Abstract
Polyamide 6 (PA6) nanocomposites with high electrical and thermal conductive were formulated via melt processing of PA6 and different inclusion of reduced graphene oxide (RGO). These nanocomposites showed small percolation thresholds, and the optimum formation of conductive links occurred with 0.5 wt% and ̴3.0 wt% of RGO respectively. RGO is effective in terms of thermal stability, causing a char of 6.8% with only 2.0 wt% RGO content. The presence of 3.0 wt% RGO enhanced thermal conductivity by 62% and reduces the peak-heat release rate to 588 and 545 kW/m2 with RGO inclusions of 1.0 and 2.0 wt%, respectively. Crystallization examination confirmed that RGO enabled the crystallization of PA6 structure mostly through speeding up the formation of crystal nuclei, reaching a maximum and smallest crystal grain extent with RGO inclusion up to 2.0 wt% that improved the generation of the most unflawed crystalline matrix. With the dynamic rheological testing, frequency-independence of G′ and abruptly decreased phase angle at the small-frequency area via RGO content of 2.0 wt% signify an alteration from liquid-state to solid-state rheological performance and validate the development of percolation link structure with RGO in the function of a crosslinking factor. Enhancement of fire-retardant characteristics of PA6 was attained due to the inclusion of RGO owing to the improvement in the PA6 structure. Morphological research showed that RGO was spread uniformly in the PA6 structure. These tests show substantial capacity for the bulk manufacture of electrically conductive polymer/RGO nanocomposites.
Introduction
Polyamide 6 is a conventional industrial polymer with superior mechanical resistance, outstanding load-bearing ability, strength to chemicals, greater self-lubricating implementation, etc.,1–3 which enables PA6 to be employed broadly in advanced engineering materials. Particularly, it is appropriate for substituting metallic substances for the fabrication of gears, bearings, and slide blocks.4–6 PA6 is a popular thermoplastic that is easy to work with and has strong chemical resistance. PA6 is used in a variety of applications, including automotive components, fibers, housings for home and power tools, electric plugs and sockets, medical implants, triboelectric nanogenerators, and many more.7,8 For PA6, the span of exterior electrical resistivity is from 1012 Ω to 1014 Ω, 9 which simply directs to the formation of static charge on the exterior sides, together with dust defect, short circuit, and trigger a flame or a blast. 10 Consequently, developed antistatic and fire-retardant characteristics are required to extend the range of PA6 applications. Graphene (Gr) was investigated owing to its interesting and incomparable physical characteristics.11–13 Hence, Gr was used in assembling several micro-electrical tools, 14 batteries, 15 particularly in the area of conductive switching, 16 bio-imaging, 17 and photocatalysis, 18 and one of the utmost intriguing features of Gr is the capacity for their usage in electronic purposes.19–21
Gr was used in nanocomposites due to the proficient thermal interface material, significant thermal regulator substance, a conductive additive, excellent thermal conductivity (k), and superior steadiness in elevated temperatures. 22 Graphite oxide, obtained through graphite oxidation, is a widely examined start-off substance for bulk production of Gr nano-layers. 23 It was confirmed that GO is easily spread in polymers that are soluble in water to boost their mechanical characteristics. 24 However, to attain electrically conductive polymer nanocomposites, a decrease of GO is needed to eliminate the majority of its oxygen-holding chemical groups and rebuild the graphitic link of sp2 links. 25 To date, there are mostly two efficient reduction methods, chemical and thermal reduction, 26 for the formulation of electrically conductive polymer nanocomposites from graphite oxide. In organic solvents, warming GO suspensions considerably decrease the GO platelets with no reduction in chemical factors.27-30 Consequently, these conductive nano-layers are employed to formulate conductive polymer nanocomposites through melt compounding. 31 Nevertheless, before the inclusion of a reducing chemical factor, the functionalization of GO nano-layers is required to deter their coagulation which stabilizes its spread in organic solvents and polymer structures. 32
The inadequate oxidation and exfoliation of graphite oxide and heterogeneous spread of Gr sheets cause small electrical conductivities.
33
Further, the chemical reduction has drawbacks of usage of huge volumes of organic solvents and reduced fitting with numerous industrial processing procedures.
