Abstract
Sodium hydrosulfite is universally applied as a reducing agent for the reduction of indigo, but also produces hazardous by-products viz. sulfate, sulfite, and thiosulfate ions with harmful effects on the environment due to their toxicity, as well as corrosive effects on the waste lines. To overcome these problems, use of an ecofriendly reducing agent, viz. alkaline catalase, along with iron (II) sulfate, was used in place of sodium hydrosulfite for dyeing of cotton with indigo. Dyed samples were characterized by attenuated total reflectance-Fourier transform infrared (ATR-FTIR) spectroscopy and scanning electron microscopy (SEM). The alkaline catalase with iron (II) sulfate reducing system produced the same reduction potential in dyebaths, reduction bath stability, surface color strength, and color fastness properties of dyed cotton compared to sodium hydrosulfite use.
Introduction
Indigo (C.I. Vat Blue 1) is one of the oldest dyes used to produce the attractive blue shade along with desired wash down property on denim. 1 It is generally applied on denim warp through the “6-dip-6-nip” technique in sheet,2,3 rope, 4 or loop 5 form using sodium hydrosulfite (Na2S2O4) as reducing agent and sodium hydroxide as alkali. 6 Sodium hydrosulfite is thermally, hydrolytically, and oxidatively unstable, prone to be oxidized by atmospheric oxygen, and the stability of its alkaline solutions reduces with the increase in temperature. Some of the by-products formed due to decomposition of sodium hydrosulfite are sulfur compounds, such as Na2S and NaSH, as well as others, which pollute the atmosphere through formation of hydrogen sulfide (H2S). At the same time, salts of sulfur in the form of sulfites and sulfates (Na2SO3, NaHSO4, Na2SO4, and Na2S2O3) contaminate sewage, lower its pH, and corrode the concrete pipes transporting the sewage.7,8 Other problems associated with the use of sodium hydrosulfite include cost, storage difficulty, and the necessary heating of stock indigo bath to complete reduction of dye.
Many reduction systems were developed for the reduction of indigo since 7000 B.C. The first of its kind was the fermentation technique, in which vegetable substances were used to liberate hydrogen for reduction of indigo. 9 Use of iron (II) salt, along with Ca(OH)2 was known since ancient times as the wooden vat or copperas method,9,10 which produces a large amount of sludge. Iron (II) salts form iron (II) hydroxide in the presence of NaOH, which acts as a reducing agent for indigo, but the degree of reduction is not optimal due to the poor solubilization of iron (II) hydroxides, ultimately resulting in poor dye strength on cotton. 11 Other earlier studied reducing systems include zinc-lime vat, 9 bisulfite zinc-lime vat,9,12 thiourea dioxide,13,14 sodium borohydride, 15 glucose with NaOH, 16 hydroxyacetone,17,18 and electrochemical reduction,19-21 all with inherent limitations. 8
Subsequently, to enhance solubilization of iron (II) hydroxides, it was complexed with ligands, such as gluconic acid, 3 tartaric acid, or citric acid and triethanolamine. 8 Iron (II) salts using gluconic acid and NaOH at 60 °C, as well as in combination with tartaric or citric acid, triethanolamine, and NaOH at room temperature (RT) showed results comparable to sodium hydrosulfite with few exceptions.7,8,22,23 A classical review of various reducing systems used in reduction of indigo has been detailed elsewhere. 24 Furthermore, to achieve effective biopolishing, the location of indigo must be restricted at the surface of cotton through ring dyeing at a pH around 11 in preferably their mono-phenolate form, obtained by reducing only one C=O group. 25
Enzymes belonging to certain classes have been found to work as reducing agents in alkaline media and may catalyze the reduction of dyes. One of these enzymes, catalase, has been found to be a potential reducing agent for reduction of sulfur dyes.26,27 Catalase belongs to the oxidoreductases class of enzyme and is used in various applications (e.g., textile bleaching and sterilization of food products and electronics). Catalase is used in industry to neutralize hydrogen peroxide (H2O2), which is a strong biocide, oxidant, and bleaching agent in the textile, pulp and paper, wood, and food indus-tries.28,29 Catalase is used to convert the residual hydrogen peroxide into oxygen and water. Immobilized thermo-alkali stable bacterial catalase-peroxidases are recommended for the treatment and recycling of textile bleaching effluent to be reused for other applications.30-32
In this work, the application of alkaline catalase with iron (II) sulfate as reducing agent for dyeing of cotton with indigo was studied. Desized, scoured, and bleached woven cotton fabric was dyed with indigo using sodium hydrosulfite and alkaline catalase with iron (II) salt separately. Box Behnken response surface design was used to analyze the alkaline catalase reduction system to achieve optimized parameters. Performance of both reduction systems were compared in terms of pH, reduction potential (mV) at various stages of dyeing, surface color strength (K/S) of dyed cotton, stability of the reduction bath, fastness properties, and tensile strength of dyed cotton. Characterization and surface morphology of dyed cotton was done by attenuated total reflectance-Fourier transform infrared (ATR-FTIR) spec-troscopy and scanning electron microscopy (SEM).
