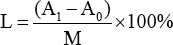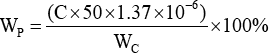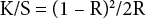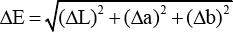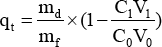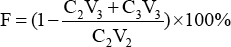Abstract
To investigate the influence of cotton wettability on its dyeing properties in a silicone non-aqueous dyeing system, alkali pretreatment was performed before dyeing. The dyeing properties and dyeing kinetics of reactive dyes in the non-aqueous system and a conventional dyeing system were compared. Alkali pretreatment significantly improved the wettability of cotton fiber by removing wax and pectin. The adsorption rate of dye increased with increased cotton fiber wettability using the non-aqueous system, while it barely changed using the conventional system. Pseudo-second-order kinetics fit well with dye adsorption for both systems. Dye fixation improved by 30% at a concentration of 2% owf, along with better levelness and color depth of the dyed cotton fiber, using the silicone non-aqueous system without salts and dispersants.
Introduction
Of natural fibers, cotton fibers are the most widely used in the textile industry. In 2019, the yield of cotton fiber was 5.89 million tons. 1 Due to the non-cellulosic impurities in cotton fibers that can influence their wettability and dyeing properties,2,3 cotton fibers must be pretreated before dyeing. Reactive dyes are extensively applied in cotton dyeing. 4 However, a large amount of water is consumed and wastewater discharged from conventional water bathed dyeing systems, brings great challenges in environmental protection and effluent treatment. 5 In recent years, researchers have begun to study new dyeing technologies to improve dye fixation, reduce the number of dye auxiliaries, and achieve salt-free dyeing. 6
Non-aqueous dyeing technologies 7 have attracted considerable attention since they offer minimum discharge of dyes and salts. Organic solvent dyeing is one of the representative dyeing technologies that may be used instead of conventional water dyeing.8,9 For example, a fully recyclable reactive dyeing system in a mixture of non-nucleophilic solvents (i.e., dimethyl sulfoxide (DMSO) and dimethylacetamide (DMAc) was designed for the sustainable textile industry. 10 However, dye fixation in an organic solvent dyeing systems is equal to or less than that of the water bathed dyeing system. Furthermore, a complex recycling system is needed to deal with the unreacted dyes after dyeing, making it unsuitable for industry use.
To simplify the dyeing medium recovery system, a new dyeing system was designed that uses organic solvents and a small amount of water during dyeing. Xia et al. studied reactive dyeing for cotton fabric in an ethanol-water dyeing system. 11 In this dyeing system, reactive dye can achieve up to a 95% uptake in a salt-free dyeing environment. However, the volume ratio of ethanol/water was difficult to control, and large amounts of ethanol cannot be stored in a dyeing plant due to the flammability and explosive properties of ethanol. Recently, a water/spent cooking oil dyeing system12,13 requiring minimal discharge was studied. High sorption and fixation of dye could be achieved without use of salts during the dyeing process. However, an alkali solution is needed to refine the oil media every time before recycling. Moreover, the cotton fibers dyed by the reusable cooking oil can cause several diseases resulting from skin contact. 14
In our previous investigations, a silicone non-aqueous medium dyeing system was systematically investigated in the reactive dyeing of cotton fabrics. 15 The chemical structure of the silicone is shown in Fig. 1a. The surface tension of the silicone non-aqueous medium is very low (19.4 dyn/cm), and it is incompatible with water. 16 Silicone has been widely used in consumer and industrial markets. 17 Researches have demonstrated that the silicone non-aqueous medium is safe to both human health and the environment.16,18 Using the silicone non-aqueous dyeing system, only 100%∼200% owf (on the weight of fiber) of water is needed during the dyeing process. 19 Owing to the water-solubility of reactive dye and the affinity of water to fiber is much stronger than that of silicone, almost all the dye solution can be adsorbed onto the cotton fiber in a salt-free dyeing environment.20,21 After dyeing, the silicone non-aqueous medium and water can be naturally separated after 2-3 h due to the low surface tension and density of silicone oil. The wastewater in the lower layer may contain a small amount of silicone medium. After oil-water separation membrane treatment, the produced water does not contain silicone medium. Furthermore, the silicone non-aqueous medium left on the fiber after dyeing can be recovered with a pressure drying machine. Therefore, almost all the silicone non-aqueous medium used can be recovered and there is no silicone effluent discharge, with no potential risks in industrial scale application.

Chemical structure of (a) silicone and (b) C. I. Reactive Red 195.
A high dye concentration can be achieved because only a small amount of aqueous solution is used in the silicone non-aqueous medium dyeing system. Since the dye is absolutely incompatible with the silicone medium, a large difference in the chemical potential of dyes is formed 12 in the silicone medium and water. Therefore, the adsorption rate of reactive dye solution from the silicone dyeing bath to the surface of cotton fiber is pretty fast. This interesting phenomenon might influence the dye leveling property 22 In this investigation, cotton fibers with various wettability properties were prepared using various pretreatments. The relationship between wax and pectin content and cotton fiber wettability was studied. Moreover, the adsorption kinetics of reactive dyes and the level dyeing properties of dyed cotton fibers were further investigated. We believe that this investigation will provide a theoretical reference for industrial use of non-aqueous medium dyeing.