34
RGO, based on the bulk fabrication on an engineering level, is decreased as the thermal treatment at temperatures to 1055°C. Oxygen chemical groups on RGO surfaces were hard to be eliminated throughout the reduction process of GO to RGO, and hence
The effect of in situ polymerization was studied on the properties of the graphene and specifically their oxidative state. 1 The conductive network structure of the conductive cellulose skeleton, as well as the interfacial interaction with PA6, were examined as well, and the results showed great potential in electronic industrial applications. 6 The polarities of RGO and PA6 macromolecules were found to be similar, which is important for maintaining the exfoliated state of the nanosheets in the PA6 matrix after polymerization, which also results in high electrical conductivity for PA6 composites due to the RGO nanosheets' well-distributed electrically conductive network. 28 The formation of an electrically conductive network, as well as the interfacial contact between PA6 chains and RGO layers, were revealed to be related to the significantly increased electrical conductivities of PA6 composites. 2
There is not much recent research into the thermal properties of Gr-based nanocomposites, particularly the thermal conductivity of thermoplastic nanocomposites. In this research, we hammered at increasing the k of Gr-based PA6 composite to comprehend its thermally conductive functions. The obtained PA6 composite with elevated k could be utilized in thermal interface substances, connectors, and other superior-behavior thermal management systems. In this study, the consistent spread of the RGO, through the melt processing, in the PA6 structure with superior melt viscosity in processing makes a quick growth of electrical conductivity of nanocomposites with a small threshold percolation. Finally, electrically conductive characteristics of the nanocomposites were researched, intending to the insight into the development mechanism of the percolation link structure. RGO and PA6 performed matched in polarity that conveys great electrical conductivity to PA6 polymer owing to properly spread the electrically conductive link of RGO nano-layers. Besides that, it was identified a profound understanding of the character of RGO in crystallization and movement/distortion histories of PA6 molecular links. We demonstrate a novel strategy to boost the fire-retardant characteristics of PA6 by the inclusion of RGO. Gr is developing like a great moment in the area of fire-retardant applications owing to its endothermic and strong structure. Therefore, the combination of RGO into the polymer structure may be considered in the function of an efficient method to get good fire-retardant characteristics in polymer and depict promising novel nanocomposites in advanced engineering materials fabrication. The novel use of RGO may have benefits over previously reported PA6 composites, indicating a feasible strategy for rapidly creating PA6/graphene composites as new nanocomposites in industrial manufacturing.
Materials and methods
Materials
Reduced graphene oxide granules were prepared by Sixth Element Materials Technology Co., Ltd., (Changzhou, China) with a median size of 55 micro-meter. The PA6 pellets (Taiwan Libaolon Co., Ltd.), were applied with no extra distillation and action.
Preparation of PA6-RGO nanocomposites
Each specimen was conveniently dehydrated in vacuo at 85°C overnight before usage. Then the required proportion of RGO was blended and melt-mixed with PA6 in Thermo-Haake Rheomix with the temperature alterations of 225–255°C at a screw speed of 85 rpm. Subsequently, the stuff fed out of the batch blender was quickly chilled to 25°C, milled into granules, compression-molded for almost 6 min at 255°C, and lastly, broken into samples for supplementary categorizations.
Characterizations
