Abstract
Indigo dyeing of cotton fiber in the silicone non-aqueous medium dyeing system was investigated to avoid the multiple dyeing of cotton yarn or fabric, high environmental pollution, and the need for multiple washing and sand washing. After fixing, the rubbing fastness of dyed fiber could be improved to 3–4 grade and the washing fastness increased 1–2 levels. The mechanism of rubbing fastness agents was also studied. Most of the wet rubbing fastness agents for indigo dyeing were waterborne polyurethanes. After fixation, a film was formed on the fiber surface. Moreover, the rubbing fastness was improved by the electrostatic interaction between the cellulosic fiber and indigo after treating with fixing agent. Indigo dyeing of cotton fiber was possible in the silicone non-aqueous dyeing system.
Introduction
Indigo is one of the most important dyes for cotton yarn dyeing; the annual global consumption is about 60,000 tons.1,2 However, traditional indigo dyeing is usually done in a water bath. In such a process, cotton textiles need multiple pad dyeing (6˜10 times) to achieve a desired color depth. 3 Since multiple dyeing may cause indigo to accumulate on the surface of the fiber, causing problems such as roll and fiber sticking during the spinning process, it is not suitable for dyeing cotton fiber. Furthermore, indigo water-based dyeing consumes a large amount of water (dyeing 1 ton of cotton yarns will consume about 60˜80 tons of water) and chemicals, resulting in a high chemical oxygen demand (COD) waste water, inorganic salts, and other chemicals such as reducing agents.4,5 Due to the monotonous color of indigo dyed warp yarns, all kinds of denim garments must be treated to obtain a variety of styles required by consumers.
Although a great deal of wastewater treatment plans have been developed, the best way to deal with these problems is to reduce the use of water and chemicals.6,7 Moreover, if indigo dyeing can be carried out on the loose cotton fibers, a series of cotton yarns with various performance, shapes, and indigo colors would be produced through spinning. Aided by changes in fiber composition, proportion, and the spinning method, and then in weaving (tatting or knitting), abundant fabric surface effects could be produced through the selection of yarn and texture variation, allowing more personalized high-quality new denim products to potentially be developed. Recently, some researchers 8 have begun to study new indigo dyeing methods to reduce waste water and chemical consumption. Reviews highlighting the significance of improving sustainability in indigo dyeing have been published, such as the development of vat dyes, the chemical modification of cotton fiber prior to dyeing, the use of eco-friendly chemical agents in dye bath, and applying a nitrogen system to reduce the use of chemical additives.9,10 All of these studies, however, are based on traditional water bath dyeing. Therefore, a sustainable indigo dyeing method that can increase the uptake of dye while saving water was investigated.
The silicone non-aqueous media dyeing system is a new green and environmentally-friendly dyeing technology. The silicone structure of D5 (decamethyl cyclopentasiloxane) is shown in Fig. 1. Researchers have shown that silicone non-aqueous media is safe enough for human and environment, and also widely used in cosmetics, body care products, and industrial application.11–13 On February 20, 2017, the European Commission submitted G/TBT/N/EU/456 to the WTO, stating that the D4 (octamethyl cyclotetrasiloxane) and D5 content of the rinse-off cosmetics should not be equal to or greater than 0.1%. As a dyeing medium, the recycling efficiency of D5 is very high,14,15 and the residual amount of D5 after drying is very low. In our previous work, silicone non-aqueous medium dyeing of cotton textiles with reactive dyes was successfully studied. In this dyeing system, reactive dyeing for cotton textile needs a small amount of water, improving the uptake of the reactive dye (close to 100%), which is relatively greater than the traditional water dyeing system.16,17 Greater than 90% dye pick-up and a deep color depth was achieved after one dyeing in the silicone non-aqueous dyeing system. 18 Furthermore, the silicone non-aqueous medium will remain on the fiber after dyeing. This part of silicone medium was recycled with a pressure drying machine. After dyeing, there is a small amount of dyeing effluent. As the surface tension of silicone medium (19.52 dyn/cm) is very low and its density is less than water, silicone non-aqueous medium and dyeing effluent can be naturally separated after 2˜3 h.
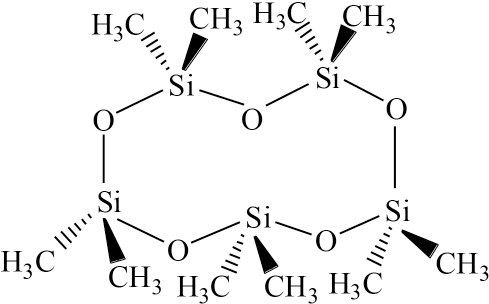
Chemical structure of silicone D5 non-aqueous medium.
