Abstract
Research on utilizing waste by-products such as coconut shell, pomelo peel, mango peel, pineapple peel, jackfruit peel, and mangosteen peel to produce activated carbon for desalination is a potential and sustainable direction. In the research, activated carbons from jackfruit peel, pomelo peel, and pineapple peel were activated with NaOH and heat-treated by the microwave method. The structure of activated carbon from agricultural by-products has some characteristics analyzed by scanning electron microscopy, Brunauer−Emmett−Teller analysis, and Fourier transform infrared spectroscopy. Remarkable results include a large surface area; porous structure with many capillaries; the surface of activated carbon having many functional groups such as −OH, −COOH, and −CO; and an amorphous structure. The best adsorption conditions were recorded with 2.794 and 2.588 mg g−1 at 5 g L−1 concentration, 0.4 g L−1 dose, 30 °C temperature, 4-h time for activated carbons from pomelo peel and jackfruit peel. Similarly, activated carbon from pineapple peel had an adsorption capacity of 2.823 mg g−1 at 5 g L−1 concentration, 30 °C temperature, 5-h time, and 0.4 g L−1 dose. The salt adsorption efficiency of activated carbon from waste sources follows the pseudo-first-order kinetic model (R2 > 0.9). The desalination process using pomelo peel and pineapple peel activated carbon followed the Langmuir and Freundlich isotherm models (R2 > 0.95), while jackfruit peel activated carbon followed the Dubinin−Radushkevich model (R2 > 0.96), indicating their potential application in brine treatment. In addition, activated carbons from these waste sources can be regenerated and reused in the desalination cycle; however, their efficiency tends to decrease after the third regeneration when using regenerators such as sodium hydroxide, water, and acetic acid.

Introduction
Climate change not only increases temperatures but also changes rainfall patterns, leading to droughts in some areas and heavy rains in others. 1 This directly affects freshwater resources. Climate change has increased temperatures and changed rainfall patterns, leading to rising sea levels. 2 Rising sea levels increase saltwater intrusion into coastal areas and rivers. Treating saline water not only helps protect water resources but can also provide clean water for daily life and production activities, contributing to ensuring water security and sustainable development. Vietnam is one of the countries with great potential for fruit production and export, thanks to its rich climate and soil conditions, including fresh fruit and processed fruit products. Fruit peels are often considered waste in the food processing process, especially in the fruit production and consumption industry.
Agricultural by-products are the main unused parts of agricultural production and food processing, including straw, fruit peels, leaves, twigs, bagasse, bean hulls, and many other wastes.3,4 These by-products are often rich in fiber, carbon, and antioxidants, and may contain high levels of bioactive compounds such as polyphenols, flavonoids, and saponins. They often contain cellulose, hemicellulose, lignin, and fermentable compounds, along with significant amounts of water, minerals, and sometimes nitrogen compounds.5 –7 Agricultural by-products are an abundant and potential source of raw materials for the production of activated carbon, a material with a large porous surface area and high adsorption capacity, widely used in water treatment, air purification, as an adsorbent, and many other applications.8 –11 It is a renewable, low-cost, and year-round source. In addition, they often have a high carbon content, which is an important factor for the carbonization process. These raw materials can easily undergo thermal or chemical treatment to convert into activated carbon with a large porous surface and strong adsorption capacity. The production process of activated carbon is carried out at high temperatures in an oxygen-deficient environment to remove unwanted components and retain the carbon part. There are two main activation methods: raw carbon being treated at high temperatures in a chemical gas environment such as steam or CO₂ to create a porous structure, or using chemicals such as KOH, H₃PO₄, or ZnCl₂ to create activated carbon with a better porous structure while increasing the adsorption capacity.6,12 With outstanding properties, activated carbon has a strong adsorption capacity for organic substances, heavy metals, and toxic chemicals in water. In addition, activated carbon has the ability to adsorb odor-causing compounds, which are used in the food, pharmaceutical, and cosmetic industries.
Activated carbon from agricultural by-products is becoming a prominent research trend in saline water treatment, due to its environmental friendliness, low cost, and the ability to utilize abundant waste sources such as fruit peels. The application of activated carbon in saline water treatment involves the ability to adsorb salt ions, heavy metals, and organic impurities, which helps to reduce salinity and improve water quality. Superior desalination performance can be achieved by using high pore volume coconut shell superactivated carbon as Capacitive deionization (CDI) electrode material, as demonstrated by Zhang et al. 13 Activated carbon pellets were also used by Thakur et al. 14 to improve the evaporation rate of seawater desalination equipment. Chen et al. 15 conducted isolated closed-loop (ICC) desalination with the presence of carbon nanotubes (CNTs) to increase the efficiency of the desalination process (>99%). Li et al. activated traditional activated carbon with KOH and used it as a capacitive deionization electrode. The desalination efficiency was high at 25.1 mg g−1 due to its porous properties, high surface area, and conductivity. 16
Reusing waste by-products not only helps to reduce waste but also creates higher economic value for agricultural products. Maximizing the potential of fruits and applying products from waste by-products will contribute to the development of Vietnamese agriculture, toward a sustainable and environmentally friendly future. Therefore, the study took advantage of waste by-products from fruit processing and consumption factories. Research focuses on the use of activated carbon derived from agricultural by-products for desalination, providing a sustainable and cost-effective alternative to conventional methods. The peels of three types of fruits (pomelo, jackfruit, and pineapple) were recovered, and activated carbon was produced. The synthesis of activated carbon by the microwave-assisted method in the presence of NaOH is one of the convenient synthesis methods, creating a porous structure and large surface area, increasing the adsorption capacity. By integrating adsorption kinetics and isotherm modeling, a comprehensive understanding of the desalination mechanism is established, laying the foundation for innovative, environmentally friendly water purification solutions. This helps to reduce the cost of the current activated carbon production process. The main product is an adsorbent material that can reduce the salinity of salt in saline water. In addition, materials synthesized from green methods contribute to reducing difficult-to-decompose waste in the environment.
