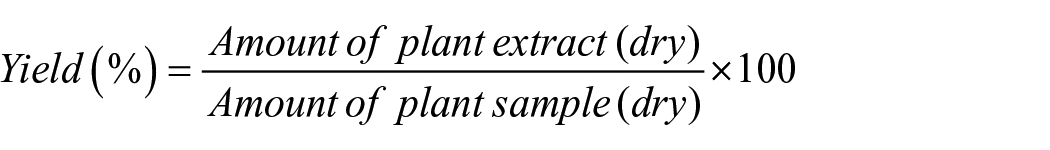Abstract
In this study, a green method was employed for dyeing silk and cotton fabrics using extracts from Beta vulgaris peels as a colorant, pomegranate rind as a bio-mordant, and microwave-assisted extraction as a green extraction technique. The dyeing process was optimized employing response surface methodology and characterization of the dyed fabric was conducted using FTIR, SEM, color strength, and fastness properties. The optimized conditions for dyeing were a solvent-to-extract ratio of 10 ml/g, temperature 70°C, and pH 6. The values of K/S calculated under optimal conditions were 2.565 for cotton fabric and 3.928 for silk fabric, with good fastness properties ranging from 4-5. Better fastness properties were obtained at low pH and a high solvent-to-solid ratio. FTIR confirmed the different functional groups including prominent peaks appearing at 1600–1700, 1546, and 1200–1300 cm−1 for Amide-I, amide-II, and amide-III functional groups in silk and 3271–3332 cm−1 (OH stretching), 2851–2915 cm−1 (C–H stretching) for cotton fabric. SEM analysis showed the characteristics of the fabric surface before and after the adsorption of the dye components. The current study revealed selected bio-waste as a new and cheaper source of coloring material. Natural dye obtained from different waste materials can be exploited as a dyeing tool for different fabrics on an industrial scale, providing a viable green alternative to hazardous synthetic colorants.
Introduction
Sustainable waste management refers to the collection, transportation, valorization and disposal of numerous sorts of wastes. The industrial use of food materials generates tons of waste materials due to processing fruits and vegetables.1–5 The disposal of these wastes is one of the major issues due to their possible harmful impacts on natural ecosystems, habitats due to greenhouse gas emissions, excessive use of water and land. One of the possible utilization of these waste materials as a sustainable and green source of natural dyes and pigments for fabrics that can reduce waste and related pollution in the environment. 6
Chemical compounds as coloring agents are known as dyes that may have synthetic or natural origin. Various organic sources, like plants, bacteria, fungi, and mildew have the potential to produce pigments. These compounds are disposable and less harmful to humans and the environment so their use in textiles has garnered increasing interest on a global scale.7,8 There are significant drawbacks in using natural pigments for dyeing, including poor repeatability, little dye fatigue and poor fastness. However, due to the favorable benefits on human health, many natural components can be utilized to create a variety of finished dyeing agents. Organic natural colors have been produced from the waste materials of many plants, including dried onion peel, nuts and walnut leaves. 9 Depending on the structure of fibers, natural colors can be applied directly or indirectly using a mordant to natural or chemical fibers. By choosing the right dyeing method or mordant, it is possible to create a wide range of colors. Natural dyes own several noteworthy characteristics such as being biodegradable, non-toxic, non-allergenic and cost-effective which give them a substantial advantage over manufactured dyes. 10
Beetroot (B. vulgaris) betalains are widely available natural colorants with a powerful practical red-violet tint that has dominated the food additives market.11,12 The cultivation of beetroot spans the world especially continents of the America, Europe, and Asia. According to a report in 2017, global beetroot production was more than 301 million tons. Western Europe has been emerging as the leading contributor with approximately 70% of the total 200-million-ton production annually. 13 While primarily consumed as a vegetable, beetroot juice is also available in the market and resulting waste can be transformed into a powder to serve as a coloring agent. 14 The manufacturing of beetroot juice, which is gaining popularity for its ability to lower blood pressure, results in significant quantities of pulp waste that predominantly end up in landfills.