Thermogravimetric and differential thermal gravimeter tester (TGA-DTG) for specimens are demonstrated on a Perkin–Elmer Diamond thermal tester from 25°C to 800°C, applying a warming proportion of 20°C.min−1 with N2 in the role of a specimen remove gas. TC is assessed ‘‘through-plane’’ technic with a Netzsch LFA 447 Nano-flash at 20°C. A heat releasing test is demonstrated on a cone calorimeter analyzer (UK, warming 40 kW/m2) according to the ISO 5660-1 technique. Differential scanning calorimetry (DSC) findings are measured on a Mettler Toledo TGA/DSC 1 tester in the N2 atmosphere and with a warming ratio of 5°C.min−1. Fourier transform infrared spectroscopy (FTIR) is tested, applying a Nicolet MAGNA-IR 750 spectrometer over the wavenumber scale of 4000-400
Results and analysis
Figure 1 shows the TGA test outcomes for PA6, RGO, and PA6-RGO nanocomposites. The thermal steadiness of RGO, PA6-RGO, and PA6 may be categorized with TGA testing. It is obvious that RGO is not thermally durable and displays a major weight loss of 11.85 wt% on a scale from 100°C to 205°C owing to deterioration of oxygen-holding chemical functions and production of CO2, CO, and H2O. The ultimate thermal degradation of 22.86% at 800°C occurs due to the elimination of additional oxygen functionalities.35,36 Associated with RGO, the deterioration procedure had no significant alteration for the specimen of PA6-RGO up to 310°C. There are two steps to the deterioration process for PA6-RGO nanocomposites. The initial deterioration step on the scale from 100°C to 310°C is begun via the elimination of functional groups of hydroxyl from the RGO surface. The weight degradation is very less than that of RGO at a similar temperature, and greater thermal steadiness causes by reactively intercalated PA6 links and intermolecular hydrogen-bond interaction among RGO layers and PA6 links. At the second deterioration step on a scale of 310°C–480°C, the weight loss of PA6-RGO is huge and superior to that of RGO owing to the deterioration of PA6. It is obvious that, though RGO has weak thermal steadiness, its PA6 nanocomposites are thermally durable. Remarkably, the quantities of remaining char of PA6 were augmented with the existence of RGO nano-layers. RGO is effective in accelerating the char development of PA6, caused in a char of 6.8% with only 2.0 wt% of graphene, more than two times superior to graphene load. GO is able in accelerating the char development of PA6. As RGO loading in PA6-RGO 2.0 wt% nanocomposite and the remaining char is 6.8%. The increased char development designates that the RGO inclusions reveal the potential in upgrading the fire-retardancy of PA6.5–7 TGA analysis outcomes for PA6, RGO, and PA6-RGO nanocomposites.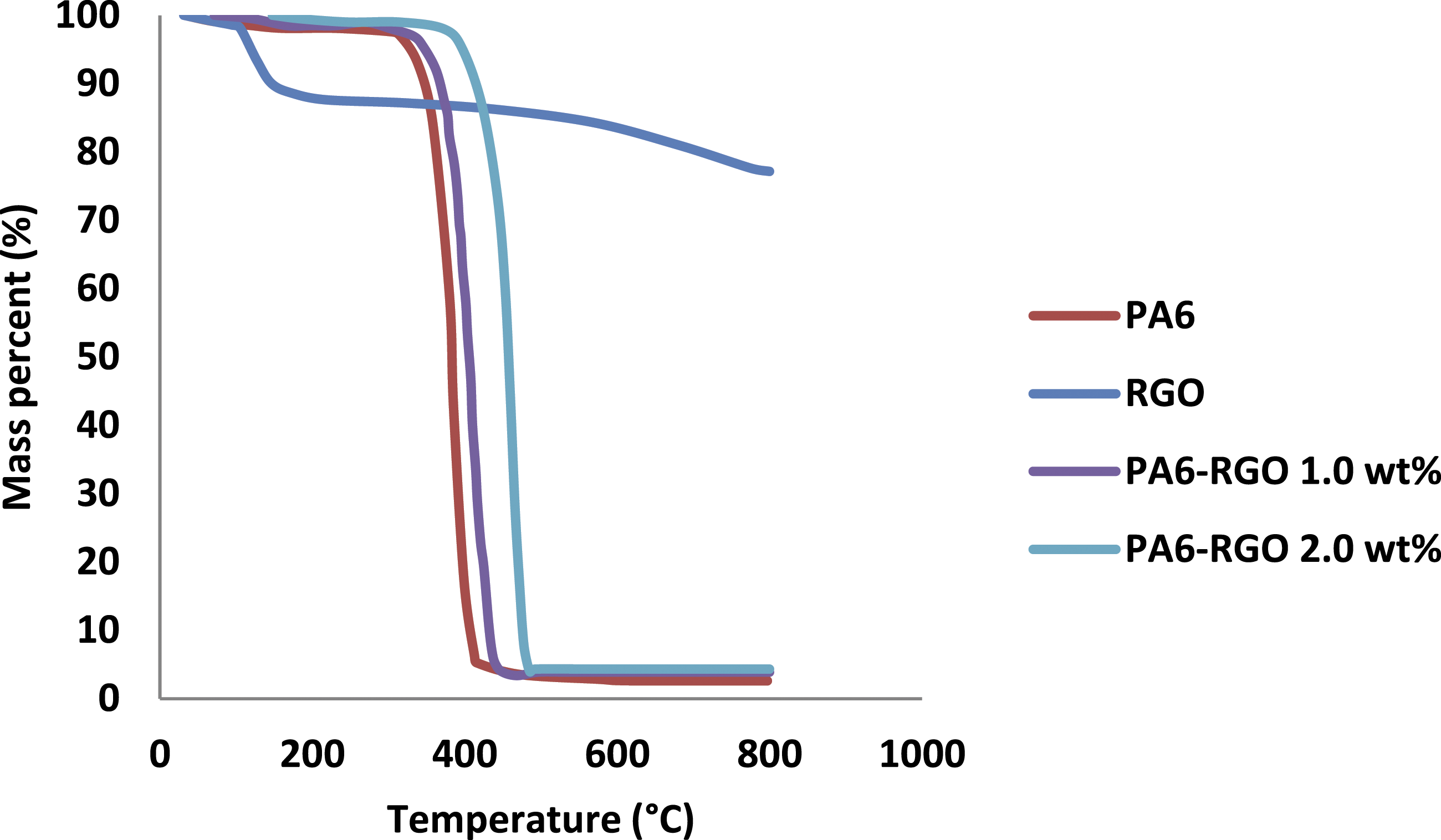
Figure 2 presents the torque alterations of PA6 and PA6-RGO nanocomposite in the blending procedure. The melting PA6 was mixed with different levels of RGO inclusion. The primary torque of the feeding point for PA6 is 46 N.m, and with the delaying of blending time, torque reduces to stabilizing torque in 85 s. The room of the blending compartment is occupied completely with the material in viscous flow condition, causing a reduction in rotor rotational resistance and torque. For PA6-RGO 1.0 wt% and 2.0 wt% nanocomposites, the primary torque of feeding point (75 N.m and 90 N.m), stabilizing torque, and the time needed (140 s and 205 s) to achieve the stabilizing torque augments correspondingly, which makes a fluidity reduction of PA6 molecular links, affected by steric barrier influence of RGO and interfacial interaction among RGO and PA6 structure. Torque alterations of PA6, and PA6-RGO nanocomposite.
Figure 3 displays the FTIR spectrums of PA6, RGO, and PA6-RGO nanocomposite to realize the surface structure of specimens. For PA6 fiber, the absorption points at 3085 Fourier transform infrared spectroscopy spectrums of PA6, RGO, and PA6-RGO nanocomposite.
Figure 4 exhibits the RS spectrums of PA6, RGO, and PA6-RGO nanocomposite to study the interaction among RGO layers and PA6.