Shao and Liu 19 studied the feasibility of indigo dyeing of cotton fiber in the silicone non-aqueous dyeing system. Results showed that indigo can achieve a deeper color depth in this dyeing system. Continuous operation of the silicone non-aqueous medium allows the reduced indigo solution repeated and full contact with the fiber; the reduced indigo solution can be captured and spread on the surface of the fiber to complete the dyeing process. However, the research on improving indigo uptake is limited. Particularly, the dry and wet rubbing fastness of indigo was poor when dyeing with the silicone dyeing system, which can cause dip roll blockage and contaminated machines, making the spinning of dyed fiber more difficult. Therefore, the dry and wet rubbing fastness of dyed fiber was investigated, and the fixation mechanism of the wet rubbing fastness agents were also systematically studied.
There is no dyeing method for cotton fiber in conventional indigo dyeing. Therefore, we dyed the fibers according to the traditional method of dyeing yarns and fabrics and compared them with the silicone non-aqueous dyeing system via the color depth, the uptake of indigo, and the rubbing fastness. Furthermore, two commonly-used wet rubbing fastness agents (A and B) were chosen for indigo dyeing, and their fixation mechanism was investigated.
Experimental
Materials
The combed cotton fiber (linear density: 1.8d, length: 28.5 mm, micronaire: A) that was used for the indigo dyeing was collected from Shaoxing Furun Dyeing and Finishing Co. Ltd. (Shaoxing, China). Indigo (granular synthetic, industrial grade) was purchased from Changzhou Lantu Chemical Co. Ltd. (Changzhou, China). The oxidation and reduction of indigo is shown in Fig. 2. Sodium dithionite, sodium hydroxide, potassium chloride, and sodium hydroxide were of analytical grade and purchased from Hangzhou Gaojing Chemical Reagent Co. Ltd. (Hangzhou, China). Silicone non-aqueous media (purity>96%) was purchased from GE Toshiba Silicone Ltd. (Jiujiang, China). Fixing agents A and B were bought from Guangzhou Asahi Chemical Co. Ltd. (Guangzhou, China).
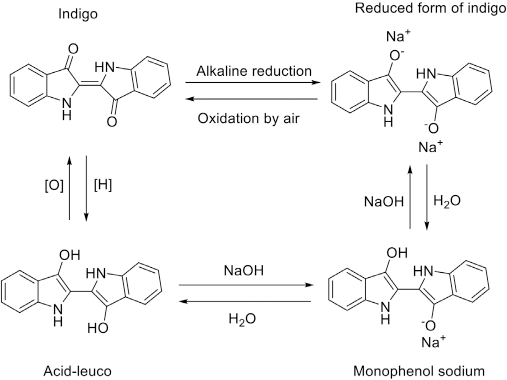
Oxidation and reduction of indigo.
Preparation of Indigo Reduced Solution
The indigo reduced solution was prepared by the addition of 6 g of indigo, 19 g of sodium dithionite, and 8 g of sodium hydroxide in 100 g of water. The reduction temperature was set at 60 °C for 20 min.
Dyeing Method
Dyeing was carried out on a Dye-24 dyeing machine (ShangHai Chain-Li Automation Equipment Co. Ltd.). The two different dyeing methods used are as follows.
Silicone Non-Aqueous Medium Dyeing System
Cotton fiber (1 g) was dyed with 1 g of indigo reduced solution, 0.8 g of water, and 20 g of silicone non-aqueous media. The dyeing temperature was raised from room temperature (RT) to 70 °C at a rate of 2 °C per min and held for 60 min. After dehydration, the cotton fiber was oxidized in the air for 12 h.
Traditional Water Bath
Cotton fiber (1 g) was dyed with 1 g of indigo reduced solution and 19 g of water. The dyeing temperature was raised from RT to 70 °C at a rate of 2 °C per min and held for 60 min. After dehydration, cotton fiber was oxidized in the air for 12 h.
After oxidation, both cotton fiber dyed in silicone non-aqueous media dyeing system and in traditional water bath were washed down at a ratio of 30:1 in water at 25 °C. Then cotton fibers were dried at 60 °C for 12 h.