Experimental
Materials
Long-staple cotton fiber (moisture regain < 5.2%, length: 28 mm, Micronaire: B) used for scouring and dyeing was purchased from Xuzhou Yunjin Cotton Co. Ltd. CI. Reactive Red 195 was purchased from Aladdin Reagent. The chemical structure of the dye is shown in Fig. 1b. Sodium hydroxide, sodium sulfate, hydrochloric acid, anhydrous sodium carbonate, and sulfuric acid were purchased from Sinopharm Chemical Reagent Co. Ltd. Galacturonic acid, ammonium oxalate, and carbon tetrachloride was purchased from Macklin Chemical Co. Ltd. Silicone non-aqueous media (purity >98%) was purchased from GE Toshiba Silicone Ltd. Fatty alcohol-polyoxyethylene ether (AEO-5) was obtained from Haining Green Universe Textile Technology Co. Ltd.
Pretreatment of Cotton Fiber
Cotton fiber (1 g) was immersed in an aqueous solution containing 3 g/L sodium hydroxide and 3 g/L AEO-5 at 90 °C for 60 min. The weight ratio of cotton to water was 1:20. After refining, the cotton fiber was rinsed twice with room temperature (RT) water, and then dried at 80 °C for 1 h. The influence of sodium hydroxide concentration, refining agent (AEO-5) concentration, refining temperature, and time on the wetting properties of cotton fibers were further investigated.
Determination of Wax and Pectin Content
The wax content of cotton fiber was determined using an extraction method. 23 Firstly, 2 g of cotton fiber was pre-dried in an oven at 100 °C for 24 h, then the cotton fiber was cut into small pieces and placed in filter paper. Carbon tetrachloride (200 mL) was added into the flask of the Soxhlet extractor to ensure that the carbon tetrachloride solvent was siphoned at least 4 times per hour, and the extraction time was 3-4 h. The wax substance contained in the flask after solvent evaporation was filtered to a specific weight in a beaker. After the beaker was dried to a constant weight, it was cooled to RT in a desiccator and weighed. The formula for the content of wax (L) is given in Eq. 1.
A1 is the mass of the wax and the beaker (g), A0 is the mass of the beaker (g), and M is the mass of the cotton fibers (g).
The pectin content on cotton fiber exists in the form of insoluble calcium, magnesium, and iron salts, which becomes soluble with monovalent cations. 3 Ammonium oxalate is a weak acid salt, and its oxalate ions have strong metal chelation, which can chelate with calcium ions to form water-insoluble pectin. 24 Therefore, 2 g of cotton fiber was extracted with 100 mL 0.5% aqueous ammonium oxalate at 90 °C After 1 h, the galacturonic acid of the sample was measured by the carbazole-sulfuric acid reaction. 25 The amount of pectin was evaluated using Eq. 2.
Wp is the pectin content per gram of fiber, C (mg/mL) is the concentration of galacturonic acid, and Wc is the weight of the cotton fiber (g). The content of pectin is 1.37 times that of galacturonic acid and 50 is the dilution ratio of ammonium oxalate.
Water Absorptivity and Wetting Time
Methods for testing the water absorptivity and wetting time of cotton fiber were based on GBT 2165.1-2008. 26 Cotton fiber (5 g) was equilibrated under standard conditions (20.0 ± 0.5 °C and relative humidity (RH) of 65 ± 1%) for 24 h, and then the fibers were cut into small pellets (0.2 g) and immersed in a beaker (the diameter was 10 cm) with 60 mL of deionized water. When the cotton fiber was fully soaked, the time was recorded.
Dyeing Process
Conventional Water-Based Dyeing System
The concentrations of Na2SO4 and Na2CO3 used were 40 and 15 g/L, respectively. The amount of dye was 2% owf. Dyeing was begun at RT using a liquor ratio (LR) of 1:30. The cotton fiber and reactive dye were both added into the water bath. Half of the Na2SO4 was added into the dyeing bath after dyeing for 10 min at 50 °C, and the remaining salt was added after the following 15 min. The temperature was then raised to 70 °C at 1 °C/min, after which the Na2CO3 was added into the dyeing bath as a fixative agent and kept for 30 min. After dyeing, the dyed sample was soaped and rinsed. Flowchart of the dyeing procedure is shown in Fig. 2.
