Abstract
Use of sodium hydrosulfite in the dyeing of cotton with vat dyes is criticized for generation of sulfur compounds leading to air and water pollution. In this study, attempts were made to dye cotton with alkaline pectinase along with iron (II) salt as an alternative formulation. A 43 Box-Behnken design was used for statistical analysis of performance for this new reducing system and to obtain optimum parameters for cotton dyeing. The results showed that alkaline pectinase along with iron (II) salt was quite effective in developing comparable dyebath potential, dye receptivity on cotton with comparable dye strength, and colorfastness, as compared to the hydrosulfite-based reducing system. Dyebath stability in presence and absence of dye also showed superior results compared to that of the hydrosulfite system.
Introduction
Vat dyes belong to a well-known carbonyl class of dyes having a coplanar and multi-ring system. These rings contribute to strengthening the Van der Waals forces between dye and substrate. 1 At present, there is no true alternative to vat dyes, due to their excellent all-round colorfastness properties, which include wash, light, perspiration, and crock fastness.2,3However, vat dyes are practically insoluble in water. But they can be converted into a water soluble form (leuco dyes) that has an affinity for cotton by reduction with a strong reducing agent like sodium hydrosulfite (Na2S2O4) and sodium hydroxide (NaOH) solubilizing agent. 4 The reduced form of the vat dye exhibits affinity to cellulosic fibers and thus becomes exhausted from the dyebath, followed by oxidation in which the exhausted dye is re-oxidized within the fiber as the oxidized insoluble form.5–7
The disposal of dyebaths and rinse water causes problems due to the formation of sulfite, sulfate, thiosulfate, and toxic sulfide from the dissociation of hydrosulfite, contaminating wastewater from dyeing plants. In addition, as a result of the considerable excess of reducing agent required to stabilize oxidation-sensitive dye baths, the wastewater may contain excess sodium hydrosulfite, which affects aerobic processes in wastewater treatment.8,9 This has led to a search for alternate eco-friendly hydrosulfite-free based reducing agents.10,11 These include application of iron (II) salts along with gluconic acid and NaOH at 60 °C, 12 and iron (II) salts in combination with tartaric or citric acid, triethanolamine, and NaOH at room temperature (RT).13–15
With the increasing demand on textile manufacturers to reduce pollution in textile coloration and finishing, the use of enzymes in the textile chemical processing is rapidly gaining wide recognition because of their eco-friendly and non-toxic characteristics. 16 Enzymes are biological catalysts that mediate virtually all the biochemical reactions that constitute metabolism in living systems. 17 They accelerate the rate of chemical reactions without themselves undergoing any permanent chemical change (i.e. they are true catalysts).18,19 Certain specific enzymes are available that can catalyze dye reduction. 20 The effectiveness of enzymes, especially those belonging to oxidoreductase and hydrolase classes, is of utmost importance in this perspective. 21
Pectinases comprise of a unique group of enzymes that catalyze the degradation of pectic polymers present in the plant cell wall. 22 They are mainly synthesized by microorganisms and plants and are defined as a heterogeneous enzyme group that hydrolyzes pectic substances—a polymer chain consisting of a rhamnogalacturonan backbone that is linked to carbohydrates and other polymers. 23
Pectinases were some of the first enzymes to be used for domestic applications. Only in the 1960s did the chemical nature of plant tissues become apparent, and with this knowledge, scientists began to use a greater range of enzymes more efficiently. 24 As a result, pectinases are finding increased use in the commercial sector as an integral part of various biotechnological applications. 25
Based on the pH requirement for optimal enzymatic activity, pectinase can be broadly classified into acidic and alkaline types. Acidic pectinase has been extensively used for the extraction and clarification of fruit juices and wines, maceration of vegetables and fruits, improvement in essential oil extraction, and enhancement of baby-food production. In contrast, alkaline pectinases are used for treatment of wastewater from the food processing industry containing pectinaceous waste, degumming of bast fibers, 26 bioscouring of cotton, retting of plant fibers, coffee and tea fermentation, paper and pulp industrial processes, and the extraction of lemon oil. 25
In the present work, the effectiveness of alkaline pectinase along with iron (II) salt was studied on the reduction and solubilization of vat dyes, thus attempting to substitute sodium hydrosulfite in the vat dyeing technology to formulate a green as well as eco-friendly process. A 43 Box-Behnken design was introduced for statistical analysis on performance of this system and to obtain optimal parameters and accuracy of the process for dyeing of cotton using alkaline pectinase along with FeSO4. The reduction potential and dye strength, as well as stability of hydrosulfite and alkaline pectinase-based dyebaths, were studied. The colorfastness of dyeing was also evaluated and compared between the two methods.
Experimental
Materials
Thoroughly pretreated cotton fabric (124 epi, 92 ppi, warp and weft: 37s, and 138 g/m2) was used in this study.
Chemicals
Alkaline pectinase was procured from Clariant. Sodium hydrosulfite (AR, 87-88%), sodium hydroxide (AR, 98%), ferrous sulfate (AR, 98%), and hydrogen peroxide (30%) were obtained from SDFCL. Ten different vat dyes of commercial grade, viz. Green XBN (Vat Green 1, C.I. 59825), Olive Green B (Vat Green 3, C.I. 69500), Brown BR (Vat Brown 1, C.I. 70800), Gold Orange 3G (Vat Orange 15, C.I. 69025), Yellow 5G (Vat Yellow 2, C.I. 67300), Red 6B/UD (Vat Red 10, C.I. 67000), Grey M (Vat Black 8, C.I. 71000), Olive R (Vat Black 27, C.I. 69005), Brown R (Vat Brown 3, C.I. 69015), and Blue BC (Vat Blue 6, C.I. 69825) were used in this study. These dyes were obtained from ATIC and Indian Dyestuff Industries.