Fixing Process
After dyeing and oxidation, cotton fibers were washed with 5% owf (on the weight of fiber) acetic acid at a liquor ratio (LR) of 30:1 in the water bath at 25 °C. After pickling, cotton fibers were fixed with 3% owf fxing agent at an LR of 30:1 in the water bath. Fixed occurred at 45 °C for 30 min. Then, the cotton fibers were pre-dried at 80 °C for 10 min and baked at 120 °C for 5 min. The dyeing and finishing process of indigo is shown in Fig. 3.
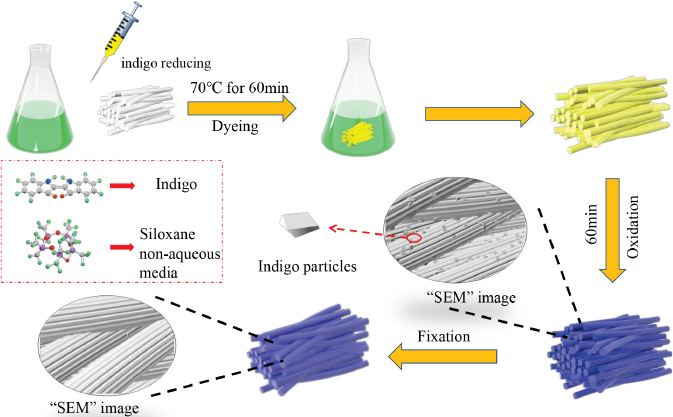
Schematic diagram of indigo dyeing in the silicone non-aqueous medium dyeing system.
Color Yield
The color values for the dyed fibers were measured using a Datacolor SF-650 Plus color testing and matching instrument that was equipped with a D65/8° source and 3-mm color measurement aperture. A cotton sample (0.5 g) was combed using a bristle comb and pressed into a 2 × 2 cm shape for measurements. Five different places were tested on the same cotton fiber, and the color data were averaged.
Rubbing Fastness
According to LVS EN ISO 105-X1219, 20 rubbing fastness was evaluated using a white fabric (100 × 40 mm) rubbed against the dyed cotton fiber under wet and dry conditions using Crockmeter 238 A (Shirley Development Laboratories Atlas InSc. United States). A cotton sample (0.5 g) was combed using a bristle comb and pressed into a 2 × 2 cm shape for measurements. After rubbing, the b* value of the stained white fabric was measured with a Datacolor SF600X spectrophotometer at the maximum absorption wavelength of 585 nm. The rubbing fastness of the sample was represented by the b* value of the stained white fabric, because the greater the b* value, the poorer was the rubbing fastness of the cotton fiber.
Light Fastness
Light fastness was determined as per the ISO 105-B0221 test method. Dyed cotton fibers (1 g) were pressed into a 4 × 4 cm shape and exposed to light continuously for 24 h.22,23
Washing Fastness
Dyed cotton fibers (2.5 g) were pressed into a 5 ×10 cm shape and sewn between the 50 × 100 multi-fiber cloth and the bleached cotton cloth. To prevent the cotton fiber agglomerating during the test, the sample was sewn again according to the fiber partition on the multi-fiber cloth. The wash fastness was evaluated according to the Marks & Spencer C4A method. 24
Fourier Transform Infrared Analysis
For Fourier transform infrared (FTIR) spectroscopic measurements in the transmission mode, the sample was tested by the KBr pellet method using a Nicolet 5700 FTIR spectrometer between 400 to 4000 cm–1 with 64 scans and a 4 cm–1 resolution. The background was scanned before the sample test, and the test results of the sample were automatically subtracted from the background.
Pyrolysis-Gas Chromatography/ Mass Spectrometry Analysis
Before testing, samples were dried at 60 °C for 48 h in an oven, and 1 g of sample was accurately weighed and placed into a sample bottle. Desiccators were used for the storage of samples prior to the analysis. Samples were stored in a desiccator before the analysis. Pyrolysis-gas chromatography/ mass spectrometry (PY-GC/MS) analysis was carried out by using a Multi-Shot Pyrolyzer EGA/PY-3030D (Frontier Laboratories, Japan). The sample was placed in a quartz capillary.24,25 Cracking was carried out at 550 °C for 30 min with a heating rate of 20 °C/s. During cracking, high-purity nitrogen gas was introduced as a carrier gas to bring out the cracking gas for the GC test.
