Abstract
The new magnetic and heat-resistant polyamide ether (PAE)/graphene oxide (GO) nanocomposites were prepared by blending PAE with modified GO. Furthermore, a novel PAE containing flexible ether, methyl, and triphenylmethane moiety in the main chain of PAE was prepared by direct polycondensation reaction of a diamine and 4,4′-(butane-1,4-diylbis(oxy))dibenzoic acid (
Introduction
Over the last years, composite materials consisting of polymeric matrix have been a topic of scientific research and industrial development. The nanocomposite materials are defined as particle-filled polymers in which at least one dimension of the dispersed particles is in the nanometer range.1–3 Polymeric nanocomposites are commonly used due to having different properties, such as thermal, mechanical, and barrier properties and flame retardancy.4–6 Specifically, aromatic polyamides exhibit excellent thermal stability and good mechanical properties, which makes them useful for their study as high-performance polymers.7,8 One of the major problems with high-temperature polymers is their poor process-ability caused by low solubility in organic solvents. The general procedure for improving the solubility and process-ability of aromatic polyamides includes the incorporation of aliphatic and heteroaromatic monomers into the polymeric backbone.9–15 Polymer-based magnetic nanocomposites have received increasing attention due to their potential applications in a wide range of industrial fields, such as cell separation and magnetic resonance imaging, magneto-optical solid devices, RNA and DNA purification, and targeted drug delivery.16–21 Carbon nanostructures have received considerable attention because of their excellent properties.22–24 Single-layer graphene was produced by mechanical exfoliation of bulk graphite 25 and by chemical vapor deposition. 26 These ways, though preferred for accurate device assembly, are less effective for bulk-scale manufacturing. 27 The most common approach to graphite exfoliation is the use of chemical oxidants as a driving force for exfoliation.28,29 Thus, incorporation of graphene to a polymer matrix has achieved a number of improved properties with potential applications in many areas, such as biosensor, antifouling coatings, supercapacitor, and medicine.30–34 In addition, loading of various functional groups into the surface of the graphene oxide (GO) could be useful to obtain better compatibility between the GO and polymer.35,36 Among the chemical groups, the amine group has high reactivity and can react with many chemicals including polymers. 37 Many researchers functionalized the GO with amine groups and found that amine-modified graphene possessed adsorption performance and excellent catalytic activity.38,39 One of the main objects of this research was to prepare a new type of heat-resistant and magnetic nanocomposite containing GO-TEPA@Fe3O4 nanoparticles in the polymer matrix. Since the addition of inorganic particles into polymers leads to improved thermal properties, we used graphene, which is the equivalent of inorganic materials in terms of increasing heat-resistance effect. Furthermore, to improve the dispersion stability of nanoparticles in the polymer, the modification of graphene surface was investigated. Interesting findings in improvement of thermal properties of the polyamide ether (PAE)/GO-TEPA@Fe3O4 nanocomposites are discussed.
Experimental
Materials
Triphenyl phosphite (TPP), 4-chlorobenzaldehyde, 2,5-dimethylphenol,
Measurements
Fourier transform infrared (FTIR) spectra were recorded on a Galaxy series FTIR 5000 spectrophotometer over the range of 400–4000 cm−1. Nuclear magnetic resonance (NMR) spectrum was recorded at room temperature using Bruker (Germany) Avance spectrometer operating at 300 MHz for proton nuclear magnetic resonance (1H NMR) and at 75 MHz for carbon-13 nuclear magnetic resonance (13C NMR) using deuterated dimethyl sulfoxide (DMSO-d6). Melting points were measured using the capillary tube method with an Electrothermal 9200 apparatus. X-ray diffraction (XRD) patterns were recorded on Philips Xpert MPD X-ray (Germany) at 40 mA (copper
Monomer synthesis
Synthesis of 4,4′-((4-chlorophenyl)methylene)bis(2,5-dimethylphenol) (
3
)
4-Chlorobenzaldehyde (0.98 g, 7 mmol), 2,5-dimethylphenol (2.13 g, 17.5 mmol), and
Synthesis of 5,5′-((4-chlorophenyl)methylene)bis(1,4-dimethyl-2-(4-nitrophenoxy)benzene) (
5
)
A mixture of 4,4′-((4-chlorophenyl)methylene) bis (2,5-dimethylphenol) (
Synthesis of 4,4′-((((4-chlorophenyl)methylene)bis(2,5-dimethyl-4,1-phenylene))bis(oxy))dianiline (
6
)
Diamine (
Synthesis of 4,4′-(butane-1,4-diylbis(oxy))dibenzoic acid (
9
)
The 4,4′-(butane-1,4-diylbis(oxy))dibenzoic acid was prepared according to the reported literature. 40
Polymer synthesis
Into a 100-mL round-bottom flask equipped with a magnetic stirrer, the equal mole of diamine (
Synthesis of nanostructures
Synthesis of GO nanosheets
GO was synthesized from graphite powder using a modified Hummer’s method. 41 Firstly, 1 g of graphite powder and 0.5 g of NaNO3 were mixed together followed by the addition of 23 mL of concentrated H2SO4. Under vigorous agitation, KMnO4 (3 g) was added gradually to keep the temperature of the suspension lower than 10°C. The mixture was stirred at 35°C for 2 h, and distilled water was added. Later, 30% H2O2 solution was added to stop the oxidation process. The GO formed was washed with distilled water and absolute ethanol, respectively, followed by filtration and drying, GO sheets were thus obtained.
Synthesis of tetraethylenepentamine-functionalized graphene oxide nano-sheets (GO-TEPA)
A suspension of GO (500 mg) in 10 mL of SOCl2 was heated to 70°C for 24 h under ultrasonic irradiation, and then, the solvent was evaporated under reduced pressure. In a 100-mL round-bottom flask equipped with a stirring bar, the obtained GO-Cl, 30 mL of THF and 400 mg of TEPA were added. The solution was stirred at 60°C for 2 h under ultrasonic irradiation again. After cooling to room temperature, the precipitate was filtered off and washed thoroughly with ethanol and dried at 60°C under vacuum.
Synthesis of magnetic tetraethylenepentamine-functionalized graphene oxide nanocomposite (GO-TEPA@Fe3O4)
GO-TEPA (100 mg) was dispersed in deionized water using ultrasonic to produce a homogeneous suspension. Then, 35 mg of FeCl2·4H2O and 50 mg of FeCl3·6H2O were dissolved in 50 mL GO-TEPA aqueous suspension. The mixture was heated under nitrogen (N2) at 90°C for 1 h. Meanwhile, 1 mL of concentrated ammonia (25%) was mixed quickly to produce a stock solution. The solution was stirred under N2 for another 1 h and then cooled to room temperature. Afterward, the precipitate was separated using a magnet, washed with ethanol several times, and dried under vacuum.
Preparation of PAE/GO-TEPA@Fe3O4 nanocomposites
PAE/GO-TEPA@Fe3O4 nanocomposite films (polyamide ether nanocomposite (PAEN) 2% and PAEN 5%) were synthesized via a solution mixing method as follows: Into a three-necked flask equipped with mechanical stirring and ultrasonic irradiation, PAE was dissolved in THF at 25°C. After dissolution completely, a calculated amount of GO-TEPA@Fe3O4 was added to the PAE homogenous solution. To control the dispersibility of GO-TEPA@Fe3O4 in the PAE matrix, constant stirring was applied at 25°C for overnight. To remove the THF solvent, the solutions were poured into petri dishes and uniformly heated. Then, PAE/GO-TEPA@Fe3O4 nanocomposite films were formed.