Cotton Dyeing using Sodium Hydrosulfite
The dyebaths were prepared with vat dye (1%, owf) pasted with TR oil and hot water was added to it. Sodium hydroxide (10%, owd) and sodium hydrosulfite (10%, owd) were added and stirred well until reduction and solubilization of dye took place (about 15 to 20 min at 60 °C). Cotton dyeing was carried out for 60 min at 60 °C at a 1:25 liquor ratio (LR). The bath was then dropped, the dyed cotton was cold washed and oxidized with hydrogen peroxide (1–2 g/L) at 50–60 °C for 20–30 min, followed by soaping and a final wash.
Cotton Dyeing using Alkaline Pectinase and FeSO4
The dyebaths were prepared with vat dye (1–1.5%, owf), alkaline pectinase (0.25–1.5%, owd), ferrous sulfate, (1–6% owd), and sodium hydroxide (10–20% owd) at 90 °C for 1 h at a 1:25 LR. The dyed fabrics were cold washed thoroughly and oxidized with hydrogen peroxide, followed by soaping and washing.
Evaluation of Dyebath and Dyed Samples
Cotton samples dyed in both the hydrosulfite- and alkaline pectinase-based dyebaths were evaluated for dye strength (K/S) and CIELab values using the Datacolor Check (Data-color International) and the colorfastness of dyed cotton was assessed according to AATCC TM16-2004 (light), AATCC TM61-2007 (wash), and AATCC TM8-2007 (crocking) using the ATIRA lightfastness tester (Paresh Engineering Works), washfastness tester (RBC Electronics), and crock-meter (Prolific), respectively. The reduction baths were evaluated for reduction potential, as well as pH, at various stages of dyeing; in a blank reduction bath, after dye reduction, and at the end of dyeing using a digital pH meter and a digital combined oxidation reduction potential (ORP) meter (Century Instruments), respectively. The redox potential was assessed at reduction/dyeing temperature using an Ag/AgCl reference electrode and a Pt-Ag/AgCl combined electrode filled up with a saturated KCl solution.
Evaluation of Dyebath Stability
Reduction baths were prepared with hydrosulfite/hydroxide and alkaline pectinase/FeSO4 separately. To study bath stability in absence of dye, the reduction baths were stored for 0, 1, 2, 4, 8, 12 and 24 h, followed by Vat Green XBN dye addition (1% owf) after the specified time of reducing agent storage, and cotton dyeing was carried out; changes in pH and reduction potential at each stage were recorded. To study the potential of stored dyebaths towards successful dyeing, hydrosulfite and alkaline pectinase reduction baths were prepared, followed by dye addition and storage up to 24 h prior to dyeing.
Statistical Analysis of the Dyeing Process
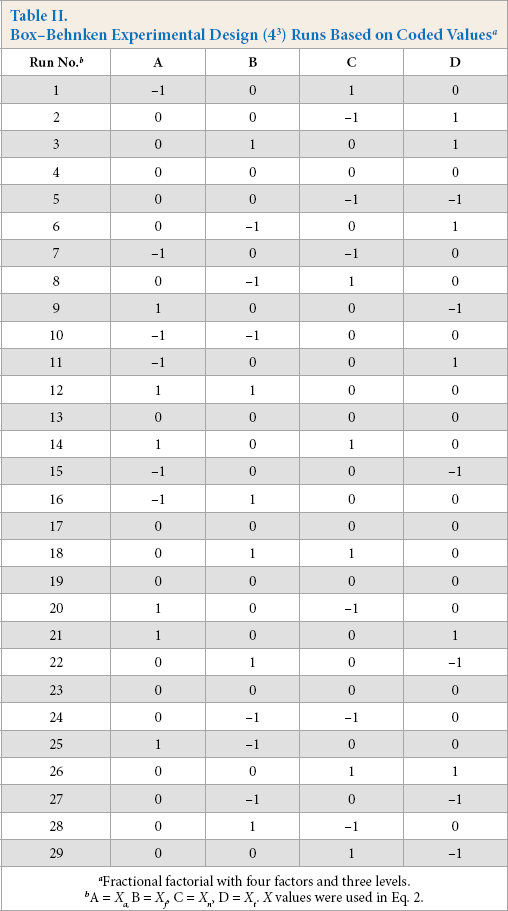
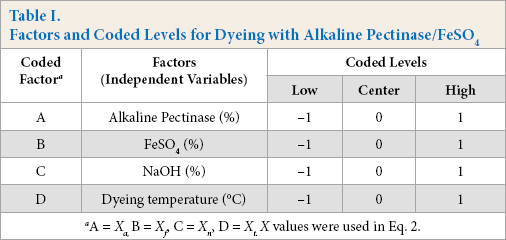
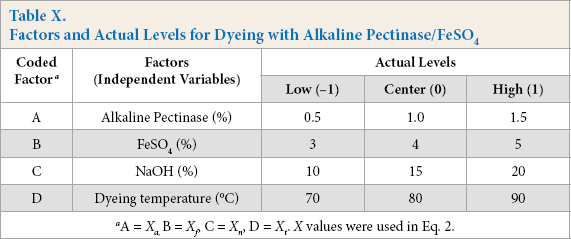
The Box-Behnken design was used to study the dye strength (K/S) as the response for cotton dyed with alkaline pectinase/ FeSO4 for optimization of process parameters. Four parameters or factors, viz. concentration of pectinase (A), concentration of FeSO4 (B), concentration of NaOH (C), and dyeing temperature (D) with respective coded values (levels) are shown in Table I.
