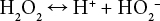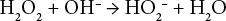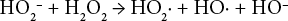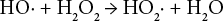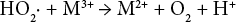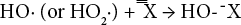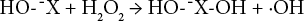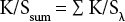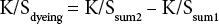Abstract
Hydrogen peroxide (H2O2) is the most commonly used bleaching agent for cotton fabric. However, the conventional H2O2 bleaching formula is not applicable for cationized cotton due to the low whiteness index after bleaching, fabric weakening, and a significant loss of cationic sites. In this work, magnesium sulfate (MgSO4) was added in the H2O2 bleaching formula to stabilize the bleaching system for cationized cotton. Additionally, a screening experiment with temperature and time, and sodium hydroxide (NaOH), H2O2, and MgSO4 concentrations as the factors, and whiteness index, bursting strength, and color strength from dyeing as the responses, was formulated. A statistical model was created using JMP software, which demonstrated how the bleaching conditions influenced the fabric properties. This procedure can be useful for bleaching cationized cotton in production settings.
Keywords
Introduction
Conventional fiber reactive dyeing of cotton is a process that uses high concentrations of salt and alkali for covalent bonding, and large amounts of water to remove hydrolyzed and unfixed dyes.1,2 As a result, effluent containing salt, alkali, and color is discharged. It has been estimated that as much as 40% of applied reactive dyes are unfixed and discharged in effluent.3,4 As consumer, brand, and retailer demands grow for sustainably produced products, so does the need for environmentally-friendly dyeing and finishing processes for cotton goods.
Cationic cotton has been touted for decades as a method to allow the coloration of cotton requiring no salt and with close to 100% dye utilization efficiency.5-9 Typically, cotton is cationized with 3-chloro-2-hydroxypropyl trimethylamine chloride (CHPTAC) reagent. From the resulting cationic charge, anionic dyes are electrostatically attracted to the fiber, negating the requirement of salt to force dye exhaustion. The literature is full of examples of researchers examining different facets of cationic cotton including cationization reaction efficiencies, methods of cationization, fastness of different dyes, effects of finishes, predicting dye recipes and cationization levels, mitigation of amine odor, and studies of how to control dye strike rates on cationic cotton.10-16
Although cationic cotton was researched for many years and showed a marked ability to reduce the environmental impact of cotton dyeing, commercial use and adoption of cationic cotton was quite limited. When cationic cotton was used commercially, it was most commonly used to produce heather fabrics, where small portions of commercially cationized fiber were blended with uncationized cotton. When dyed, the cationized cotton preferentially took up dye, resulting in a heather or mélange fabric.
A new initiative in cationic cotton production uses a continuous cationization process 17 resulting in a greige, highly-cationized fiber, at very difficult to obtain cationization levels. As a result, any safety risks associated with cationization, and the variability associated with fabric cationization, are removed from the mill. Additionally, dark colors are easily obtained due to the high level of cationization. This approach to using cationic cotton drastically simplifies the process of obtaining cationic cotton for commercial production of bright shades, because the fiber was treated in greige form. However, the cationic cotton must be bleached to provide a white base and remove any motes or trash that are readily seen on bright shades.
In most published research, the process for cotton cationization was scouring/bleaching and then cationization in fabric form. However, in fiber cationization production, the fiber preparation steps add significant cost to the fiber cationization process. For commercial fiber cationization, the fiber is typically cationized in greige form without a scour or bleach step, minimizing the cost of cationization. Therefore, in cases where the cationized fiber is used to dye bright shades that require bleaching, an efficient bleaching process is needed for pre-cationized cotton fiber.
Bleaching of cationic cotton received very little attention in the literature because most applications or cationization of cotton are performed on scoured or bleached cotton, and for most dark shades on cationic cotton, a simple alkaline scour is all that is required before dyeing. It is generally accepted that the active species responsible for the peroxide bleaching of cotton is the formation of the perhydroxyl anion (Eq. 1). 18
Because disassociation is very low, the peroxide bleaching of cotton is typically carried out at high temperature and alkalinity. A simplified representation of the reaction of hydrogen peroxide in an alkaline media is shown by Eq. 2.18,19
Eqs. 3–7 show the suggested reaction of hydrogen peroxide bleaching under alkaline conditions. 18
M is a transition metal, =X is a chromophore, and ¯X is an oxidized chromophore.