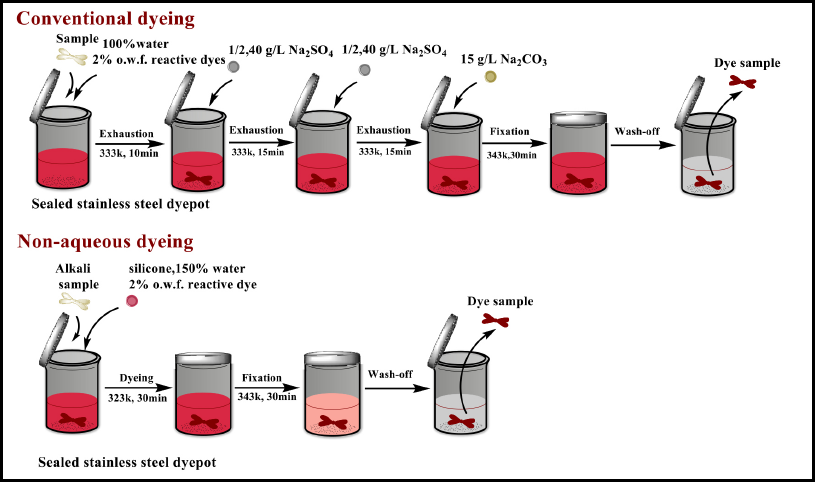
Reactive dyeing for cotton fibers using the conventional water-based dyeing and the silicone non-aqueous medium dyeing systems.
Silicone Non-Aqueous Medium Dyeing System
Firstly, the cotton fibers with various wetting properties were prepared with different pretreatment formulas. These cotton fibers were then dyed with 2% owf of reactive dye and 150% owf of water at an LR of 1:30. The dyeing began at RT for 30 min. Then the temperature was raised to 70 °C at a rate of 2 °C/min and held for 30 min. After fixation, the dyed fibers were washed with a soap solution containing 2.0 g/L of Na2CO3 and 2.0 g/L of soap fakes (Zhejiang Green Universe Textile Technology Co. Ltd.) at 95 °C for 15 min and washed thoroughly with hot water (60 °C) for 10 min and RT (25 °C) water for 10 min. Flowchart of the dyeing procedure is shown in Fig. 2.
Evaluation of Dyeing Quality
Color Measurement of Dyed Cotton Fibers
A Datacolor SF600X spectrophotometer (D65 illuminant, specular included, 10° observer angle) was used to assess the color strength (K/S value), which was calculated by the Kubelka-Munk equation (Eq. 3), the K/S values of ten different positions on each dyed sample were measured.
According to Eq. 4, the color difference (ΔE) was used to estimate the level of dyeing of the dyed cotton fiber. The CIE L*, a*, and b* values of ten different points on each dyed sample were measured to calculate the ΔL, Δa, and Δb values.
Uptake of Reactive Dye
The amount of dye absorbed on the cotton fiber (qt, mg/g) was calculated using Eq. 5.
md (mg) is the total dye mass, mf (g) is the cotton fiber mass, C0 and C1 are the concentration (g/L) of dye before and after dyeing, and V0 and V1 are the volume (mL) of the initial dye solution and the total volume (mL) of the extraction solution, respectively.
Fixation of Reactive Dye
Dye fixation was evaluated using Eq. 6.
F is the dye fixation rate, C2 and C3 (g/L) are the concentrations of initial dye and dye in the washing solution, respectively, and V2 and V3 are the volumes (mL) of initial dye bath and the raffinate, respectively.
Dyeing Kinetics
The dyeing kinetics of reactive dye on cotton fibers in both silicone non-aqueous medium dyeing systems and conventional water-based dyeing systems were studied. The adsorption of Reactive Red 195 on cotton fiber was performed at 30 °C for 1∼80 min. The pseudo-first-order 27 and pseudo-second-order 28 models were used to stimulate the dye adsorption behavior.
Results and Discussion
Influence of Pretreatment on Cotton Fiber Wetting Properties
The dyeing process in the silicone non-aqueous medium system is different from that of the conventional water bath dyeing system. Only a little water is used during dyeing, so the wettability of the cotton fiber will influence dye adsorption and penetration. 29 Therefore, the pretreatment method that is suitable for the silicone non-aqueous media dyeing system should be investigated.
As shown in Fig. 3a, when no alkali was used during pretreatment, it was difficult to remove the non-cellulosic impurities, leading to the poor wettability of cotton fibers. The wetting time of cotton fiber decreased when alkali was added to the pretreatment formula. Moreover, the wetting time of cotton fiber was about 1.0 s when the concentration of NaOH was 3 g/L. However, the wettability of cotton fiber did not significantly improve if the concentration of alkali was increased to 4 g/L. In Fig. 3b, the wetting time of cotton fiber decreased from 15.2 to 1.0 s when the concentration of AEO-5 was increased from 0 to 3 g/L. However, when the concentration of AEO-5 increased from 3 to 4 g/L, the cotton fiber wettability barely changed. From Fig. 3c, the cotton fiber wettability improved enormously with increased temperature, obtaining an excellent wetting time at 90 °C. As shown in Fig. 3d, the wetting time decreased from 6.1 to 1.0 s when the pretreatment time was extended from 30 to 60 min, and the wetting time reached the minimum when the pretreatment time was 60 min. As a result, the pretreatment temperature and time should be 90 °C and 60 min, respectively.