Factors and Coded Levels for Dyeing with Alkaline Pectinase/FeSO4
A =
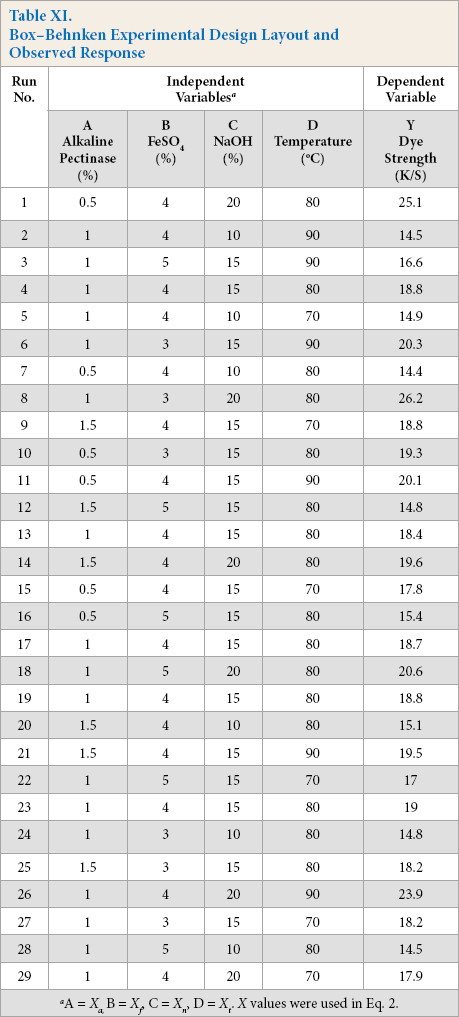
Using these four parameters as factors, each with three levels, a 43 Box-Behnken design was run to get a set of data (run), consisting of 29 total runs with four replicates at the central point. The design run results are given in Table II. Results were analyzed with response surface plots and equations were created for responses at a 95% confidence level. Individual, as well as interaction, effects of the process factors on K/S values were examined. The accuracy of the model generated was evaluated by the coefficient of determination (R 2 ). Response surface figures were analyzed for understanding the effect of individual parameters (factors) on response (i.e., K/S values). The regression equation was determined accordingly.
All design formations and statistical analysis were done with Design Expert 9 software. A quadratic polynomial was used to analyze the relationship K/S values (response) with four independent variables (factors) for the Box-Behnken design runs.
Data (response results) were analyzed using statistical tools to analyze the individual and interaction effect of the process factors on K/S values. Software was used to yield a regression equation at 95% confidence level (Eq. 1) and response surface figures were formed.
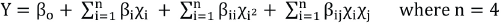
Y represents the predicted response function, β0 is an intercept, βi, βii, and βij are the coefficients of the linear, quadratic, and interactive terms of regression equation, and χi, χii, and χij represent the coded independent variables, respectively. The regression coefficients were then used to make statistical calculations to generate dimensional response surface figures from the regression models. The fitted polynomial equation was expressed as response surface figures to visualize the relationship between the response and the experimental levels of each factor by 3-D response surfaces figures. These figures display the relative influence of the factors and help to predict experimental results for other combinations. The accuracy of the model was checked by the resulting R 2 . When R 2 approaches unity, the empirical model fits the actual data. P-values of less than 0.05 were considered to be statistically significant. A lack of ft test was analyzed to check the adequacy of the model. Two techniques (i.e., response surface figures and regression equation techniques) were used to predict the optimal combination of factors that produces a maximum K/S value.
Results and Discussion
Sodium Hydrosulfite Reduction
Cotton was dyed with ten different vat dyes (1% owf) using Na2S2O4 (10% owd) and NaOH (10% owd), followed by oxidation with H2O2. The reduction potential and pH of baths at different stages of dyeing, as well as dye strength (K/S) of cotton are shown in Table III.
Dyebath Status and Dye Strength using the Sodium Hydrosulfite System
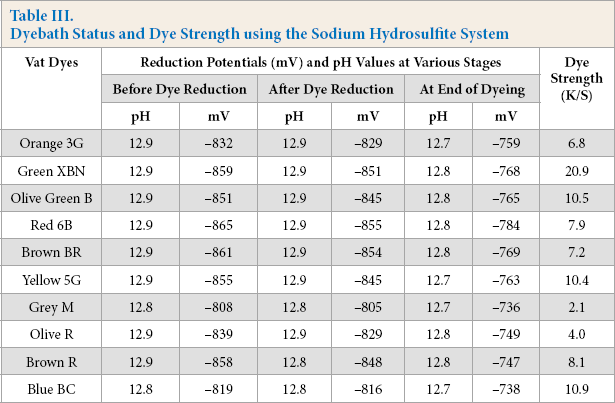
The pH of the baths at different stages of dyeing were found to be mostly identical, showing adequate alkalinity of the bath (Table III). The reduction potential of the baths remained on the high side, with a slight decrease after dyeing; adequate for reduction of vat dyes and retaining their reduced state throughout dyeing. K/S values of the samples were as expected. These K/S values were accepted as control values for the study of enzyme-mediated vat dye reductions.
Vat Dyeing using Alkaline Pectinase
In our earlier work, we established that alkaline pectinase can reduce sulfur dyes at 90 °C, thus proving its dye reduction abilities. 27 To develop a new enzyme-based reduction method, dyebaths were prepared with alkaline pectinase (0.25% owd) and NaOH (10% owd). Dyeings were carried out at 60 °C. The reduction potential and pH of the bath, as well as the resulting K/S values on dyed cotton, are given in Table IV.