Apart from the peel and pomace, the aerial parts of beets (leaves and stalks) are usually discarded after beet processing but contain valuable phytochemicals. 15 Hence, finding valuable uses for beet processing waste can play a crucial role in minimizing waste generation, aligning with the concept of zero waste. 16 The beetroot peels in addition to the complete taproot also contain comparable amounts of nutrients, bioactive compounds and natural dyes. 17 In recent research, there has been a particular emphasis on exploring the potential applications of various chemical components obtained from red beet waste. These studies were conducted with the primary objective to investigate the extraction and utilization of phenolic compounds and betalains present in beetroot waste that can have significant commercial value.18,19 Tannins are natural compounds found in many plants that have excellent dyeing properties because they can bind well with textile fibers. Utilizing tannin-rich sources like tree bark, leaves, and agricultural leftovers is a sustainable and eco-friendly option for dyeing textiles. 20 Re-using waste materials containing tannins from industries such as winemaking, brewing, and leather processing not only helps the environment but also promotes circular economy practices.21,22 In addition to providing vibrant colors, they also offer benefits like UV protection and antimicrobial properties to textiles. 23 Exploring tannins as a renewable and versatile resource for natural dyeing is a promising avenue. 24
The residual material obtained after the extraction process of beetroot, commonly known as pomace, is often perceived as a waste. Pomace typically accounts for approximately 15%–30% of the initial beetroot volume after squeezing. Beetroot waste contains various dyeing agents including betanin, isobetanin, 2, 17-bidecarboxy-neobetanin, betanidin, 2-O-glucosyl-betanin and isobetanidin.25,26 Betalains are brittle pigments that can change color due to changes in pH or enzymatic hydrolysis when exposed to light.27,28 Betacyanins (bright colors) and betaxanthins, two types of carotenes are responsible for the beetroot’s red hue. Among betacyanin compounds, betanin is the primary component responsible for the vibrant color. Betanin can be extracted from beetroot and utilized as a red food dye commonly referred to as “beetroot red,” which is assigned the E number E162. 29 Indicaxanthin on the other hand imparts yellow to brown color and can be an alternative to yellow dye. 30
Biomordants refer to natural substances derived from living organisms that are utilized in the dyeing process to enhance the colorfastness and bonding of dyes to textiles. 31 These sustainable alternatives to synthetic mordants offer eco-friendly solutions for the textile industry. Mordants used in the dyeing process enhance the chemical bond between the fiber and dye, directly influencing the color strength and fastness. 32 Bio-mordants can be derived from various sources such as plants, fungi, and bacteria. Common bio-mordants include polyphenols from plant extracts, which can form stable complexes with metal ions, and microbial-derived extracellular polymeric substances that exhibit metal-binding properties. 33 The use of bio-mordants not only addresses environmental concerns associated with traditional metal-based mordants but also contributes to the development of greener and more sustainable practices in textile dyeing. 34 The exploration and application of bio-mordants align with the growing demand for eco-conscious and socially responsible approaches in the textile and fashion industries. Pomegranate peel contains polyphenolic compounds gallic acid, catechin, ellagic acid, and apigenin-7-glucoside and anthocyanins that could act as bio[mordants] for textile dyeing.35–37
Recent research has increasingly focused on using plant-based dyes for sustainable dyeing of protein-based textiles like wool and silk. Natural dyes from natural sources like henna and jujube leaves have shown potential in improving color depth and fastness, especially when combined with mordants.38,39 Techniques like ultrasonic waves and plasma treatment have further enhanced dye uptake and performance. 40 Additionally, pomegranate leaves have been explored as a viable dye source, enabling effective dyeing without synthetic mordants. 15 These approaches not only improve dyeing efficiency but also offer eco-friendly alternatives with added antimicrobial properties to the textiles.41,42
Scientists have increasingly embraced modern methodologies over conventional techniques to streamline process expenses, reduce labor requirements, and minimize chemical usage. 43 Contemporary approaches such as microwave, ultrasonic, gamma, ultraviolet, and plasma-assisted extraction are now being employed for the conservation of natural dyes and coloring, marking a shift from traditional methods to environmental friendly green techniques.44–46 In comparison with conventional extraction techniques, the economic benefit of microwave extraction of plant-based bioactive chemicals seems to be rather realistic, as it requires less time and electricity. 47 In microwave-assisted extraction (MAE), the selection of the extraction solvent is primarily based on factors such as security, cost and environmental friendliness. MAE has many advantages reduced equipment size, reduced thermal gradients and an increase in extract yield as compared to other conventional techniques. 8
Cotton fibers consist of 88%–97% cellulose, with the remaining components being waxes, proteins, and pectin. Cotton fibers are composed of a long molecular chain called cellulose, which features hydroxyl groups (-OH) within its molecular structure. On the other hand, silk fiber obtained from insects such as silkworms is primarily composed of fibroin, a natural protein. 48 Therefore, fibroin is the most suitable term to describe the protein found in silk fibers. Although the colorants possess functional groups crucial for dyeing fabric, it is reported that textiles do not exhibit any affinity for the dyes derived from both pulp and red beet peels (known as betalains). Nevertheless, under specific circumstances, betalains can effectively and durably dye protein-based textiles, creating vibrant and long-lasting colors. To adhere the pigment to the fabric, mordanting is done using metallic or bio-mordant. 49
Keeping in view previously reported data, the current study was designed to develop a method for utilizing beetroot waste as a natural alternative to hazardous chemical dyes. The innovation of this study is to develop a dyeing method for selected fabrics using food waste that will be a resource-efficient process in alignment with global organic textile standard (GOTS) and natural organic dye standard (NODS) criteria and repurpose discarded plant materials to create environmentally friendly dyes for textiles.50,51 To keep the process sustainable, MAE was selected as a green extraction technique and bio-mordant was used instead of chemical mordant making the process an inexpensive green procedure for cotton and silk dyeing. To achieve the best outcomes of the designed experiment at a large scale, conditions of the dyeing process were optimized using response surface methodology (RSM). The current study is a motivation for the scientific community and industrial officials to explore a reliable, resilient, and eco-friendly technique for fabric dyeing utilizing beetroot waste and pomegranate rind through a comprehensive green method.