Materials and Methods
Materials
Thoroughly desized, scoured, and bleached plain woven cotton fabric (Vardhman Textiles Ltd., Baddi, India, EPI 126, PPI 84, warp 40s, weft 33s, and GSM 135) was used in this study.
Chemicals
Alkaline catalase (EC 1.11.1.6) and indigo (C.I. Vat Blue 1) were supplied by Clariant and Dystar, Mumbai, India respectively. Iron (II) sulfate (AR grade, 99.5%), sodium hydroxide (AR grade, 96%), sodium hydrosulfite (87–88%), and hydrochloric acid (37%) were procured from SD Fine Chemicals.
Preparation of Pad Liquor
Sodium Hydrosulfite and NaOH System
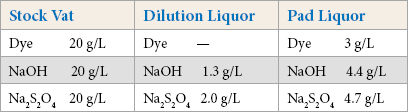
A stock vat and dilution liquor were used to prepare the required concentration of dye solution. For preparing the stock vat, the required amount of sodium hydroxide (NaOH) and dye was added in 100 mL water and heated up to 50 °C. The required amount of sodium hydrosulfite (Na2S2O4) was added and the solution was maintained for 15–20 min for complete reduction of indigo.
For preparing the dilution liquor, the required amount of NaOH and Na2S2O4 was added in 1 L of water at RT and stirred well until a clear solution was obtained.
A pad liquor of 3 g/L indigo was prepared from these two solutions. The dilution liquor (567 mL) was added to 100 mL of reduced stock vat to make the padding liquor solution. The concentrations of dye, NaOH, and Na2S2O4 were duplicated (as per BASF guidelines) to prepare a control solution.
Iron (II) Sulfate—NaOH System
Pad liquor was prepared using FeSO4 (2% owf) and NaOH (4%), followed by addition of indigo (3 g/L). Reduction was carried out at 80 °C for 15–20 min, followed by dyeing via padding.
Alkaline Catalase and Iron (II) Sulfate System
Pad liquor was prepared in the same way as the iron (II) sulfate—NaOH system, replacing the hydrosulfite with alkaline catalase.
Dyeing of Cotton with Indigo
Cotton was dyed with reduced indigo from all reduction systems using a “6-dip-6-nip” padding operation. This includes dipping cotton in dye liquor for 30 s, followed by padding at 1 kg/cm2 pressure for a 75-80% dye pickup, and airing for 1 min to complete a “1 dip 1 nip” cycle. Cotton fabric was dyed with six such consecutive cycles, with the final airing for 3 min converting the reduced dye on the fabric to its oxidized form. The dyed samples were then thoroughly washed in hot water.
Statistical Analysis of Dyed Cotton
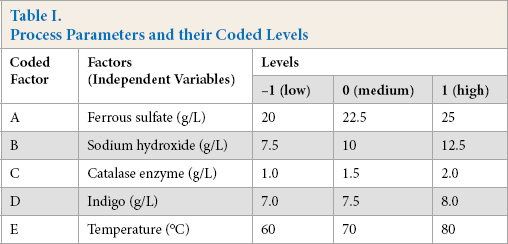
A Box-Behnken response surface design was used to analyze and optimize the process parameters. This included identifying the best suitable combinations of parameters and their levels, resulting in a K/S value equivalent to that obtained from the hydrosulfite system. Five dyeing parameters were studied viz. the concentration of FeSO4, NaOH, alkaline catalase, indigo, and temperature as factors, and their coded values as levels (-1, 0, +1). Using these five parameters (factors), each with three levels, a 35 Box-Behnken design was run to get a set of data (run), consisting of a total of 46 runs with six replicates at the central point. Results were analyzed with response surface plots and equations were formed for response at a 95% confidence interval. Response surface fig-ures were analyzed for understanding the effect of individual parameter (factor) on the surface color strength (K/S).