Results and discussion
Monomer synthesis
The new aromatic symmetrical diamine monomer
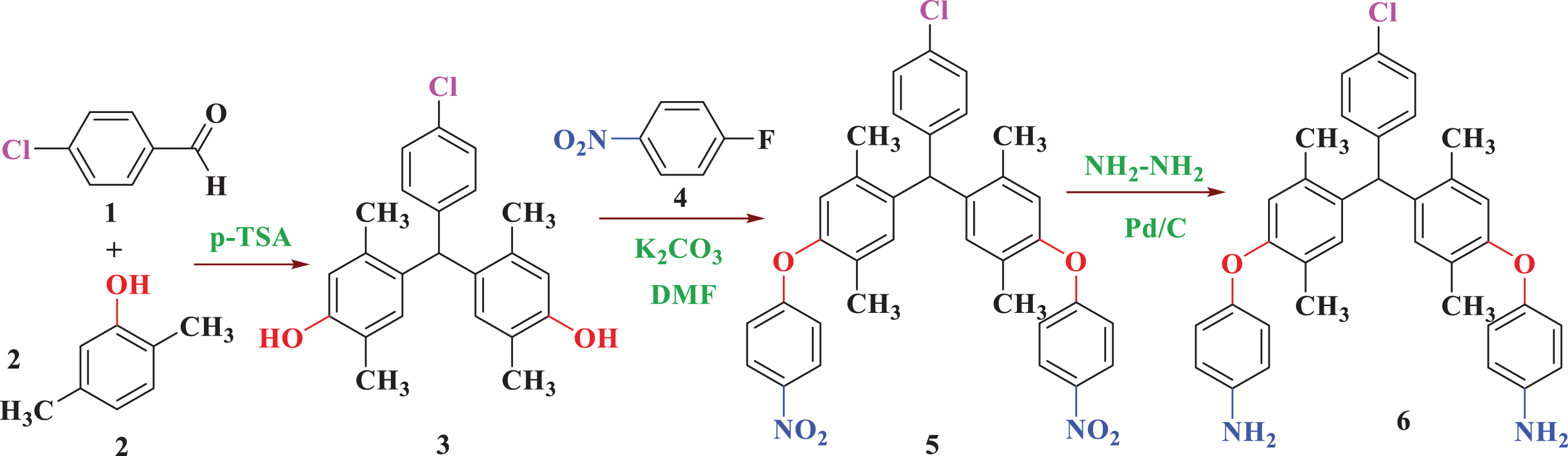
The synthetic pathway of 4,4′-((((4-chlorophenyl)methylene)bis(2,5-dimethyl-4,1-phenylene))bis(oxy))dianiline (
Figure 2 shows the FTIR spectra of diamine monomer
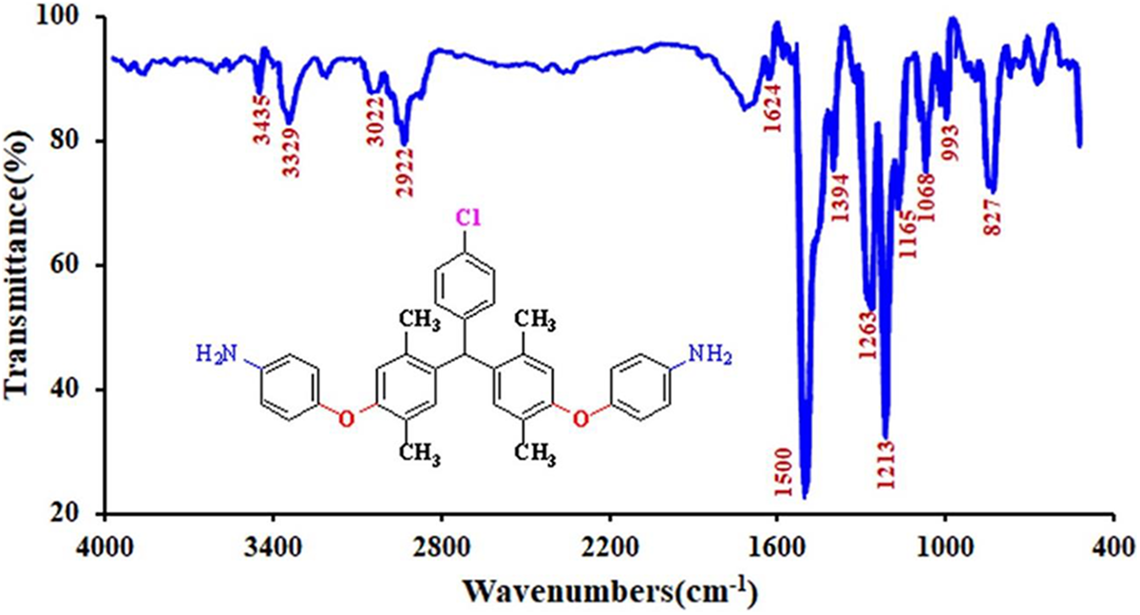
FTIR spectrum of 4,4′-((((4-chlorophenyl)methylene)bis(2,5-dimethyl-4,1-phenylene))bis(oxy))dianiline (
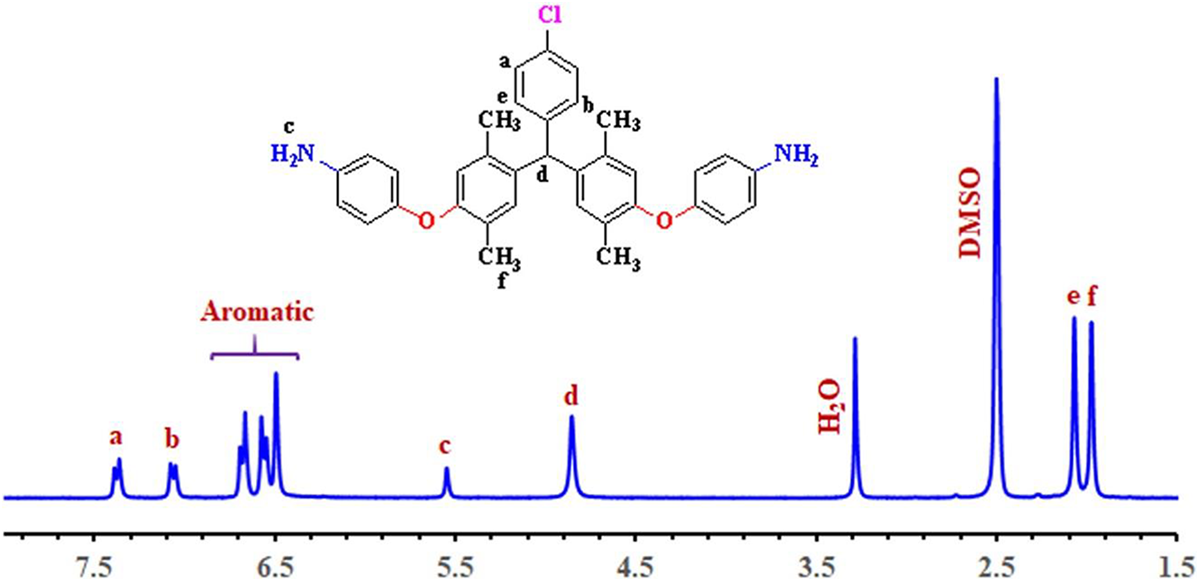
1H NMR spectrum of 4,4′-((((4-chlorophenyl)methylene)bis(2,5-dimethyl-4,1-phenylene))bis(oxy))dianiline (