GC/MS analysis was carried out using a 7890B/5977AGC/MS chromatograph (Agilent Technologies, USA). The pyrolysis interface and the GC port were kept at 280 °C, and the cleavage product was separated by a DB-5MS GC column (30 m × 0.25 mm × 0.25 μm) with a split ratio of 100:1. The carrier gas content in the column was maintained at 0.98 mL/min, and the initial temperature was increased at a rate of 1 °C/min after 1 min of incubation. The temperature was raised from 60 °C to 140 °C at a rate of 1 °C/min, and then raised to 300 °C at a rate of 20 °C/min for 8 min. The ion source was analyzed for positive ions at 230 °C in full scan mode. The relative content of each major component was calculated by using the chromatographic peak area normalization method and the National Institute of Standards and Technology (NIST) library.
Field Emission Scanning Electron Microscopy Analysis
Firstly, the fixing agent was diluted to 400, 100, 80, 60, and 20 g/L and coated onto a silicon wafer (0.5 × 0.5 cm) using spin coating. Then, field emission scanning electron microscopy (FESEM) analysis was performed using a VLTRA55 (Carl Zeiss SMT Pte. Ltd., Germany) instrument. Cotton fibers and the silicon wafer with fixing agent were adhered to the aluminum stage using a conductive paste, and then the surface of the sample was coated with a platinum layer. The microscope used an accelerating voltage of 15 kV, with a beam spot sizes of 15 and 20mm.
Surface Zeta Potential
After fixation, the surface charge on the cotton fiber was measured using a solid surface zeta potentiometer (SurPASS, Anton Paar, Austria). The electrolyte solution was potassium chloride (KCl) at a concentration of 0.001 M. The pH value was 7.28 (same as the pH of the fixation process) and streaming current mode was used during testing.
Solution Zeta Potential
The zeta potential of the fixation solution was tested using a 90Plus Zeta Nanoparticle Size and Zeta Potential Analyzer (Nano Brook 90plus Zeta). The fixation agent (1 g) was dissolved in 250 mL of water and the solution was tested under the water phase electrode.
Results and Discussion
Dyeing System Comparison
To evaluate the new dyeing method, cotton fibers were dyed in both the silicone non-aqueous medium dyeing system and traditional dyeing system. The K/S values of cotton fibers are shown in Fig. 4.

K/S values for cotton fibers dyed with indigo: (a) one time in water base, (b) six times in water base, (c) twelve times in water base, and (d) one time in silicone non-aqueous medium system.
As shown in Fig. 4, the K/S value of cotton fiber gradually increased with the number of dyeings in the traditional water bath. The K/S value of cotton fiber increased to 20.99 when the number of dyeings was twelve in the traditional water bath. However, the K/S value of dyed sample was 25.09 when the cotton fiber was dyed one time in the silicone non-aqueous medium dyeing system. It was then investigated whether the silicone non-aqueous medium influenced the dyeing property of the indigo reducing solution. The reducing solution of indigo was put in the silicone non-aqueous media for 60 min at 70 °C, followed by cotton fiber dyeing. Cotton fiber that was dyed in this manner had the same dyeing properties (K/S value = 25.17 ± 0.21, dry rubbing fastness (b* value) = –5.19 ± 0.09, and wet rubbing fastness (b* value) = –6.50 ± 0.03) as the cotton fiber that was immediately dyed with the indigo reducing solution. This confirmed that the reduced indigo solution was stable. The dyeing parameters and the silicone non-aqueous medium didn't influence the dyeing performance of the cotton fiber.
The important factor that influences the color depth of dyed sample is the final uptake of dye.26–28 If the dye could not be adsorbed on the cotton fiber, with most of the indigo remaining in the dye bath, a low uptake of indigo results. 29 In the silicone non-aqueous medium dyeing system, only 180% owf of water is used during dyeing. Moreover, this part of water can be completely adsorbed by the cotton fiber because it can adsorb 300% owf of water. As a result, all indigo reduced solution can be adsorbed by cotton fiber. This implies that the final uptake of indigo is higher in the silicone non-aqueous medium dyeing system. Conversely, 100% owf of indigo dye reduced solution is dissolved into water, the concentration of indigo will be reduced, and cotton fiber can't adsorb so much of water for the dyeing in traditional water bath.30,31 Therefore, only a small portion of indigo can diffuse to the surface of cotton fiber. We usually use a lower concentration of indigo reduced solution in the traditional dyeing of cotton fabric, so the cotton fabric needs multiple dyeings using the traditional water bath (the lower uptake of dye).
