Abstract
The natural cationic colorant berberine was used to dye cationic dyeable polyester (CDP) fibers. Its dyeing properties, namely color strength (
Introduction
The application of natural dyes for textile dyeing has a long history over thousands of years. For instance, it is believed that cochineal colorant was applied to leather materials and silk more than 4000 years ago in China.
1
However, synthetic dyes have been developed at an astonishing tempo over the past 150 years. Recently, due to the increase of worldwide health and environmental awareness, a resurgence of interest in natural dyes has occurred to provide eco-friendly textile materials. Thus, there is a renewed interest in the study of natural dyes for textile dyeing (e.g., almond shell,
2
annatto,
3
hazelnut,
4
cochineal, and
Berberine, the extract of
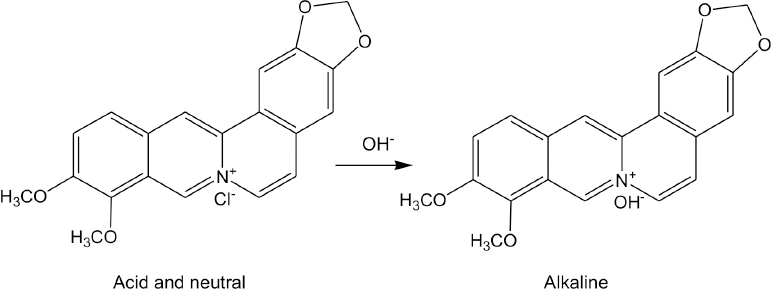
Chemical structure of berberine chloride under different pH conditions.
In this study, the effects of dyeing pH, temperature, time, and berberine concentration on the dyeing properties of berberine on CDP fibers were evaluated. Simultaneously, its adsorption properties were investigated in terms of dyeing thermodynamics and kinetics.
Experimental
Materials
Cationic dyeable polyester (CDP) loose fiber (1.5 dtex/38 mm) was obtained from Jiangsu Green Peafowl Chemical Fiber Co. Ltd. Berberine chloride (99%) was purchased from Aladdin Reagent Co. Ltd. Glacial acetic acid, phosphoric acid, and boric acid (Sinopharm Chemical Reagent Co. Ltd.) were analytical grade and were used to prepare buffer solutions.
Dyeing Methods
All dye solutions were prepared using berberine powder, Britton-Robinson buffers, and distilled water. The liquor ratio (LR) was maintained at 50:1. The pH was adjusted using Britton-Robinson buffers (H3PO4-HOAc-H3BO3/ NaOH). The dyeing process has been presented in Scheme 2. After immersing CDP fibers in dye solutions at room temperature (RT), the system was heated to 40 °C at a maximum rate. The dye bath temperature was then increased to 100 °C at 1 °C/min, held there for 60 min, and then cooled to 70 °C at 3 °C/min. At the end of dyeing, the dyed samples were removed, rinsed thoroughly in tap water, and dried in the open air.
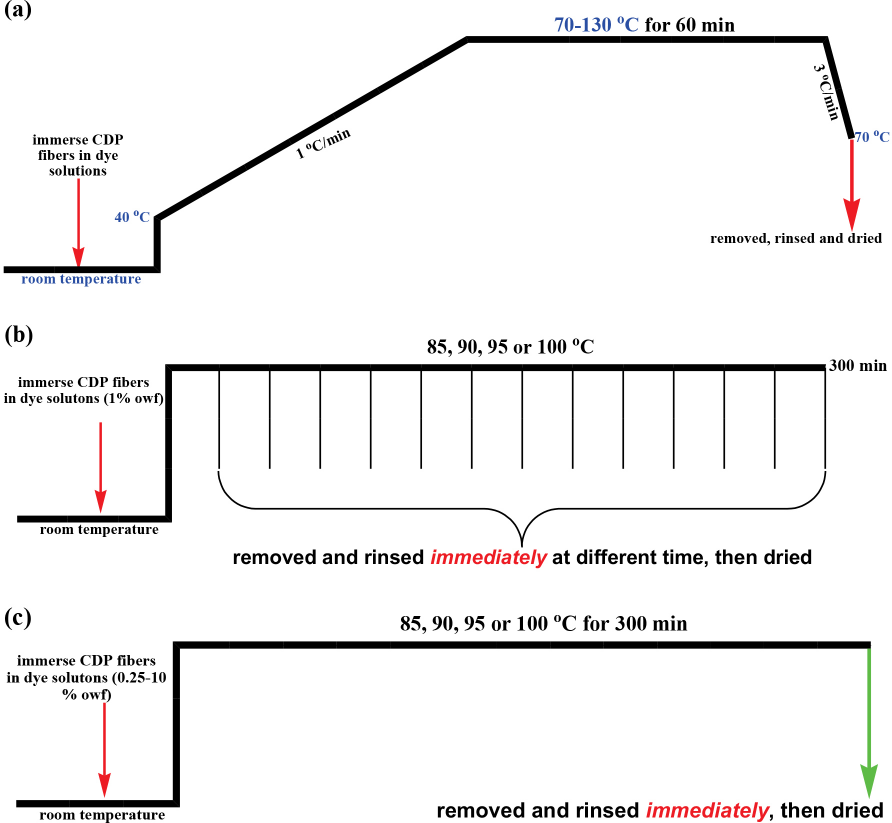
Dyeing process of (a) various levels of dyeing pH, temperatures, time, and concentrations of berberine, (b) kinetic dyeing conditions, and (c) thermodynamic dyeing conditions.