Materials and methods
Pretreatment of materials
B. vulgaris peels and pomegranate rind were obtained from the caterer’s firm (Zahoor Sons Caterers, Lahore). The colorants and bioactive compounds were extracted from peels and pomegranate rind to be utilized as a bio-mordant. Figure 1 shows the structural representation and functional groups present in peels, silk and cotton. Both plant materials were washed with water and shade dried for 7 days and converted into powder form (100 mesh size) using an electric grinder (Model Anex AG-640). The powdered material was stored in sealed plastic bags and refrigerated till further use. Plain cotton fabric (70 GSM cotton fabric with warp count (Ne) 60 and weft count (Ne) 60, the warp and weft densities as 23.62 ends/cm and 23.62 picks/cm, respectively) and silk fabric (GSM value of 60, warp count (Ne) 40, and weft count (Ne) 30, the warp density is 0.015 ends/cm, and the weft density is 0.02 picks/cm) were purchased from the local textile market in Lahore, Pakistan.

Microwave-assisted extraction of dye
A household microwave oven (LG Intellowave Sensor model MS-3242DPS) was used to perform microwave-assisted extractions using distilled water as a green solvent. Single-factor experiments were designed to find out the best possible ranges for the extraction procedure keeping solvent-to-solid ratio (ml/g), time (min), temperature (℃) and microwave power (W) as variables. For each experiment, the selected solvent-to-solid ratio (10–25 ml/g) was added to a 250 ml beaker and each beaker was microwave irradiated at selected power (160–800 W), time (2–5 min) and temperature (20℃–80℃) (Figure S1). To prevent overheating of the solvent and evaporation, the microwave extraction process was carried out in pulse mode with periodic cooling using ice water following the reported method and turned off the heating for 30, 15 s and the process was continued for a time duration as per the experimental design. 53
After extraction, the plant material was filtered, extract was dried, weighed and was used directly as sustainable dyestuff. Quantification was done and percentage yields were calculated as follows:
The mordant solution was prepared in distilled water using powdered pomegranate rind at a solvent-to-solid ratio of 30 ml/g by maceration for 3 days at room temperature. Afterward, the solution was filtered, dried and quantified. For mordanting purposes, 50 g extract was dissolved in 1 L of water and was filtered. 54 The dyestuff Beta vulgaris peel extract (BV-PE) and mordant solutions were stored in the refrigerator for further use.
Experimental design for dyeing fabric
The design of experiments and statistical analysis of responses were performed using Design Expert software (version 7.0, Stat-Ease Inc., USA). In this work, the impact of three key operational factors on the optimization of the bio-mordant treatment process and color strength of cotton/silk fabric dyed with BV-PE was assessed using RSM and Box-Behnken design. 55 Before planning the experimental runs, preliminary research was conducted to identify the most affecting variables and establish a practically acceptable range for each parameter. The selected independent variables and their coded level are shown in Table 1.
Selected experimental ranges for extraction of dye from BV-PE.

Mordanting and dyeing process
The fabric was initially washed with water before being utilized for the dyeing procedure (Figure 2). For the preparation of fabric for dyeing a reported method was followed. 56 Scouring of the fabric was done by washing the fabric with sodium carbonate and detergent solution for an hour. The bleaching was performed at pH 10–10.5, temperature 80°C–90°C for 1 h using 10 g/l hydrogen peroxide (H2O2), and 2 g/l NaOH as a bleaching agent. The resulting scoured and bleached fabric was then cut into 12 in × 18 in pieces for subsequent dyeing applications.

Schematic representation of mordanting and dyeing.
