Abstract
This work reports on two debated points related to the metal content of cotton fiber and its influence on processing. The first issue is if the metal levels of raw fibers are naturally deposited during fiber development or if the levels are influenced by weathering and harvesting conditions present after boll opening. This was tested by harvesting bolls just as they were opening and after the opened bolls were allowed to field age. The second issue relates to the importance of metal levels on fiber dyeability. Results indicate that the metal levels of newly-opened cotton were not appreciably different from those of aged cotton bolls and that the fiber metal levels after scouring and bleaching had little correlation with dye uptake. Additionally, some metal levels exceeded those previously reported and the environment appeared to have a stronger influence on fiber Ca and Mg levels than did cultivar differences.
Introduction
The textile performance of cotton is largely influenced by the length, strength, and development of the fiber, which is ∼95% cellulose. 1 The non-cellulosic components of cotton fiber, such as proteins, pectins, waxes, sugars, pigments, and metals, are present in low quantities but still influence cotton processing. 2 Some metals are absorbed into the plants as nutrients, such as potassium (K), calcium (Ca), phosphorus (P), and magnesium (Mg). The amount of these metals present in the cotton plant and fiber are dependent on the developmental stage of the crop and the metal content and conditions of the soil. 3
Some studies have shown that the metal content of cotton fiber varies by growing location. 4 Metal uptake by crops is dependent on a number of factors, such as availability of the metal to the plant, moisture, pH, and root conditions. 5 Although small in quantity, the uptake of certain metals is vital to plant growth and development. Cotton root growth is strongly influenced by P uptake. 6 Proper K uptake is necessary to prevent shorter and lower yielding cotton plants. 7
Most research on metals and cotton production has focused on the development of total biomass and the crop as a whole. However, Leffler and Tubertini 8 examined the differences in metal distribution throughout the plant components, including the fiber and burr, as a function of plant and fiber maturity and development. The bulk of metal accumulation in the fiber was shown to occur in the first six weeks post-anthesis, with distribution changing between burr and lint as a function of water redistribution.
Washing, scouring, and bleaching are known to reduce the metal content of cotton fibers in varying degrees.9-11 On raw cotton fibers, K is usually present in the highest concentration. However, after alkaline scouring, most K is removed, as are several of the other metals that are present in lower levels. Ca and Mg, however, are not fully removed by scouring. P has not been considered in any depth in prior reports.
It has been proposed that residual metals can interfere with bleaching and dyeing processes. 12 Because of this, expensive sequestering reagents are often used as a precaution in finishing and dyeing processes. 13 Scouring and bleaching are performed before dyeing; hence, it is the residual metal left on the fibers after these steps that is of potential interest relative to dyeing. A few reports have focused on the effects these preparation steps have on fiber metal levels.10,11 These reports each test the treatment of a single cotton sample; hence variability in the effectiveness of these treatments among different cotton samples is largely unknown. Gamble, 11 using fiber from a single cotton bale, found that a water or ethylenediaminetetraacetic acid (EDTA) treatment of the fibers before bleaching resulted in significant differences in dye uptake. The EDTA wash was found to remove Ca and Mg more completely than did the water wash. Curiously though, the EDTA-treated fabrics did not take up the dye as effectively as the water-washed fabrics, which seems to run counter to the proposal that surface metals impede dyeing. The proposed explanation for the dyeing differences was that the interaction among Ca and Mg, pectin, and the cellulose in cotton is complex and that some combinations of these may interfere with dye uptake. 11 Controlled experiments that show a direct correlation between surface metals levels present at the time of dyeing and dye uptake are largely lacking.
The current work has two goals. The first goal is to determine if the levels of metals typically measured on raw cotton samples were reflective of what occurs biologically as part of the boll development process, or if environmental factors (e.g., dust deposition in the field, harvesting practices, or rainfall) contributed significantly to the metal levels of harvested fibers. The second goal was to test if differences in the metal levels of scoured and bleached cotton affect dye uptake. For the first objective, cotton bolls were harvested just as the bolls were opening and after the opened bolls were allowed to stand in the field after several weeks. For the second objective, a series of different fiber samples were sourced from the National Cotton Variety Tests (NCVT), and the metal levels were determined on the raw, scoured, and bleached samples. By looking at a population of samples, it was expected that the degree of metal variation was likely to be similar to that observed in commercial processing.