As to the 13C NMR spectra shown in Figure 4, the resonance signals of all carbon atoms of diamine monomer (
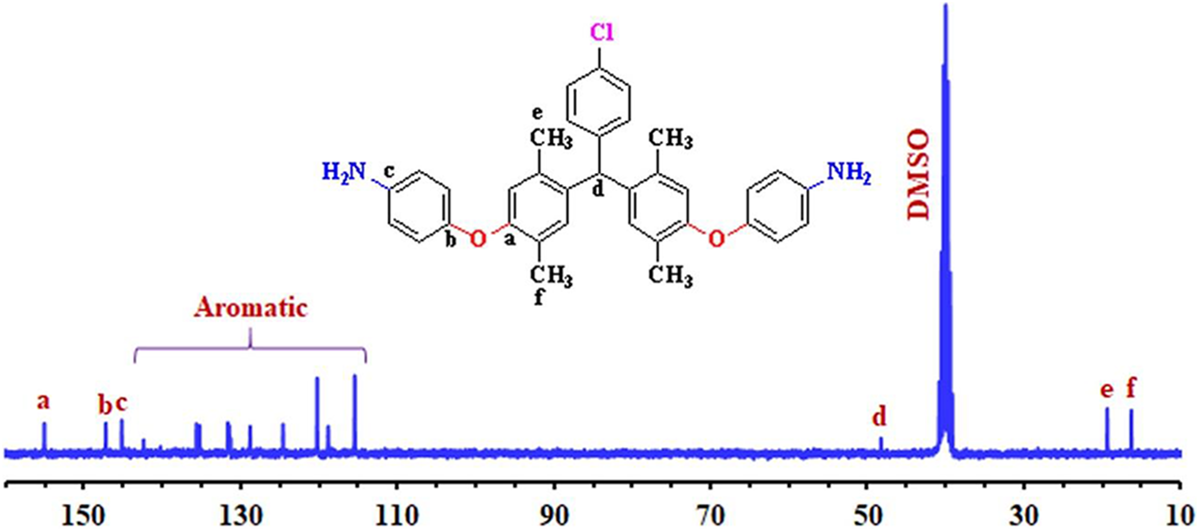
13C NMR spectra of 4,4′-((((4-chlorophenyl)methylene)bis(2,5-dimethyl-4,1-phenylene))bis(oxy))dianiline (
Polymer synthesis and characterization
General procedure for the preparation of PAE
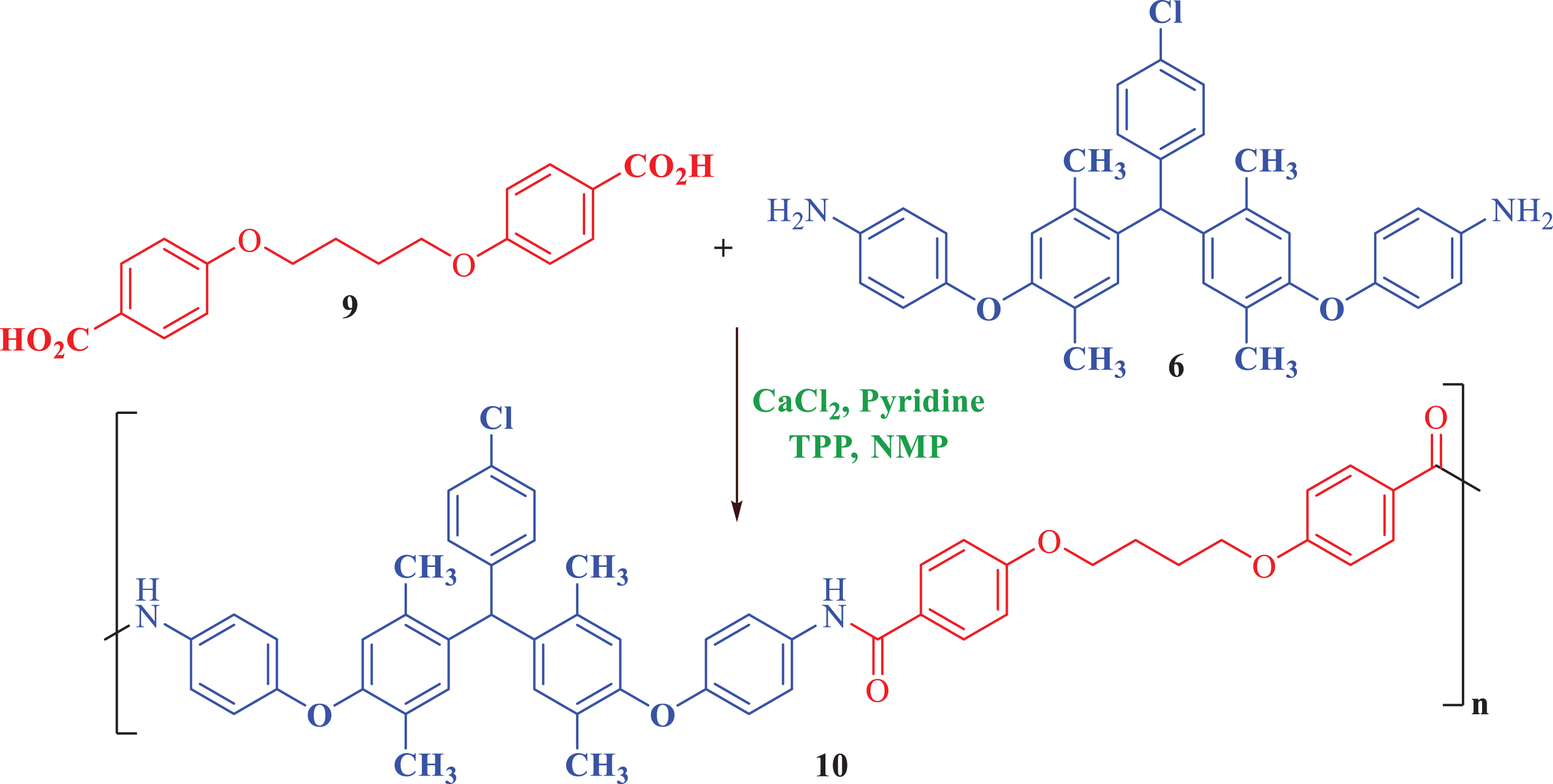
Synthesis route of PAE (
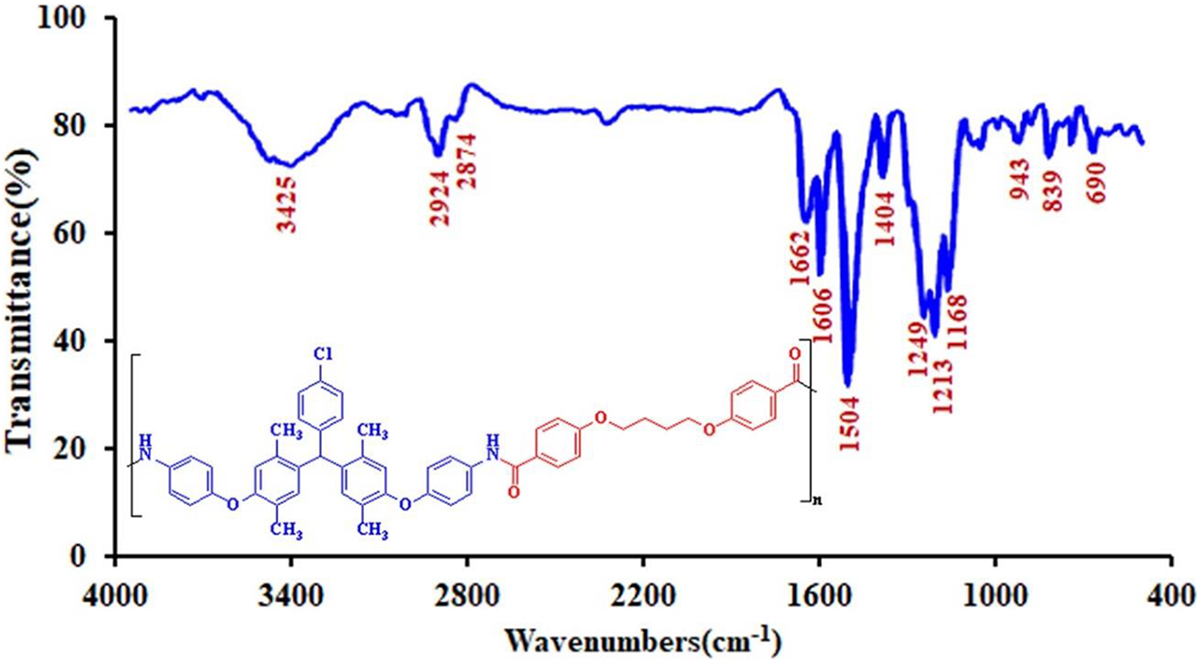
FTIR spectra of the PAE (
Figure 7 shows 1H NMR spectrum of PAE (
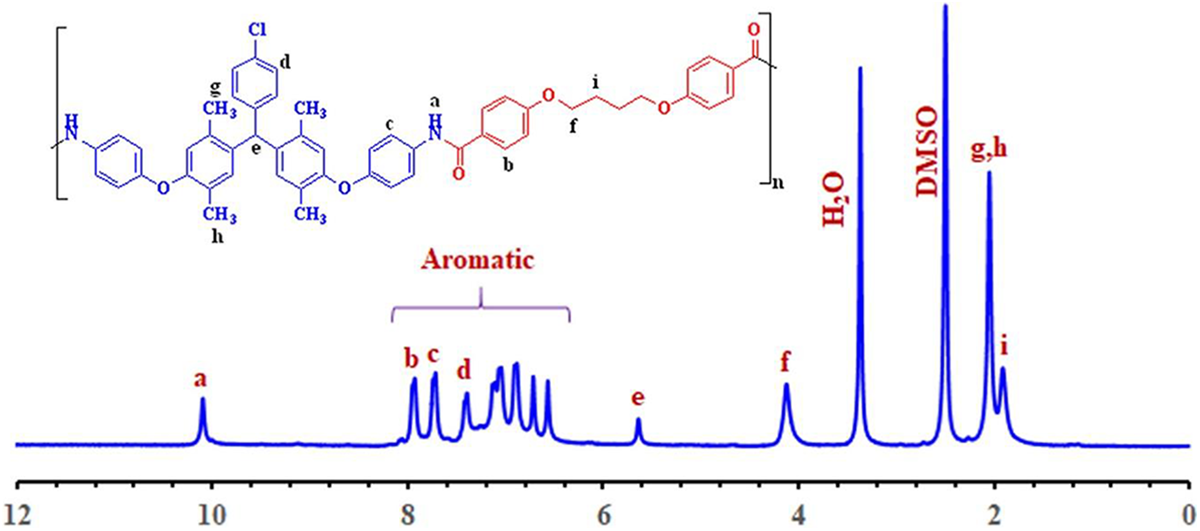
1H NMR spectra of the PAE (
The organosolubility behavior of the PAE (
Synthesis and characterization of nanostructures