Influence of Alkaline Pectinase and NaOH on Dyebath Status and Dye Strength with Vat Green XBN (1%)

The reduction potential of the dyebaths at different stages of dyeing did not rise beyond –206 m V, although the pH remained on the remarkably high side (12.8–13.0). Vat dyes require a reduction potential of -750 mV or higher for their reduction and dyeing. In this case, because of absence of reduction of vat dyes, no dyeing took place.
Vat Dyeing using Alkaline Pectinase and FeSO4
The activity of certain enzymes can be enhanced by adding FeSO4 to reduction baths. The FeSO4 performs two functions here; it forms Fe(OH)2, which in turn gets partially solubilized in the bath in the presence of excess alkali, and the synergetic reduction of vat dye, possibly by contributing to the gross reduction potential of bath. Thus, FeSO4 was added to the alkaline pectinase-based reduction baths to enhance the reduction potential. Dyebaths were prepared with alkaline pectinase (0.25% owd), FeSO4 (1% owd), and NaOH (10% owd) at 60 °C for 1 h. The dyebath status at different stages of dyeing and the corresponding K/S values on dyed cotton are shown in Table V.
Influence of FeSO4 on Dyebath Status and Dye Strength with Vat Green XBN (1%)
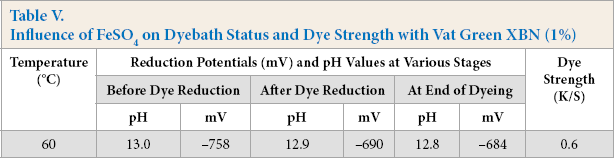
The reduction potential of the dyebaths at different stages of dyeing rose up to –758 m V, and the pH was on the high side (12.8–13.0) throughout. Due to a substantial rise in reduction potential, dyeing took place to some extent, which was obvious from the K/S value of the dyed sample. Enzymes exhibit maximum performance at specific temperatures. Alkaline pectinase may not work fully at 60 °C, and as a consequence, partial reduction of dye caused the poor dye strength of cotton. In subsequent studies, dyebath parameters, such as concentrations of pectinase, FeSO4, and NaOH, as well as time and temperature of dyeing, were varied initially to see their influence on the dyeability of cotton before statistical analysis was performed.
Selection of Factors and Levels for Design
To study the reduction potential of the new alkaline pectinase-based system using FeSO4 for successful dyeing of cotton, the dyeing parameters (factors) were initially tested within a certain range to check their capabilities, dye strength (K/S) values were checked, and these results were fed into to the Box-Behnken design as factors and levels.
First, the dyebath temperature was varied at 30, 40, 50, 60, 70, 80, and 90 °C, keeping the other parameters unchanged. The dyebath status at different stages of dyeing and the corresponding K/S values of dyed cotton are shown in Table VI.
Influence of Dyeing Temperature on Dyebath Status and Dye Strength
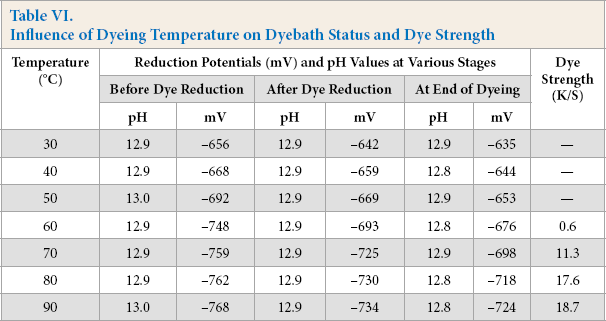
The new reducing system could not generate the required reduction potential in the dyebath at temperatures below 60 °C, with no reduction, and therefore, no cotton dyeing. Increased temperature progressively increased the dyebath reduction potential and the maximum dye uptake took place at 90 °C (Table VI); this temperature was accepted for further study. Pectinase is a cultured bacterial enzyme, frequently applied in scouring of cotton to remove hemicelluloses at boil; increased temperature enhanced its activity by improving the dyebath reduction potential.
To check the concentration of alkaline pectinase, dyebaths were prepared with Vat Green XBN dye (1% owf), alkaline pectinase (0.25–1.5% owd), FeSO4 (2% owd), NaOH (10% owd), temperature of vatting and dyeing at 90 °C, and a dyeing time of 1 h. The dyebath status at different stages of dyeing and corresponding K/S values of dyed cotton are shown in Table VII.
Influence of Alkaline Pectinase on Dyebath Status and Dye Strength
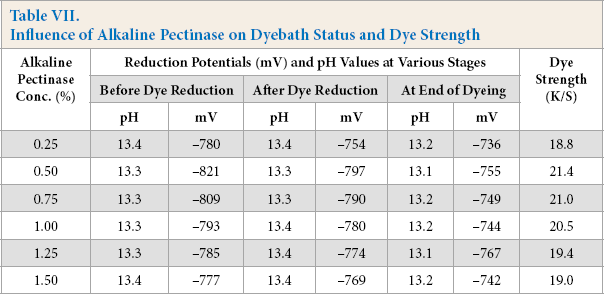
With an increase in alkaline pectinase concentration up to 0.50%, there was a proportionate increase in the reduction potential of the dyebaths, followed by a gradual decrease at greater alkaline pectinase concentrations, while the pH remained unchanged. The maximum K/S value of dyed cotton followed the same trend as the reduction potential (Table VII). Enzymes are effective at low concentrations; an increase in concentration beyond a certain limit failed to further improve the reduction potential and K/S value.