H2O2 is a fairly active oxidant that acts both on pigments and cellulose. It is well known that the degree of polymerization of cellulose can be lowered with H2O2 bleaching due to the formation of oxycellulose. Additionally, with the formation of oxycellulose, chain cleavage is possible through β-alkoxyl elimination. 20 To control the decomposition of peroxide and cellulose damage, stabilizers must be added to the bleach bath. However, as this research demonstrates, conventional H2O2 bleaching of cationic cotton causes rapid decomposition of H2O2, causing severe cellulosic fiber degradation and cleavage of cationic dye sites. This necessitates the need for a commercially-viable H2O2 bleaching process for cationic cotton.
H2O2 bleaching of cotton, paper, and pulps was widely studied and reported.21-24 The role and use of magnesium in alkaline peroxide bleaching systems was likewise widely studied for its use in bleach bath stabilization.25,26 In this study, we investigate H2O2 bleaching of cationic cotton using magnesium sulfate (MgSO4) for bath stabilization. A statistical model examining bleaching temperature, time, and sodium hydroxide (NaOH), H2O2, and MgSO4 concentrations as the factors, and whiteness index, bursting strength, and color strength from dyeing as the responses, is presented.
Experimental
Materials
The fabric used for bleaching is a cationized, 100% 30/1 single jersey greige cotton fabric provided by ColorZen. According to the manufacturer, the cotton fibers were cationized using CHPTAC before spinning and knitting. A 100%, 30/1 untreated greige cotton jersey fabric from Cotton Incorporated was used as a control. Sultafon D, a nonionic scouring agent, was obtained from Bozzetto Inc. Marlube CMN, a polyacrylamide lubricant, and Marquest PB, a H2O2 bleach bath stabilizer without magnesium were both obtained from Marlin Chemical Co. Reagent grade 50% sodium hydroxide (NaOH), 35% H2O2, and 50% citric acid were all obtained from Brenntag. ReagentPlus grade anhydrous magnesium sulfate (MgSO4, ≥99.5%) was obtained from Sigma-Aldrich. Invazyme CAT, a stabilized liquid catalase enzyme to remove residual peroxide in the bleached fabric, and the reactive dye Novacron Red FN-R (C.I. Reactive Red 238) were obtained from Huntsman International. Reagent grade 0.1 N potassium permanganate (KMnO4), 0.1 N and 20% sulfuric acid (H2SO4) were obtained from Reagents. A 0.04% phenol red indicator was obtained from LabChem Inc.
Bleaching
Bleaching was carried out in a Mathis laboratory Labomat beaker dyeing machine at a 10:1 liquor ratio (LR). All chemicals and auxiliaries were added into the bleaching bath initially, including 1-10 g/L of 35% H2O2, 1-10 g/L of 50% NaOH, 0-1 g/L of MgSO4, 1 g/L of Sultafon D, 1 g/L of Marlube CMN, and 0.4 g/L of Marquest PB. The greige cotton fabrics were bleached at various target temperatures with a ramp rate of 3 °C/min for a desired bleaching time. When bleaching was completed, the bleach bath was titrated immediately and the bleached cotton fabric was rinsed thoroughly with water and dried in a forced-air oven for 2 h at 60 °C.
CIE whiteness index (WI-CIE) of the fabrics were measured using an X-Rite Color i7 benchtop spectrophotometer with Color iControl software. The conditions used for measurement were illuminant D65, 10° standard observer, 25-mm aperture, specular included, and UV excluded. While measuring, the fabric was folded two times and each sample was measured three times by changing the measuring point and angle randomly to calculate the average value.