Experimental
Cotton Samples
To determine if environmental factors have an important influence on the metal levels of raw harvested cotton samples, newly-opened and aged bolls were collected. Tree upland cultivars, Fibermax (FM) 1911, Deltapine (DP) 1321, and Phytogen (PHY) 499, were grown in a large field plot in 2018 with several other cotton experiments in New Orleans, LA, USA. Bolls of each cultivar were hand collected as they were opening in early September. A second set of marked bolls were then collected after the opened bolls were allowed to stand in the field for three weeks. Tree replicate bolls from separate plants were collected for each cultivar and each field condition.
The freshly-opened bolls were placed in plastic specimen jars and were allowed to dry in a desiccating cabinet to limit the potential for further metal contamination. Fiber was ginned with a laboratory roller gin with no other pretreatment, as the fibers harvested in this state showed no visible signs of debris. The separated fibers were allowed to equilibrate at laboratory conditions (20 °C ± 5 °C and 55% ± 5% relative humidity) prior to metals analysis.
To study the effect of residual metals on fiber dyeability, two sets of fibers were subsampled from the 2016 NCVT samples. One set consisted of 10 cultivars that were grown in Lubbock, TX, USA (Table I). The second set consisted of a single cultivar (PHY 499) that was grown in 14 different locations (Table I). Both sets exhibited some visible foreign matter and were processed through a Shirley analyzer (two passes) to remove debris, and the fibers were blended to ensure sample uniformity.
NCVT Cotton Samples Used in this Work
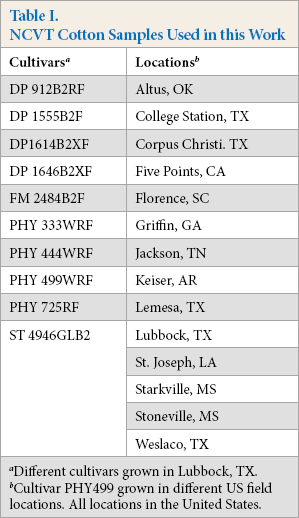
Different cultivars grown in Lubbock, TX.
Cultivar PHY499 grown in different US field locations. All locations in the United States.
Scouring and Bleaching
The NCVT samples were scoured and bleached to prepare them for dyeing. For scouring, 10 g of each raw cotton sample was weighed into a 250 mL flask with 200 mL of a 1% NaOH aqueous solution and two drops of a Triton X-100 surfactant solution (10% w/v). The preparations were placed in a Lab-Line orbital shaker bath operated at 200 rpm, and the samples were heated to 80 °C for 1 h. The flasks were then removed from the bath and allowed to cool to room temperature (RT). The solutions were decanted and the fibers were repeatedly rinsed in deionized water until a neutral pH was reached. The fibers were then dried overnight in a vacuum oven at 50 °C and allowed to equilibrate to RT before bleaching.
For bleaching, 5 g of each scoured sample was dispersed into a 100 mL solution of 0.35% H2O2 and 0.1% NaOH. This mixture was heated to 100 °C for 1 h in the same orbital bath operated at 200 rpm. After allowing the sample to cool and decanting the solutions, the fiber samples were then rinsed repeatedly with deionized water until a neutral pH was reached. The samples were then dried in the same manner as after scouring, having no sequestrant included.
Dyeing
The scoured and bleached fiber samples were dyed with C.I. Reactive Blue 19 dye. Each sample (3 g) was added to a 250-mL wide-mouth flask with 50 mL of water, 25 mL of a 2.4 g/L aqueous dye solution (equivalent to a 2% owf (on weight of fiber) dye treatment), and 2 drops of a 10% (w/v) Triton-X surfactant solution. Samples were placed in the orbital shaker bath at 200 rpm. The bath temperature was increased slowly over 30 min from 30 °C to 60 °C. Once the temperature reached 60 °C, 50 mL of a 2% (w/v) solution of sodium sulfate was added in three equal portions over a 10 min period. After the salt addition, the sample was maintained at 60 °C for an additional 30 min. Then, 25 mL of a 1% (w/v) sodium carbonate solution was added in three portions over 10 min, and the samples were held at 60 °C for another 60 min. The dye solution was then poured from the bottles, and the fiber sample was removed, allowed to cool for 15 min, and washed repeatedly with water to remove salt, alkali, and unreacted dye. The sample was then returned to the bottle with 250 mL of deionized water and 2 drops of the Triton-X solution with the orbital shaker again operating at 200 rpm and 95-98 °C for 20 min to remove any hydrolyzed dye from the fiber. The samples were removed from the bath, and the wash water was decanted. The sample was then rinsed with cold deionized water. The dyed fibers were dried overnight in a vacuum oven at 60 °C, then allowed to equilibrate at RT before color measurement.