The synthesis process of GO-TEPA@Fe3O4 nanocomposite was schematically illustrated in Figure 8. Synthesis of GO was achieved by placing graphite in concentrated acid (H2SO4) in the presence of an oxidizing agent (KMnO4). To improve its dispersion in the polymer matrix, GO was modified with TEPA. Then, the magnetic tetraethylenepentamine-functionalized graphene oxide (GO-TEPA@Fe3O4) prepared via homogeneous suspension of GO-TEPA in deionized water and then alkaline hydrolysis of the Fe3+ and Fe2+ ions.
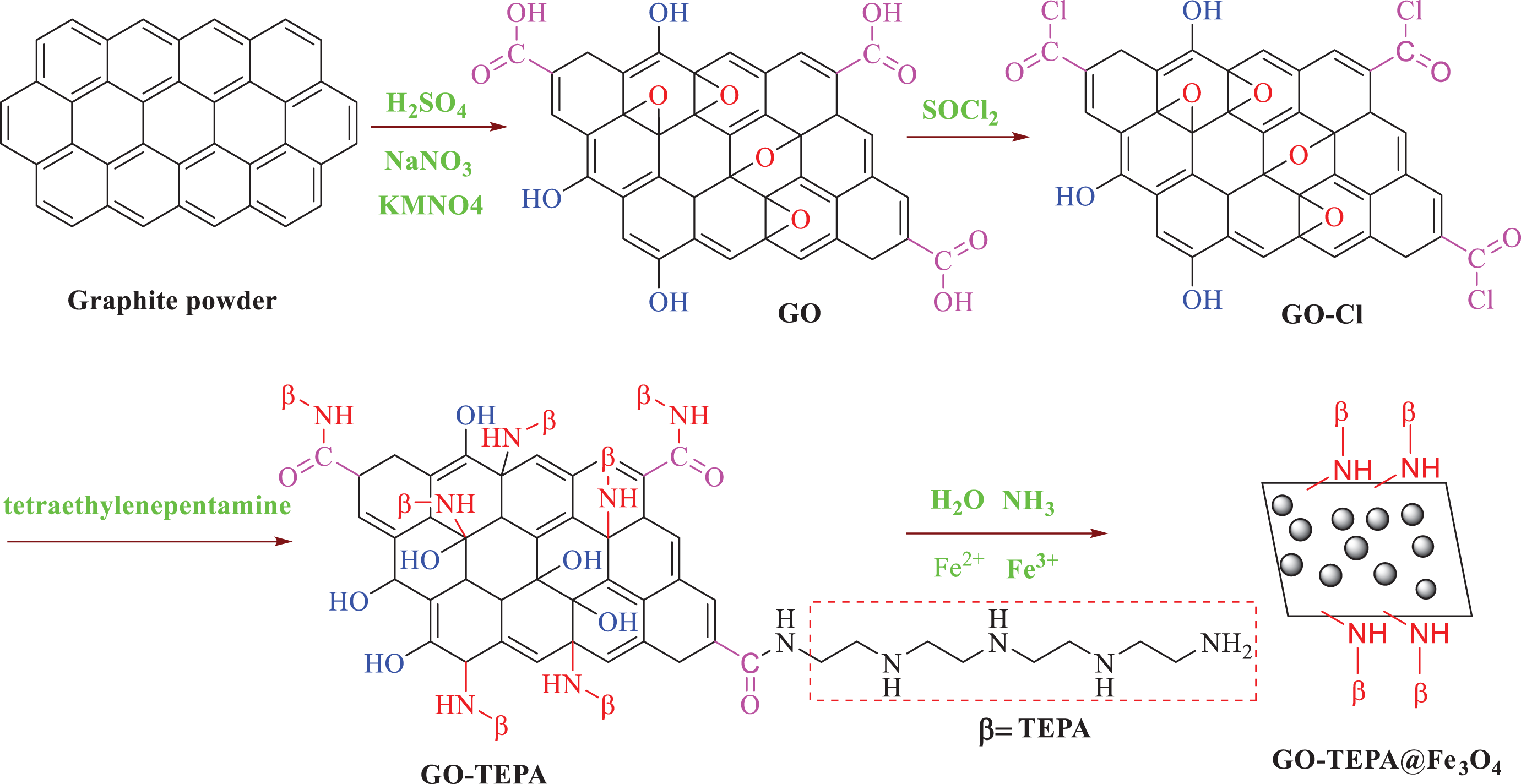
Preparation of GO-TEPA@Fe3O4 nanocomposite.
