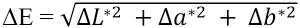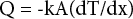Abstract
A green reduction processes for graphene oxide using carob extract is reported in this work. In this study, graphene oxide (GO) nanosheets were synthesized using the improved Hummer's method and applied to polyamide fabric thorough the simple dip coating method. Then, the graphene oxide was reduced with a chemical reduction process using carob extract as a green reducing agent to give the reduced graphene oxide (RGO) material. The reduction time was studied. The structure, morphology, and thermal behavior of the material was characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), and thermogravimetric analysis (TGA), respectively. The electrical resistivity results clearly revealed that the GO coated polyamide fabric was successfully converted to the RGO coated polyamide fabric with the effective elimination of oxygen containing functional groups.
Introduction
Functional textile materials have recently attracted attention for their electrical conductivity, antibacterial, fire retardant, superhydrophobic, and ultraviolet blocking properties.1,2 Electrically conductive textiles are used in various application fields such as smart and e-textiles, chemical sensing, wearable electronics, energy conversion and storage, 3 healthcare, 4 , bio-monitoring, 5 strain sensors, 6 and electromagnetic shielding.7,8 Several strategies have been developed to fabricate conductive textiles.9,10 For example, use of metallic fibers, 11 extrusion of fibers with conductive particles such as carbon nanotubes or carbon black, 12 and in-situ synthesis of conductive polymers on the fabric surface.9,13 However, these techniques require complex processes, expensive materials, and pre-functionalization, and have some disadvantages such as lacking uniform coating, and flexibility and durable wear resistance, which increases cost of production. 11 One possible application is the coating of textiles with the nanoparticles, which can be dispersed well in coating solutions.10,14,15
The use of graphene and its derivatives has opened a new era in the field of physics and materials science. 16 Graphene is an attractive material for textile substrates due to its unique and multifunctional properties such as compatibility, high transparency, light weight, high surface area, high mechanical properties, and excellent thermal and electrical conductivities. 17
Graphene is a single atomic layer of sp2 carbon atoms and has excellent electronic, mechanical, optical, and thermal properties.18–20 Graphene sheets exist in nature and we can exfoliate them from their precursors. 20 The exfoliation of graphite to graphene can be realized either physically or chemically. 21 The chemical route is comprised of two stages: the oxidation of natural graphite fakes, and the reduction reaction to produce reduced graphene oxide. The chemical oxidation of graphite offers a simple way to get exfoliated monoatomic thick graphene oxide (GO) nanosheets. 22 Due to the formation of oxygenated functional groups such as –OH and –COOH on graphite oxide, it exhibits good hydrophilicity and hence excellent dispersion in water, unlike graphene and graphite fakes. Therefore, it becomes much easier to exfoliate into monolayers of GO by simple stirring or mild sonication.23,24 However, GO is electrically insulating, so it has to be chemically reduced afterwards to produce reduced graphene oxide (RGO).11,16 The introduced oxygen functional groups can be easily removed by reduction mechanisms converting GO to reduced graphene oxide (RGO), with improvement in mechanical strength and electrical conductivity. 23 RGO is electrically conducting due to the restored sp2 bond networks. 11 Chemical reduction of GO to obtain RGO is unique and attractive because of its inexpensive large-scale production capacity.20,22,25
There is a challenge of producing high-quality graphene in large quantities using low cost and environmentally-friendly methods. 26 The removal of the oxygen moieties is a vital topic that determines the properties of the ultimate product. Therefore, finding a suitable reducing agent is very important. Various reducing agents have been applied to convert GO to RGO. 27 One of the negative aspects of the chemical reduction method is the highly toxic nature of the reducing agents (e.g., hydrazine, dimethyl hydrazine, sodium borohydride, and hydroquinone). The existence of trace amounts of such toxic agents could have a harmful effect, particularly in bio-related applications. 28
As an alternative, a number of green reductants have been introduced within the last decade. They include organic acids, plant extracts, microorganisms, sugars, antioxidants, amino acids, and proteins. Many reports on plant extracts have been published, including leaves, fruits, and flowers. 27 Mushroom extracts,29,30 carrot juice, 31 coconut water, 32 rose water, 33 green tea, 34 eucalyptus leaf extract,35,36 cinnamon, 37 grape extract, 38 miswak extract, 39 hibiscus, 40 and spinach 41 are some examples. Plant extracts have been used as the reducing agents for GO, as they have large organic molecules, such as polyphenolic compounds, that are readily oxidizable, so they can interact with the GO sheets to give RGO. 42
The carob tree (Ceratonia siliqua L.) has been widely grown in the Mediterranean region for centuries and is also widespread on almost all continents (Europe, Africa, Australia, Asia, North America). The main components of the carob tree are the pods and the seeds. The chemical composition of carobs is well-known—carob pods contain high amounts of carbohydrates, polyphenolic and antioxidant compounds, insoluble dietary fibers and minerals, and low amounts of proteins and lipids.43–45 Previous studies elucidate the antioxidant property of carob pods in detail.44–47 Carob leaf extract as a reducing agent for the synthesis of silver nanoparticles, 48 magnetic iron oxide nanoparticles, 49 magnetite nanoparticles, 50 cerium oxide nanoparticles, 51 and copper oxide nanoparticles 52 were reported. Because of the antioxidant properties of carob, it may act as a good reducing agent for the reduction of GO. Therefore, the aim of the present study is to investigate the reduction of GO-coated polyamide fabric using carob powder.