Evaluation of Dyebath and Dyed Cotton
K/S values of dyed cotton were evaluated using computer color match (Datacolor Check, Datacolor International), and color fastness properties viz. light, wash, and rubbing were evaluated using AATCC TM16-2004, TM61-2007, and TM8-2007, respectively, while reduced dyebaths were evaluated for pH and reduction potential using a digital pH with ORP meter (Century Instruments) at various stages of dyeing (i.e., before and after reduction of dye as well as at the end of dyeing).
Estimation of Dye Uptake
To study the quantity of indigo uptake on cotton after each dip and nip, a known weight of dyed cotton was dissolved in dimethyl sulfoxide (DMSO) to extract indigo from dyed cotton. The extract was analyzed using a UV-Vis spectrophotometer (Perkin Elmer) to evaluate absorbance and subsequently to grams of indigo vs. 100 g of cotton.
Stability of Reduced Dyebaths
To study the stability of baths in presence and absence of indigo, blank reduction baths were prepared. One set of these blank baths (containing only reducing liquor and no indigo) of both the sodium hydrosulfite and alkaline catalase reduction systems were stored, evaluated for reduction potential and pH after specific time of storage (0–24 h), followed by addition and reduction of indigo in these baths, and dyeing. This was for studying the stability of baths in the absence of dye. In another set of reduction baths, indigo was added during preparation of bath, reduction was carried out and stored for specific time (0–24 h), after which the bath potential and pH were evaluated, and dyeing was performed. This was for studying the storage stability of baths in the presence of indigo.
Attenuated Total Refection–Fourier Transform Infrared (ATR-FTIR) Spectroscopy
Attenuated total reflection–Fourier transform infrared (ATR-FTIR) spectra of white cotton fabric as well as dyed cotton were obtained on an Alpha eco-ATR spectrometer (Brucker) equipped with a zinc selenide (ZnSe) crystal using 16 scans/sample in the range of 4000 to 500 cm–1, at a resolution of 16 cm–1.
Scanning Electron Microscopy (SEM)
Surface morphology of dyed cotton was characterized using scanning electron microscopy (SEM, Zeiss EVO 50) at a voltage of 10 kV and 5k magnification.
Tensile Strength of Cotton Fabric
A Universal testing machine (Aimil Ltd.) was used for measuring the tensile strength of cotton using the ASTM D5035 test method after dyeing with both reducing systems.
Results and Discussion
Dyeing of Cotton with Sodium Hydrosulfite
Cotton was dyed with indigo using the “6-dip-6-nip” padding technique, followed by oxidation and washing. The K/S value of dyed cotton was 22.40 at λmax = 590 nm. The range of pH and mV were 12.2 to 12.8, and –640 to –780, respectively at various stages of dyeing.
Dyeing of Cotton with Alkaline Iron (II) Salts
The reduction potential of indigo baths prepared with indigo, iron (II) salt, and NaOH was –810 mV at the start of dyeing and –731 mV at the end, resulting in the complete reduction of indigo. The pH was in the range of 12.7 to 12.4 throughout. In spite of this, the dye strength (K/S) on cotton was only 11.5, which was almost half that obtained using the hydrosulfite system.
Dyeing of Cotton with Alkaline Catalase
Attempts were made to dye cotton with indigo the same way using alkaline catalase in place of sodium hydrosulfite. The reduction potential of the bath was only in the range of –450 to –580 m V. In contrast, indigo requires a reduction potential around –750 mV and greater for reduction. It is known that enzyme activity is enhanced with the addition of metals in the dyebath.33,34 Because of this, indigo baths were formulated with alkaline catalase and iron (II) sulfate as the reducing agent. Cotton was dyed in this bath following the same procedure that was used in the hydrosulfite system. It was observed that the reduction potential of the bath increased to around –750 mV with the reduction of indigo. Consequently, the cotton was dyed at lower K/S value than expected. To improve the K/S values, the dyeing parameters were varied, and reduction baths were prepared with dyeing of cotton from each bath in an attempt to reproduce the dye strength on cotton obtained using the hydrosulfite system. After thorough studies on the influence of different parameters, the optimal ranges were: concentration of FeSO4 (20–25 g/L), NaOH (7.5–12/5 g/L), alkaline catalase (1–2 g/L), indigo (7–8 g/L) and temperature between 60 to 80 °C. The actual values of levels are given in Table I.