Metal Determination
The raw, scoured, and bleached fiber samples were microwave digested. Each sample was weighed (0.50 ± 0.02 g) into a Teflon digestion tube and 20 mL of a nitric acid-water solution (1:1 v/v) was added. The tubes were allowed to sit for 10 min (pre-digest), then were capped and placed in a CEM MARS6 (Mathews, NC, USA) microwave digester. The instrument temperature was increased to 200 °C over 15 min, then held for 20 min, and followed by cooling for 35 min, which was found to result in complete digestion of the fibers. Eight samples were processed per digester cycle. Each digest solution (4 mL) was diluted to 50 mL total volume with nanopure water in 50 mL plastic centrifuge tubes. The digestions were replicated three times.
A Teledyne Leeman Labs (Hudson, NH, USA) inductively-coupled plasma optical emission spectroscopy (ICP-OES) was used for the determination of fiber metal content. A blank consisting of 4% nitric acid in water was run before all standards. Standard curves were prepared from purchased standard solutions of K, Ca, and Mg prepared at 1, 5, 10, and 20 ppm, and P at 0.1, 0.5, 1, and 5 ppm, with each solution prepared in 4% nitric acid. The ICP was operated in axial mode. Samples were analyzed in a random order, and an instrument rinse with nanopure water was run before each sample. Metal levels were calculated from the measured values and the standard curves, and the results were then calculated in ppm based on fiber weight and the sample dilutions. Each digested sample was analyzed three times, with the values averaged to give the best estimate for the sample.
Color Analysis
Fiber color was determined for the bleached and dyed samples with a Gretag Macbeth (New Windsor, NY, USA) Color Eye model 7000A benchtop spectrophotometer. Color was determined in the CIE Lab color system and differences due to dyeing were determined as ?E calculated using Eq. 1.
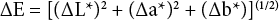
Five measurements were taken on each fiber sample and were averaged to give the best values for that sample. Measurements were performed in a random order.
Fiber Property Measurements
All samples were analyzed for fiber properties using a High Volume Instrument (HVI, Uster Technologies) and the Advanced Fiber Information System (AFIS). Each sample was analyzed five times by HVI and three times by AFIS, and the values were averaged.
Statistics
Differences in boll metal levels were tested for each cultivar with ANOVA and Tukey's range test (α = 0.05) based on the three boll replications described previously. The individual cultivar data was also pooled to examine the overall differences between the two boll states. Similarly, the NCVT raw, scoured, and bleached fibers were analyzed by ANOVA and Tukey's range test (α = 0.05) for metal differences among locations (n = 14) and among varieties (n = 10) based on the three prepared and treated sub-samples. Correlation analysis was performed to examine the relationship between the content of individual and total metal levels of the bleached fibers and fiber properties with dyed fiber color (L*, a*, and b*), differences in bleach and dyed color (ΔL*, Δa*, and Δb*) and ΔE values. Similar analyses were conducted with the fiber properties (i.e., maturity, fineness, and micronaire). All statistical testing was conducted with Minitab 18.1 (State College, PA, USA).
Results
Boll Harvesting Study
Ca, K, Mg, and P levels of the harvested cotton bolls were similar to the levels reported in prior studies on raw cotton fibers (Table II).12,14 K was present in the highest amount; the other three metals were present at 5 to 10 fold lower levels. ANOVA indicated that there were no significant differences in the levels of each metal between the freshly harvested and field aged bolls for each cultivar. Additionally, no significant differences were observed in the levels of the measured metals among the three cotton varieties for either boll condition. Pooling of the individual cultivar data did show a significant reduction (P = 0.009) of the K level for the aged bolls compared with the freshly-opened bolls. No other significant metal differences were found for the pooled data.