In this study, GO was deposited on the surface of the poly-amide (PA)6,6 fabric through a dip coating method. Then, the GO-coated PA6,6 fabric was reduced using a green reductant for conversion into the electrically conductive RGO fabric. PA is one of the most widely used polymers in the textile industry and PA6,6 is a popular commercial fiber. PA6,6 has a high modulus and strength, abrasion resistance, and dimensional stability, as well as a relatively low cost and easy recycling. These specifications are important for technical textile applications. 53 Besides the above characteristics, PA6,6 was chosen because of the lack of literature about coating with GO. GO is highly dispersible in aqueous media due to the presence of various oxygen containing functional groups and presents a negative charge in aqueous solutions. 54 The hydrophilicity of GO allows it to be readily soluble in water for adsorption, resulting in the strong adherence onto the surface of PA fabric. 55 PA contains amide groups in its structure and also contains free amine groups at the end of its polymeric chain. 54 GO functional groups, such as carboxyl, carbonyl, hydroxyl, and epoxy groups, can react with amine and carboxyl groups of PA6,6 fibers in the fabric. 56 The reduction of GO in a colloidal state results in an increase in hydrophobicity caused by a decrease in polar functionality on the surface of the sheets. 57 The direct applicability of RGO is limited because of the elimination of oxygen functionalities and increased hydrophobicity. Therefore, it is more convenient to apply GO in the form of an aqueous dispersion to the fabric surface.
Experimental
Materials
All chemicals were of analytical reagent grade and used without further purification. Graphite fakes were purchased from Sigma Aldrich. Hydrogen peroxide (H2O2, 35%), sulfuric acid (H2SO4, 95–98%), phosphoric acid (H3PO4), potassium permanganate (KMnO4), and hydrochloric acid (HCl, 37%) were purchased from Merck. Carob powder was purchased from a local market. Distilled water was used throughout the experiments.
Procedures
Synthesis of GO
Graphene oxide was synthesized from fake graphite by the improved Hummer's method. 19 Briefly, a 9:1 mixture of concentrated H2SO4/H3PO4 (400 mL) was added to a mixture of graphite fakes (3 g), and KMnO4 (18 g). The reaction mixture was heated to 50 °C and stirred for 12 h. The reaction mixture was then cooled to room temperature (RT) and poured onto ice (400 mL) containing 30% H2O2 (6 mL). The resulting suspension was washed by repeated centrifugation (each at 8000 rpm for 30 min), first with 400 mL of 1 M HCl and 200 mL of ethanol (2×), and then with distilled water until a pH of 4–5 was achieved. The obtained solid product was dried overnight in an oven at 60 °C.
Coating of PA fabric with GO
PA6,6 warp knitted fabric, kindly supplied by Kadifeteks, Istanbul, Turkey (172 g/m2, 30 x 30 cm), was treated in a bath with a liquor to goods ratio (LR) of 15:1 containing Perlavin OSV nonionic washing agent (Textilchemie Dr. Petry GMBH, Germany) for about 30 min at 60 °C to remove impurities. The fabric was then washed with excess water and dried at RT.
The PA6,6 fabric was treated with the cationic surfactant cetyltrimethyl ammonium bromide (CTAB, Sigma Aldrich) to charge the fabric surface positively. The treatment was applied using 5 g/L of CTAB at a 50:1 LR in a total volume of 750 mL at 90 °C for 1 h. The fabric was then rinsed with distilled water and dried at RT.