Materials and methods
Chemicals
Raw materials such as pomelo peel, jackfruit peel, and pineapple peel were collected from Mekong Food Processing Co., Ltd. (Vietnam). The fruit peels were washed to remove dirt, impurities, and damaged parts. The peels were dried at a temperature of about 60−80 °C until they were completely dry for easy carbonization. The raw material samples, after being dried and crushed into small sizes, were used for the material synthesis process.
Sodium hydroxide (NaOH ⩾ 96%), sodium chloride (NaCl ⩾ 99.5%), and hydrochloric acid (HCl 73%) were obtained from Xilong Chemical Co., Ltd. (Shantou, China).
Activation process of activated carbon
The initial materials (such as pineapple peel, pomelo peel, and jackfruit peel) were crushed to increase the contact surface area. The raw materials were soaked in a solution of water and NaOH at a ratio of 1:1:1; the mixture was mixed well and incubated for 30 min. NaOH has the effect of expanding the pore structure of the material and removing organic impurities. After soaking in NaOH, the raw material is filtered and dried at 100 °C for 24 h. Next, the raw material is put into a microwave oven for activation at a power of 600W (190–220 °C) for 2 min. 17 After microwave activation, HCl (1M) is used to neutralize and remove excess NaOH. The activated carbon will be washed with distilled water several times until it reaches a neutral pH (pH 7). Then, the carbon is dried at 100 °C for 24 h. 18
Characterization
Structural characterization of activated carbon by scanning electron microscopy (SEM), Brunauer−Emmett−Teller (BET) analysis, and Fourier transform infrared spectroscopy (FTIR) methods can provide insight into the surface structure, surface area, and chemical functional groups of the material. Specifically, SEM helps determine the size and shape of pores, as well as their distribution on the surface of activated carbon, which is performed on a Hitachi S-4800 (Japan) instrument. JASCO FT/IR-6000 (Japan) is used to analyze the chemical bonds and functional groups on the surface of activated carbon in the range of 4000−400 cm−1. BET analysis provides important information on the specific surface area (m2 g−1), pore volume, and pore size distribution. Material samples are analyzed on a Micromeritics 2020 (USA) instrument.
Desalination process in simulated water environment
First, saline water is standardized with NaCl solution at different concentrations. The concentration of the adsorbent can be adjusted depending on the experimental requirements. Weigh an amount of activated carbon and add it to 100 mL of simulated saline solution at an appropriate ratio. This mixture can be shaken for a certain time for the adsorption process to take place. The adsorption process is affected by many different factors such as time (1−30 h), concentration (1−8 g L−1), and mass of the adsorbent material. These factors affect the rate and efficiency of the adsorption process, as well as the saturation capacity of the adsorbent material. After the adsorption time, the solution is filtered to remove the activated carbon. The sample of the solution after filtration will be used to analyze the remaining salt concentration. The reaction is performed on a JeioTech—Korea device, with a maximum speed of 200 rpm. After the adsorption process, the activated carbon can be reused by washing with acetic acid, water, or NaOH solution, and then drying to check the reuse ability and compare with the initial adsorption efficiency. The Hanna HI98325 device is used to determine the desalination parameters after the adsorption process, and the adsorption capacity Qe is calculated based on the following formula: Qe = ((Co −Ce) × V)/W, 19 in which Ce and Co are the concentrations after adsorption and the concentrations before adsorption (mg/L), V (L) is the volume of the colored solution, and W (g) is the mass of the adsorbent.
Results and discussions
Preliminary evaluation of activated carbon from agricultural by-products
Preliminary investigations on activated carbons from waste by-products such as sawdust, coconut shell, jackfruit shell, pomelo shell, pineapple shell, rambutan shell, and mangosteen shell can help evaluate the adsorption potential of each type in treating different types of salts or pollutants (Figure 1). Each type of shell has a characteristic composition and structure, which can affect the surface area, pore structure, and chemical functional groups on the surface of the activated carbon. Figure 4(a) shows that all activated carbons from different waste by-product sources show desalination potential, but the best desalination potential is in activated carbons from jackfruit, pomelo, and pineapple shells with desalination capacities of about 1.1 to 1.3 mg g−1. To achieve desalination efficiency, the influencing factors such as adsorption time, activated carbon dosage, and initial salt concentration were specifically determined for each type of activated carbon.

Desalination capacity of activated carbon from agricultural by-products.