Cotton Fiber Calcium, Potassium, Magnesium, and Phosphorus Levels for Newly Opened and Field Aged Bolls a
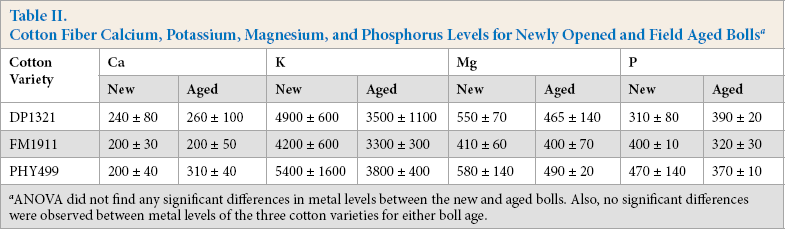
ANOVA did not find any significant differences in metal levels between the new and aged bolls. Also, no significant differences were observed between metal levels of the three cotton varieties for either boll age.
NCVT Fiber Samples
The first set of NCVT samples included 10 different cultivars grown in Lubbock during the 2016 crop year. In these samples, K was by far present at the greatest level (Table III), with the levels of Ca and Mg present at ∼20-25% the level of K. P was present at about half the level of Ca or Mg. The differences in the relative levels of the metals for the Lubbock grown samples and the three varieties grown in New Orleans suggest that environment had a significant effect on the relative levels of the different metals.
Raw Cotton Metal Content of 2016 NCVT Varieties Grown in Lubbock, TX a
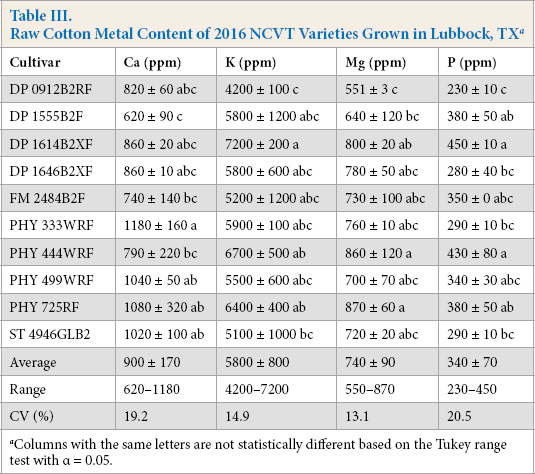
Columns with the same letters are not statistically different based on the Tukey range test with α = 0.05.
For the purpose of developing a set of fibers with different metals levels for dyeing, the variability in the metal levels was of interest. Ca levels varied by a factor of two from 620 to 1180 ppm, and the population of cultivar samples exhibited a coefficient of variation (CV) for Ca of about 19%. P levels, although smaller in magnitude, also varied by a factor of about two (230 to 450 ppm) and the population of samples had a similar C V. Greater levels of K and Mg have been reported by others,12,14 but the variation of K and Mg values was comparable to prior reports,12,14 with CV values of 15% and 13%, respectively. Overall, the relative differences among the Lubbock-cultivar samples were fairly modest, a result that was similar to that of Mullins and Burmester, 15 who evaluated Ca, Mg, and sulfur uptake by cotton and found few differences within four cultivars.
The second set of NCVT samples were of a single cultivar (PHY499) grown in 14 different environments across the cotton belt (Table IV). These samples exhibited a range of K levels (4100 to 7500 ppm) similar to that observed for the Lubbock grown NCVT samples and to those reported in prior literature.12,14 P levels covered a slightly greater range of values (300 to 620 ppm) than did the Lubbock samples. In contrast, the Ca and Mg values exhibited greater ranges, with the Mg values varying between 460 and 1260 ppm, and the Ca values exhibiting values between 760 and 4490 ppm. High levels (>3000 ppm) of Ca have been noted in prior reports. 16 Although not outside the range of previously reported values, the sample from Five Points, CA, USA also recorded the greatest levels of the other three measured metals (Table IV). In part, because of the Five Points sample, CVs for the PHY499 populations of samples were considerably greater than the CVs for samples from cultivars grown in Lubbock (Table III). The overall results suggest that environmental differences had a greater effect on raw cotton fiber values than did genetic differences.