Process Parameters and their Coded Levels
Putting these levels into the Box Behnken design resulted in 46 separate runs. The indigo baths were prepared based on these sets of parameters and levels, cotton was dyed, and K/S values were evaluated. The results are shown in Table II.
Responses of Box-Behnken Analysis
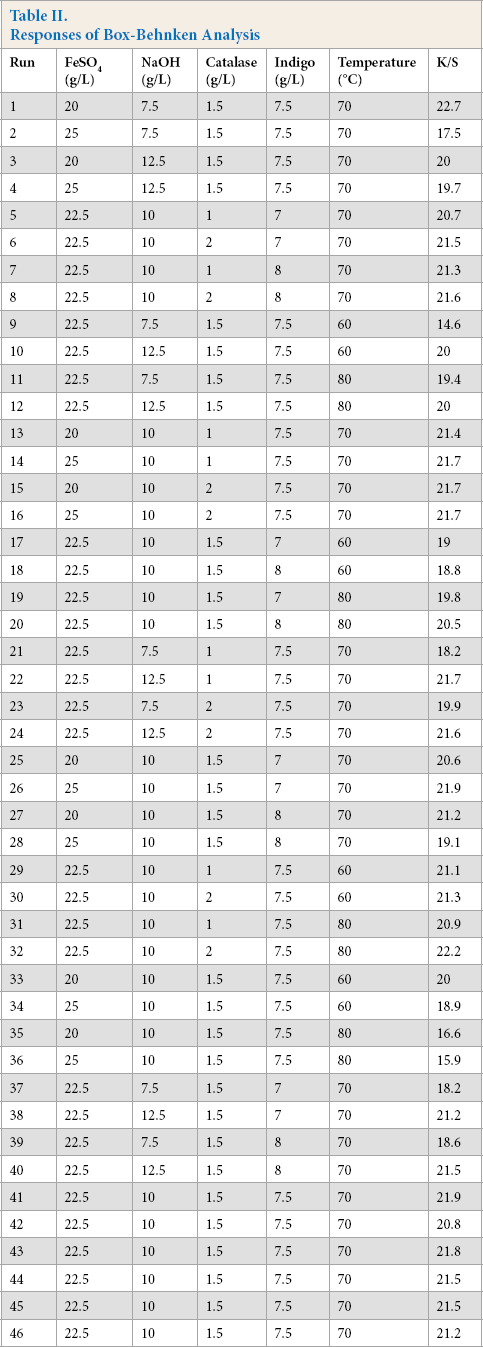
The range of K/S values was 14.63 to 22.7, and the maximum K/S value was obtained if the bath was prepared as per Run 1 in Table II. It is to be noted that a 3 g/L indigo bath resulted in a K/S value of 22.4 using the hydrosulfite system. Furthermore, the color difference (ΔE) value between samples dyed in hydrosulfite and alkaline catalase systems was nominal and within limits (1.6).
Influence of Dyeing Parameters on K/S Values
The influence of process parameters viz. concentration of FeSO4, NaOH, catalase, indigo, and temperature on K/S values were investigated using the Box Behnken design and response surface methodology. The influence of parameters on surface color strength (K/S), corresponding to two factors interaction and cubic effect with R 2 = 0.89, was observed.
The model was significant at a 95% confidence level as the value of Prob>F was less than 0.05. In this case, NaOH was the significant model factor. The model equation in coded form is shown in Eq. 1.

This equation predicts the theoretical K/S of dyed samples for given dyeing parameters.
Influence of FeSO4 and NaOH Concentrations
The combined effect of FeSO4 and NaOH concentrations on K/S at a specific alkaline catalase concentration, dye concentration, and temperature (medium level) is shown in Fig. 1a. At a lower level of NaOH (7.5 g/L) and FeSO4 (20 g/L) concentrations, the maximum K/S value of 22.7 was obtained. On increasing the concentration of FeSO4, the K/S value of cotton decreased. Increased concentration of FeSO4 might have decreased the concentration of NaOH in the dyebath due to the formation of insoluble Fe(OH)2, affecting the solubility of reduced indigo.