A meta-mordanting procedure was carried out at different conditions as per the design given in Table 2 and the control sample was dyed using only pomegranate peels without BV-PE. The fabrics were simultaneously mordanted and dyed for 1 h with beetroot peel extract using pomegranate rind as bio mordant (Mordant solution to dye solution ratio as 50:1). The dye bath temperature was raised gradually (1°C per min) to higher temperatures as per experimental designs and maintained there for 60 min. The BV-PE is an acidic colorant since the dyestuff is naturally acidic and has a pH of 5–6. To alter the pH of the solution under various acidic and alkaline conditions, acetic acid 10% solution and NaOH 10% solution were employed, respectively. After 60 min of dyeing 2 g/l soda ash was added (as a dye fixer) and left in a dyeing bath for 10 min. After that, the dyed samples were rinsed with water and air-dried (Figure 2). 57
Effect of different optimization factors on color strength (K/S) values and shades of dyed fabric samples.

Fourier transform infrared spectral analysis (FTIR)
Interactions between functional groups and bonding between fabric, dye and bio-mordants were investigated using a Bruker FTIR (model αT) spectrophotometer in the spectral range of 4000–500 cm−1 with a single-bounce diamond crystal ATR. Measurements were conducted at a resolution of 2 cm⁻¹ and ATR-FT-IR spectra were directly recorded from the textile sample. The sample was positioned on the ATR crystal, pressure was applied, and the ATR-FT-IR spectrum was then scanned.
Color fastness and strength measurements of dyeing fabric
The rubbing fastness properties of dyed fabric were measured based on ISO 105X12:2016 (dry and wet; length and width wise) and color strength (K/S) was measured (using Datacolor D850 spectrophotometer) according to AATCC Evaluation Procedure 6, (single wavelength; 410 nm, temperature; 20.1℃ and humidity 65.1%) at National Textile Research Center, Textile Physical and Chemical Laboratory, National Textile University Faisalabad, Pakistan. The following equation was used to calculate the color strength (K/S) of each colored test sample:

where R is the observed reflectance at the wavelength of maximum absorbance, K is the absorption coefficient, and S is the light scattering coefficient.
Scanning electron microscopy
Scanning electron micrographs were taken using FEI Nova 450 Nano SEM at Syed Babar Ali School of Science and Engineering, LUMS Lahore Pakistan to examine the effects of dyeing and bio-mordanting procedures on the surface morphology of cotton fibers.
Results and discussion
The maximum yield obtained in the initial single-factor experiment was calculated as 29%. The best yields were obtained at 320 microwave power, 3 min time, 50℃ temperature and 20 ml/g solvent-to-solid ratio.
Analysis of dyed fabric (cotton/silk)
Test samples of dyed cotton/silk fabric were prepared as per design and were dyed using the methodology given in Table 1. The dependent variables and corresponding response (color strength: K/S) of the dyed samples are shown in Table 2. Statistical models of the procedure and the impacts of three independent parameters on the color strength of dyed cotton/silk textiles were constructed and are presented as 3D response surface plots in Figure 3.

Three-dimensional response surface plots for the effect of different variables on K/S: (a–c) for cotton and (d) silk fabric.
Factors affecting K/S values for cotton fabric
Effect of pH on K/S values
In this study, it was observed that the highest K/S value (5.61) was obtained at pH 4 (Temp: 30°C and solvent-to-solid ratio: 15 ml/g). Low pH values favor the dyeing of cotton fabric and a periodic decrease in K/S values was recorded with increasing pH beyond 7. 58 The pigment present in B. vulgaris peel extract is betalains (nitrogen-containing compounds) including red-violet Betacyanins and yellow betaxanthin imparting red-violet and yellow color respectively. The pH affects betalains color. Betacyanins degrade at pH < 4, beyond 7.0 and show no affinity for fabric (washed off easily) while betaxanthin imparts color at the 2.2–7.0 pH range. 59 At higher pH, the compounds tend to degrade and the K/S value also decreases. The dyeability of materials is therefore greatly influenced by the pH values of the dyeing bath.60–62 It has been reported that low pH values can improve the dyeability of pigments, but it dramatically decreases by increasing pH. The dye’s anion has a complicated nature, and when it is attached to cotton cloth, additional types of interactions happen in addition to ionic forces. Since the utilized dye has hydroxyl groups and is water soluble, it would interact with the protonated groups of cotton or cellulose fabric through an ion exchange process at an acidic pH. An increase in the pH of the medium causes a significant increase in repulsion between anions of dye and cellulose hence lower dyeability in the case of pH 8 may be attributed to the enhanced desorption of the dye as its ionic bond strength is decreased. 63
Effect of temperature on K/S values