The fabric was then immersed into a 2 mg/mL aqueous dispersions of GO (20:1 LR, 300-mL total volume) and heated for 30 min at 60 °C. Because of the strong adsorption, the fabric was quickly coated by the GO. Then, the coated fabric was kept in an oven at 70 °C for 30 min. The coating process was repeated five times in order to increase the GO adsorption on the fabric. The obtained fabric was coded as GOF.
Reduction of GOF Fabric with Carob Extract
The extract was prepared using an ultrasonic bath extraction method. Carob powder (5 g) was added to 100 mL of distilled water and the mixture was bath sonicated for 1 h at RT. 58 Then, the mixture was centrifuged at 4000 rpm for 10 min and the supernatant was used for the chemical reduction process of GO.
To investigate the effect of reduction process on the electrical conductivity of the coated fabrics, three different process durations (6, 12, and 24 h) were used at a 50:1 liquor to goods ratio. The temperature of the reduction process was kept at 95 °C. 59 – 61 The resulting fabrics were rinsed with distilled water to remove the remaining reducing agent. The fabrics were dried in an oven at 90 °C. The obtained fabrics after 6, 12, and 24 h reduction process were coded as C6-RGOF, C12-RGOF and C24-RGOF, respectively.
Analytical Methods
The X-ray diffraction (XRD, Shimadzu LabX XRD 6100) spectra of the synthesized GO, RGO, and graphite fakes were performed at RT using the Cu Kα radiation source (λ = 0.1540 nm). Scanning electron microscopy (SEM, FEI Sirion) was used to observe the morphology of the coated samples. The surfaces of the samples were coated with gold at 1.5 kV for 100 s before analysis. An attenuated total reflection Fourier transform infrared spectrometer (ATR-FTIR, Perkin Elmer Spectrum Two) was used at 650-4000 cm–1 for infrared spectra. The thermal behavior of the samples was determined by thermogravimetric analysis (TGA, Perkin Elmer Termal Analyzer STA6000). TGA was carried out under a nitrogen flow at a heating rate of 10 °C/min from 30 to 600 °C.
Color coordinates were determined with a Datacolor SF600+ instrument, using a small area view (SAV) aperture and specular included (SI) mode, and the color differences were calculated in accordance with the CIELab system with D65/10° observer values. In the CIELab system, the lightness L* represents the darkest black at L* = 0 and the brightest white at L* = 100, a* represents redness (a* is +)/ greenness (a* is -), and b* represents yellowness (b* is +)/ blueness (b* is -). a* and b* represent true neutral gray values at a* = 0 and b* = 0. The color difference (ΔE) was determined using Eq. 1. 15
The surface electrical resistivity of the fabrics was measured using a standard four-point probe. The setup consists of a sourcemeter (Keithley 2450 Sourcemeter) and a four point probe station (Everbeing Intl. Corp.). Each sample was measured at least five times and the average value was calculated. The heat conduction apparatus (H940, PA Hilton Ltd.) was used to measure the thermal conductivity of the samples.
The thermal conductivity coefficient of samples was calculated according to Eq. 2. 62
Q is the heat flow (W), A is surface area (mβ), x is the thickness of sample (m), T is the temperature difference (K), and k is the thermal conductivity coefficient (W/m·K) of the sample. The bursting strength of the warp knitted fabrics was measured using an AutoBurst (SDL Atlas) instrument according to the ISO 13938-1 standard. 63
Results and Discussion
XRD Analysis
XRD spectra were used to characterize the structure of synthesized GO. As shown in Fig. 1, fake graphite shows a strong and sharp peak at 26.5° corresponding to an inter-layer spacing d = 0.34 nm. The XRD spectra of synthesized GO exhibits a sharp peak at 10.2°, which corresponds to an interlayer spacing d = 0.86 nm. The interlayer spacing of synthesized GO is much larger than that of graphite due to the introduction of hydroxyl, epoxy, carboxyl, and carbonyl groups on the basal planes and edges of GO sheets. 64 This result reveals the successful transformation from graphite to GO. After the reduction process of GO using carob extract as a green reductant, the (002) reflection peak of GO disappeared and a significant peak was observed at 25.8°, indicating the formation of RGO layers.