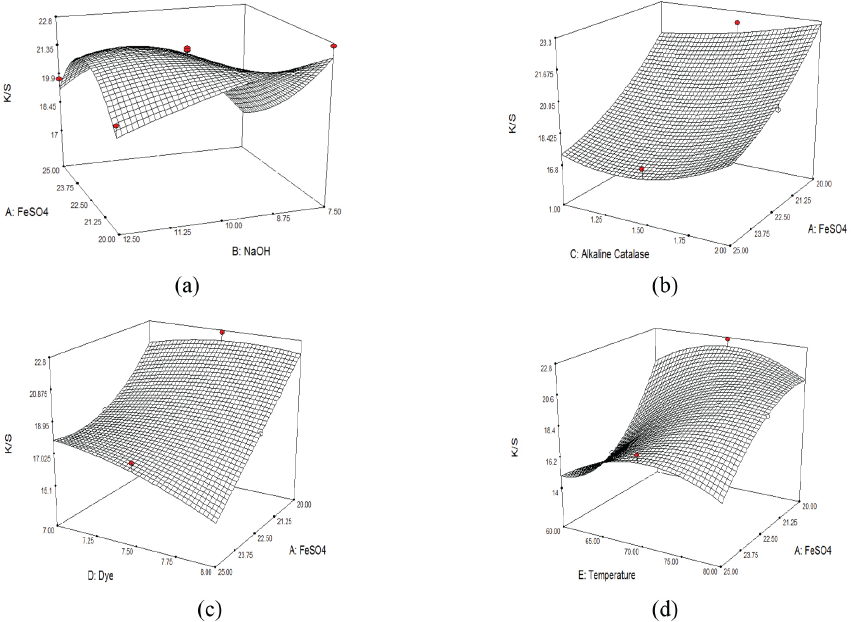
(a) influence of FeSO4 and NaOH concentrations, (b) influence of FeSO4 and catalase concentrations, (c) influence of FeSO4 and indigo concentrations, and (d) Influence of FeSO4 concentration and temperature on K/S values.
Influence of FeSO4 and Catalase Concentrations
The combined effect of FeSO4 and alkaline catalase concentrations on K/S values at a specific dye concentration, temperature (medium level), and NaOH concentration (lower level) is shown in Fig. 1b. A lower level of FeSO4 concentration (20 g/L) and a medium level of catalase concentration (1.5 g/L) resulted in a maximum K/S value of 22.7, with the reduction potential of the bath in the range of –640 to –710 mV before and after the reduction of dye, as well as at the end of dyeing. On increasing the concentration of FeSO4, the K/S of dyed cotton decreased to 17.5, which may be due to the partial reduction of indigo. FeSO4 reacts with NaOH to form Fe(OH)2, therefore, the concentration of sodium hydroxide (lower level) was decreased in the dyebaths.
Influence of FeSO4 and Indigo Concentrations
The combined effect of FeSO4 and indigo concentrations on K/S at a specific catalase concentration, temperature (medium level), and NaOH concentration (lower level) is shown in Fig. 1c. A lower level of FeSO4 concentration (20 g/L) and a medium level of indigo concentration (7.5 g/L) gave a maximum K/S of 22.7. The K/S values of dyed samples continued decreasing at increasing FeSO4 concentrations to 25 g/L for lower, medium, and upper levels of indigo concentration.
Influence of FeSO4 Concentration and Temperature
The combined effect of FeSO4 concentration and temperature on K/S values at specific enzyme, dye (medium level), and NaOH concentrations (lower level) is shown in Fig. 1d.
A maximum K/S value of 22.7 was achieved at a lower level of FeSO4 (20 g/L) and a medium level of temperature (70 °C). The lowest K/S value (14.6) was achieved at a lower level of temperature (60 °C) and a medium level of FeSO4 concentration (22.5 g/L). In this case, catalase was probably not completely activated to reduce indigo.
Indigo Uptake and Surface Indigo Dye Strength on Cotton
Indigo uptake (grams of indigo/100 g cotton) after each dip/ nip was evaluated and is shown in Fig. 2a; respective K/S values after each dip/nip against that of indigo uptake is shown in Fig. 2b. K/S values and dye uptake both increased proportionally. Although the final K/S value was nearly identical in both reducing systems, the total dye uptake was greater in the catalase system despite having a lower K/S value with the catalase system after the first dip/nip compared to that for hydrosulfite. The same K/S value in both cases, with variations in the dye uptake, permitted more diffusion of dye into cotton for the catalase system.