The characteristic FTIR spectrum of GO, GO-TEPA, and GO-TEPA@Fe3O4 is depicted in Figure 9. In all curves in Figure 9, the absorption bands at around 3400 and 3440 cm−1 can be attributed to the O–H group vibrations stretching of the GO. The absorption peak at 1739 cm−1 and the strong peak between 1028 cm−1 and 1107 cm−1 related to C=O and C–O stretching, respectively. Compared with GO, two absorption peaks at about 2852 and 2926 cm−1 are assigned to the C–H bonds stretching vibrations, which confirm the presence of TEPA on the surface of GO-TEPA nanosheets (Figure 9(b) and (c)), and GO-TEPA@Fe3O4 shows a characteristic absorption of Fe-O bond at about 592 cm−1, which confirms the presence of Fe3O4 nanoparticles (Figure 9(c)). Considering results, the surface modification of GO-TEPA@Fe3O4 nanocomposite was successful.
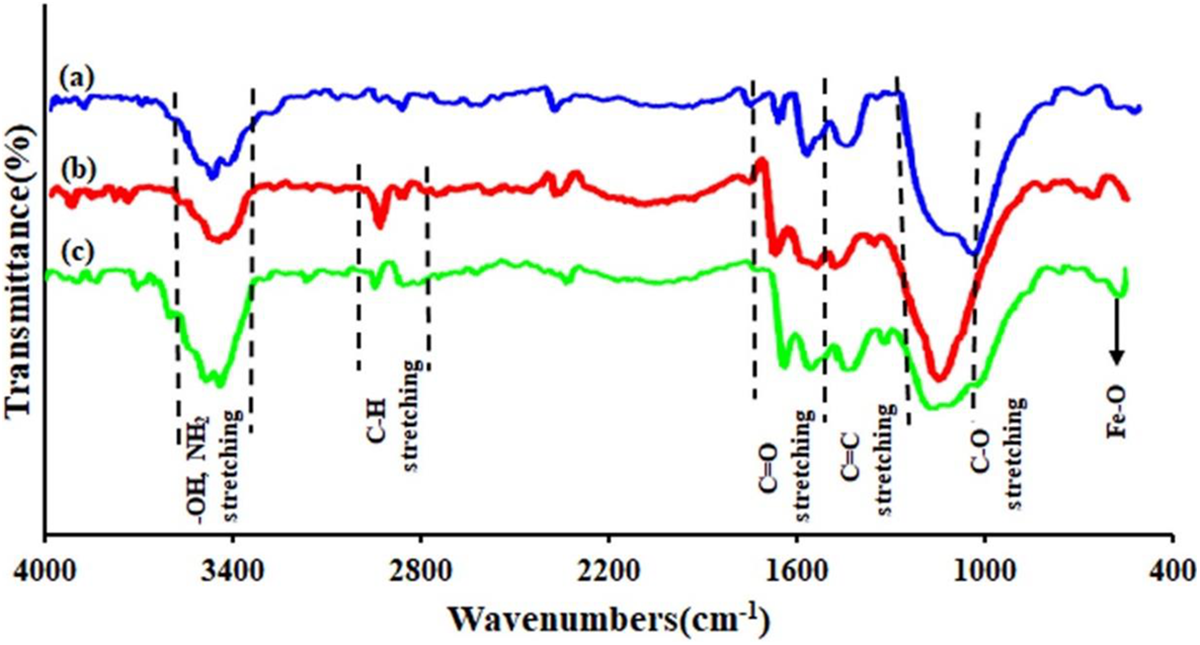
The comparative FTIR spectra for (a) GO, (b) GO-TEPA, and (c) GO-TEPA @Fe3O4 nanosheets.
The EDX spectrum of GO-TEPA@Fe3O4 (Figure 10) shows iron, nitrogen, carbon, and oxygen. The obvious attendance of Fe signal in the EDX spectrum demonstrates that GO-TEPA@Fe3O4 have been successfully functionalized by Fe3O4 nanoparticles.
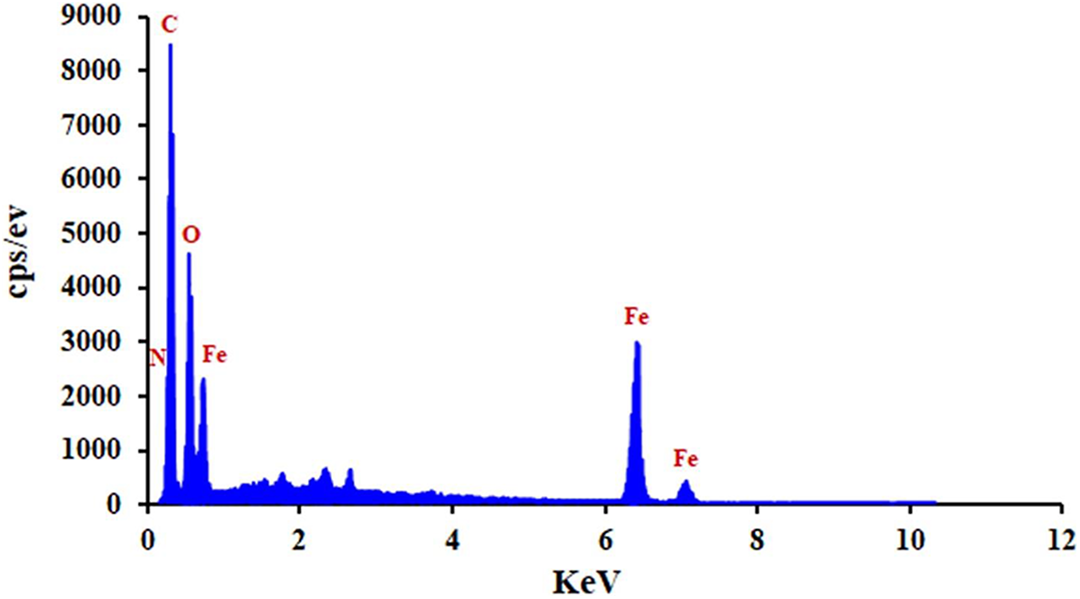
EDX spectrum of GO-TEPA@Fe3O4 nanocomposite.