Raw Cotton Metal Content of 2016 NCVT PHY499 Grown in 14 Locations a
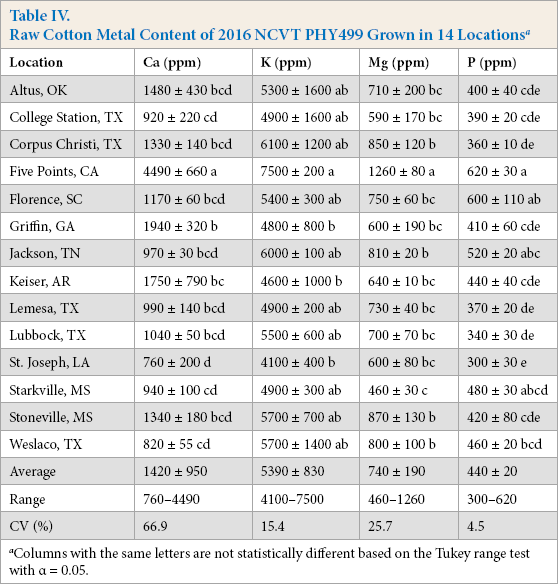
Columns with the same letters are not statistically different based on the Tukey range test with α = 0.05.
Scouring removes surface impurities from cotton fibers and is known to reduce metal levels.11,16,17 This was observed for both sets of NCVT samples. Scouring reduced fiber K levels by >97% of their initial values (not shown). Ca, Mg, and P levels were also reduced, but less so, and significant levels remained on the fibers. After souring, Mg and P levels of the Lubbock-cultivar samples varied between 130 and 360 ppm and 40 and 230 ppm, respectively (Table V), corresponding to an average reduction due to the scouring treatment of 65% and 60%. Ca levels were reduced by the lowest amount, with values that varied from 460 to 870 ppm, representing an average reduction of 23%. The PHY499-location samples exhibited similar metal reductions with scouring; the Mg samples were lower on average by 65%, the P values lower by 67%, and the Ca levels reduced by 39% (Table VI). Scouring also appeared to reduce the Ca levels of the Five Points sample to within the range of values exhibited by the other samples. Curiously, while Gamble noted that scouring reduced the Ca level of his fibers by a similar degree, almost no reduction in the level of Mg was found. 11
Scoured Cotton Fiber Metal Content of the 2017 NCVT Varieties Grown in Lubbock, TX a
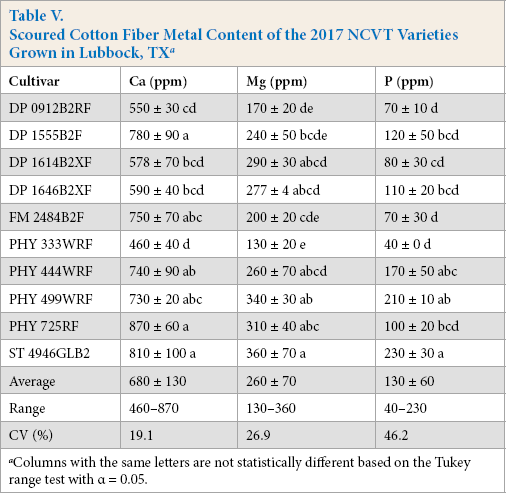
Columns with the same letters are not statistically different based on the Tukey range test with α = 0.05.
Scoured Cotton Metal Content of 2017 NCVT PHY499 Grown in 14 Locations a
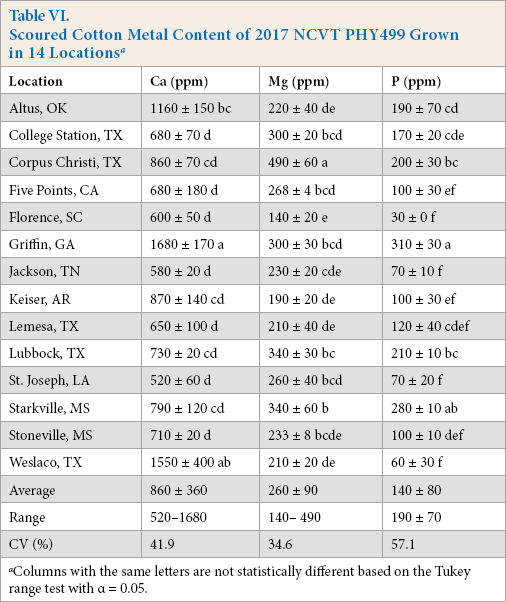
Columns with the same letters are not statistically different based on the Tukey range test with α = 0.05.