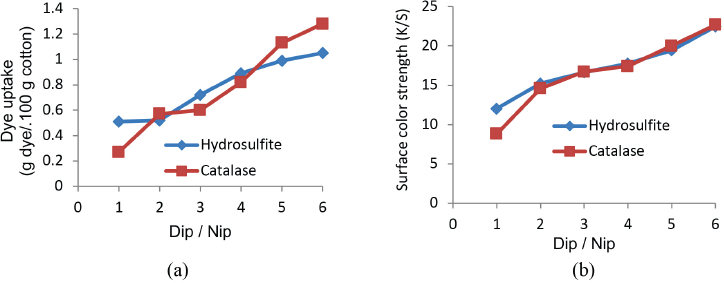
(a) Indigo uptake and (b) surface color strength (K/S) after each dip/nip.
Stability of Reduction Baths
In the Absence of Dye
The stability of the reduction baths prepared with Na2S2O4 or alkaline catalase with iron (II) salt was studied in the absence of indigo for up to 24 h, followed by the addition of indigo. Although both reducing systems retained their reducing capability for up to 24 h to some extent, an effective reduction potential was found only at 0 hours for obtaining a maximum K/S value. There was a progressive drop in mV and pH in both reduction baths at increased times. K/S values gradually decreased with increased storage time for both reduction systems as shown in Fig. 3a.
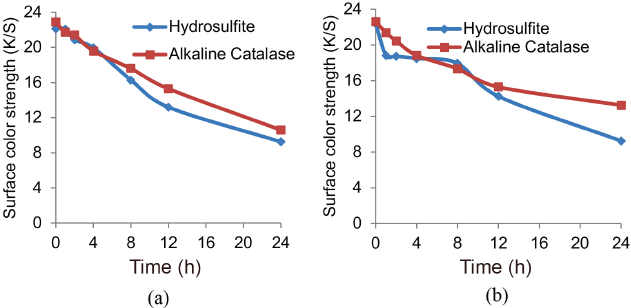
Surface color strength (K/S) of cotton after storage of baths for a specific time. (a) K/S of sample dyed from baths where indigo was added after storage and (b) K/S of sample dyed from baths stored along with indigo.
In the Presence of Dye
In this case as well, both reducing systems retained their reducing capability for up to 24 h to some extent. There was a steady drop in mV and pH of both reducing baths with increased time and proportionally decreased K/S values as well, as shown in Fig. 3b. Reduced dyebaths showed maximum K/S values at 0 h.
Characterization
SEM Analysis of Dyed Cotton Fabric
The objectives of SEM analysis were to evaluate morphological differences among undyed and dyed cotton fabric using sodium hydrosulfite or alkaline catalase with iron (II) sulfate as reducing agents. The SEM images of undyed cotton, as well as cotton dyed using sodium hydrosulfite or catalase systems, are shown in Figs. 4a–c, respectively. The images show marginal damage on the surface of cotton dyed using the sodium hydrosulfite system that was not prominent using the alkaline catalase system. The white spots on the dyed cotton were those of indigo deposited on the surface.

SEM images of (a) undyed cotton, (b) dyed cotton using sodium hydrosulfite, and (c) dyed cotton using alkaline catalase along with iron (II) sulfate reducing systems.
FTIR-ATR of Dyed Cotton
FTIR-ATR spectra were obtained using a Brucker Eco-Alpha spectrophotometer. Spectra of dyed cotton with Na2S2O4 and alkaline catalase were compared with undyed cotton (reference) as shown in Figs. 5a and b respectively. The absorption bands were mainly observed in the ranges of 3869 to 2850 cm–1 and 1623 to 522 cm–1.
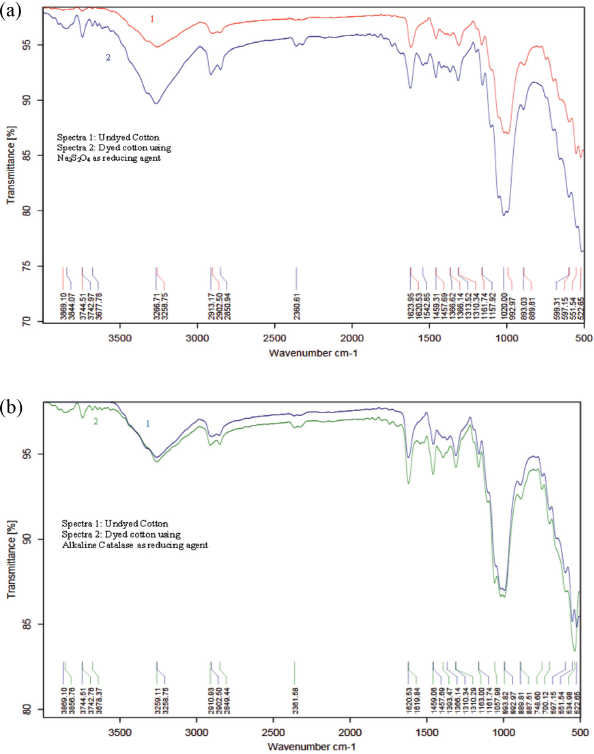
FTIR-ATR spectra of undyed cotton, as well as cotton dyed using (a) sodium hydrosulfite and (b) alkaline catalase along with iron (II) sulfate reducing systems.