It has earlier been reported that an increase in the temperature of the dye bath may decrease color strength and have a negative influence on K/S values. 64 Maximum K/S value was observed as 5.61 at 30°C and minimum as 2.34 at 70°C. The color strength rises as the dyeing bath temperature rises to 30°C, and when the dyeing temperature is raised further, the strength of K/S gradually decreases, which is attributable to the dye’s poor stability at higher temperatures. In this investigation, we found that dye adheres to the surface of cellulose fibers and aggregates. By raising the temperature of the dye solution, the aggregate number is reduced. The dye clusters disintegrated as a result of an increase in temperature. Another possible explanation is the exothermic nature of the reaction between the dye and the cotton fabric, which might lessen the affinity of the dye molecules for the cotton fiber and lessen fatigue as the temperature rises. 65 In the current study, the response surface plot (Figure 2) shows an interactive correlation between pH and temperature of the dyeing process and its influence on K/S values. Betalains exhibited stability within a pH range of 4–5 but are susceptible to degradation when exposed to elevated temperatures, indicating their sensitivity to heat. 66 At pH 4 and temperature 30°C, the maximum K/S value was observed as 5.61. As both temperature and pH increase, K/S values of cotton fabric decrease because of possible degradation of dye molecules (betalains) (Figure S3). As a result, degradation products are released at higher pH which is the reason for the decrease in the adsorption of dye on fabric.66–68 This observation is in accordance with the reported studies that moderate temperature and low pH are optimal conditions for dyeing using BV-PE. 52
Effect of solvent-to-solid ratios on K/S values
In this study, it was observed that an increase in solvent-to-solid ratio increases the K/S values of the dyed cotton fabric up to some extent. The highest K/S value is observed at 15 ml/g solvent-to-solid ratio as 5.61 for the cotton sample. At very low solvent-to-solid ratios, accumulation of dye molecules occurs at the surface of fabric as a result of the formation of aggregates while very high solvent-to-solid ratios do not cause significant sorption of dye on cellulose fabric.63,69 The initial dye concentration is one of several variables that affect adsorption. The increase in initial dye concentration results in an increase in a concentration gradient, which causes a rise in the amount of dye absorbed onto cloth to the greatest extent before becoming constant at the saturation point. 70 In this study, the solvent-to-solid ratio has a relatively minor effect on K/S values compared to other independent variables. The observation suggests that an optimum ratio is achieved early; beyond this point, increasing the solvent-to-solid ratio yields minimal improvement in K/S values, as dye dispersion and adsorption are already maximized, and further dilution may even reduce dye concentration and color strength.
Factors affecting K/S values for silk fabric
Effect of pH on K/S values
For silk samples, the highest K/S value (5.42) was obtained at pH 4 (Temp: 30°C and solvent-to-solid ratio: 15 ml/g) and the lowest was observed at pH 8 (Temp: 50°C and solvent-to-solid ratio: 20 ml/g). BV-PE dyeing agent has a functional group (-COO-) and these groups retain as COO- in an acidic environment at pH 4. These COO- groups of dye molecules react with the protonated amino group (NH3+) of fabric under acidic conditions (at pH 4), which is key for enhancing dye fixation onto the silk fiber as shown in the equation below. 71



A decrease in K/S value with increasing pH can be described based on the mechanism of interaction between dye and fabric (Figure 3(d) and (e)). Bio-mordants have demonstrated comparable performance when compared to chemical mordants. This can be attributed to the presence of conjugated benzene rings and -OH groups as the binding sites. These binding sites interact with the -C=O and -OH groups of the colorant from bio-mordant extract, as well as the amide linkage (-CO=O and -NH2) of silk. This interaction leads to the formation of additional hydrogen bonds during the development process. 72 Temperature had no discernible impact on K/S values while dyeing silk in correlation with pH and a linear decrease was observed when pH and solvent-to-solid ratio increased.
Effect of temperature on K/S values
It is reported that direct dyeing processes are greatly influenced by the temperature of the dyeing bath as heating results in enhancing the kinetics of the process but only up to a certain level.73,74 In the case of silk dyeing, the K/S value decrease when the temperature ranged from 30°C to 70°C. The combined effect of dyeing temperature and pH has been presented in Figure 3(d). K/S values dropped as dyeing temperature and pH increased as a result of thermal as well as chemical degradation of dye molecules and cellulose fiber. 1 The combined effects of thermal and chemical degradation at elevated temperatures and pH levels are in according with the previously reported studies where increase in pH and temperatures collectively impair the dyeing efficiency and durability of the color on cellulose fibers, resulting in reduced K/S values. 75
Effect of solvent-to-solid ratio on K/S values
The highest K/S value (5.42) was obtained at 15 ml/g solvent-solid-ratio (Temp: 30°C and pH 4). An increase in solvent-to-solid ratio showed a decrease in the K/S values of dyed silk fabrics. The initial dye concentration is one of several variables that affect adsorption. 69 As the solvent-to-solid ratio increases, the K/S value on silk shows a slight decrease. This indicates that a higher solvent-to-solid ratio (greater dilution) may lead to reduced color strength on the silk. As the solvent-to-solid ratio rises with a fixed amount of dye and fabric, the color intensity diminishes. This is due to the dilution effect; adding more solvent at the same dye concentration lowers the dye availability in the solution, which reduces dye uptake by the fabric and results in decreased color intensity. 76
Data analysis (ANOVA) and optimization
For cotton fabric
The results revealed that the quadratic model best fits the studied dependent variable. The sequential p-value (<0.0001) and the difference between the predicted R2 and the adjusted R2 (less than 0.2) indicate that the model is significant. By focusing on the model maximizing the adjusted R2 and the predicted R2, the quadratic model was the best-fitted model for describing the effects of the variables on the K/S value response in this natural dyeing process and these effects are presented as 3D response surface plots (Figure 3).