While the amount of metals was reduced in these samples, the relative degree of variation of the metal content among the different samples increased in most cases. For example, the CVs of the Lubbock-cultivar scoured samples were 29% for Mg, 19% for Ca, and 47% for P. The same CVs for the PHY499-location samples were 32% for Mg, 41% for Ca, and 59% for P. Comparison of these reductions with prior reports indicates that the effectiveness of scouring to reduce metal levels could be quite variable.
Bleaching further reduced metal levels (Table VII). 11 After bleaching, no samples contained >11 ppm levels of K (not shown). The bleaching treatment also further reduced Ca levels and lowered the levels of Mg and P, but to a much smaller degree than for Ca. For the Lubbock-cultivar samples, the combined scouring and bleaching treatments reduced the average P levels by 80%, Mg levels by 79%, and Ca levels 53%, from their initial raw fiber values. Similar percent reductions were observed for the PHY499-location samples (Table VIII). These reductions were 84%, 80%, and 67%, for P, Mg, and Ca, respectively. In effect, the bleaching step helped lower the levels of the Ca in the scoured fibers to almost a similar degree as that for the other three metals.
Scoured and Bleached Cotton Fiber Metal Content of the 2017 NCVT Varieties Grown in Lubbock, TX a
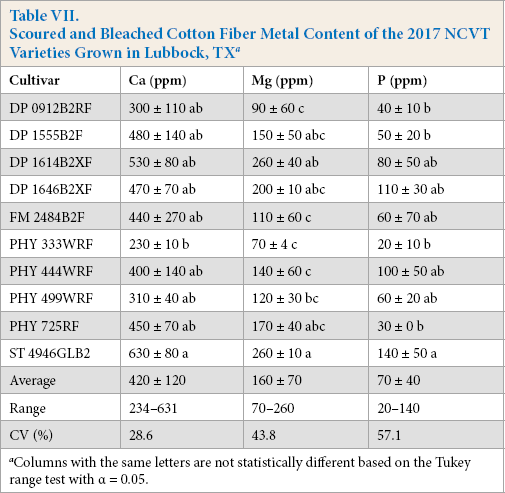
Columns with the same letters are not statistically different based on the Tukey range test with α = 0.05.
Scoured and Bleached Cotton Metal Content of 2017 NCVT PHY499 Cultivar Grown in 14 Locations. a
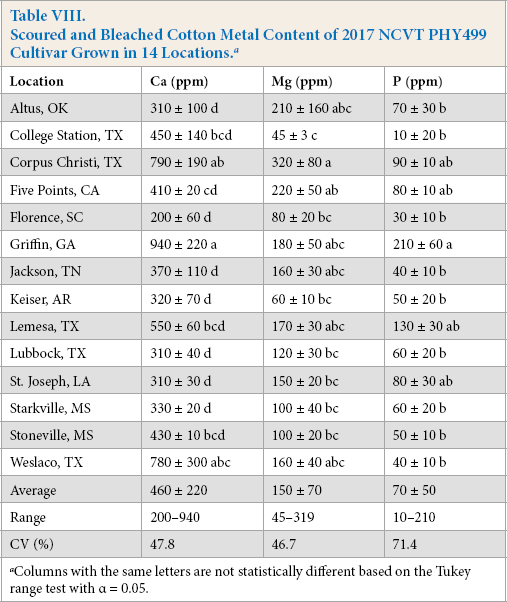
Columns with the same letters are not statistically different based on the Tukey range test with α = 0.05.