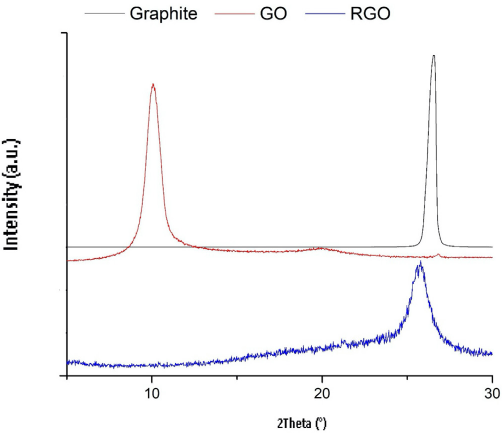
XRD patterns of graphite, synthesized GO, and reduced graphene oxide using carob extract.
SEM Analysis
Fig. 2a shows the SEM micrograph for the untreated PA fabric. The fabric had a very smooth surface. Fig. 2b shows the micrograph for the GO-coated PA fabric. After GO was coated on the fabric, the porous network of fabric was closed by GO sheets. The GO sheets coated on the fibers uniformly and closely, indicating that the GO sheets were well connected with the fibers. After the reduction process, Fig. 2c reveals the presence of RGO sheets on the surface of the PA6,6 fabric.

SEM images of samples (a, d) Polyamide fabric, (b, e) GOF, and (c, f) C-RGOF.
ATR-FTIR Analysis
Fig. 3 shows the ATR-FTIR spectra of the PA fabric, GO-coated PA fabric, and the reduced sample using carob powder extract. The ATR-FTIR spectra of PA6,6 fabric showed peaks at 1634 and 1532 cm–1, which respectively correspond to N–C=O and C–N–H vibrations.65 The presence of amide groups in PA6,6 was well reflected as an intense band at 3435 cm–1, related to the N-H stretching vibrations. The strong C-H stretching and bending vibrations of CH2 groups, vibrations of CH3 groups relative to PA terminal chains, and C-N stretching appeared at 2852, 2923, 1433, 1368, and 1000–1300 cm–1, respectively. 53
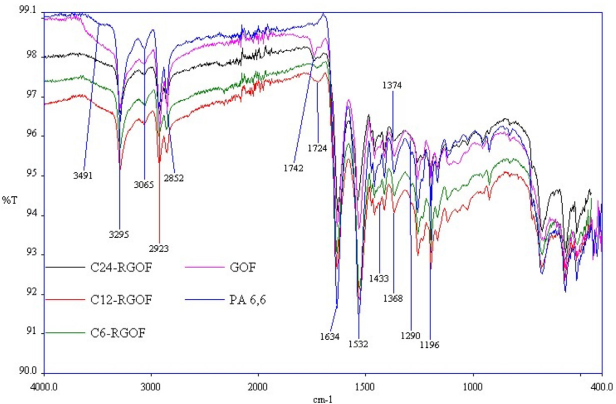
ATR-FTIR spectra of the samples.
After coating GO on PA6,6, the spectrum of the GOF sample showed new peaks at around 1742 and 1709 cm–1 attributed to the carbon skeleton vibration of graphene. The characteristic C=O stretching vibration peak of GO appeared at 1742 cm–1. Another indication was the widening of hydroxyl (OH) band (3600-3200 cm–1) in the GO-coated PA6,6 fabric as the GO is oxidized and is terminated with hydroxyl groups.12,66 All these significant changes suggested the attachment of GO on the PA6,6 fabric.
In comparison to the PA6,6 fabric, the ATR-FTIR spectrum of C24-RGO fabric had no substantial variation, but the intensity of the peaks at 1634 and 1536 cm–1 decreased, which suggested partial removal of the oxygen functionalities. The broad peak around 3400 cm–1 cannot be seen in the C24-RGOF sample, which indicates that the hydroxyl groups were removed. Also, the peaks occurred at 1742 and 1709 cm–1 after coating of GO disappeared after the reduction process using carob extract for 24 h.
TGA Analysis
The thermal stability of the PA6,6 fabric, GO coated fabric, and GO coated fabric reduced with carob extract was examined using thermogravimetric analysis (TGA) and is shown in Fig. 4. The TGA curves of all samples were similar and showed a weight loss around 100 °C due to evaporation of adsorbed water. The GOF and C24-RGOF samples showed a weight loss around 150–200 °C, mainly due to the decomposition of the oxygenated groups. The main weight loss of all samples occurred between 400 to 480 °C. This was attributed to the thermal decomposition of PA6,6, which caused a main chain breakdown with the formation of NH3, H2O, CO, CO2, and hydrocarbons. 67 The amounts of PA6,6 residual char increased with the reduction process using carob extract. The residuals at 600 °C of PA6,6 fabric, GOF, and C24-RGOF samples were 2.38%, 5.05%, and 6.60%, respectively. The residual char of the C24-RGOF sample at 600 °C was greater than that of the PA6,6 fabric.