Wet and dry rubbing fastness are important properties for indigo dyeing. 32 As shown in Table I, after dry and wet rubbing of the traditionally dyed fabric, the b* value of white test fabric was –0.40 and –0.76 respectively, indicating that good rubbing fastness was achieved. However, the dry and wet rubbing fastness became poor with increased number of dyeings. For example, the b* value of white test fabric was –7.05 and –8.83 respectively when the number of dyeings was twelve using the traditional system. However, the rubbing fastness of fiber dyed in the silicone non-aqueous medium dyeing system was good, because the b* value of fabric after dry and wet rubbing fastness testing was –5.21 and –6.49, respectively. Furthermore, the color depth of the dyed fiber was 25.09, which was deeper than that using traditional water dyeing system with twelve dyeings (20.99). Therefore, indigo dyeing can achieve a better color depth and rubbing fastness in the silicone non-aqueous medium dyeing system than that in the traditional water dyeing system.
Rubbing Fastness of Indigo Dyed Cotton Fiber using Traditional Bath and Silicone Non-Aqueous Systems
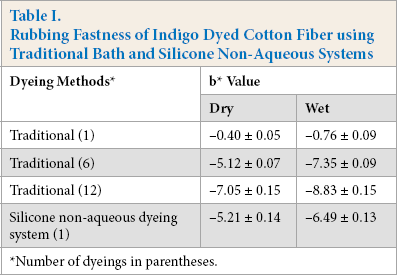
Number of dyeings in parentheses.
Effect of Fixing Agents on Rubbing Fastness
For denim, indigo-dyed cotton fibers require spinning, weaving, and other processes. The wet rubbing fastness of dyed fiber will influence these processes.32,33 Two different fixing agents were chosen to study their influence on the rubbing fastness of the dye. As shown in Fig. 5, the b* values of the white test fabric increased from –5.41 for the dyed control fiber not treated with fixing agent, to –2.86 and –0.98 for dyed fiber treated with fixing agents A and B, respectively. Thus, either fixing agent can significantly improve the rubbing fastness of the dyed fiber. Furthermore, the K/S value of the fixing agent treated cotton fiber was almost unchanged from the untreated dyed control fiber, indicating that the fixing process had no effect on the indigo color depth.
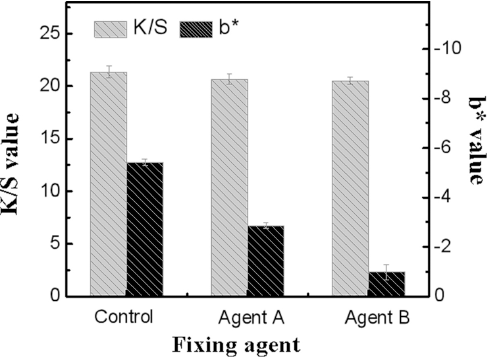
Effect of fixing agents on color depth and rubbing fastness of dyed cotton fibers.
Effect of Fixing Agents on Light and Washing Fastness
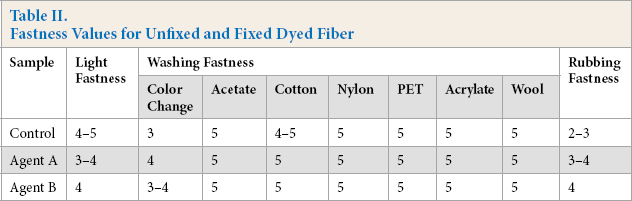
The light and washing fastness of the fixed and unfixed dyed cotton fibers are shown in Table II. Both fixing agents gave good results, with rating levels of 3–4 or 4 for light and rubbing fastness. Fixing agent A had a better washing fastness to color change than fixing agent B, while washing fastness to acetate, cotton, nylon, PET, acrylate, and wool was as good as unfixed. Therefore, the rubbing fastness of the dyed fiber improved after fixing. Comparing the rubbing fastness between the fixing agent A and the fixing agent B, fixing agent B gave better results on the dyed cotton fiber.