24
The characteristic G point is placed at 1588 Raman spectra alterations of PA6, RGO, and PA6-RGO nanocomposite.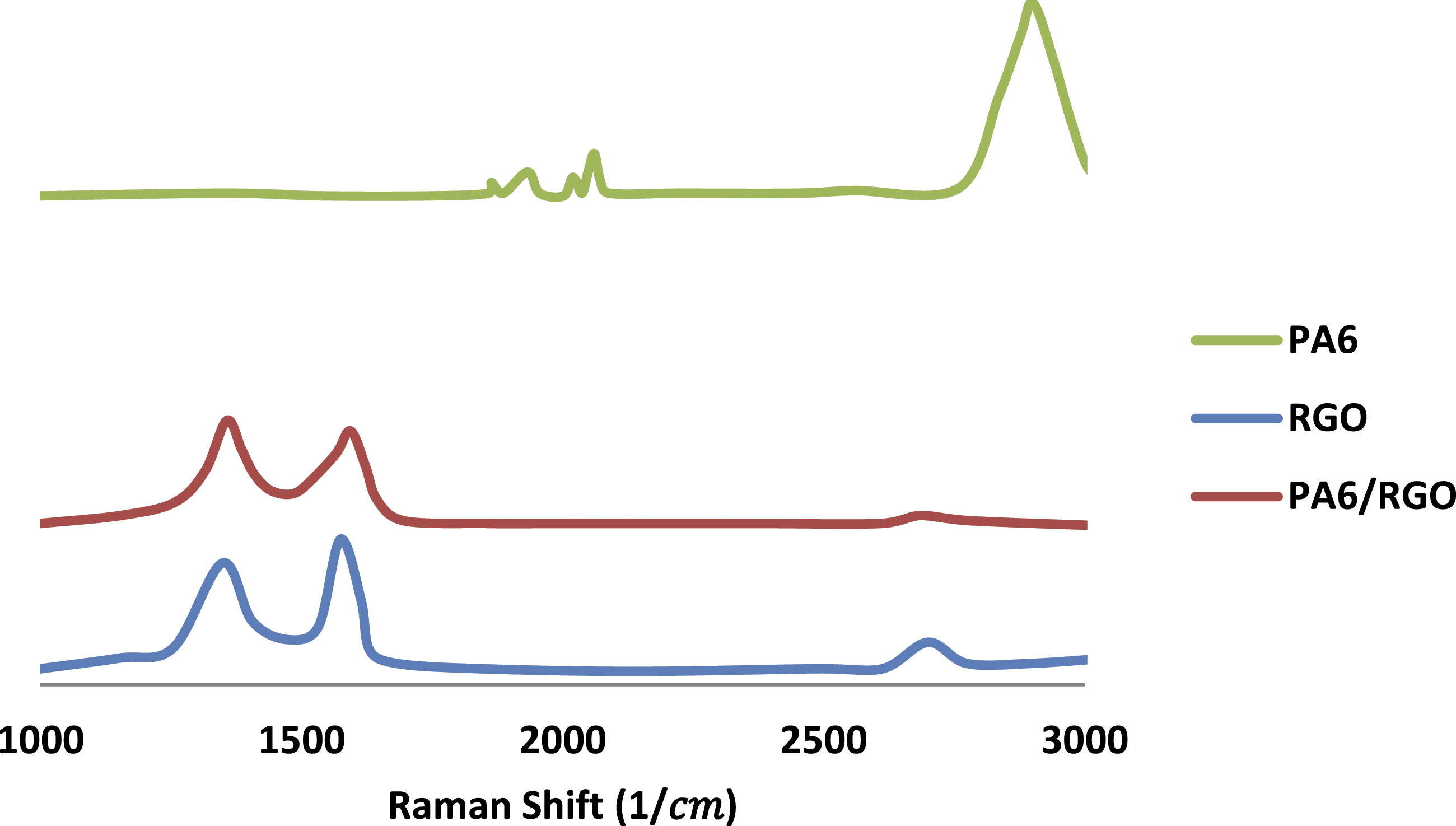
Figure 5 and Table 1 reveal the XRD spectrums of PA6, RGO, and PA6-RGO nanocomposites. XRD testing is accomplished to assess the crystal structure of PA6 and PA6-RGO nanocomposites. As indicated in Figure 5, the representative diffraction point of the RGO, a wide point focused at 24.5° that relates to (002) was monitored, revealing that the attained specimen was composed of arbitrarily arrayed RGO sheets with an uneven form.
12
For PA6, monitored diffractions at 2θ=20o and 23o are attributed to α1 and α2 crystalline structures and are labeled as (200) and (002/202) reflections correspondingly.
23
It may be monitored that there are no further diffraction points in XRD of PA6-RGO nanocomposites excluding diffraction points assigned to the PA6 phase, signifying that RGO does not any effect on the crystal structure of PA6. But, the diffraction peaks sites and intensity in nanocomposites alteration a bit, showing that the inclusion of RGO has a minor effect on the crystal grain extent of nanocomposites. Besides, no obvious point was monitored to the accumulation of RGO s, showing that stacking of RGO in PA6 structure is not ordered and near to single-sheet level. The crystallinity (Xc, XRD) and crystal extend (Lhlk) are obtained by the below formulas, accordingly X-ray diffraction spectrums of PA6, RGO, and PA6-RGO nanocomposites. X-ray diffraction of PA6-RGO nanocomposites with different RGO loadings.