The CV of the bleached Lubbock samples for Mg, Ca, and P levels were 47%, 42%, and 55%, respectively, and the CVs for the PHY499 samples of the Mg, Ca, and P levels were 47%, 47%, and 69%. Hence, while the treatments reduced the levels of metals, the percent standard deviation or a measure of the variability of the samples significantly increased relative to the raw fiber samples. Because the variation of metals levels was comparable to prior reports and the variation only tended to increase with scouring and bleaching, the combined samples were believed to represent a range of differences likely to be observed by commercial processors of cotton.
To have the greatest range of variation in bleached metal content, the two sets of NCVT samples were combined for dyeing. All samples were dyed with Reactive Blue 19. Because the bleached samples might have significant differences in color that might influence dyed color, color measurements were made on the bleached and dyed fibers. The bleached fibers differed in color (L* from 89.6 to 93.0; a* from −0.75 to −0.38, and b* from 2.3 to 5.1). Dyeing both significantly altered the color values and yielded a wider range of color values (L* from 42.6 to 52.3; a* from -2.75 to 0.77, and b* from -39.3 to -43.2).
Comparisons between the individual metals (Ca, Mg, or P) and the total metal content with the individual bleached or dyed cotton color values or the ΔE values yielded few significant correlations (Table IX) with most Pearson-R values being <|0.30|. The sole exception was between the bleached fiber Ca content and its b* value, where the Pearson R value was +0.643 and was highly significant (p < 0.001). However, while the Ca level correlated with the yellowness of the bleached fibers, after dyeing, the effect was lost as this Pearson-R value was 0.101 (p = 0.689).
Pearson-R Values between Bleached Fiber Metal Levels and Fiber Color Before and After Dyeing a
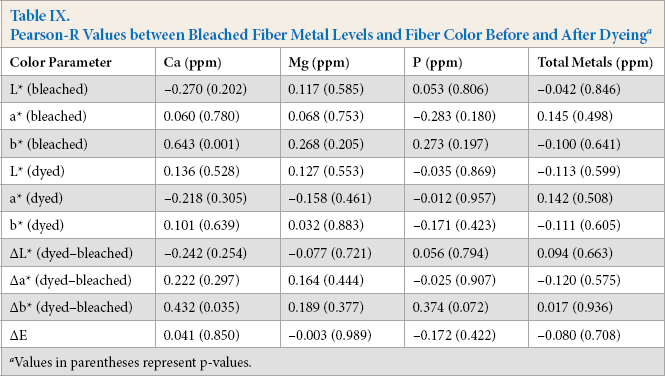
Values in parentheses represent p-values.
Measured fiber properties showed only a typical range of values (fineness: 179-207 mtex, maturity ratio: 0.96-1.05, and micronaire: 4.3-5.3), and the values indicated that the fibers were fairly average without extreme properties. No significant correlations were observed with the Reactive Blue 19 dye fiber color measurements used in this study, and these fiber physical properties (data not shown); hence, fiber properties did not appear to be a complicating factor that might influence the interpretation of fiber color variations with metal content.
Although the primary focus was on relating the metal levels of the bleached fibers to the dyed color values, a few other correlations were noted within the data. There were significant correlations among the bleached fiber metal levels. The Pearson-R between the Ca and Mg levels was +0.581 (p = 0.003), which might be related to a similar complexing that occurs with residual pectin that was not removed by the pre-dyeing treatments. Additionally, however, although seemingly unrelated, there was also significant correlations between P levels and both Ca (Pearson R = +0.629, p = 0.001) and Mg (Pearson R = +0.541, p = 0.006) levels. The mechanism responsible for these trends is unknown, but may be related to metal buried deep within the cellulose fibrils that was not removed by alkaline scouring treatments.
Color data for bleached and dyed cotton fibers with the color change ΔE by cultivar in Lubbock, TX and by location are available from the author upon request.
Discussion
Some prior literature has stated that cotton fiber metals levels are affected by field conditions after boll opening and during harvesting, due to dust and mineral deposition, and from contamination due to various plant and field debris.16,18 In this study, no large differences were found in the metal levels of newly-opened bolls and opened bolls allowed to age in the field for three weeks. K, Ca, Mg, and P fiber levels of these bolls were within the range of values previously reported for these metals. Hence, most of the metal present on cotton fibers appears to occur as a part of biological processes and deposition during fiber and boll development. Metal levels did not appear to be particularly sensitive to environmental conditions that occur after boll opening. While normal harvesting of the several field experiments were occurring in the field over the three weeks of study, the field did experience three significant rainfalls, and these may account for the statistically-lower K levels that were detected when the individual cultivar data was pooled. Still, given the reported large effect that water washing has on fiber K levels,9,17 the slight reduction suggests that even substantial rainfall only had minimal effect on changing the metal levels of the fiber within field bolls. This does not mean that instances do not occur where the fiber metal levels can be influenced by contact with dust or debris or weather events, but only that under fairly typical field conditions, these appear to be secondary influences.