Characteristics of activated carbon from agricultural by-products
The peaks in the FTIR spectrum of activated carbon usually represent functional groups or chemical bonds present on the surface of activated carbon samples from pomelo peel, jackfruit peel, and pineapple peel (Figure 2). The 1087 cm−1 peak is characteristic of C–O or C–O–C bonds, related to ether groups or carboxyl groups (–COO–) present on the surface of activated carbon. 20 These groups may be due to impurities or oxidized groups formed during processing. The 1215 cm−1 peak indicates the presence of C–O groups in compounds such as phenols and esters. This is also a sign of oxidized functional groups on the surface of activated carbon, which helps improve the adsorption capacity due to the polarity of this group. The 1604 cm−1 peak or peaks in this region usually correspond to the vibration of C=C bonds in aromatic rings or carbonyl groups (C=O) linked to the ring system. 21 This may indicate that your activated carbon contains aromatic ring structures or carbonyl groups. The peak at 2881 cm−1 is due to C–H bond stretching vibrations in methylene (–CH₂–) or methyl (–CH₃) groups. These groups may come from carbonized organics that retain some short-chain structure. The broad peak at 3420 cm−1 is an O–H bond stretching vibration, which may be associated with hydroxyl (–OH) groups or bound (adsorbed or surface-bound) water on the activated carbon.22,23 This is a common feature of activated carbons, indicating hygroscopicity and polar functional groups. Analysis of these peaks indicates that your activated carbon may contain functional groups such as ether, phenol, carbonyl, methylene, and hydroxyl.24,25 These functional groups contribute significantly to the adsorption properties of activated carbon, especially in the adsorption of polar or ionic compounds.

FTIR spectrum of activated carbon from agricultural by-products.
SEM images of activated carbon from jackfruit peel, pomelo peel, and pineapple peel activated with NaOH based on the microwave method will show the surface structure of activated carbon after activation (Figure 3). The results show the appearance of many pores and micropores on the surface of activated carbon. These pores can be uneven in size and shape; they can be spherical or oval, reflecting the development of micropores and mesopores after activation by NaOH. Compared with other activation methods, combining NaOH with microwaves helps to generate heat quickly, promoting the dehydration and decomposition of cellulose and lignin. 26 SEM shows cracked and perforated areas, which may be due to the evaporation of organic compounds during activation, making the surface of the carbon more complex and with more surface area.

SEM image of activated carbon from agricultural by-products: pomelo peel (a), and pineapple peel (b), and jackfruit peel (c).
Analysis of the BET surface area results of activated carbons from pineapple peel, pomelo peel, and jackfruit peel showed a clear difference in the surface area of each type (Figure 4). Pomelo peel had the highest BET surface area (682.37 m2 g−1), indicating effective pore formation when activated with NaOH and microwave treatment, corresponding to a type II isotherm indicating that the activated carbon material has large pores. Jackfruit peel with a BET surface area of 443.72 m2 g−1 may have an intermediate fibrous structure and chemical composition between pomelo peel and pineapple peel, resulting in a high surface area, but still lower than pomelo peel. Pineapple peel had the lowest surface area (102.26 m2 g−1), possibly due to the structure of the parent material having little pore formation ability or ineffective reaction with NaOH. Similarly, jackfruit peel and pineapple peel followed a type IV isotherm with medium pores. The NaOH activation process creates pores by breaking the bonds between lignin, hemicellulose, and cellulose.27,28 In pomelo and jackfruit peels, this process is more effective, possibly due to the difference in the structure and thickness of the fiber layers, which allows NaOH to penetrate more easily and create pores. In pineapple peels, the reaction rate with NaOH may be less effective, resulting in less pore formation. Microwaves generally promote rapid and uniform thermal decomposition in materials, leading to the expansion and development of pores. However, the absorption and response to microwaves of each peel may be different. Pomelo peels may react more strongly to microwaves, expanding existing pores, while pineapple peels may not absorb enough energy to achieve the same effect. 29
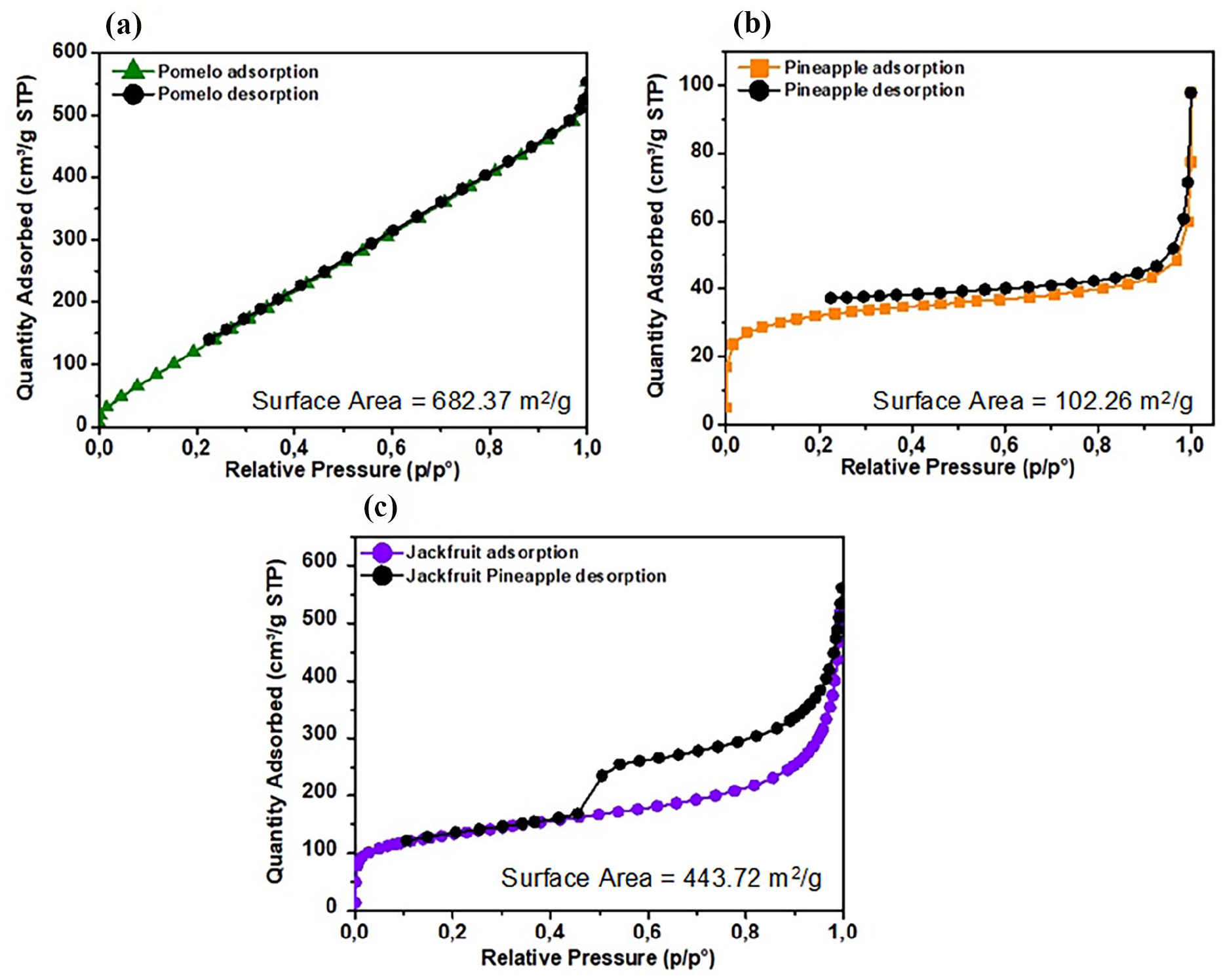
N2 adsorption-desorption isotherm curves of activated carbon: pomelo peel (a), and pineapple peel (b), and jackfruit peel (c).
Desalination of activated carbon from agricultural by-products