To evaluate the effect of pH and temperature on the color strength (
To determine the kinetics adsorption properties, a series of experiments at a constant berberine concentration (1% owf) and constant temperature (85, 90, 95, or 100 °C) at pH 5 over different time periods were carried out. To determine the thermodynamics adsorption properties, a series of experiments at various berberine concentrations (0.25-10% owf) and constant temperature (85, 90, 95, or 100 °C) at pH 5 for 12 h were carried out.
Analysis
Percent Exhaustion
To calculate the
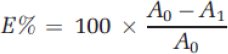
Color Strength
The
Berberine Concentration on CDP Fibers
A standard concentration plot of berberine was obtained (Eq. 2).
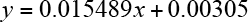
y is the absorbance at λmax of berberine and x is the concentration of berberine.
The concentration of berberine on CDP fibers (
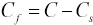
As the dyeing procedure finished, the absorbance at the λmax of berberine (425 nm) in the original bath (
Colorfastness
Washfastness of the dyed samples was tested using AATCC TM61-2013. 16 Sample pieces were prepared with a 50 × 150 mm multifiber test fabric sewn, stapled, or suitably attached centered along one 50-mm edge of the test specimen and in contact with the face of the material. Using the 3A version of the test method, the test specimens were washed at 71 °C for 45 min. Color change and staining of the test specimens were evaluated by comparison with the AATCC Gray Scale.
Results and Discussion
Effect of pH on K/S and E%
A set of experiments were designed to evaluate the effect of pH on
Effect of pH on
These results may be due to the electropositivity of berberine at various pH values and the dissociation of sodium sulfonate salts on the CDP fibers in the dye bath. Higher pH dyeing conditions are favorable to the dissociation of the sodium salts. The dissociation of sulfonic acids is much weaker than the sodium salts of sulfonates. Therefore, the resulting number of available anionic sites on CDP fibers under alkaline conditions is relatively greater than those under acidic conditions. The cationic dye berberine in its carbinol form above pH 9 is not colorless, and decreasing the pH would normally increase the electropositivity of berberine.
Effect of Dyeing Temperature on K/S and E%
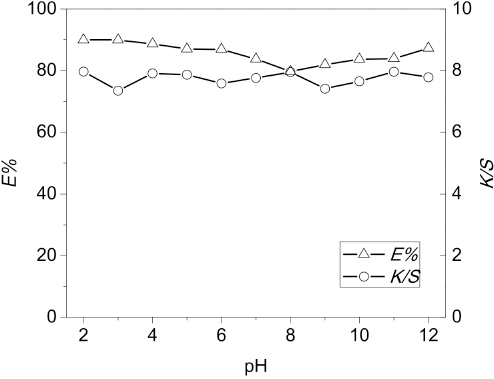
The next step was to assess the effectiveness of temperature (
Effect of dyeing temperature on
The glass transition temperature (Tg) of CDP fibers was analogous to that of PET fibers (67 to 125 °C) due to having similar supramolecular structures. Normally, the Tg values of amorphous, crystalline, and crystalline-oriented PET fibers are 67, 81, and 125 °C, respectively. The results showed that the