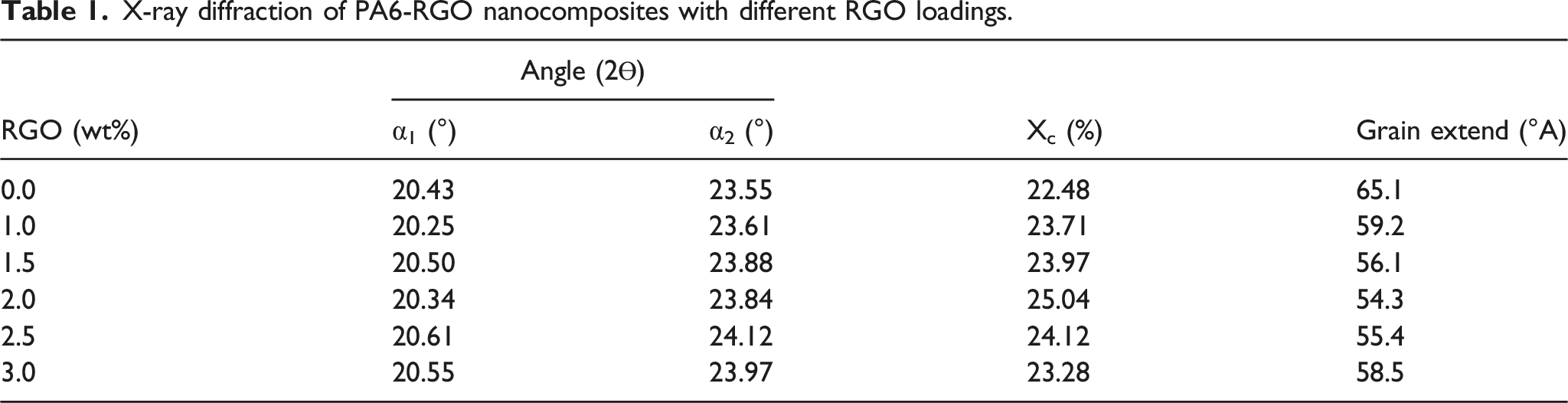
Figure 6 demonstrates the electrical conductivity of PA6 and PA6-RGO nanocomposites. For PA6-RGO nanocomposites, a sudden augment of about 6 orders of magnitude in electrical conductivity is monitored on a narrow scale with RGO load from 0 to 0.5 wt%. It may be noticed that the electrical conductivity of nanocomposites augments quickly with RGO content, and even with the inclusion of 1.0 wt% RGO, the nanocomposite alterations from an insulator to semiconductor, showing a wonderful conductive link slightly generated at RGO loadings close to the percolation threshold. With additional augment of RGO up to 2.0 wt% loading, the growing level of nanocomposite conductivity reduces slightly, showing a great conductive link generated at upper RGO loading. Above 2.0 wt% RGO, the conductivity has finally enhanced to equilibrium amount, reaching the higher of 1.15 × 10−2 S.m−1 with 3.0 wt% RGO load, which shows the final development of the ideal conductive link. Therefore, the electrical conductivity of nanocomposites enhanced quickly to equilibrium amount, causing the development of the appropriate conductive link at this content of RGO. Figure 6 also reveals the thermal conductivity alterations of PA6 and PA6-RGO nanocomposites. The nanocomposites with appropriate thermal conductivity have many uses in thermal interface substances, connectors, and other elevated-performing thermal managing systems.
16
The thermal conductivity of PA6-RGO nanocomposites enhances continuously with RGO inclusion. The thermal conductivity of PA6-RGO nanocomposite loading 3.0 wt% RGO enhances by 62% (from 0.31 to 0.5 (W. m−1.K−1)) associated with PA6. In nanocomposites, as thermal energy is moved mostly in form of phonons, weak coupling in vibration modes on the filler-polymer interface conveys substantial thermal resistance.
28
Chemical bonding among the filler and the polymer may effectively decrease the acoustic phonon spreading at the interface. Electrical conductivity and thermal conductivity of PA6, and PA6-RGO nanocomposites.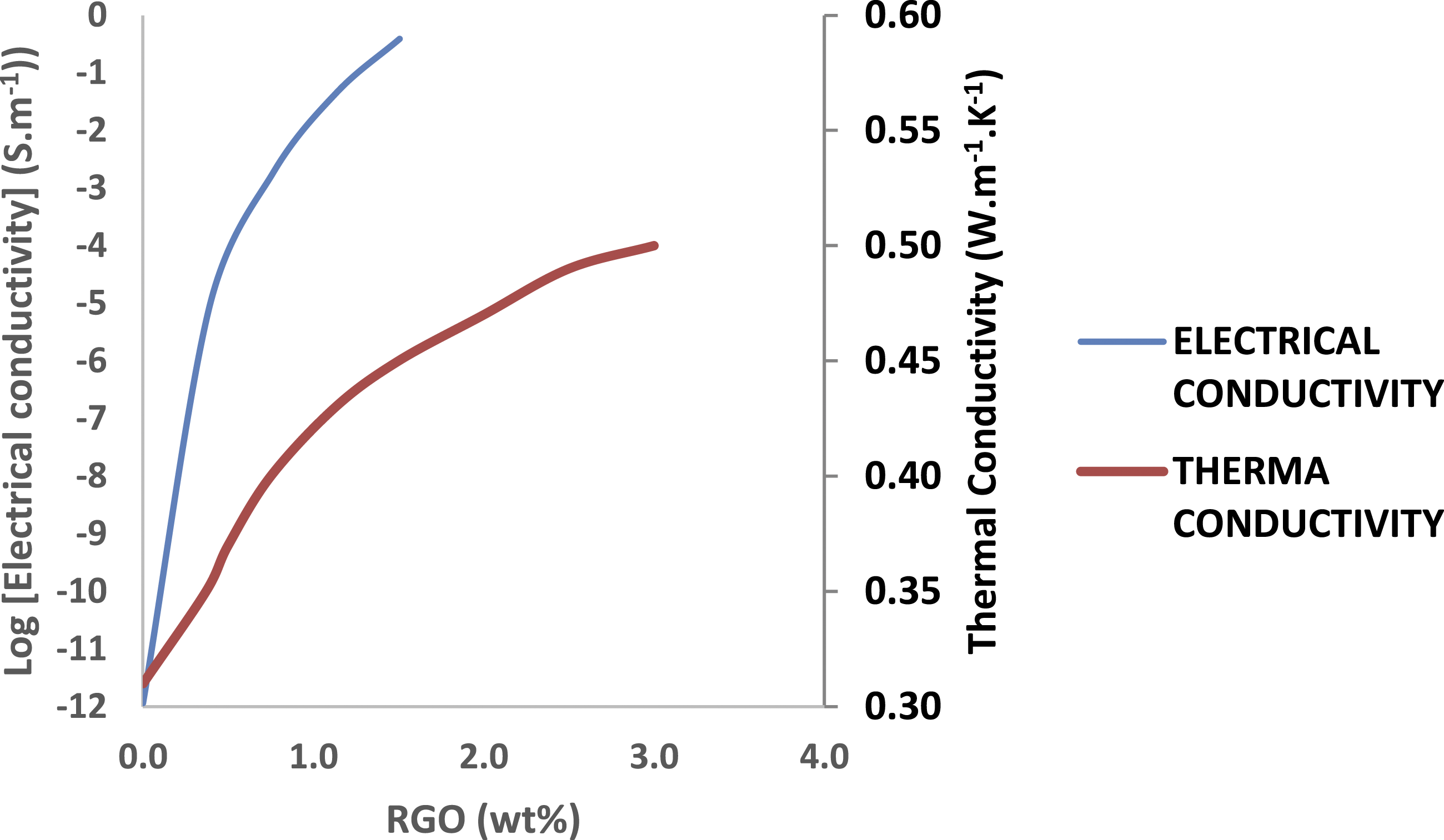