The pH of the solution has an important influence on the desalination process of activated carbon because it affects the surface and adsorption capacity of the carbon. At different pH values, the surface of activated carbon can be negatively or positively charged. At low pH, the surface of activated carbon is easily positively charged, while at high pH, the carbon surface will be negatively charged. Therefore, the pH of the solution will affect the adsorption capacity of activated carbon for ions with opposite charges. Activated carbon has good adsorption capacity for salt ions (such as Na⁺ and Cl⁻) through electrostatic interactions and chemical bonds. Normally, each type of activated carbon and each salt ion will have an optimal pH range to achieve the highest adsorption efficiency. During the desalination process, changes in pH can also affect the stability and structure of functional groups on the surface of activated carbon, affecting the regeneration capacity and long-term performance of the carbon (Figure 5(a)). The pH of the solution was investigated in the range of pH 2 to pH 10, the desalination capacity reached from 0.6 to 0.8 mg/g, and the adsorption capacity almost reached the threshold in the environment of pH 6 and pH 8. Based on the previous results of the research group, the pHpzc values of jackfruit peel, 30 pineapple peel, and pomelo peel activated carbon were 4.25, 4, and 4.4, respectively. When the pH of the solution is greater than the pH at the zero charge point (pHpzc), the surface of the activated carbon will be negatively charged. This occurs due to the loss of protons from acidic functional groups (such as −COOH) on the surface of the activated carbon when the pH increases higher than the pH. As a result, the surface of the activated carbon has a better ability to adsorb positive ions, due to the electrostatic attraction between the negative surface and the positive ions in the solution, which effectively supports the desalination process.

Factors affecting the desalination process of activated carbon from pomelo peel, pineapple peel, and jackfruit peel: pH (a) and pHpzc (b).
The activated carbon dosage is an important factor affecting the desalination capacity of activated carbons from pineapple, pomelo, and jackfruit peels. Figure 6(a) shows that as the activated carbon dosage increases, the total surface area and the number of available adsorption sites also increase. This increases the contact between Na+ and Cl− ions and the carbon surface, allowing for more effective removal of salt ions in the solution, especially at high salt concentrations. The desalination efficiency of activated carbons derived from pomelo and pineapple peels increased as the adsorption dosage rose from 0.2 to 0.8 g L−1. In contrast, the activated carbon from jackfruit peel achieved its maximum desalination efficiency at a dosage of 0.4 g L−1, after which the efficiency declined with further increases in dosage. After a certain threshold, further increases in the activated carbon dosage may not result in significant desalination efficiency. When the amount of Na+ and Cl− ions in the solution reaches equilibrium with the adsorption capacity of the carbon, the adsorption sites are gradually saturated, and the salt removal capacity will reach the maximum threshold. At this time, the desalination efficiency will increase slowly even though the carbon dosage increases. Using too high a dosage of activated carbon not only does not significantly increase the adsorption capacity but also increases the operating and raw material costs. The effect of salt concentration on the adsorption capacity of activated carbon will help determine the optimal conditions, ensuring high efficiency and cost savings for saltwater treatment applications. Figure 6(b) shows that when the NaCl concentration increases to 5 g L−1, the number of Na+ and Cl− ions in the solution increases, creating a higher ion pressure, causing these ions to rapidly move into the pores and surface of the activated carbon. At this stage, the adsorption capacity of the carbon increases rapidly because the adsorption sites on the surface are still abundant and the surface area is not saturated. At concentrations of 6 and 7 g L−1, many of the adsorption sites on the carbon have begun to be occupied by salt ions. Na+ and Cl− ions compete more to access the remaining pores, leading to gradual saturation. This slows down the adsorption process because the activated carbon no longer has enough sites to adsorb all the Na+ and Cl− ions, so the adsorption capacity increases at a slower rate. When the concentration of 8 g/L is reached, although the activated carbon is close to saturation, some large pores and unadsorbed functional groups may still be available, leading to an increasing adsorption capacity, but with high variability. However, further increases in salt concentration beyond this threshold may reduce the economic efficiency and optimization for the desalination process.