The synthesized GO-TEPA@Fe3O4 nanocomposite was observed with SEM (Figure 11). FE-SEM images indicates that the Fe3O4 nanoparticles on the surface of modified GO (GO-TEPA) are approximately spherical and the average diameter of the nanoparticles is 40 nm. The results obtained for GO-TEPA@Fe3O4 nanocomposites express that the agglomeration of Fe3O4 nanoparticles has occurred. The agglomeration role in the interfacial properties of the matrix is much important because it reduces the interfacial area between matrix and nanoparticles, which lastly result in the poor mechanical properties of samples.

FE-SEM images of GO-TEPA@Fe3O4 nanocomposite.
The PAE/GO-TEPA@Fe3O4 5% and PAE/GO-TEPA@Fe3O4 2% nanocomposite films were prepared according to the Yamazaki method. 44 Then, the PAEN 5% and PAEN 2% were produced by solution intercalation method, in which different amounts of GO-TEPA@Fe3O4 were mixed with appropriate amounts of PAE solution to yield particular nanocomposite concentrations. The resultant nanocomposites (PAE/GO-TEPA@Fe3O4 5% and PAE/GO-TEPA@Fe3O4 2%) were characterized using FE-SEM, XRD, UV-Vis spectrometry, TGA, and VSM.
To investigate the quality of the dispersion of GO-TEPA@Fe3O4 nanocomposite in the polymer matrix and the morphology of the nanocomposites, FE-SEM images of the surface of the PAE/GO-TEPA@Fe3O4 5% nanocomposite have been utilized (Figure 12). From the SEM results, it can be seen that GO-TEPA@Fe3O4 have been well dispersed within the PAE matrix with very small aggregations. Also, spherical Fe3O4 nanoparticles are observed on the nanocomposite surface.

FE-SEM images of PAEN 5% nanocomposites.
The crystallinity of the GO-TEPA@Fe3O4, PAEN 2%, and PAEN 5%, was examined by XRD characterization (Figure 13). Seven diffraction lines were observed in the representative XRD pattern of GO-TEPA@Fe3O4 at 2
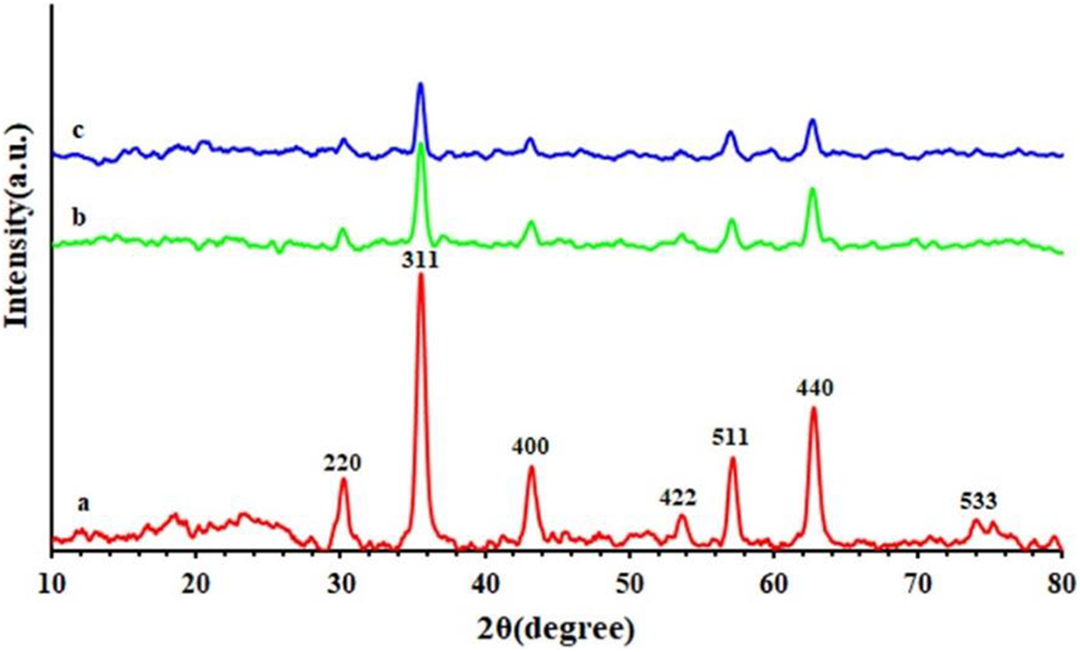
XRD curves of (a) GO-TEPA@Fe3O4, (b) PAEN 5%, and (c) PAEN 2% nanocomposites.
UV-Vis spectra of PAE, PAEN 2%, and PAEN 5% were shown in Figure 14, PAE exhibits strong UV-Vis absorption bands at 250 and 290 nm, assignable to π–π* transitions. Also, the PAEN 5% and PAEN 2% nanocomposites exhibit UV absorption bands at 250 and 295 nm and 252 and 300 nm, respectively. A redshift is observed at the maximum wavelength of both nanocomposites, which can be attributed to the strong interactions between filler nanoparticles and polymer matrix. According to the results, PAENs containing magnetic nanoparticles have optical properties, such as PAE.
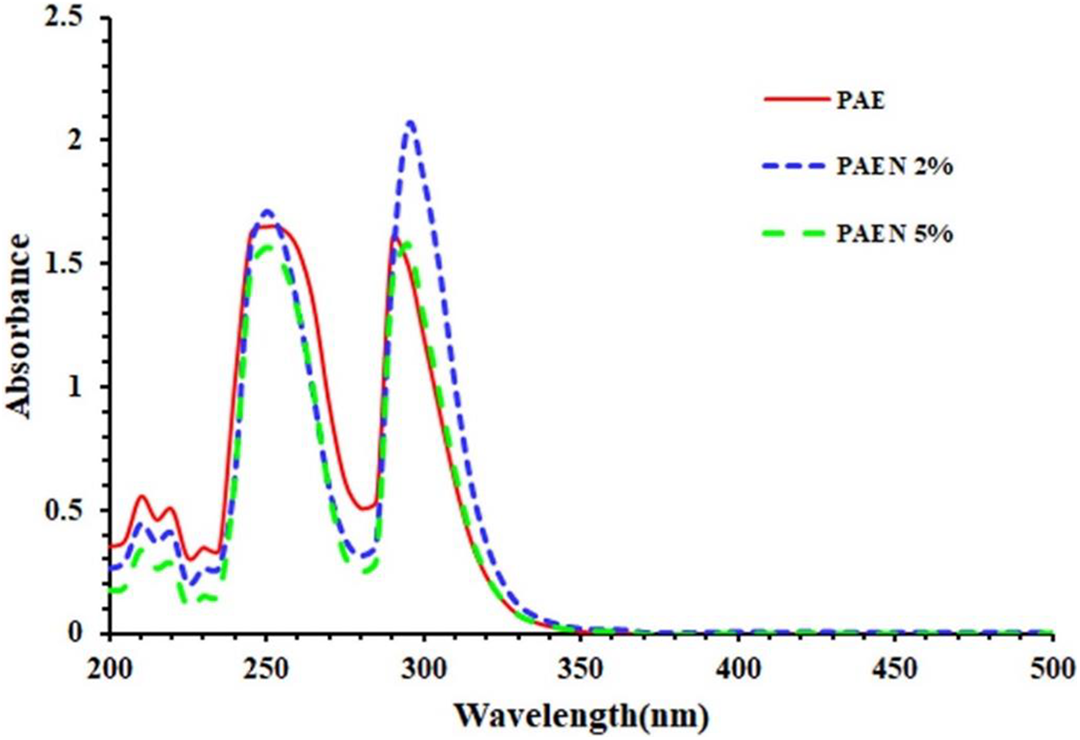
UV-Vis spectra of PAE and their magnetic nanocomposites.