Analysis of variance was carried out to determine the important factors and interactions in the dyeing process. ANOVA helps to determine whether statistically significant differences exist between the mean values of two or more independent groups. 77 The model is suggested to be significant by the model’s F-value of 112.91. The significant factors identified include A, B, AB, BC, A², and B², all of which play crucial roles in the dyeing process. The model’s high R² value of 0.9932 suggests that the model fits the experimental data extremely well, demonstrating excellent predictive power. Additionally, the lack of fit p-value of 0.0826, being higher than 0.05, indicates that the model adequately fits the data without significant deviation, further confirming its reliability. 78 The Predicted R² of 0.9121 is in good agreement with the Adjusted R² of 0.9844, as the difference is less than 0.2, which is acceptable. Adequate precision, which assesses the signal-to-noise ratio, is 40.952, well above the desirable threshold of 4, indicating a strong signal. This model is reliable for navigating the design space effectively. 46
The equation in terms of coded factors is useful in predicting experimental data as responses for given levels of each independent variable. The created model illustrating the mathematical relationship between the coded elements and K/S is shown in equation (2).

For silk fabric
The results showed a linear model best fit for the studied dependent variable. The sequential p-value (<0.0001) and the difference between the predicted R2 and the adjusted R2 (less than 0.2) indicate that the model is significant (Table 3). Analysis of variance suggested the linear model to be best fitted and significant based on the model’s F-value (46.02). Based on p-values significant model terms were A (pH) and C (solvent-to-solid). A high R2 value (0.91) verified the suggested model’s adequate adherence to the trial data. The final equation in terms of coded significant factors for K/S (silk) was found as:

Coefficient data for responses of dyed fabric samples.

The results for ANOVA (Table 3) indicate the linear model for silk shows an R² of 0.9139 and an adjusted R² of 0.8941, which still explain a large portion of the variation, with a model p-value of less than 0.0001. 78 The lack of fit p-values for both fabrics (0.0826 for cotton, 0.1053 for silk) indicate that both models are well-suited to the data. The predicted R² of 0.8380 is in good agreement with the Adjusted R² of 0.8941, (difference of less than 0.2). Adequate precision is 22.289 (indicative of signal-to-noise ratio) which is well above the recommended threshold of 4, indicating a strong and adequate signal. 79 The simplified linear model for silk suggests that factors like pH, temperature, and solvent-to-solid ratio have more straightforward effects on color strength in silk compared to cotton, where interactions and quadratic effects play a more significant role.
