacity of 160.91 mg/g for Cd(II) ions can be achieved. Moreover, the magnetic ferrite was a cost-effective adsorbent for the removal of heavy metal according to the cost analysis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the National Natural Science Foundation of China (21707167) and the Fundamental Research Funds for the Central South University for the support of this study.