To determine the effective FeSO4 concentration, dyebaths were prepared with Vat Green XBN dye (1% owf), alkaline pectinase (0.50% owd). FeSO4 (1–6% owd), NaOH (10% owd), temperature of vatting and dyeing at 90 °C), and a dyeing time of 1 h. The dyebath status at different stages of dyeing and corresponding K/S values of dyed cotton are shown in Table VIII.
Influence of FeSO4 on Dyebath Status and Dye Strength
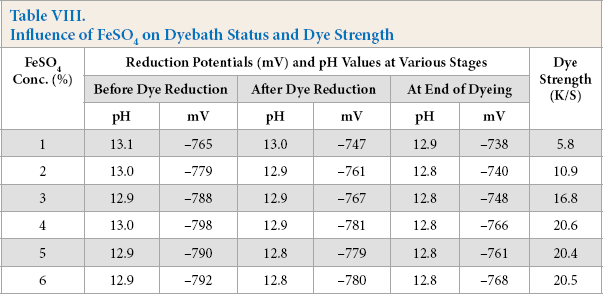
An increase in the concentration of FeSO4 increased the reduction potential of the dyebaths proportionately, while keeping the pH unchanged (Table VIII). The K/S values of dyed cotton also increased up to a 4% FeSO4 concentration, beyond which it remained almost unchanged.
Application of FeSO4 can consume NaOH to form Fe(OH)2, thus requiring excess NaOH for efficient dyeing. Dyebaths were prepared with Vat Green XBN dye (1% owf), alkaline pectinase (0.50% owd), and FeSO4 (4% owd). The concentration of NaOH was varied (11–20% owd), temperature of vatting and dyeing at 90 °C, and a dyeing time of 1 h. The dyebath status at different stages of dyeing and the corresponding K/S values of dyed cotton samples are shown in Table IX.
Influence of NaOH on Dyebath Status and Dye Strength
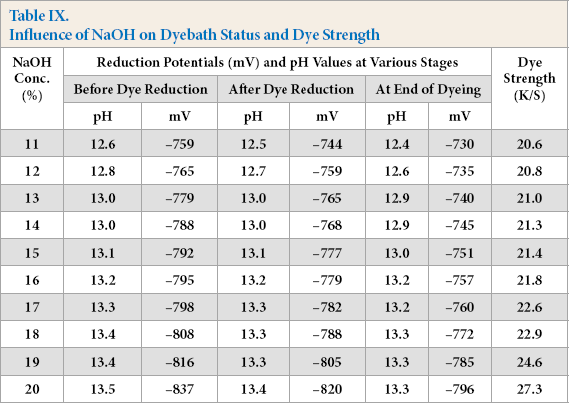
Increased NaOH concentration proportionately increased the reduction potential and pH of the dyebaths as expected (Table IX). The K/S values of dyed cotton gradually increased with increased NaOH concentration up to 20% owd. NaOH concentration must be kept high to obtain the maximum solubility of reduced vat dye, to maintain higher pH, in addition to the NaOH consumed by FeSO4 conversion to Fe(OH)2.
Experimental results showed at least comparable efficiency of alkaline pectinase-based dyebaths as compared to the hydrosulfite system. Reduction potentials in all the three stages of dyeing (–759 to –837 mV) indicated complete dye reduction. The K/S results confirmed reduction and solubilization of dye using alkaline pectinase.
Parameters for selection of factors and levels for design were alkaline pectinase (0.5–1.5% owd), FeSO4 (3–5% owd), NaOH (10–20% owd), and temperature (70–90 °C).
Box-Behnken Experimental Design
A 43 Box-Behnken fractional factorial experimental design was run with four factors and three levels each (Table X), consisting of 29 runs with four replicates for optimization of parameters in the alkaline pectinase system. For all design runs, dye strength (K/S values) as response was obtained from empirically-derived CIELab values (Table XI).
Factors and Actual Levels for Dyeing with Alkaline Pectinase/FeSO4
A =
Box—Behnken Experimental Design Layout and Observed Response
A =
Statistical Analysis
Statistical analysis was carried out for the obtained dye strength (K/S values) for 29 runs (Table XI) using the Box-Behnken experimental design. Response surface methodology (RSM) represents the effects of the four independent factors and their interaction with K/S values. The response surface figures (Figs. 1a–f) show the K/S values (dependent variable) of cotton dyed using alkaline pectinase as a function of two factors, while the third factor was kept at a constant center value level.