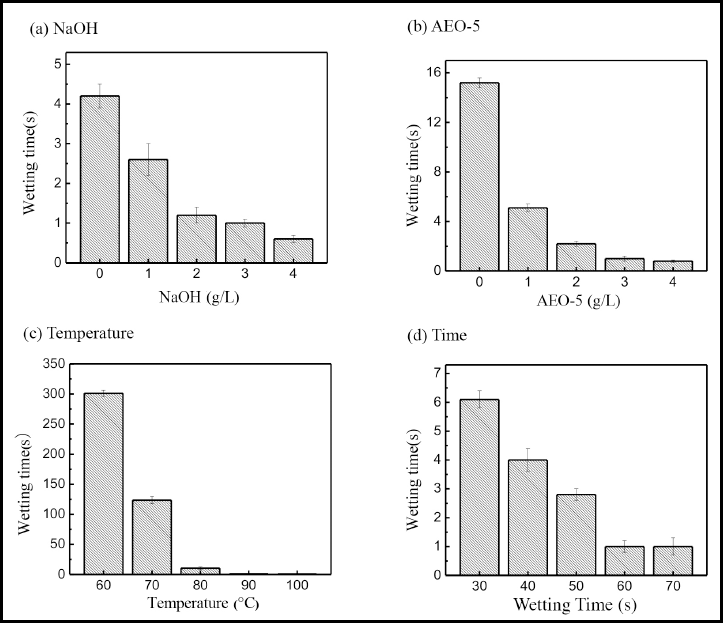
Effects of (a) NaOH concentration, (b) AEO-5 concentration, (c) refining temperature, and (d) time on cotton fiber wettability.
Effect of Pretreatment on Wax and Pectin Content
As shown in Fig. 4a, compared with the use of no alkali, the removal rate of wax and pectin increased from 42.3 to 63.4%, and 84.7 to 95.4%, respectively, when the concentration of NaOH was 4 g/L. The wax was saponified and emulsified with alkali agent at high temperature. 3 Pectin could react with alkali agent to form sodium pectinate, which was easily washed of from the cotton fiber.3,30 From Fig. 4b, the wax removal rate showed a linear increase with the increase in AEO-5 concentration. On one hand, AEO-5 (a non-ionic surfactant) had good penetration, emulsification, and dispersion effects. 31 Therefore, AEO-5 could effectively reduce the surface tension of the aqueous solution, allowing the alkali solution to penetrate into the fiber interior, and the non-cellulosic impurities could then be emulsified in aqueous solution. On the other hand, the non-cellulosic impurities were dispersed in the solution under mechanical force. 32 From the results of Fig. 4c and d, the pretreatment time and temperature also had a great effect on the wax and pectin removal rate. When the pretreatment temperature was at 60∼70 °C, the wax and pectin removal rate was poor because wax and pectin could not be emulsified at a lower temperature. 33 From the pretreatment time, it took 60 min to remove the wax and pectin on the cotton fiber.
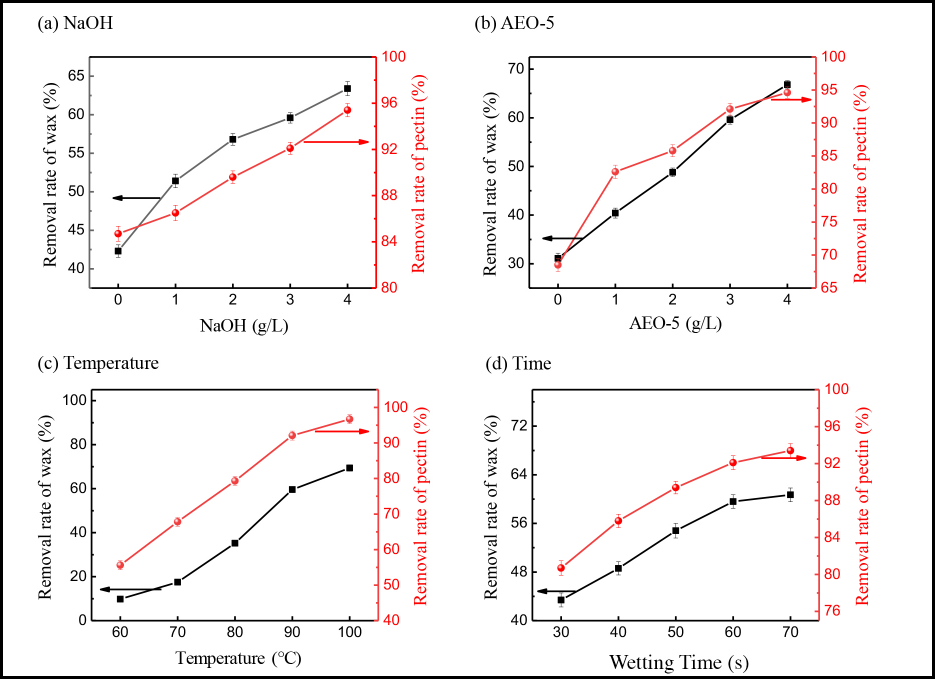
Effects of (a) NaOH concentration, (b) AEO-5 concentration, (c) exhaust temperature, and (d) time on wax and pectin content.