Optimization and validation of the data
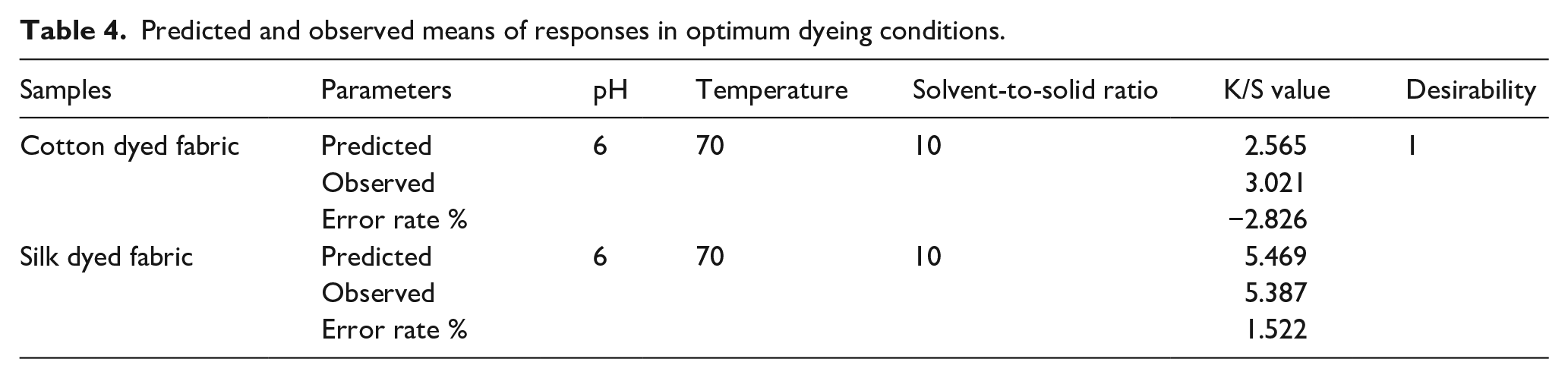
Using the optimization feature of Design Expert software, the ideal circumstances for the bio-mordant treatment procedure and achieving the highest K/S after dyeing with beetroot peels were projected. All process parameters’ values were chosen to fall “in range” keeping the solvent-to-solid ratio as “minimum” in order to keep the process sustainable. The optimum condition was suggested as follows; solvent-to-solid ratio of dye 10 ml/g, 70°C temperature, pH 6 (using fixed bio-mordant concentration 10 g/100 ml and curing time 60 min) with desirability as 1 and results are reported in Table 4. The maximum predicted value of K/S under the optimal condition was 2.565 for cotton fabric and 3.928 for silk fabric.
Predicted and observed means of responses in optimum dyeing conditions.
Surface analysis of undyed and dyed cotton fabric
The change in the surface morphology of the undyed and dyed cotton fabrics was examined using the SEM technique. SEM images (Figure 4(c) and (d)) highlight significant differences in surface structure and dye adherence between untreated cotton (a and b) and bio-mordanted dyed cotton. Raw cotton fabric surface’s convolution was found to be smooth and without any obvious physical alterations. The results are displayed, and the undyed fabric is smooth and unique with minimal surface deposits. 80 The colored fabric, shows increased surface roughness with visible dye particles embedded along the fibers. This pattern suggests strong dye attachment, likely aided by the bio-mordant, which enhances dye bonding to the cotton surface. 81 The SEM images clearly illustrate the effectiveness of the bio-mordanting dyeing method, with variations in surface morphology indicating the interaction between the natural dye and the cotton fiber structure. 82

(a and b) SEM images of raw cotton fabric at different magnifications with an accelerating voltage of 3.00 kV and (c and d) SEM images of (bio-mordanted) dyed cotton fabric at different magnifications.
It can be predicted that the physical or mechanical properties of fiber will remain unaffected due to dyeing with BV-PE at different temperature ranges as 40°C–70°C.59,83 No change in surface features of the mordanted/dyed fabric in comparison with undyed fabric that is, no cracks or voids were observed on the surface after dyeing that are usually considered responsible for fiber properties. These results confirmed that dyeing with coloring agents from natural waste can be safe and innocuous use with different bio-mordants. 84
Fourier transform infrared (FTIR-ATR) spectroscopy analysis
FTIR analysis on dyed cellulose provides valuable information about the chemical composition, bonding, and structural changes induced by the dyeing process, aiding in the understanding and quality control of dyed cellulose materials. 85 Major absorptions in FTIR spectra of cotton fabric are shown in Figure S2. The broad free -OH stretching peaks of cellulose (cotton fibers) appear at 3271−3332 cm−1. When cotton is colored with BV-PE dye, the peak frequency is the same as when it is undyed, but only the peak intensity increases toward higher wavenumbers, signifying a change in the hydrogen bond. A peak appearing at 2851–2915, 1644, and 1425 cm−1 are associated with C-H and CH2 stretching, adsorbed water/moisture and CH2 symmetric bending of the cellulose respectively. 86
Peaks for (C-H) stretching; (CH2) wagging and (C-H) stretching appear at 1314–1363 cm−1 while characteristic vibrations of the anti-symmetrical bridge of C-O-C stretching are shown at 1158 cm−1 and peak shift are observed in this region which indicates involvement of these functional group in making H-bonding with cellulose. Primary alcohol stretching and C-O-C (1-4)-glycosidic linkage frequency were observed at 1016 and 891 cm−1 respectively. Peak intensity reduction at 891cm-1 indicates bonding of dyeing phyto-constituents on the fabric surface. New peaks appearing in the dyed fabric at 1050 and 1106 cm−1 are associated with C-O stretch and C-O-C aryl conjugated rings. These functional groups are reported in pomegranate tannins and bio-mordants with fabric surfaces. 87
In the FTIR spectrum of silk minor changes in peaks were observed. Peaks appearing at 3380–3450 cm−1 are associated with free -OH and N-H stretch. Peaks at 1018 and 1143 cm−1 are combination peaks for C-N stretching, N-H, and C-H. 88 Peaks appearing at 1600–1700, 1546, and 1200–1300 cm−1 are associated with Amide-I, amide-II, and amide-III functional groups respectively.89,90
Fastness properties of dyed
Color fastness to rubbing
The results in the current study showed the maximum rating for dry rubbing of dyed cotton fabric as 4–5 and wet rubbing as 3–4 while the minimum rating for dry rubbing was found to be 4–5 and wet was 2. The silk fabric’s maximum dry rub rating was 4–5 and its maximum wet rub rating was 3–4. The minimum rating for dry rubbing was 4–5 and for wet rubbing 3.