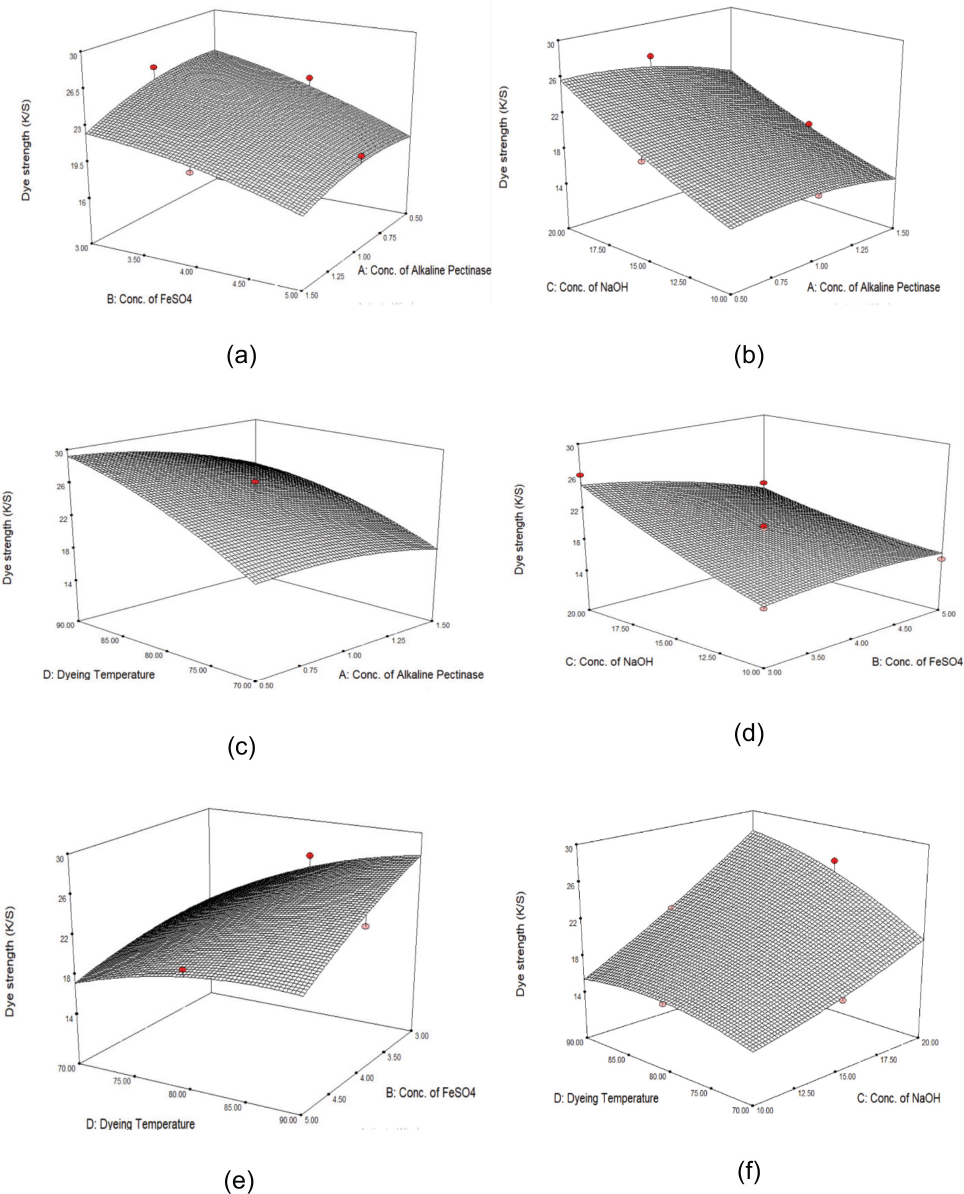
Response surface figures of dye strength (K/S values) data obtained (a) with combined effects of alkaline pectinase and FeSO4 concentrations, (b) alkaline pectinase and NaOH concentrations, (c) alkaline pectinase concentration and temperature, (d) FeSO4 and NaOH concentrations, (e) FeSO4 concentration and temperature, and (f) NaOH concentration and temperature on cotton dyed with Vat Green XBN dye (1% owf) using alkaline pectinase along with iron (II) salt.
All four factors showed significant influence on K/S values. The results were analyzed using both surface plots and the regression method. Regression equations in terms of coded values for run number 8 in Table XI showed the maximum K/S value. These results were compared with the results of the regression equation to obtain the optimized parameters for cotton dyed with this alkaline pectinase system.
Regression Data Analysis
Data was analyzed using statistical tools to obtain a regression equation for the response variable dye strength (K/S value). The regression equation for K/S values (R 2 = 0.9526 and F-value = 20.1) is given in Eq. 2.
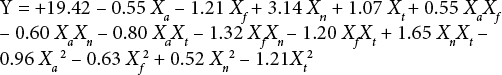
The model adequacy was checked by an F-test and determination coefficient R
2
. The regression model showed that this regression model was significant with an F-value of 20.1 for K/S values implying the model's significance, as the F-value was less than that of the calculated F-value. There was only a 0.01% chance that a model F-value this large could occur due to noise.
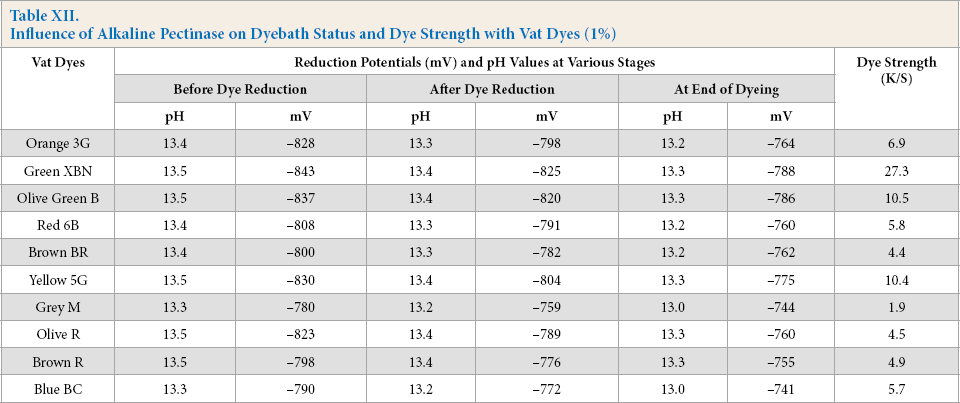
In the next stage, to check efficiency of these optimized parameters using Vat Green XBN, dyebaths were prepared with ten different vat dyes (1% owf), alkaline pectinase 1%, (owd), FeSO4 (3% owd), NaOH (20% owd), temperature of vatting and dyeing at 80 °C, and a dyeing time of 1 h. The dyebath status and corresponding K/S values on dyed cotton are shown in Table XII.