Fastness Values for Unfixed and Fixed Dyed Fiber
FTIR Analysis
Use of fixing agent A and B improved the rubbing fastness and washing fastness of indigo dyed cotton fiber. However, the fixing mechanism of these fixing agents was unclear. FTIR spectroscopy analytical technique was used to help understand the fixation process. The FTIR spectra of agents A and B are shown in Fig. 6. The broad band observed in the 3300–3400 cm–1 region showed a typical O-H stretching mode on the carbon chain.34–36 The bands at 2870 and 1450 cm–1 were due to -CH2 vibration states, and the strong hydrogen band at 1720 cm–1 was attributed to the stretching vibration of C=O in amide compounds. The peaks found at 1600 and 766-785 cm–1 were due to C-H stretching vibrations of benzene. Also, bands at 1540 and 1300˜1030 cm–1 were due to the stretching vibrations of -NH and C-O-C group, 37 respectively. By comparing the FTIR spectra of agents A and B, B showed two characteristic peaks at 785 and 856 cm–1, which were different from those of A at 766 and 816 cm–1, indicating the positions of the substituents on the benzene ring may be different for the two fixing agents.
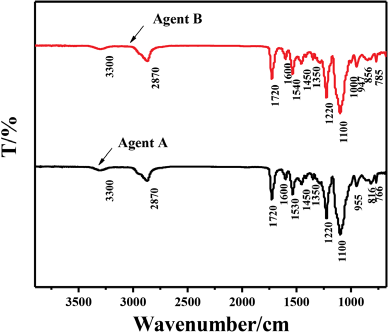
FTIR analysis of fixing agents.
The infrared spectra of fixing agents A and B were basically consistent. According to the FTIR spectrum of polyurethane, it was found that there were similarities between the FTIR spectra of fixing agents A and B and the spectrum of polyurethane.
PY-GC/MS Analysis for Fixing Agents
PY-GC/MS can break cross-linked polymeric materials down into smaller molecules and provide information about the chemical components of polymers.38,39 PY-GC/MS analysis of fixing agents A and B are shown in Fig. 7. The most important compounds are listed during the cracking process in Tables III and IV. Compounds were identified by using a standard database.
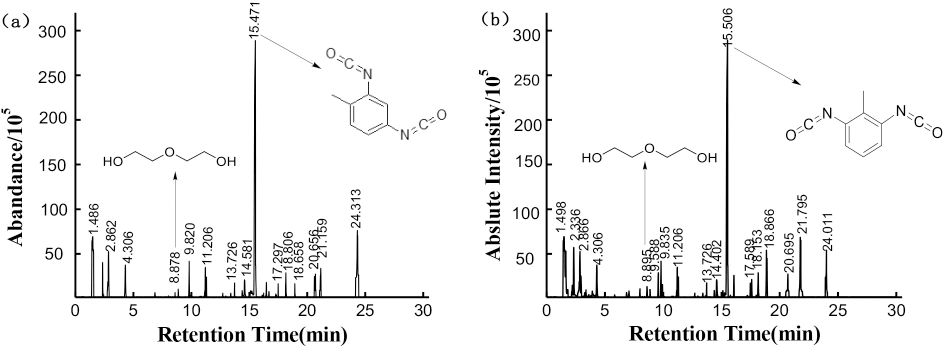
PY-GC/MS analysis of fixing agents (a) A and (b) B at 500°C.
Some strong signals of ethers, alcohols, and isocyanates were observed during the pyrolysis of agents A and B. Due to the high alcohol content, the pyrogram of fixing agent B contained more polyether fragments. Based on the cracking compounds, the polyol monomers of fixing agent A and B were both polyethylene glycol. After cracking, the polyol fragment peaks can be seen in many places, indicating that fixing agents A and B were polyether polyurethane.40,41
Although there were some similar peaks for these two fixing agents, some peaks were different. For example, the chain extender of synthetic polyurethane was diethylene glycol, which was detected at a retention time of 8.895 and 8.878 min in Table III and Table IV, respectively. Moreover, the obvious difference between these two fixing agents were their cyanate monomer. For fixing agent A, this was toluene-2,4-diisocyanate, which was detected at 15.471 min in Table III. For fixing agent B, it was toluene-1,3-diisocyanate, which was detected at 15.506 min in Table IV. Therefore, the PY-GC/MS results showed the molecular structure information of these two fixing agents. The different fixing effects observed when using A or B might be due to the different isocyanate monomer used in each.