A strong spectral band was found in the range of 3869 to 2900 cm–1, which was due to stretching vibrations of O-H and C-H bonds, and the peak around 3266 to 3258 cm–1 was due to the stretching vibration of R-OH in cellulose. This peak also included inter- and intra-molecular hydrogen bonds.35-37 The peak around 2913 to 2849 cm–1 was due to symmetrical and asymmetrical stretching of -CH2 groups in cellulose.38,39 Typical bands in the range of 1623 to 522 cm–1 were observed. The absorption band at 1623 to 1619 cm–1 was characteristic for the stretching of C=C. 37 The peaks at 1459, 1393, 1366, 1310, 1161, and 1057 cm–1 corresponded to deformation or stretching vibrations of C=O, C–H, C–O–C, C–O, C—N, C=C, and N-H groups in cellulose as well as indigo.40-42 In Fig. 5b, differences were observed in the spectrum of dyed cotton using alkaline catalase. There were changes in the absorption band in the range of 748 to 700 cm–1, which was assigned to in-plane bending of methyl groups in cellulose. 39
The spectra of both sodium hydrosulfite and alkaline catalase systems dyed cotton were nearly identical. Thus, it was concluded that no chemical changes occurred using the alkaline catalase system instead of sodium hydrosulfite.
Tensile Strength of Dyed Cotton
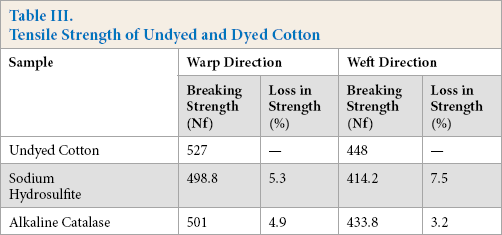
Reduction and oxidation of indigo may lead to loss in tensile strength of cotton.38,42 Tensile strength studies on dyed and undyed cotton revealed that a minimal loss in tensile strength occurred in both warp and weft directions, which was greater when the sodium hydrosulfite system was used (Table III).
Tensile Strength of Undyed and Dyed Cotton
Fastness Performance
Performance of light, rubbing and wash fastness of indigo dyed cotton using the Na2S2O4 and catalase systems were evaluated. The light, wash, and rubbing fastness ratings remained in the ranges of 6, 4–5, and 4–5, respectively. for both Na2S2O4 and alkaline catalase reducing systems.
Conclusions
In this study, application of alkaline catalase with iron (II) salt as a reducing agent for dyeing of cotton with indigo was reported. Alkaline catalase with iron (II) salt showed almost identical surface color strength (K/S) for an indigo concentration of 7.5 g/L compared to that using the hydrosulfite system with an indigo concentration of 3 g/L but a greater dye uptake was found for the alkaline catalase system due to higher diffusion of indigo into the cotton. Stability of dyebaths in both the absence and presence of indigo showed nearly similar results for up to 24 h, but the maximum dye strength was obtained at 0 h (i.e., dyeing immediately after reduction of indigo in the bath). However, alkaline catalase-based baths showed comparatively better stability. The colorfastness of dyed cotton was comparable for both reducing systems. No severe damage on the surface of dyed cotton was found for the alkaline catalase-based reducing system.
Sodium hydrosulfite is a highly toxic reducing agent and can be substituted with alkaline catalase with iron (II) salt for reduction of indigo, thus making the indigo dyeing process ecofriendly. However, the pH of the alkaline catalase-based baths requires further investigation to reduce the diffusion of indigo into cotton and to obtain comparable surface color strength at lesser concentration of indigo.