Influence of Alkaline Pectinase on Dyebath Status and Dye Strength with Vat Dyes (1%)
The reduction potential was adequate to reduce vat dyes completely for Gold Orange 3G, Green XBN, Olive Green B, Yellow 5G, and Olive R at 1% owf dye concentrations. The K/S values using these dyes gave equivalent or better results than those obtained using the hydrosulfite system. The remaining dyes—Red 6B, Brown BR, Grey M, Brown R. and Blue BC—showed lower K/S values compared to those from the hydrosulfite system (Fig. 2). K/S values on cotton for these dyes were around 33% less, but it was not an indication of low dyebath reduction potential (Table XII)—no dye reduction would have been observed. It is difficult to explain why K/S values were reduced for some dyes despite having adequate reduction potentials. This technology requires further study. Hence for these dyes, the dye concentration was increased from 1% to 1.5%. The resulting K/S values for these dyes is shown in Fig. 3. The K/S values of these dyes gave equivalent or better results than that for cotton dyed with vat dyes using the hydrosulfite system, except for Brown BR and Brown R.
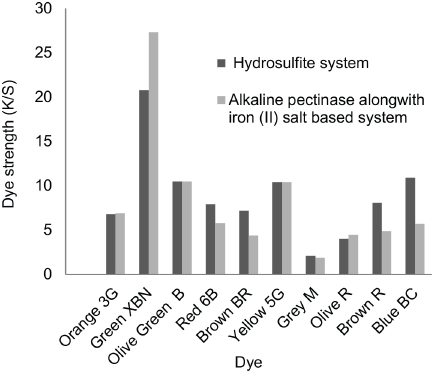
Comparison of K/S values on cotton in hydrosulfite and alkaline pectinase-based reduction baths for 1% shade.
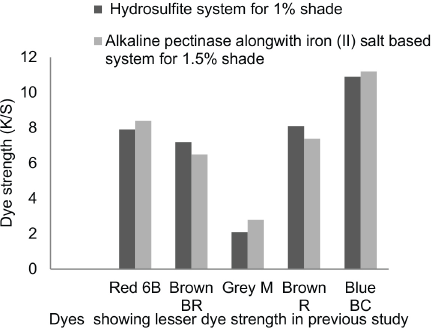
Comparison of K/S values on cotton in hydrosulfite system for 1% shade and in alkaline pectinase-based reduction baths for 1.5% shade of selected dyes.
Dyebath Stability
Without Dye
The stability of reduction baths prepared with hydrosulfite, and alkaline pectinase and iron(II) salt, were studied separately in the absence of dye. As stated earlier, the reduction baths were prepared for both systems and were stored for specific times. The change in pH and reduction potential were recorded. Vat Green XBN dye (1% owf) was added, the dyebath heated up to 80 °C, and dyeing carried out for 1 h.
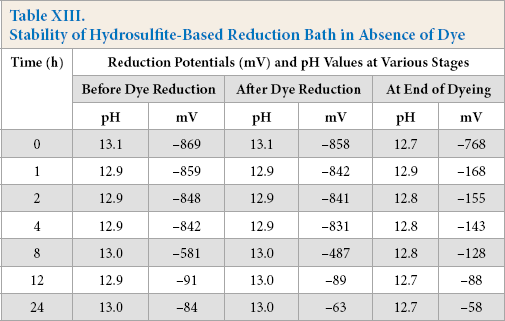
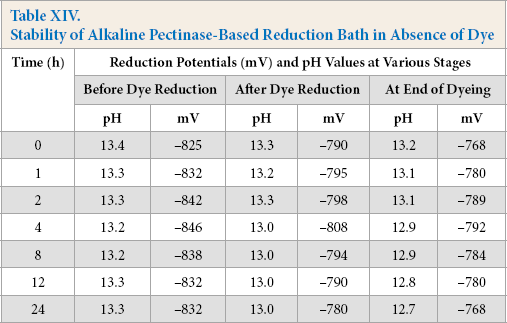
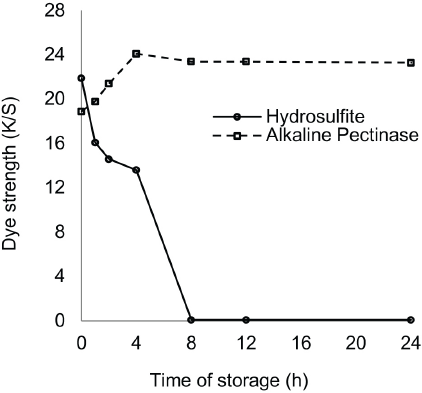
Hydrosulfite-based reduction baths showed decreases in reduction potential over time and maintained their reducing capability for 4 h. In contrast, the alkaline pectinase-based reduction baths retained their reducing capability successfully throughout 24 h of storage in absence of dye, because of the non-oxidizing nature of pectinase (Tables XIII and XIV). For the hydrosulfite-based baths, the reduction potential was maximal at the time of bath preparation (i.e., 0 h), but thereafter, showed a gradual decrease with increased storage time. The reduction potential values of baths remained quite effective for dye reduction up to a storage time of 1 h, though the reduced status could not be maintained until the end of dyeing. A storage time of 1 h was the limit for complete dye reduction and maintaining reduced status until the end of dyeing as shown by K/S values (Fig. 4). However, alkaline pectinase-based baths showed stability for up to 4 h, and then gradually decreased until 24 h, although the pH remained as high as 12.5-13.5 throughout. K/S values were also at their maximum after a storage time of 4 h (Fig. 4).