To analyze the thermal stability of the PAE and PAEN films, we carried out thermogravimetric analysis (TGA) and DTG characterizations under nitrogen atmosphere, which are described in Figure 15 and summarized in Table 1. It can be observed that the PAE and their nanocomposites were a mass loss ranging from about 240°C and continuing until about 480°C.
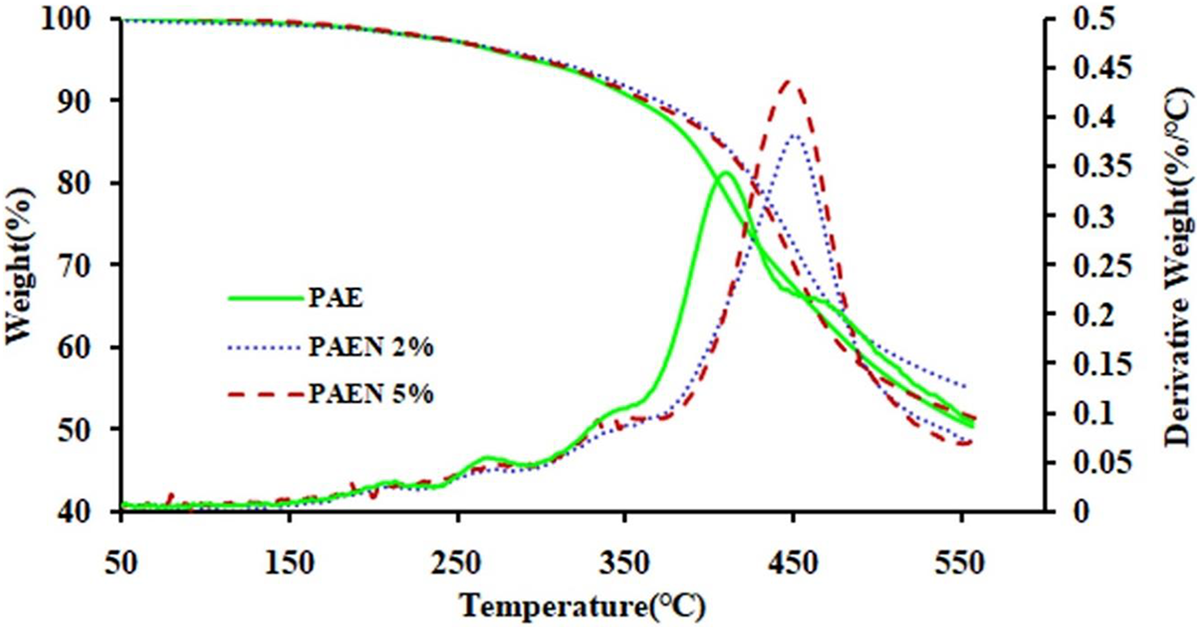
TGA and DTG curves of PAE and their magnetic nanocomposites.
Thermal characteristic data of the PAE and nanocomposites.
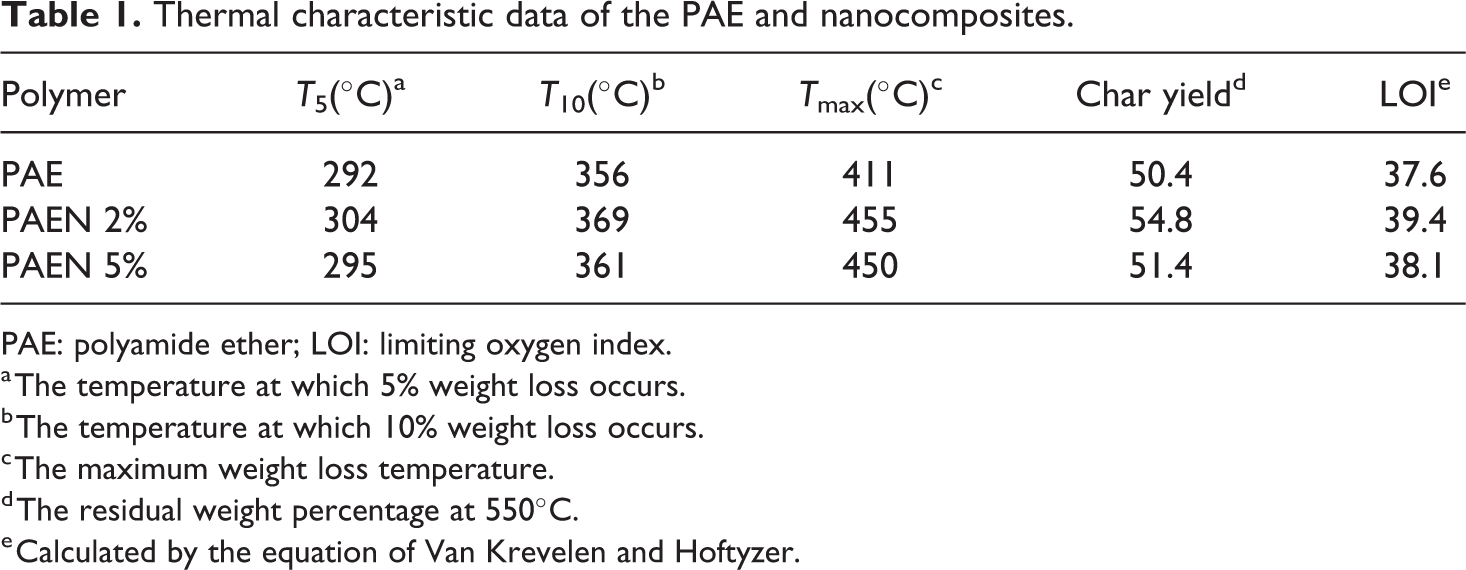
PAE: polyamide ether; LOI: limiting oxygen index.
a The temperature at which 5% weight loss occurs.
b The temperature at which 10% weight loss occurs.
c The maximum weight loss temperature.
d The residual weight percentage at 550°C.
e Calculated by the equation of Van Krevelen and Hoftyzer.
The thermostability of the polymer and corresponding nanocomposites (PAE/GO-TEPA@Fe3O4 2% and 5%) were evaluated by TGA under nitrogen atmosphere at a heating rate of 10°C min−1. The corresponding weight loss temperatures of 5% (
The limiting oxygen index (LOI), which is the minimum concentration of oxygen gas in an atmosphere consisting of a mixture of oxygen and nitrogen, is employed to compute the flammability of a material. Char yield can be used as criteria for evaluating LOI of the polymers in accordance with Van Krevelen and Hoftyzer equation 46
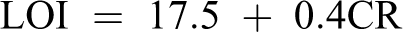
The PAE, PAEN 2%, and PAEN 5% having LOI values were calculated from their char yield at 550°C. Therefore, the samples can be classified as self-extinguishing materials (Table 1).