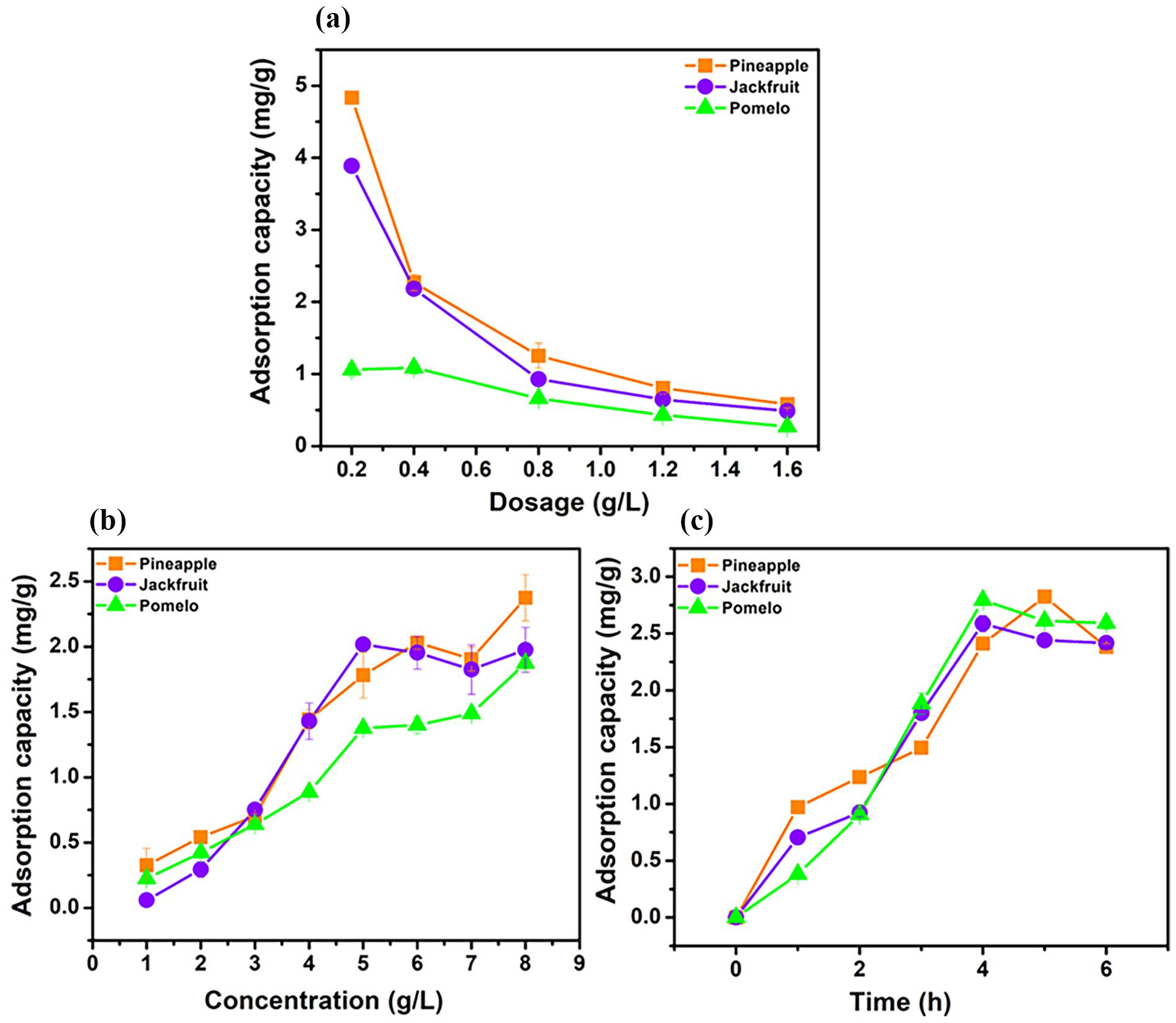
Factors affecting the desalination process of activated carbon from pomelo peel, pineapple peel, and jackfruit peel: dosage g L−1 (a), concentration g L−1 (b), and time hour (c).