Relationship between Fiber Wettability and Wax/Pectin Content
The previously described pretreatment can effectively remove the wax and pectin on the cotton fiber and improve the fiber wetting properties. Fig. 5 shows the relationship between fiber wettability and wax and pectin removal rates. The wettability of cotton fiber greatly improved with the increased wax removal rate. The wetting time of cotton fiber decreased from 15.2 to 0.6 s when the wax removal rate increased from 31.1 to 63.4%. However, as the wax removal rate continued to increase, the wetting time of cotton fiber only changed slightly.
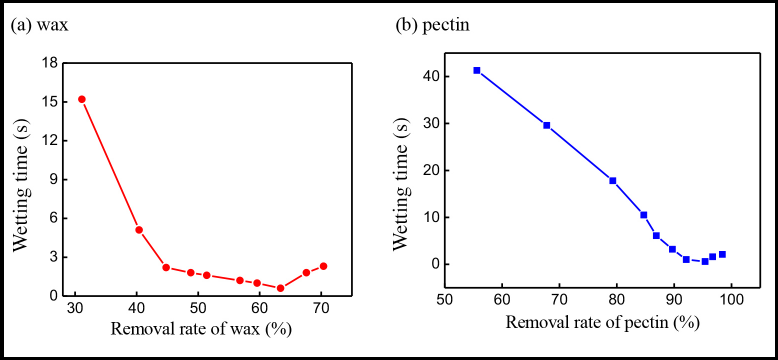
Effects of (a) wax and (b) pectin removal rates on cotton fiber wetting time.
As shown in Fig. 5b, compared with wax removal, the removal effect of pectin was more obvious. When the removal rate of pectin was 55.6%, the wetting time of cotton fiber was 41.3 s. With the increase in pectin removal rate, the wetting time of cotton fiber gradually decreased. The wetting time of cotton fiber was 0.6 s when the pectin removal rate was 95.4%, indicating that the resulting cotton fiber had good water adsorption properties. The wetting time of cotton fiber only slightly changed when the pectin removal rate was further increased. As a result, both the content of wax and pectin on cotton fiber directly influenced the fiber wettability. When the wax and pectin content was lower, the cotton fiber wettability was better. As for wax, the wettability of cotton fiber increased with decreased pectin content. The pectin is located between wax and fiber, which acts as a “glue” that binds the wax to the fiber. 34 After removing the pectin, the wax can be released. This explains why the pectin was removed more than the wax.
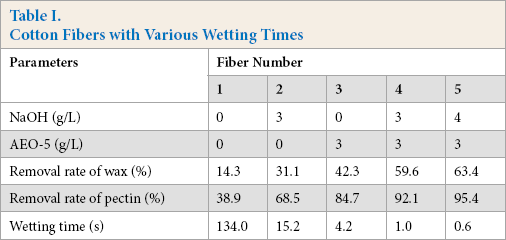
According to the previous results, cotton fibers with various wettabilities were chosen for dyeing in the silicone non-aqueous medium dyeing system, and a conventional water bath dyeing system was used as a comparison (Table I). The relationship between the wettability of cotton fibers and its dyeing properties were thereby further investigated.
Cotton Fibers with Various Wetting Times
Comparative Dyeing Adsorption of Cotton Fibers with Various Wettabilities
As shown in Fig. 6a, the adsorption rate of Reactive Red 195 increased gradually with the improvement of cotton fiber wetting properties using the silicone non-aqueous medium dyeing system. When the cotton fiber wetting time was 134 s, the dye adsorption rate was slow, and the equilibrium dye adsorption quantity was only 11.93 mg/g. As the wetting time of cotton fiber decreased to 15.2 s, the dye adsorption rate increased dramatically, and the equilibrium dye adsorption quantity increased to 14.51 mg/g, which was 1.22 times that of the relatively hydrophobic cotton fiber. When the cotton fiber wetting time was reduced to less than 1s, the equilibrium adsorption quantity of the reactive dye increased to about 20 mg/g, which indicated that the dye uptake was nearly 100%. This interesting phenomenon was mainly because the better the cotton fiber wettability, the stronger the ability to absorb water. 35 Therefore, the dye solution dispersed in the dyeing bath could be adsorbed by the cotton fiber.