To further confirm the thermal stability trend observed from TGA and DTG thermograms, an analysis of the heat-resistance index is performed on the PAE and nanocomposites. The heat-resistance index (
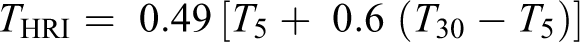
Using the thermograms shown in Figure 15, the heat-resistance index was calculated (Table 2) for PAE and nanocomposites. From the
Temperature values at 5% and 30% weight loss and heat-resistance index of the PAE and nanocomposites.
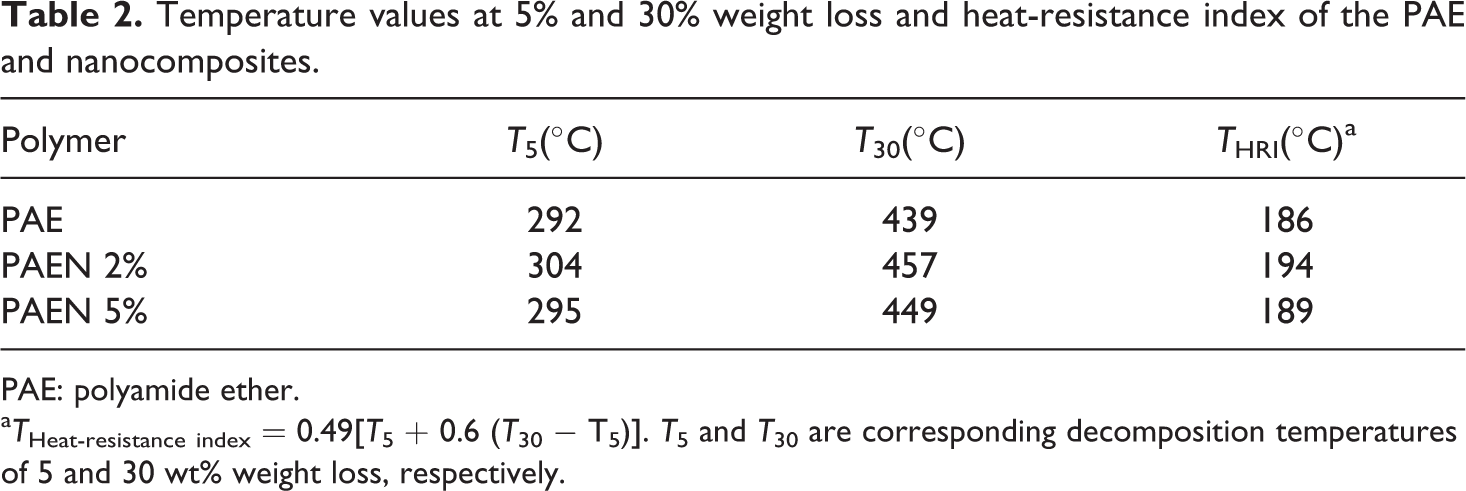
PAE: polyamide ether.
a
Figure 16 shows the magnetization (M) versus the applied magnetic field (H) (M-H curves or hysteresis loops) of PAEN 5% and PAEN 2% measured at room temperature for the PAENs with different loading amounts. The hysteresis curve allows determination of the saturation magnetization (Ms), remanent magnetization (Mr), and coercivity (Hc). The magnetization of samples could be completely saturated at high fields of up to ±8000.0 Oe and the specific Ms of the PAEN 2% and PAEN 5% nanocomposites were about 1.33 and 2.34 emu g−1, respectively, which increased with the increased loading amounts of GO-TEPA@Fe3O4. It has been reported that the ferromagnetic nanoparticles exhibit superparamagnetic behavior when the particle size decreases to below a critical value. 48 The nanocomposite samples PAEN 2% and PAEN 5% illustrate very small Mr and Hc as magnetic nanocomposites; Mr = 0.18 and 0.34 emu g−1, and Hc = 74 Oe for both of them, indicate that PAEN 2% and PAEN 5% have superparamagnetic behavior.
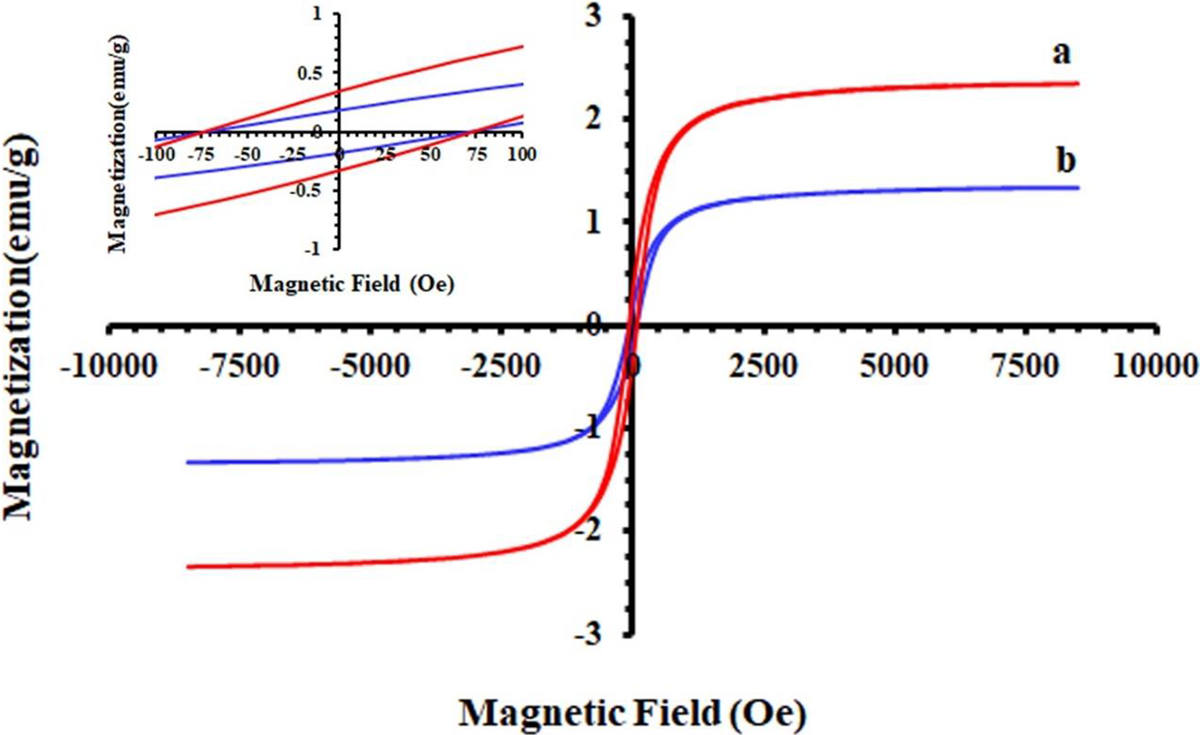
Magnetic hysteresis loops of magnetic nanocomposites (a) PAEN 5% and (b) PAEN 2% samples.
Conclusions
We have prepared heat-resistant and magnetic PAE/GO-TEPA@Fe3O4 2% and PAE/GO-TEPA@Fe3O4 5% nanocomposites. The new PAE derived from 2,5-dimethylphenol (