An optimization test could involve varying the immersion time in a fixed concentration of 5 g L−1 NaCl solution and then determining the contact time of the activated carbon with the NaCl solution to achieve the optimal salt adsorption capacity (Figure 6(c)). As the adsorption time increased, the initial desalination capacity increased rapidly within the first 4 h, with adsorption capacities reaching 2.794, 2.588, and 2.411 mg g−1 for activated carbons of pomelo peel, jackfruit peel, and pineapple peel, respectively. This could be due to the fact that the active surfaces of the activated carbon were still open and easily exposed to salt ions in the solution, resulting in a fast adsorption rate. During this period, many salt ions could easily be trapped on the surface or inside the pores of the carbon. However, after about 4 h, the adsorption process began to slow down and gradually reached equilibrium. This phenomenon is due to the fact that the number of available adsorption sites has gradually decreased, and the salt ions in the solution have to compete with each other to occupy the remaining sites. At this stage, the adsorption capacity reaches equilibrium because the adsorption rate and desorption rate become equivalent, and there is no longer a significant increase in the amount of salt adsorbed. The interaction time of activated carbon in the salt solution (NaCl) and then the contact time during desalination greatly affect the salt adsorption capacity. If the time is too short, the salt ions may not have enough time to contact and be effectively adsorbed on the activated carbon surface. However, if the time is too long, the salt ions may reach a saturated state on the carbon surface, reducing the adsorption efficiency.
In the desalination process using activated carbon, popular adsorption isotherm models such as Langmuir, Freundlich, Temkin, and Dubinin−Radushkevich (D-R) are often applied to describe the adsorption capacity of salt ions (Na+ and Cl−) on the surface of activated carbon. Based on the results of Table 1 and Figure 7, the compliance with both Langmuir and Freundlich isotherm models of pineapple peel and pomelo peel activated carbons with correlation coefficients R2 > 0.95, while the correlation coefficient R2 of jackfruit peel activated carbon only reached 0.8608 and 0.8715. The Freundlich model shows that the surface of the material may contain adsorption sites with different energies. This may be due to the heterogeneous structure of the material surface, especially if the material is from biological sources or activated from agricultural wastes. This heterogeneity makes the adsorption process more flexible and can take place at sites with different binding energies, leading to the formation of multilayer adsorption at high solution concentrations. The Langmuir model shows that some adsorption sites of the material can behave like monolayers, with independent and homogeneous adsorption sites. These sites can be saturated at certain concentrations, which is consistent with the Langmuir model describing the early stages of the desalination process well. When the solution reaches higher concentrations, the process can switch to multilayer adsorption, demonstrating the suitability of the Freundlich model. The agreement with both models shows that the desalination process is not only limited by physical mechanisms but can also be influenced by some chemical mechanisms. Adsorbates, such as salt ions, can interact strongly with sites of high binding energy and weaker with sites of lower binding energy, creating a wide range of interactions and allowing adsorption to occur simultaneously on monolayer and multilayer sites. With a correlation coefficient above 0.9, the Temkin model also describes the salt adsorption process on activated carbon quite well. This suggests that the interaction between salt ions and the activated carbon surface may be related to the gradual decrease in adsorption energy with the degree of surface coverage, consistent with the assumption of the Temkin model. At the same time, with a correlation coefficient greater than R2 > 0.94 of the two activated carbon samples, the D-R model shows a high degree of suitability in describing the salt adsorption process on activated carbon. This suggests that the adsorption process is likely physical in nature and that the activated carbon surface has heterogeneous properties, consistent with the assumptions of the D-R model (Figure 8).
Data of adsorption isotherm constants of desalination process on activated carbons.


Nonlinear models for isothermal model for desalination process at room temperature. Jackfruit peel (a), pineapple peel (b), and pomelo peel (c).

Desalination mechanism of activated carbon from agricultural waste (grapefruit peel, jackfruit peel, and pineapple peel).
The adsorption kinetics was evaluated using four kinetic models (pseudo first-order, pseudo second-order, Elovich, and Bangham) shown in Table 2 and Figure 9. The desalination process was carried out under specific conditions such as NaCl concentration, pH, and activated carbon dosage over time. The adsorption mechanism according to the pseudo-first-order kinetic model reflects the physical adsorption process, in which the adsorbed molecules or ions adhere to the material surface by weak interactions such as Van der Waals forces. This shows that the adsorption process does not depend much on chemical reactions or electron exchange but is mainly related to the initial adhesion. Based on Table 2, the correlation coefficients R2 of the Pseudo-first-order (PFO) model are 0.9106, 0.8996, and 0.9201, respectively, for activated carbons of pineapple peel, pomelo peel, and jackfruit peel, which are higher than those of the other models. Since it is a physical adsorption process, the pseudo-first-order model usually shows a high initial adsorption rate when there are many vacant sites on the surface. 31 This rate then decreases as the sites are filled, and the system approaches equilibrium. The constant k1 of the pseudo-first-order model helps to measure the adsorption rate of the material. The larger the k1 value, the higher the initial adsorption rate. This is useful in selecting materials that can desalinate quickly in a short time. At the initial stage of the adsorption process, when there are many vacant sites on the adsorption surface, the amount of adsorbed substance increases rapidly. The adsorption rate is proportional to the concentration of the substance to be removed in the solution, and an increase in the initial concentration will promote a faster adsorption rate. Since the physical interactions are weak, adsorption equilibrium is reached quickly when the accessible surface sites are filled. Then, the adsorption rate gradually decreases and approaches equilibrium, in which the adsorption and desorption rates are balanced. An important kinetic aspect is interparticle diffusion, which governs the rate at which NaCl ions penetrate into the internal pores of the activated carbon. The pseudo-first-order kinetic model is consistent with interparticle diffusion as an important mechanism. The high correlation coefficients (>0.94) for the D-R isotherm and (>0.9) for the Temkin isotherm model indicate that both physical and chemical adsorption mechanisms are involved. In the initial stage, rapid adsorption occurs as Na+ and Cl− ions adhere to the outer surface. Following the intermediate stage, the adsorption rate slows down as ions diffuse into the mesopores and micropores. This stage is mainly controlled by interparticle diffusion kinetics. Finally, adsorption reaches equilibrium when the ion concentrations in the solution and on the activated carbon surface are equal.