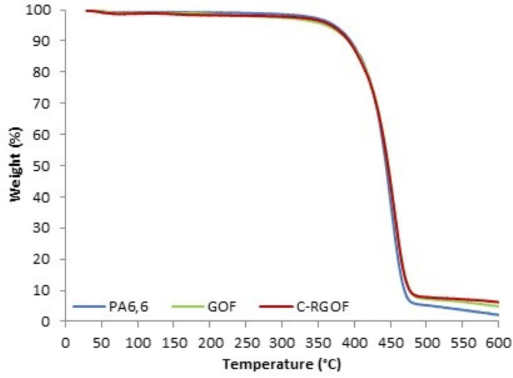
TGA curve of the samples.
Color Measurement
As a fast and easy way for determination of deposition and reduction of GO, the color of the fabric can be considered. The color changes of the PA6,6 fabric after GO coating and reduction processes were determined by reflectance spectra and color coordinates. The reflectance spectra of samples within a 360 to 700 nm range are given in Fig. 5. The reflectance spectra showed that the RGO-coated fabrics were close to each other in color. However, the lowest reflectance percentage was obtained with the increase in reduction time to 24 h.
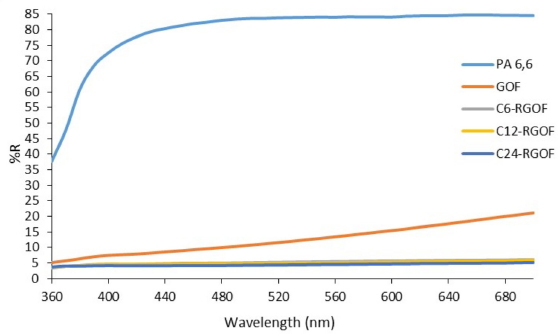
Reflectance spectra of the samples.
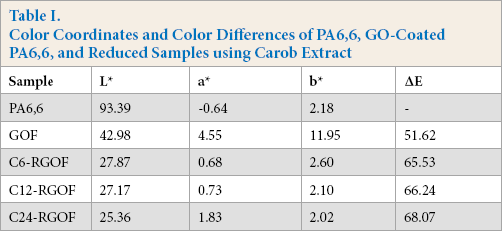
The color coordinates and the color differences of the samples are given in Table I. From the results, it can be seen that the reduction of GO-coated fabric using carob extract can change the color significantly and indicates the adsorption of reduced graphene oxide on the PA6,6 fabric. The L* value of the PA6,6 fabric decreased from 93.39 to 42.98 after GO coating, and with the reduction process at different durations, the lightness continued to decrease from 27.87 for C6-RGOF to 25.36 for C24-RGOF. There were also changes in a* and b* values of fabrics. With the GO coating on the PA6,6 fabric, the a* value became positive and showed a strong increase in the redness. Also, the significant increase in b* value indicates increased yellowness. The color of the PA6,6 fabric changed from white to yellow-brown after GO coating. However, when the reduction process takes place, the a* and b* values of the fabrics decreased dramatically. Additionally, the ΔE values showed the color differences of the GO-coated and RGO-coated PA6,6 fabrics compared to the untreated PA6,6 fabric. The ΔE value of the GOF sample showed a high degree of color difference from the untreated PA6,6 fabric. Also, the ΔE value of the RGOF samples increased with increased reduction time. The greatest color difference was obtained with the sample reduced with carob extract for 24 h. The obtained data from the reflectance spectra and the color coordinates showed the successful and effective deposition and reduction of the GO on the PA6,6 fabric.