Meanwhile, a plateau of
Sorption Rates
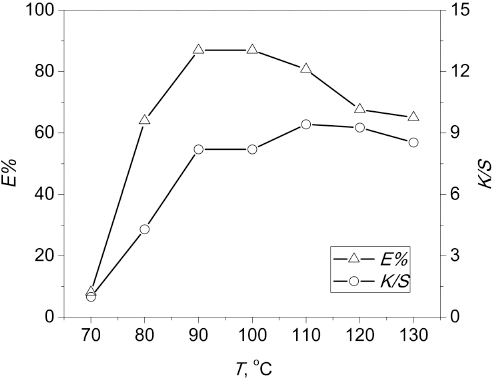
The effect of dyeing time (
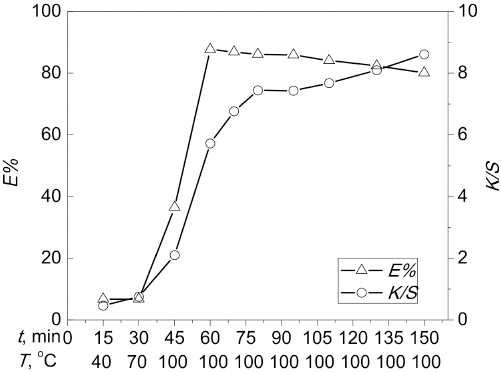
Effect of dyeing time on
Build-Up Properties and Effect of Dye Concentration on Exhaustion
A further study investigated the build-up properties and effect of berberine concentration (0-5% owf) on the
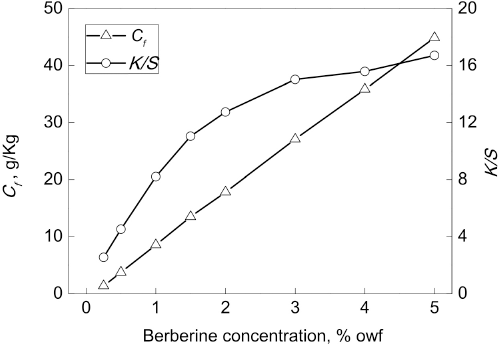
Effect of berberine concentration on
Kinetic Studies
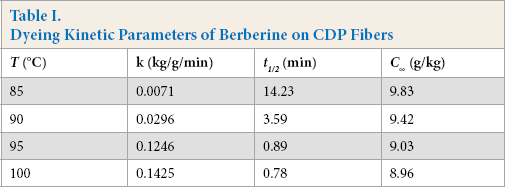
Kinetic curves at varying temperatures (85-100 °C) were generated to analyze the dye adsorption properties (Fig. 5, Table I). The
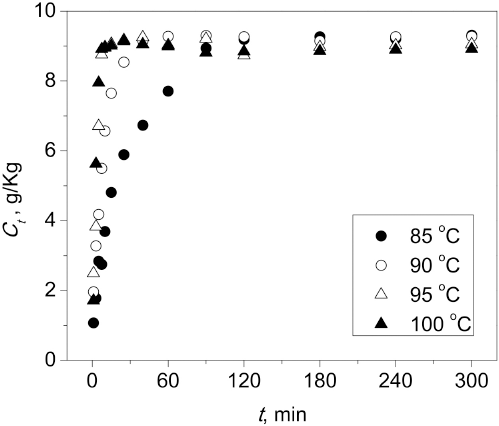
Dyeing kinetic curves at various temperatures.
Dyeing Kinetic Parameters of Berberine on CDP Fibers
Adsorption Isotherms
To investigate the dyeing mechanism of berberine on CDP fibers, adsorption isotherms at different temperatures were prepared (Fig. 6). The adsorption isotherms were analogous to the Langmuir type. However, a comparison of Langmuir and Freundlich models was carried out to confirm. The dye concentrations on the fibers at equilibrium (
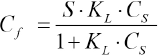
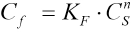
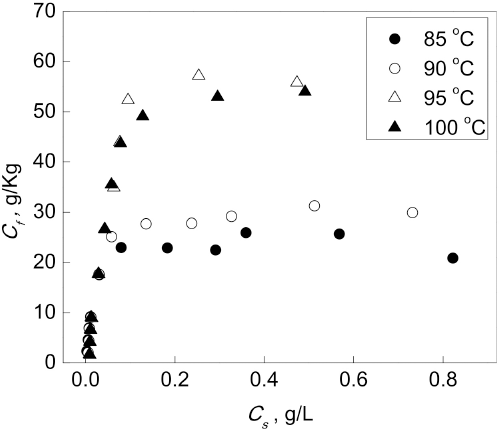
Adsorption isotherms of berberine for CDP.
Furthermore, the R 2 values of the two types at varying temperatures are tabulated in Table II. The R 2 value of the Langmuir model is always greater than that of the Freundlich model. The adsorption isotherms in this study were in good accord with the Langmuir type, indicating that the berberine interacts with the anionic sites of the CDP fibers in the form of ionic bonds.
R
2
of Freundlich Type and Langmuir Type Adsorption Isotherms, and
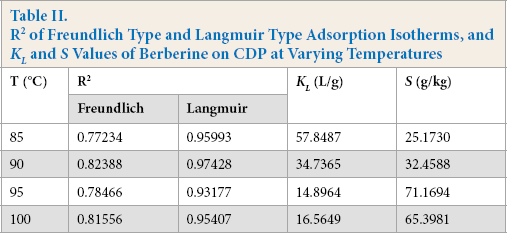
The Langmuir affinity constant and Langmuir adsorption saturation of berberine on CDP at varying temperatures are also presented in Table II. As the temperature increased,
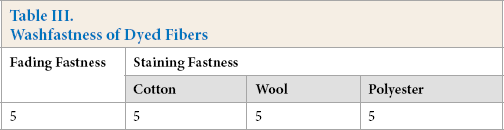
Washfastness
The berberine dyed CDP had excellent washfastness (Table III). The staining fastness of cotton, wool, and polyester fabrics was all grade 5. And the fading fastness and staining fastness of dyed CDP was also grade 5. The results showed that stable ionic bonds were formed between berberine and CDP fiber.
Washfastness of Dyed Fibers
Conclusions
Berberine, a natural cationic dye, was used to dye CDP fibers successfully. The
Footnotes
Acknowledgements
This work was partly supported by the key scientific research project in 2017 of Department of Science & Technology of Henan (172102310466), the doctoral funded project of Henan University of Engineering (Dkj2018005), and the young teachers foundation of the Institution of Higher Learning in Henan province (2018GGJS152)”.