Mordanting improved the fastness characteristics of colored cellulose materials. Mordanted colored textiles have better fastness qualities than un-mordanted fabrics because bio-mordants may chelate dye molecules that are forming complexes in the dyed fabric. 91 Pomegranate rind contains many polyphenolic compounds that possess the ability to bind both fabric and dye molecules and these functional groups of the biomordant (-OH groups), interact with the -OH groups of the betalains present in beetroot peel extract, as well as the -OH groups of the cellulosic fabric. 92 This interaction involves additional hydrogen bonding, creating a soothing and firm coloration with unique tints and enhanced color strength.91,93 Pomegranate peel contains natural tannins and other compounds that enhance the binding of the dye to the fabric fibers. As a biomordant, it offers an eco-friendly alternative to synthetic mordants, helping to fix the BV-PE dye onto silk and cotton. 94
Table 5 represents the fastness values of the dyed sample. All samples of cotton and silk fabric show good/very good values or ratings in dry staining, which is a 4–5 rating while in wet staining some samples gave moderate to good fastness. It has been observed that as the solvent-to-solid ratio increased, colorfastness properties were improved (from 2 to 4). However, at a very high solvent-to-solid ratio wet staining fastness decreases because of the dilution effect and reduced interaction between the dye and the fabric. 95 In dyeing processes, the solvent is used to dissolve or disperse the dye molecules, allowing them to evenly penetrate and adhere to the textile fibers but when there is a high ratio of solvent-to-solid (dye or fabric), several other factors affect wet staining fastness like reduced affinity and limited interaction of dye molecules with fabric. 96
Color fastness to crocking (ISO 105X12:2016) for cotton/silk fabric.

Conditions: Temperature: 20.9°C, Humidity: 64.5%, Force: 9 N, Finger dia:16 mm, Soak %: 95%–100%.
In the case of silk fabric, a lower pH (4) and a lower solvent-to-solid ratio improved the fastness properties and showed high K/S values.
Conclusions
Communities globally are presently opposing the use of azo and benzidine synthetic dyes due to their carcinogenic, non-biodegradable, and hazardous effects on health and the environment. This study addresses the urgent need for sustainable alternatives by using natural dyes from food waste materials, such as beetroot and pomegranate peels. The process, which employs water as a solvent and microwave-assisted extraction (MAE), is eco-friendly and scalable. Optimal dyeing conditions pH 4, 30°C, and a 10 ml/g solvent-to-solid ratio produced golden orange and brown shades on cotton and silk fabrics. The deepest colors were observed on cotton treated with pomegranate peel as a bio-mordant through a pre-mordanting technique. Color analysis, based on K/S values, showed higher dye absorption in acidic conditions and at lower solvent-to-solid ratios, while color-fastness ratings confirmed the effectiveness of these natural dyes. The study, following GOTS and NODS standards, demonstrates that natural dyes from food waste are viable for eco-friendly textile dyeing and hold significant promise for sustainable, large-scale production.
Supplemental Material
sj-docx-1-jef-10.1177_15589250241309010 – Supplemental material for Response surface optimized green approach for silk and cotton dyeing using Beta vulgaris peels extract as a dye and pomegranate rind as bio-mordant: Waste to craft
Supplemental material, sj-docx-1-jef-10.1177_15589250241309010 for Response surface optimized green approach for silk and cotton dyeing using Beta vulgaris peels extract as a dye and pomegranate rind as bio-mordant: Waste to craft by Hafiz Nawaz Hussain, Faiza Imtiaz, Arfaa Sajid, Umer Younas, Qaisar Manzoor, Wissem Mnif, Arif Nazir, Samiah H Al-Mijalli, Munawar Iqbal and Nimra Ehsan in Journal of Engineered Fibers and Fabrics
Footnotes
Acknowledgements
The authors are thankful to the Deanship of Graduate Studies and Scientific Research at University of Bisha for supporting this work through the Fast-Track Research Support Program. The authors express their gratitude to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R158), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.