Compounds Identified in Pyrograms from Py-GC/MS Analysis of Fixing Agent A *
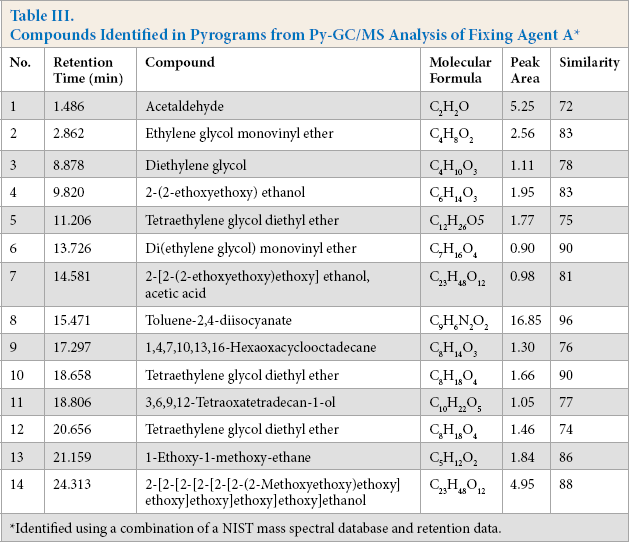
Identified using a combination of a NIST mass spectral database and retention data.
Compounds Identified in Pyrograms from Py-GC/MS Analysis of Fixing Agent B *
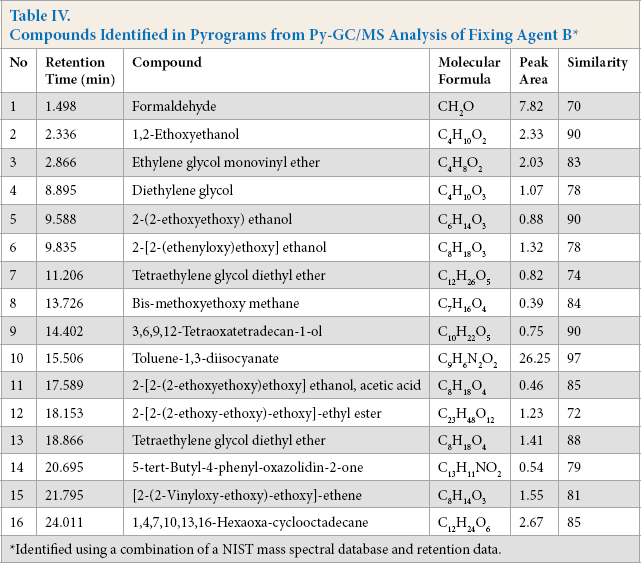
Identified using a combination of a NIST mass spectral database and retention data.
SEM Analysis
The surface morphology of fibers may influence the rubbing fastness of indigo. As shown in Fig. 8, the surface morphology of dyed cotton fiber that was not treated with fixing agents showed some deep grooves and slender concave grooves (Fig. 8a), indicating that the surface of untreated fiber was rough, uneven, and with massive lumps.42–44 Due to the high concentration of indigo in the silicone non-aqueous dyeing system, dye aggregates may form and load on the cotton fiber surface, resulting in a surface that was not smooth. However, the surface of cotton fibers were smoother after treating with the fixing agent A (Fig. 8b). Although the large particles of dyes disappeared, there were still some smaller dye particles. Fig. 8c showed that almost no indigo aggregates were on the cotton fiber surface after treating with fixing agent B. The reason may be that the film forming property of fixing agent B was better than that of fixing agent A (Fig. 9).

SEM image of cotton fibers before and after fixation: (a) dyed, untreated control fiber sample, (b) sample treated with fixing agent A, and (c) sample treated with fixing agent B (magnifcation 20,000×).

Scanning electron microscopy image of spin-coated fixing agent.
Film forming properties of the two fixing agents were tested by coating them on silicon wafers. The two fixing agents were spun coated on the silicon wafers at various concentration. As shown in Fig. 9, the integrity of the film formed on the surface of silicon wafer became worse with decreasing fixing agent concentration. The film formed by fixing agent B was more complete than that of fixing agent A at the same concentration. For example, when the concentration was 400 g/L, there were only two voids on the film of B, but many voids were visible on the film of A. As the concentration decreased to 20 g/L, the voids on the film of fixing agent B increased, but the continuity of the film was still good.
The SEM images showed that the polyurethane with different reactive monomers could self-crosslink on the fiber surface to form macromolecules under certain conditions. The film mechanism of the fixing agent is shown in Fig. 10. The fixing agents were adsorbed on the cotton fiber surface, and then formed a crosslinked membrane after dehydration and drying. As a result, indigo dyes were difficult to remove by rubbing if the fibers were covered with a film. Moreover, the cyanate monomer of fixing agent B (toluene-1,3-di-isocyanate) had excellent film forming properties, when comparted to fixing agent A, and, therefore, gave better fiber dye rubbing fastness.