Data of adsorption kinetic constants of desalination process on activated carbons.

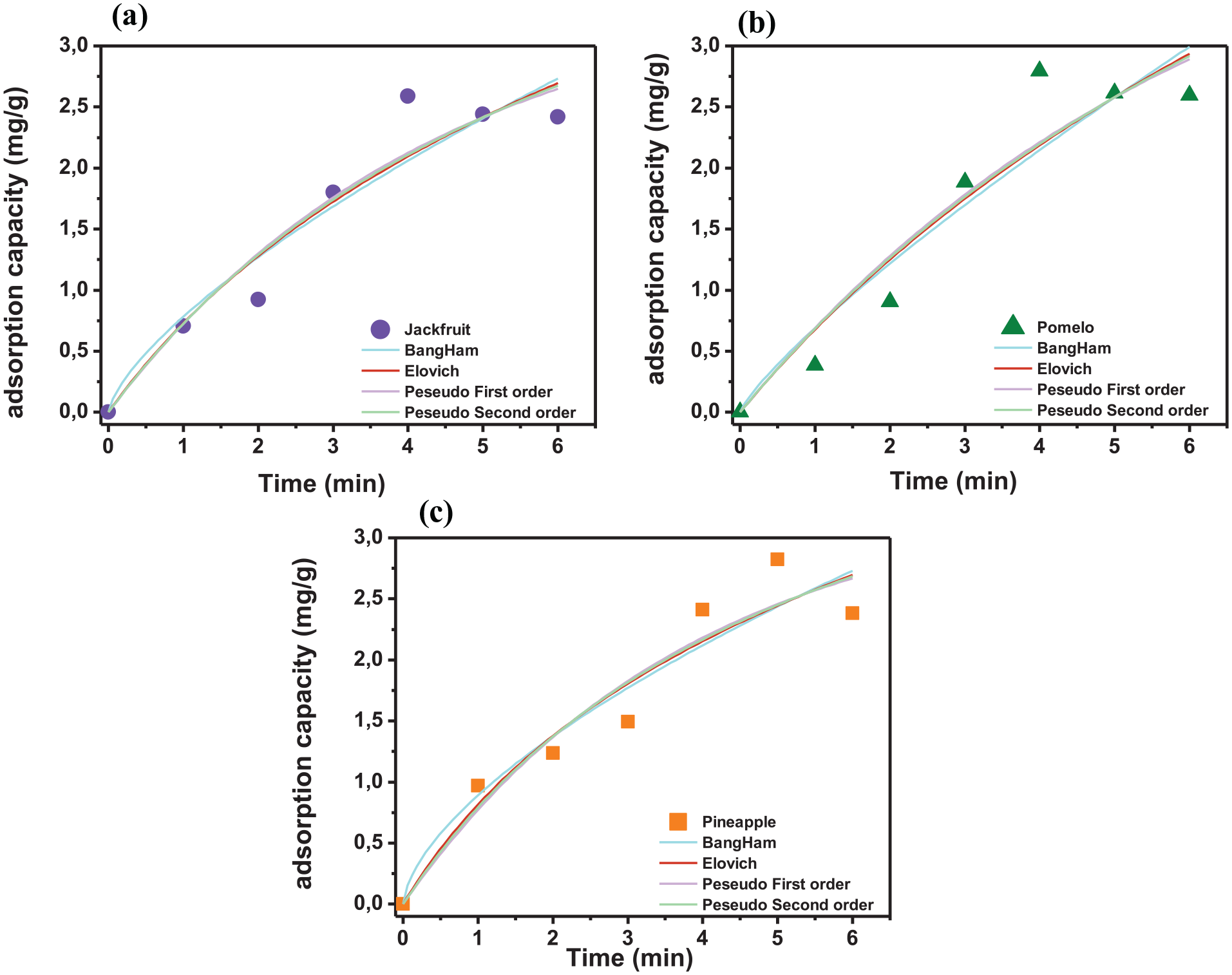
Nonlinear models for adsorption kinetics of desalination on activated carbon. Jackfruit peel (a), pomelo peel (b), and pineapple peel (c).