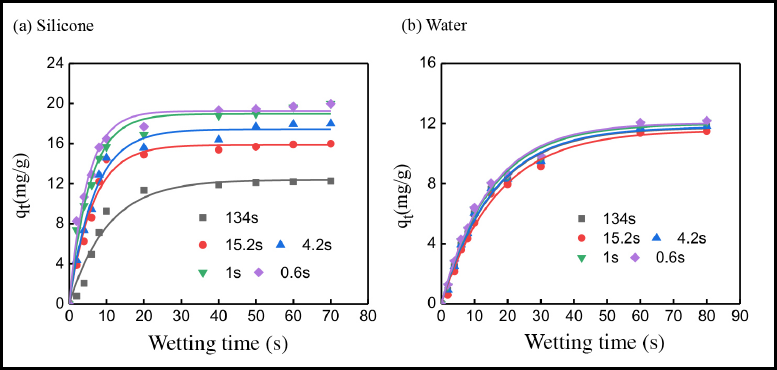
Dyeing adsorption of cotton fibers with various wettability in (a) silicone non-aqueous and (b) conventional water-based dyeing systems.
Compared with the silicone non-aqueous medium dyeing system, the equilibrium adsorption quantity of Reactive Red 195 was around 12 mg/g (Fig. 6b) in the conventional water bath dyeing system. In the water-based dyeing system, the dye adsorption equilibrium took about 80 min, which was obviously longer than that of a non-aqueous dyeing system. The dye uptake rate curves of cotton fibers with various wet-tabilities almost overlapped using the conventional system, indicating that the cotton fiber wettability had no effect on the adsorption of reactive dye in the water bath. Reactive dyes are water-soluble dyes, 4 and most reactive dyes remain dissolved in the water phase, so it is difficult for dyes to adsorb on cotton fiber in aqueous solution.
Influence of Cotton Fiber Wettability on Dye Adsorption Kinetics
Pseudo-First-Order Kinetics
Pseudo-first-order kinetics27,36 was used in this study to simulate the adsorption behavior of dyes was designed by Lagergren and was calculated using Eq. 7.
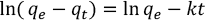
qe (mg/g) is the mass of the dye adsorbed on the cotton fiber at equilibrium, qt (mg/g) is the mass of the dye adsorbed on the cotton fiber at time t (min), and k (g/mg·min) is the adsorption rate constant.
The R2 correlation coefficient represents the matching degree of pseudo-first-order kinetics for the simulation of dye adsorption behavior. The higher the correlation coefficient, the better the fit. As shown in Fig. 7 and Table II, the line plots did not ft the pseudo-first-order kinetics model well. The R2 correlation coefficients for the various wetting cotton fibers using non-aqueous an d water bath dyeing systems were less than 0.98. Therefore, pseudo-first-order kinetics was not suitable for evaluating the adsorption behavior of reactive dyes on cotton fibers with various wettability.
R2 Values of Pseudo-First-Order Reactive Dyeing Kinetics for Cotton Fibers with Various Wetting Times in Silicone and Water-Based Dyeing Systems
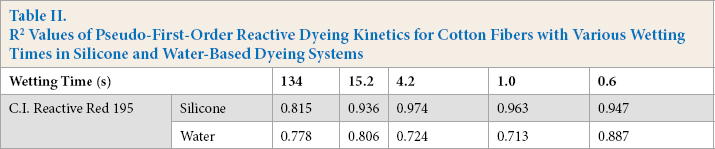
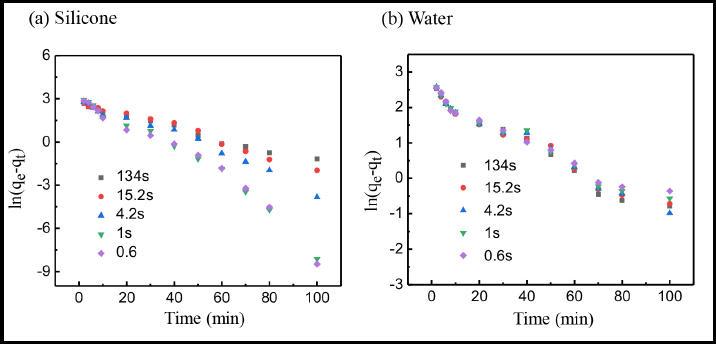
LineMar fitting of pseudo-first-order dyeing kinetics in (a) sillicone non-aqueous and (b) conventional water-based dyeing systems.
Pseudo-Second-Order Kinetics
Next, the use of pseudo-second-order kinetics was investigated.
The pseudo-second-order kinetics model, which had been system atically studied for the liquid-phase adsorption of reactive dyes,28,37 was calculated using Eq. 8.
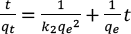
qe (mg/g) is the mass of dye adsorbed on the cotton at equilibrium, qt (mg/g) is the mass of dye at time t (min), and k2 (g/mg·min) is the adsorption rate constant.
k2 and qe can be calculated by the slope and intercepts using Eqs. 9 and 10, respectively. The half-dyeing time t0.5, an index to express the dyeing rate, can be calculated using Eq. 11.