Stability of Hydrosulfite-Based Reduction Bath in Absence of Dye
Stability of Alkaline Pectinase-Based Reduction Bath in Absence of Dye
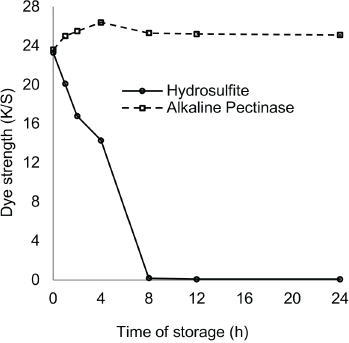
Stability of dyebaths in absence of dye.
With Dye
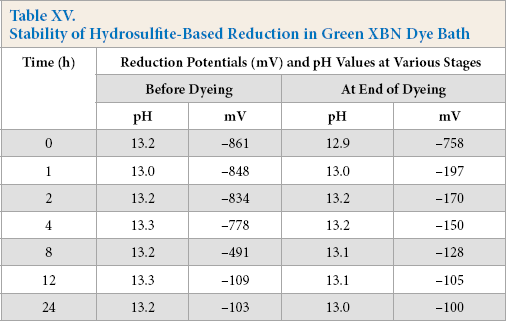
Reduction baths were prepared for both hydrosulfite and alkaline pectinase systems; dye was added and stored up to 24 h. The hydrosulfite system dyebaths showed the required reduction potential throughout dyeing up to a storage time of 4 h; beyond which, the starting reduction potential remained adequate enough, but the end potential (after dyeing) decreased progressively (Table XV), indicating the gradual decrease in reduced dye stability towards the end of dyeing. In contrast, alkaline pectinase-based reduction baths showed better long term dye stability. The baths showed a good potential up to a storage time of 24 h (Table XVI) with comparable dye strength. The comparison of K/S values on dyed cotton from all stored baths can be seen in Fig. 5.
Stability of Hydrosulfite-Based Reduction in Green XBN Dye Bath
Stability of Alkaline Pectinase-Based Reduction in Green XBN Dye Bath
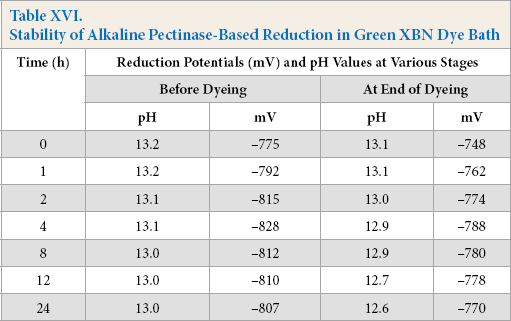
Stability of dye baths in presence of dye.
Fastness Performance
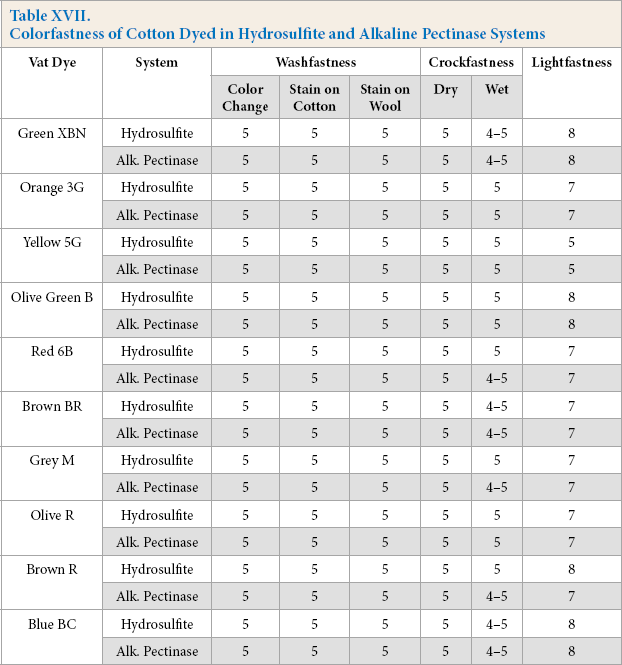
Colorfastness of cotton dyed in hydrosulfite and alkaline pectinase systems were evaluated. The results are shown in Table XVII. The lightfastness of dyed cotton remained very good to excellent and almost the same in both systems for the ten dyes under study, except for Yellow 5G dye. Washfastness, and dry and wet crockfastness, results gave identical, excellent fastness properties for both hydrosulfite and alkaline pectinase systems, except for Brown R dye. Overall, the colorfastness of cotton dyed using the alkaline pectinase system showed comparable performance with those dyed with the hydrosulfite system.
Colorfastness of Cotton Dyed in Hydrosulfite and Alkaline Pectinase Systems
Conclusion
Application of alkaline pectinase in combination with FeSO4 resulted in complete reduction and solubilization of vat dyes. The dyebath status in terms of reduction potential and dye strength (K/S values) of cotton was found to be similar, with little variation, to those obtained using the hydrosulfite system. The stability of the pectinase/FeSO4 reduction baths in the absence and presence of dye showed excellent stability for 24 h. The colorfastness of the dyed cotton fabrics were also identical with those obtained from the hydrosulfite system, except for Brown R dye. Sodium hydrosulfite used for vat dye reduction can be successfully substituted with alkaline pectinase/FeSO4, thus making the vat dyeing process eco-friendly. However, in spite of the high dyebath reduction potential, complete dye reduction and uptake did not occur for few dyes, for which the dye concentration was increased to 1.5% to give comparable K/S values to those found for the other test dyes. The reasons behind this require further investigation.