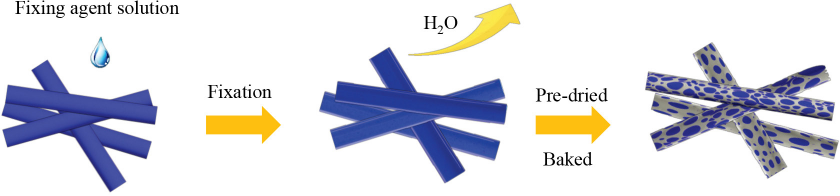
Schematic diagram of film mechanism of fixing agent for indigo dyeing.
Surface Zeta Potential
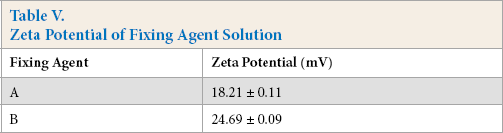
The surface zeta potential of the fixed fiber was measured to determine its influence on rubbing fastness. As shown in Fig. 11, the control cotton fiber (dyed and untreated with fixing agent) showed a low negative charge (–25.56 mV). However, the surface charge on cotton fiber increased to –20.73 mV and –21.30 mV after being treated with fixing agents A and B, respectively. It could be concluded that the zeta potential of these two fixing agent solutions were positive (Table V).45–47 The surface charge on cotton fiber after being treated with fixing agent A increased more than the cotton fiber after being treated with fixing agent B even if fixing agent A had a smaller zeta potential than fixing agent B, indicating that there was a greater amount of fixing agent A on the cotton fiber. Based on Fig. 9, the film formation of fixing agent B was better than that of fixing agent A at the same concentration and the effect of fixing agent B on improving the rubbing fastness was better than fixing agent A. It was apparent that the film-forming property had a greater influence on the rubbing fastness than the iconicity of the fixing agent. In conclusion, both of these fixing agents increased the zeta potential of the cotton fiber, so the film formation property was the major effect on improving the rubbing fastness.
Zeta Potential of Fixing Agent Solution
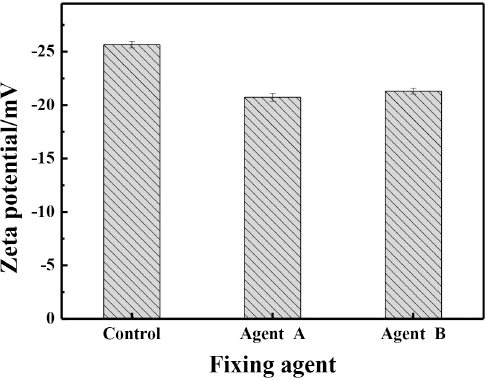
The surface zeta potential of fibers after fixation.
Cotton is a cellulosic fiber containing many hydroxyl groups (-OH) in its molecular structure. The surface charge of the fiber treated with fixing agents was greater than that of the control fiber, resulting in the increased electrostatic interaction between the cellulosic fiber and indigo.
Consequently, less dye was removed from the treated fiber during friction because of stronger interaction between fiber and indigo.48,49 Therefore, the rubbing fastness of dyed fiber could be improved by the fixing process.
Conclusions
In conclusion, indigo non-aqueous medium dyeing of cotton fibers can achieve greater than 90% dye uptake after just one dyeing, which makes deeper indigo dyeing of cotton fibers possible. The rubbing fastness of cotton fiber that was dyed using the silicone non-aqueous dyeing system was better than that dyed using the traditional water system. The rubbing fastness of indigo dyed cotton was greatly improved by fixing agent treatment. The fixing agents improved the washing fastness of the dyed cotton fiber, but had a little influence on the light fastness.
The FTIR spectrum of fixing agents A and B showed them to be waterborne polyurethanes. The chemical components of these two agents were studied by PY-GC/MS during the polymer pyrolysis. These results indicated that both fixing agents had the same chain extender and polyol monomer, although the substituent position of isocyanate (-N=C=O) was different on the benzene ring. This gave each fixing agent different film-forming properties. Furthermore, the zeta potential of fixing agents showed that both fixing agents were cationic and the electronegativity of the fiber decreased after fixation. These favorable results demonstrated that the deep dyeing of indigo on cotton fiber was achievable using the silicone non-aqueous dyeing system, and the resulting rubbing fastness was better than that achieved by the traditional water dyeing system.