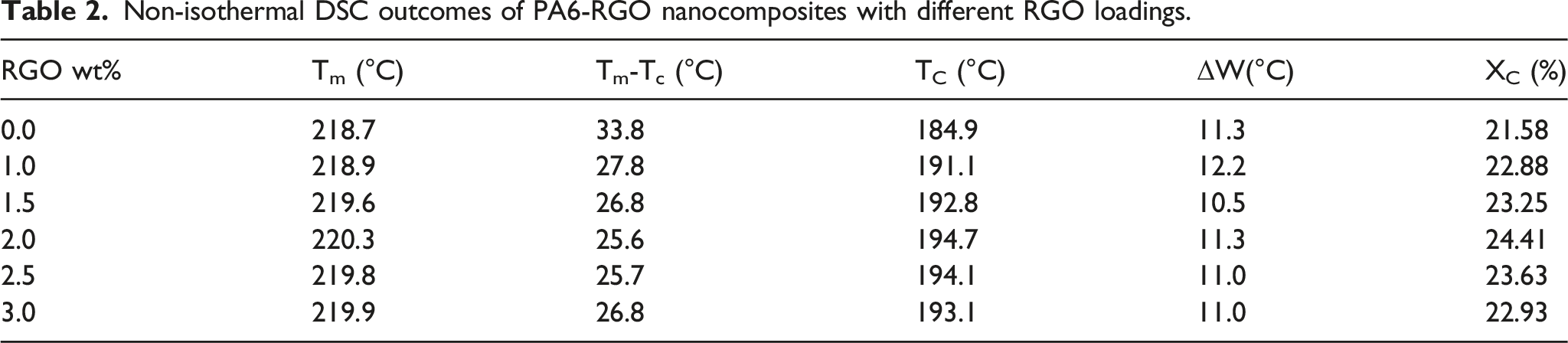
Figure 7(a) and (b) and Table 2 exhibit the DSC test outcomes for PA6, and PA6-RGO nanocomposites in endothermic and exothermic conditions. DSC data displayed in Figure 7 displays that for control PA6 and PA6-RGO 1.0 wt% an exothermic point is monitored among 210 and 230°C, with locations at 224°C and 226°C correspondingly. This summit point is attributed to the melting point of substance
16
especially the melting of Ɣ phase crystals.36,37 The 2°C smaller melting temperature of bound PA6 in RGO that is associated with the nanocomposites and control polymer is probable a cause of the length of PA6 links grafted onto the RGO surface and is monitored to be owing to Gr hindering the link spread in PA6, causing in a smaller molecular weight.26,35 The physical characteristics of crystalline polymers are directed by super-molecular morphology that consecutively is regulated by the crystallization procedure.20,21 Therefore, the research of crystallization performance is essential for creating structure-property correlations in PA6-RGO nanocomposites. The non-isothermal DSC alterations of PA6 and PA6-RGO nanocomposites at a warming and cooling level of 15°C.min−1 are shown in Figure 4, from that crystallization temperature (Tc), melting temperature (Tm), half-point width (ΔW), and crystallinity (Xc, DSC) may be resulted, as exist in Table 2. Table 2 indicates that alterations of Tm for PA6 in nanocomposites are not very clear, although, Tc of PA6 enhances as loading of RGO enhances till to 2.0 wt% and then reduces. The super-cooling (Tm-Tc) reduces at first and then enhances. The crystallinity (Xc) of PA6 shows the trend of first enhancing, achieving the highest at 2.0 wt% load of RGO, and then reducing, showing the nucleation influence of RGO on PA6 as a heterogeneous nucleating chemical factor at a smaller RGO load.