Color Coordinates and Color Differences of PA6,6, GO-Coated PA6,6, and Reduced Samples using Carob Extract
Electronic Properties
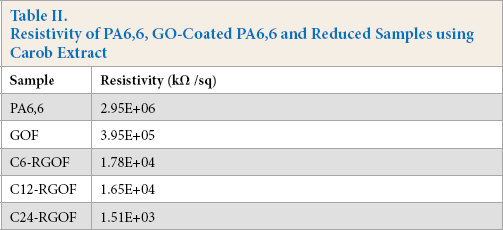
The electrical resistivity of PA6,6 fabric, GO-coated fabric, and RGO fabrics prepared using carob extract were measured and given in Table II. The PA6,6 fabric exhibited extremely high resistivity (106 kΩ/sq). The resistivity decreased after the GO coating and the average value of 3.95 × 105 kΩ/sq for the GOF sample was obtained. The resistivity decreased further after reduction processes using carob extract with different durations. The resistivity decreased to 104 kΩ/sq when the GO-coated PA6,6 fabric reduced with carob extract for 6 h. Increasing the reduction time from 6 h to 24 h, the resistivity of C24-RGOF sample was measured to be 1.51 × 103 kΩ/sq. The increase in reduction process duration can enhance the electrical conductivity of PA6,6 fabric.
Resistivity of PA6,6, GO-Coated PA6,6 and Reduced Samples using Carob Extract
Termal Conductivity
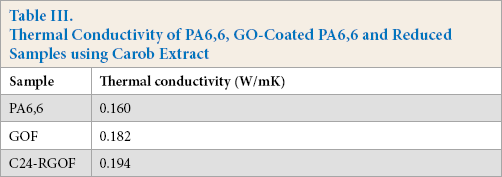
The thermal conductivity results are given in Table III. The thermal conductivity increased after each process. The thermal conductivity of GO-coated fabric showed an increase when compared with the untreated PA6,6 fabric. There was a 13.75% enhancement in thermal conductivity for the GO-coated PA6,6 fabric. For the RGO-coated fabric, the thermal conductivity increased to 0.194 W/mK with a 21.25% enhancement when compared to the PA6,6 fabric.
Termal Conductivity of PA6,6, GO-Coated PA6,6 and Reduced Samples using Carob Extract
Bursting Strength
The bursting strength of PA6,6, GO-coated PA6,6, and the reduced samples using carob extract were tested and average results are given in Fig. 6. The bursting strength of PA6,6 warp knitted fabric was 508.1 kPa, while the bursting strengths of GO-coated and RGO- coated fabrics were 503.3 and 517.3 kPa, respectively. Therefore, the GO coating and reduction process had no negative effects on the mechanical properties of fabrics.
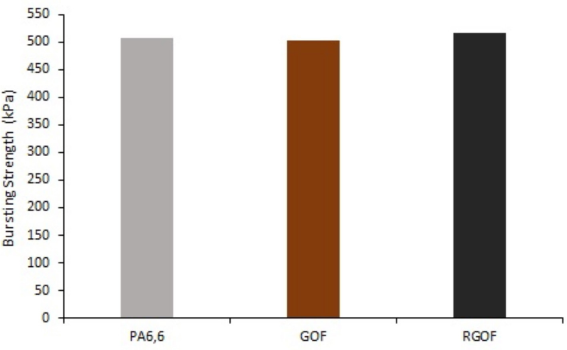
Bursting strength of the samples.
Conclusions
In this study, the reduction of graphene oxide (GO)-coated polyamide (PA)6,6 warp knitted fabric using carob extract as a green reductant was investigated. This investigation demonstrated that carob powder extract could act as the green reductant to efficiently reduce GO into reduced graphene oxide (RGO). The XRD characterization showed the successful conversion of graphite to GO and GO to RGO. In addition, the SEM and ATR-FTIR characterizations conducted on GO- and RGO-coated fabric samples showed deposition of GO- and RGO-sheets on the PA fabric. The color of the PA6,6 fabric changed from white to yellow-brown after GO coating and finally turned brown-black after the reduction with carob powder extract.
One of the most important parameters that characterizes RGO is the electrical conductivity. The reduction of GO-coated PA6,6 fabric using carob powder extract at different durations has affected the electrical conductivity in a positive way. The electrical resistivity mainly depended on the reduction time and decreased from 106 to 103 kΩ/sq. The GO coating of PA fabric and reduction with carob extract of the GO-coated PA fabric changed the mechanical properties only slightly. The main advantages of carob powder extract against the commonly-used reducing agents such as hydrazine hydrate is its cost-effectiveness and presence in large amounts in nature.1,3,11–13,17,54,56 The process is environmentally friendly and can be used for cost-effective mass production.
Future work such as the optimization of the concentration of carob extract and possible functional applications of carob RGO-coated PA fabric are undergoing.
Footnotes
Acknowledgement
This work was supported by the Research Fund of the Marmara University. Project number FEN-C-DRP-141118-0592.