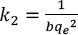
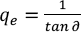
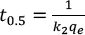
Where tab ∂ and b denote the slope and intercept of the pseudo-second-order kinetic model, respectively.
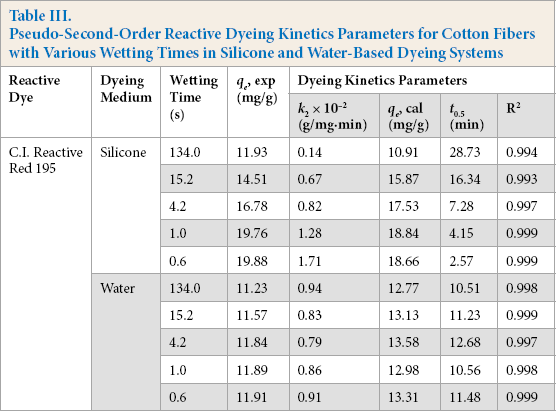
In Fig. 8 and Table III, the line plots ft the pseudo-second-order kinetics model well, and the R2 correlation coefficients for these two dyeing systems were both greater than 0.993. Furthermore, the calculated equilibrium adsorption quantities of reactive dye qe (cal) were similar to the experimental values for both silicone non-aqueous and water bath dyeing systems, indicating that pseudo-second-order kinetics could be used to describe the adsorption behavior of the reactive dye on cotton fiber.
Pseudo-Second-Order Reactive Dyeing Kinetics Parameters for Cotton Fibers with Various Wetting Times in Silicone and Water-Based Dyeing Systems
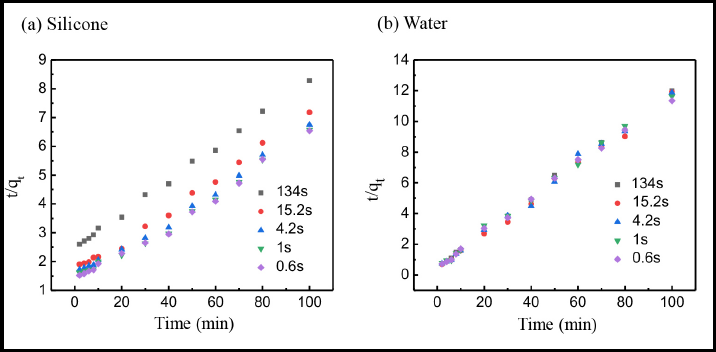
Linear fitting of the pseudo-second-order dyeing kinetics in (a) silicone non-aqueous and (b) conventional water-based dyeing systems.
As shown in Table III, for the silicone non-aqueous dyeing system, the k2 value increased from 0.14 × 10−2 to 1.71 × 10−2 (g/mg·min), when the wetting time of fiber decreased from 134.0 to 0.6 s, and the t0.5 value decreased from 28.73 to 2.57 min. These results indicated that the adsorption rate of the reactive dye increased with increased cotton fiber wettability. For the silicone non-aqueous medium dyeing system, the concentration of dye between dyeing bath and cotton fiber was quite different,15,38 and the reactive dye was absolutely incompatible with the silicone media, 39 resulting in the dye quickly transferring from the dyeing media to the cotton fiber. It is easier for the reactive dye to permeate into the fiber when the wettability of cotton fiber was good. 40 Compared with the silicone non-aqueous dyeing system, the k2 value was between 0.79∼0.94 × 10−2 (g/mg·min) and the t0.5 value barely changed when the wetting time of cotton fiber decreased from 134 s to 0.6 s in the conventional water bath dyeing system, indicating that the wettability of cotton fiber had little influence on the adsorption rate of the reactive dye when the conventional dyeing system was used.
Effect of Cotton Fiber Wettability on Level Dyeing
As shown in Fig. 9, the influence of cotton fiber wettability on the level dyeing of reactive dye cotton fiber dyeing using the silicone non-aqueous dyeing system was investigated. The K/S value of the dyed cotton fiber increased with increased cotton fiber wettability, implying that more dye could be adsorbed and react with cotton fiber that had greater wettability. For level dyeing, the color difference (ΔE value) decreased with improved cotton fiber wettability. Relatively good leveling of dyed cotton fiber was obtained when the wetting time of cotton fiber was less than 20 s. A minimal ΔE value was achieved when the wetting time of cotton fiber was 1s. The maximal ΔE value was greater than 1 at a fiber wetting time of 134s, resulting in very poor leveling. According to Liu et al., 13 the reactive dye molecule could not be absorbed when the wettability of cotton was poor, and dye aggregates on the cotton fiber surface prevent the dye from diffusing from the dyeing media into the cotton fiber interior.