23
With additional growth of RGO load, abruptly enhancing the viscosity of the system reduces the mobility of the PA6 link and inhibits the shaping of huge crystalline domains due to restricted space and sturdy limits imposed on PA6 links, which causes a reduction of Xc. Differential scanning calorimetry analysis outcomes for PA6, and PA6-RGO nanocomposites, (a) Endothermic, (b) Exothermic. Non-isothermal DSC outcomes of PA6-RGO nanocomposites with different RGO loadings.
The cone calorimeter test outcomes for PA6 and PA6-RGO nanocomposites, formulated by melt mixing are displayed in Figure 8, and Table 3. It may be monitored that PA6 fires quickly after burning and a peak of heat release rate (PHRR) amount is 663 kW/m2. The inclusion of RGO (1.0 and 2.0 wt%) into PA6 makes the PHRR reduce to 588 and 545 kW/m2. From Figure 8, the total heat release (THR) alterations show the same tendency as heat release rate (HRR), and THR values of PA6-RGO are decreased from 142.6 MJ/m2 (PA6) to 113.6 and 102.5 MJ/m2. From Table 3, it may be noticed that the inclusion of the RGO has a small effect on the time-to-ignition (TTI) of the nanocomposites, due to the low contents. Concerning the total smoke production (TSP), all the specimens loading RGO show superior TSP associated with PA6. This phenomenon is probably owing to the incomplete combustion of PA6. RGO is not permeable to any gas molecules. The two-dimensional construction of RGO inside the PA6 forms a tortuous path that deters the molecular diffusion through the PA6 and accordingly reduced penetrability. The orientation of the platelets increases the barrier characteristics perpendicular to their alignment, while the upper platelet aspect correlates with augmented barrier resistance. So, the inclusion of RGO increases the gas barrier characteristics of PA6. The fire-retardant characteristic of PA6 nanocomposites has become a serious concern in industrial areas. Owing to its endothermic and strong layered structure, Gr performs like physical barriers to decrease the diffusion of gases and deterioration products, hence preventing the supply of oxygen and playing the role of fire-retardant. The degree of spread and interfacial interactions among RGO and polymers are major thoughts for progress in fire-retardants of nanocomposites. Heat release rate and THR test outcomes for PA6, and PA6-RGO nanocomposite. Cone analysis outcomes for PA6 and PA6-RGO.

Figure 9(a–c) displays the complex viscosity (9-a), elastic modulus (9-b), and viscous modulus (9-c) test outcomes for PA6, and PA6-RGO nanocomposites. The molecular link tangle performance of PA6 nanocomposites with different RGO loading was searched by dynamical rheological outcomes. The complex viscosity like a variable of frequency denotes a pseudo-plastic fluid feature of PA6 flow performance which was monitored.
4
The small-frequency complex viscosity enhances considerably in the existence of RGO, particularly above 2.0 wt%, and the Newtonian plateau of viscosity alterations vanishes slowly, indicating an outstanding shear-thinning performance.
5
Figure 9(b) and (c) show the elastic modulus (G′) and viscous modulus (Gʼʼ) of specimens with various RGO loadings. At small frequencies, PA6 displays a conventional polymer terminal performance with scaling characteristics of G’ ∼ ω 1.3 and Gʼʼ ∼ ω 0.87 owing to its entirely stress-free polymer links.
6