To enhance the reuse of activated carbon in desalination with NaOH, H2O, and C2H4O2, a combination of these substances allows for efficient regeneration by alternating between basic, neutral, and acidic washes, maximizing the activated carbon’s reuse potential (Figure 10), typically utilized in the washing stage to eliminate adsorbed ions from the activated carbon surface, particularly cations like Na+ that may adhere to the carbon surface during desalination. NaOH aids in restoring the adsorption capacity by eliminating these ions and decomposing organic residues. Water serves as a solvent to facilitate the migration of reagents and thorough rinsing, aiding in the removal of any residual salts or reagents from the activated carbon, ensuring its readiness for reuse. On the contrary, acetic acid can be employed in the acid washing stage to neutralize any remaining alkalinity from the NaOH and assist in desorbing weakly adsorbed anions. Acetic acid offers a gentler acidic wash that can also aid in regenerating functional groups on the carbon surface without the necessity for harsh treatments. The decrease in desalination efficiency over reuse cycles suggests potential issues with the regeneration process. Some ions may strongly bind to the activated carbon and resist removal even with NaOH and acetic acid. This can lead to a reduction in available surface area and active sites over time. Prolonged exposure to chemicals like NaOH and acetic acid, even in mild concentrations, can gradually degrade the structure of the activated carbon, diminishing its porosity and adsorption capacity. Throughout multiple reuse cycles, the functional groups responsible for adsorption may deteriorate, weakening the carbon’s ability to bind salt ions. Consequently, the effectiveness of the reuse process may diminish after numerous cycles, resulting in salt or adsorbent residues remaining on the carbon. Therefore, further research is needed to modify the surface of activated carbon with functional groups (e.g. amine groups, sulfonic groups, or carboxyl groups) to improve ion selectivity and desalination performance; utilize nanomaterials (e.g. graphene oxide, CNTs) to create composite materials to enhance adsorption capacity; evaluate performance using actual brackish or seawater samples to understand the effects of competing ions and organics; and conduct long-term cycling tests to evaluate performance degradation mechanisms and stability.

Regeneration process of activated carbon with different solvents. C2H4O2 (a), NaOH (b), and H2O (c).
The highlight of this study is the use of agricultural waste (e.g. coconut shell, grapefruit peel, mango peel) to produce activated carbon, minimizing waste to the environment. Agricultural by-products are inexpensive and widely available, which helps reduce production costs and can be an affordable alternative to commercial activated carbon that is sustainable and environmentally friendly. Some activated carbons derived from biomass (e.g. jackfruit peel, grapefruit peel, and pineapple peel) show relatively good salt adsorption efficiency. Activated carbon can be regenerated with NaOH, H₂O, and C₂H₄O₂ (acetic acid), allowing for multiple reuses, extending the life of the adsorbent, and reducing operating costs. Adsorption-based desalination does not require high energy inputs such as reverse osmosis or thermal distillation, making it suitable for decentralized water treatment, especially in rural areas. However, there are some limitations in the desalination process; the efficiency can decrease over many reuse cycles. Activated carbon can adsorb other organic/inorganic impurities, reducing the selectivity for salt. The presence of competing ions (e.g. heavy metals, organics) can reduce the desalination efficiency. Some agricultural activated carbons may have lower mechanical strength than commercial activated carbons, leading to breakage or reduced shelf life. Variability in agricultural raw materials can lead to inconsistent adsorption properties. Batch adsorption processes may require long contact times for optimal salt removal. Activated carbon from agricultural by-products is a promising, low-cost, and sustainable desalination method. However, challenges related to adsorption efficiency, regeneration, and scalability need to be addressed for practical implementation in large-scale water treatment systems.
Conclusions
Activated carbon from pomelo peel, jackfruit peel, and pineapple peel waste was synthesized using the microwave method and activated with NaOH. The activated carbon has an amorphous structure, with many pores on the surface, and irregular stimulation and shape. At a temperature of 30 °C, at a concentration of 5 g L−1, a dosage of 0.4 g, and a time of 4 h, the adsorption efficiency of activated carbons from grapefruit peel and jackfruit peel was 2.794 and 2.588 mg g−1, respectively. Similarly, at a concentration of 5 g L−1, a temperature of 30 °C, a time of 5 h, and a dosage of 0.4 g L−1, the adsorption capacity of activated carbon from pineapple peel was 2.823 mg g−1. The desalination process of activated carbon follows the pseudo-first-order kinetic model, indicating that the adsorption process is fast and effective. The data indicated that the activated carbon fits the Langmuir and Freundlich isotherm models, suggesting a good adsorption capacity of pineapple peel and pomelo peel activated carbon. In particular, the D-R model has a correlation coefficient greater than 0.94, and the Temkin model also has a correlation coefficient greater than 0.9, indicating the high suitability of activated carbon in salt adsorption. Although activated carbon can be reused in the desalination process, the adsorption efficiency decreases significantly from the third reuse cycle onward when regenerated with NaOH, H₂O, and C₂H₄O₂. Thus, activated carbon is a potential material for water desalination, but attention should be paid to the reuse method and frequency to maintain high adsorption efficiency during long-term use.
Footnotes
Ethical consideration
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Thi Cam Quyen Ngo and Thi Kim Ngan Tran: Writing—original draft. Van Thuan Tran and Thi Kim Ngan Tran: Writing—review & editing. Thanh Dat Do and Thi Kim Oanh Nguyen: Data curation. Thi Cam Duyen Nguyen: Conceptualization.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Science and Technology Incubation Program for Youth (STIY), managed by the Youth Promotion Science and Technology Center of the Ho Chi Minh City Communist Youth Union and the Department of Science and Technology of Ho Chi Minh City, under contract number “01/2024/HĐ-KHCNT-VU.”
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding authors upon reasonable request.