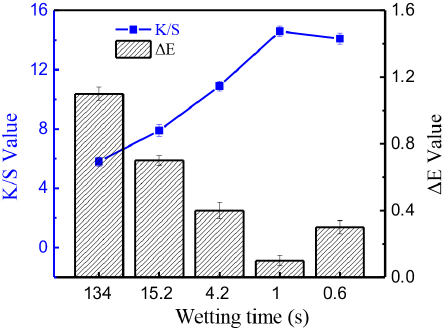
K/S values and levelness (ΔE) determinations of cotton fibers with various wettabilities.
As shown in Fig. 10, the fixation of Reactive Red 195 in the silicone non-aqueous medium dyeing system was as high as 90% without adding any salts when the cotton fiber had a good wettability. However, dye fixation was only 60% with the help of a large number of salts in the conventional water bath dyeing system. Therefore, reactive dye fixation of the reactive dye in the non-aqueous dyeing system was significantly greater than in the conventional dyeing system. The high concentration of the reactive dye solution has a high affinity for the cotton fibers,20,41 resulting in a greater final dye uptake (the dye uptake in the conventional water bath system was about 70%). On the other hand, the water content (150% owf) in the non-aqueous dyeing system was much less than that of the conventional dyeing system. As a result, the hydrolysis rate of reactive dyes in non-aqueous medium dyeing was less than that of conventional dyeing.
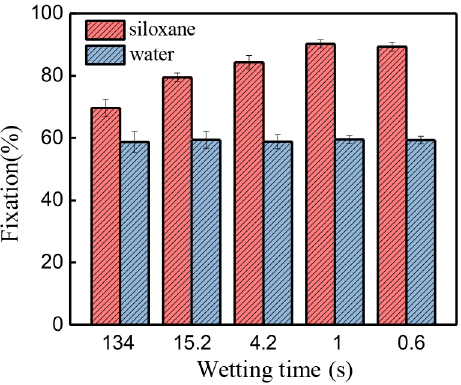
Dyeing fixation of cotton fibers with various wettability in silicone non-aqueous and conventional water dyeing systems.
Cost of Cotton Reactive Dyeing using the Silicone Non-Aqueous Dyeing System
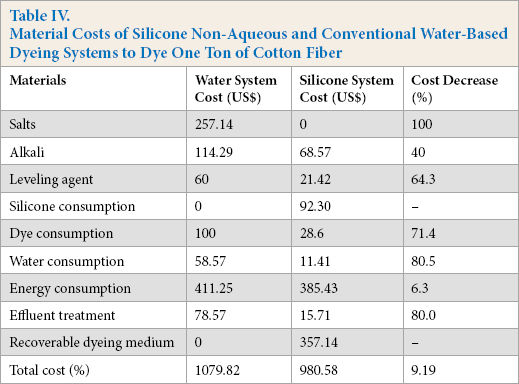
Compared to the conventional water-based dyeing system, less dye and chemicals were consumed using the silicone non-aqueous dyeing system. Material cost savings based on the current research is shown in Table IV. Owing to the ultra-high utilization of dye (above 90%) in the silicone non-aqueous dyeing system, the cost of dye consumption can be decreased by 71.4%, which would also lead to the cost reduction of effluent treatment (80%). In addition, zero salts and a small amount of alkali are needed to improve the dye fixation in the non-aqueous dyeing system. Almost all dye would transfer from the non-aqueous medium to the cotton fiber due to the incompatibility between the non-aqueous medium and the dye. In the non-aqueous dyeing system, only a little water is needed to swell the cotton fiber and dissolve the dye, which would lead to a water consumption reduction of 80%. Although the consumption and recycling cost of silicone increased the cost of silicone non-aqueous medium dyeing, the total cost was less than that of the conventional water-based dyeing system (9.19%).
Material Costs of Silicone Non-Aqueous and Conventional Water-Based Dyeing Systems to Dye One Ton of Cotton Fiber
Conclusion
This study investigated the influence of different pretreatment formulas on the wettability of cotton fibers and quantitatively analyzed the relationship between wettability, and wax and pectin content, on the cotton fiber and its dyeing properties using Reactive Red 195 dye. The wettability of cotton fiber improved with the increased wax and pectin removal rate. Using the silicone non-aqueous dyeing system, the dye adsorption rate increased with the increase in cotton fiber wettability, while cotton wettability had practically no effect when the conventional water bath dyeing system was used. Pseudo-second-order kinetics was suitable for evaluating the adsorption kinetics of the reactive dye in both silicone non-aqueous and conventional water bath dyeing systems. There was a positive correlation between levelling and wetting properties using the non-aqueous dyeing system. Moreover, dye fixation was up to 90% without addition of salts for the silicone non-aqueous medium dyeing system, which was 30% greater than that for the water bath dyeing system. As a result, better level dyeing and color depth of the dyed cotton fiber was obtained. This study has great significance on improving the fixation and level dyeing using reactive dyes on cotton fibers with the silicone non-aqueous medium dyeing system.