The RGO loading conducting sudden enhances in both G′ and Gʼʼ, and the terminal performance at small frequencies vanishes with 2.0 wt% load of the RGO. Based on the pronounced alterations of G′ and Gʼʼ, it is surmised that terminal to non-terminal alteration for PA6-RGO nanocomposites, proving a development of interconnecting link of RGO nano-layers in nanocomposites. The frequency dependence of G′ at small frequencies fails with the growth of RGO loads. At RGO loads higher than 2.0 wt%, the approximate frequency-independence of G′ directs the alteration from liquid-state to solid-state rheological performance. Complex viscosity (a), elastic modulus (b), and viscous modulus (c) test outcomes for PA6, and PA6-RGO nanocomposites.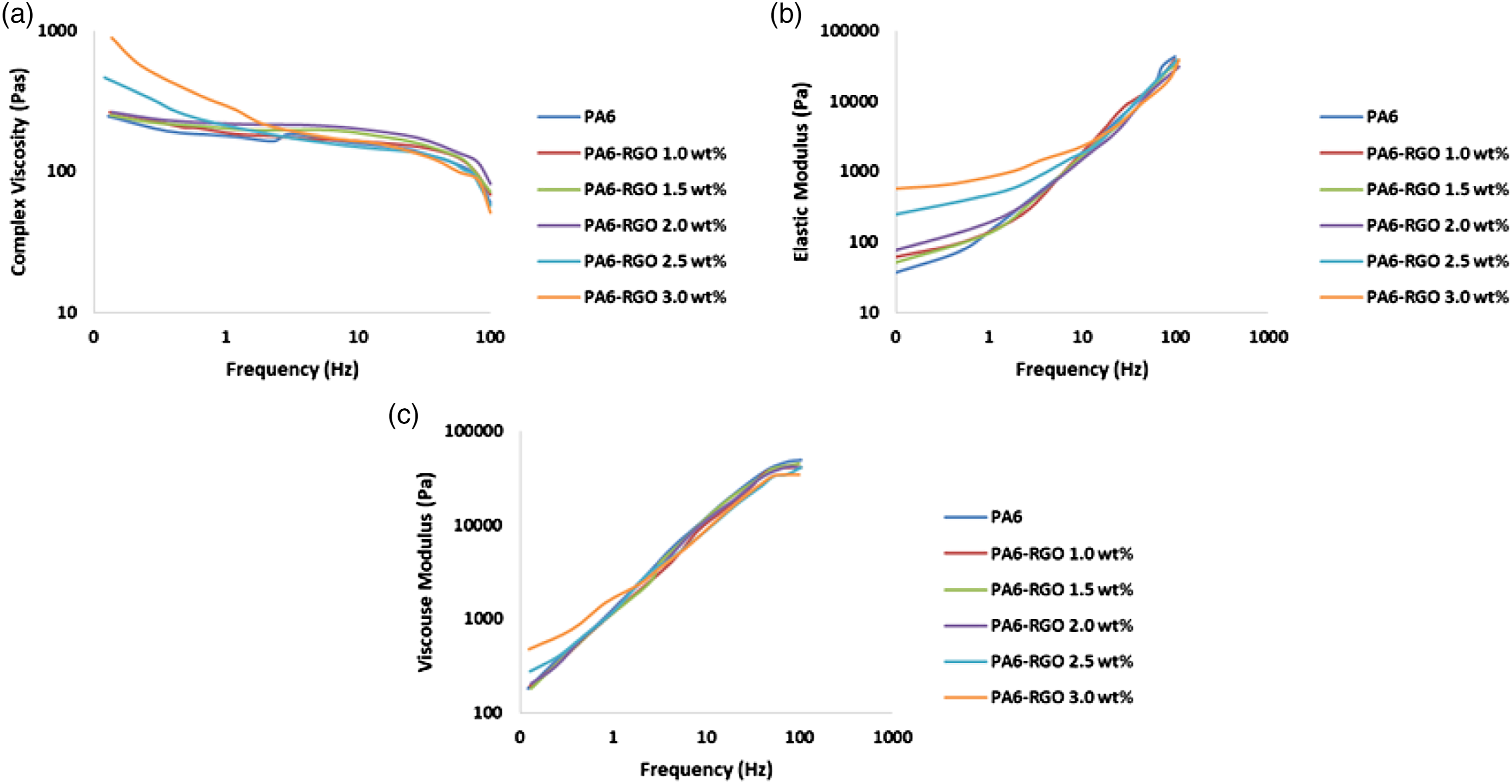
Figure 10(a) indicates SEM depiction on a cross-section of hot-pressed PA6. To directly assess the general spread and exfoliation of RGO nano-layers in PA6, Figure 10(b) indicates the SEM photo of the freeze-fractured surface of the PA6-RGO nanocomposite. It is monitored that RGO nano-layers were exfoliated and spread properly in nanocomposite structure and no huge coagulates existed. The exfoliation of RGO allows the development of an interconnected 3D-dimensional conducting link in the polymer structure. Hence, the electrically conducting characteristic of the PA6-RGO nanocomposite with a small percolation threshold may be related to stabled exfoliation and spread of thermally RGO nano-layers in the PA6 structure. Moreover, even spread of the exfoliated RGO nano-layers was attained, which is associated with the fact that RGO still bore some remaining polar chemical functional groups on the RGO surface; however, it practiced a thermal reduction at 1055°C
9
that enabled and stabled spread of RGO nano-layers in polar PA6 structure. TEM is applied to deliver immediate signs-on development of electrically conductive link of RGO (the black area) inside PA6 (the white area). A further concentrated electrically conductive link causes in the nanocomposite with a percolation threshold of 2.0 wt% in Figure 10(c). The development of the conductive pathways at such a small RGO load is produced by reactive intercalation of PA6 molecules in RGO nano-layers that advance the spread of RGO in the PA6 structure. At an upper content (3.0 wt%), the RGO nano-layers are linked to generate an appropriate and following conductive pathway inside the polymer structure in Figure 10(d). (a) SEM of PA6 (b) SEM of PA6-RGO 1.0 wt% (C) TEM of PA6-RGO 2.0 wt%, (d) TEM of PA6-RGO 3.0 wt%.
Conclusions
PA6-RGO nanocomposites were formulated via the melt-mixing procedure. The influence on the development of crystallization and link structure on electrically conductive characteristics of nanocomposites was studied. Frequency-independence of G′ and then abruptly decreased phase angle at the small-frequency area with a load of 2.0 wt% specifies the change from liquid-state to solid-state rheological performance. Dynamic percolation performance of nanocomposites was searched and indicated that thermal conductivity, electrical conductivity, and mechanical characteristics of nanocomposites were enhanced with the development of the electrically conductive link. Other characteristics like thermal steadiness and fire-retardant were also realized progress of nanocomposites with the extremely small loading of the RGO. The covalently bonded RGO nanostructures successfully improved the fire-retardant characteristics as the fillers in PA6 from the PHRR and THR outcomes. A morphological analysis displayed that RGO was spread well in PA6. Our outcomes designate that the progress in fire-retardancy of RGO nanocomposite may be due to the barrier effect of its link. The RGO nanocomposites open a new strategy to be applied to a wide range of polymer materials as an effective filler to advance the flame safety of polymers. However, the current approach outcomes have shown that RGO is extremely hopeful for the future applications of industrial polymers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