The metal levels of the NCVT samples indicated that, for Ca and Mg, location differences tended to produce a wider range of levels than did genetic differences as measured by either the range of the values or by the CV of the sample populations (Tables III and IV). The uptake or deposition of Ca appears to be particularly sensitive to environment, as exhibited by the extremely high level of Ca (>4400 ppm) that was found in the Five Points, CA sample. Most prior studies have found Ca levels to be between 600 and 1500 ppm, and prior California sourced samples all have noted Ca levels below 1000 ppm.12,14 Soil pH is relatively high in Five Points, CA, (around 8), which may reflect higher levels of calcium carbonate (or limestone) and that may influence the availability and uptake of Ca by plants. 19 There also appeared to be more variability in the Mg levels of the samples grown in different locations than in the samples generated from different cultivars. For Mg, however, the range values appeared to be comparable to the range of values previously reported in older studies.12,14 K levels found in this study were essentially the same as in prior reports. Little information is available on P levels in cotton, so few comparisons with prior data can be made.
As noted by others,10,11 scouring and bleaching reduced metals levels significantly. K levels were lowered by >95% to levels that were unlikely to have an important influence on dyeing properties. Varying levels of Ca and Mg were removed, but significant levels were retained, as noted by others, although the pattern of removal with successive treatments appeared to be somewhat different than previously reported. This suggests that some variability existed in the effectiveness of the treatments to remove metals for different fiber samples, which contributed to the variation in the metal levels present on fibers entering the dyeing process. This likely justifies the use of sequestrants during scouring and bleaching treatments.
No correlations were found between the levels of Ca, Mg, or P on the dyeability of the fibers. Hence, this exhaustive dye system was not affected by the metal differences represented within the samples. Of course, several factors can influence these results. If fibers with greater metal levels were dyed, perhaps differences would be observed. Other dye treatments might be more sensitive to metal levels. Also, continuous throughput processing systems with relatively short contact times that are not as exhaustive may be more sensitive to metal levels.
Curiously, a significant positive correlation was found between the bleached fiber b* values and their Ca levels. Similar correlations were not apparent with Mg or P levels. Unfortunately, we did not have sufficient fiber to test the color of the scoured only or raw fibers to see if a similar correlation could be found prior to the bleaching process. This result was a little contrary to a result by Gamble, 11 who that found that an EDTA treatment between the scouring and bleaching step removed more Ca than did a simple water wash, but the b* values of the bleached fabrics were essentially comparable despite their large differences in Ca levels. The difference points to the many factors that can influence the final dyed product, including the growth conditions, the field and harvesting product of the fibers, their various pretreatments, and the many dyes and dyeing systems that are used in cotton textile processing.
Conclusions
No significant differences were found on the metal levels between freshly-open bolls and opened bolls that were allowed to stand in the field for three weeks during harvesting season, which suggests that weathering, winds, rain, and physical harvesting practices had a secondary effect on the metal levels of cotton fiber. Metal levels of cotton fibers decreased with progressive fiber treatment (i.e., scouring and bleaching), and significant correlations were found between the Ca, Mg, and P levels of the scoured and bleached fibers. Yet, in this study under these preparation and dyeing conditions, there appeared to be no direct relationship between the levels of metals on the bleached fibers and the color of the exhaustively-dyed fibers. Although these experiments may be in conflict with some current beliefs, the results of this single system cannot be used to suggest that metals levels will never affect dyeing, but only that not much of an effect was apparent for this fairly-standard dyeing system that is often used in bench research.
Footnotes
Trade names are used solely to provide specific information. Mention of a trade name does not constitute a warranty or an endorsement of the product by US Department of Agriculture to the exclusion of other products not mentioned.