Titration
The bleach baths, both before and after bleaching, were titrated to calculate the content of alkali and H2O2 using a condensed titration method based on AATCC TM98 and AATCC TM102. 27 First, 20 mL of deionized water, 5 drops of phenol red indicator, and 10 mL of the bleach bath was added into a flask. While shaking the flask, a 0.1 N H2SO4 solution was added dropwise until one drop changed the color of the solution from red to greenish-yellow. Ten the same solution was used for H2O2 determination after adding 20 mL of 20% H2SO4 to the flask. The solution was titrated with 0.1 N KMnO4 solution to a faint pink color that lasts at least 30 s. The volume of the 0.1 N H2SO4 and 0.1 N KMnO4 used for titration were recorded and used for calculating the alkali and H2O2 present in the bath.
Bursting Strength Measurement
The bleached cotton fabrics were tested for bursting strength using a SDL Atlas M229P PnuBurst bursting strength tester. The test was done according to the standard test method for bursting strength ASTM D3786, 28 but due to the limited amount of fabrics, each sample was only measured five times and the average was recorded.
Estimation of Cationization Level by Dyeing
To estimate the amount of cationic sites on the cationized cotton fabrics before and after beaching, cationized cotton fabrics were dyed with 6.25% owf of Reactive Red 238 at 71 °C for 20 min at a 25:1 LR. The dyed samples were cold rinsed in a sink, boiled for 5 min in a steam kettle, and dried at 60 °C in a forced-air oven for 2 h. Since no salt or alkali was added during the dyeing, the only mechanism for holding the dyes to the fabric was through ionic bonds with the cationic sites. Thus, with the excess amount of dye applied, a higher color strength value of the dyed fabric indicated more cationic sites on the fabric. To measure the colorimetric data and K/Ssum value of dyed fabrics, the same spectrophotometer and settings were used as described in the bleaching section. K/Ssum is defined by Eq. 8.
λ is 360-750 nm, at 10 nm intervals.
Since the bleached fabrics have different K/Ssum values before dyeing, K/Sdyeing was used to better represent the depth of color obtained during dyeing, as shown in Eq. 9.
K/Ssum1 is the color strength of the fabric after bleaching, and K/Ssum2 is the color strength of the dyed fabric.
Results and Discussion
Conventional H2O2 Bleaching
First, the cationized cotton fabric was bleached at 60 °C to 100 °C for 25 min using a 10:1 LR and a conventional H2O2 bleaching recipe with 4 g/L of 35% H2O2, 4 g/L of 50% NaOH, 1 g/L of Sultafon D, 1 g/L of Marlube CMN, and 0.4 g/L of Marquest PB. Also, an uncationized 100% cotton fabric was bleached with the same formula at 100 °C for 25 min.
Table I presents the effect of temperature on the alkalinity and H2O2 consumption of the bleaching bath, as well as on the WI-CIE and bursting strength values of the bleached cotton fabrics as compared to the greige fabrics. Since the cationized and uncationized cotton fabrics had different yarn and knitting parameters, their initial whiteness index and bursting strength values were different. To make a fair comparison, the bursting strength of the bleached fabric was compared to the corresponding greige fabric to calculate the percent strength loss due to bleaching.
Effect of Bleaching Temperature on Cationized Cotton Fabric
As presented in Table I, the conventional bleaching recipe and process at 100 °C successfully bleached the uncation-ized cotton fabric with high WI-CIE and increased bursting strength values. The gain in strength may result from the shrinkage of the fabric during bleaching and the increase in fabric density. However, for cationized cotton, bleaching at high temperature resulted in relatively lower WI-CIE values and significant fabric strength loss.
The current understanding of the H2O2 bleaching mechanism is that NaOH activates H2O2 to liberate perhydroxyl anion (HO2?), which can react as a nucleophile to attack the conjugated double bond chromophore of the pigments in greige cotton. However, if the bleaching system is over activated and the liberation rate of HO2? is too high, the HO2? anion may damage the fabric and form oxygen directly without bleaching the natural chromophore. The high consumption of NaOH and H2O2, and the loss in strength of bleached cationized cotton fabric, indicated that the presence of cationic cotton over activated the H2O2 in the system. Lowering the bleaching temperature for cationized cotton significantly reduced the chemical consumption and strength loss, but further decreased WI-CIE values. Thus, lowering the bleaching temperature was not able to efficient-ly stabilize the bleaching system and a stabilizer was added.
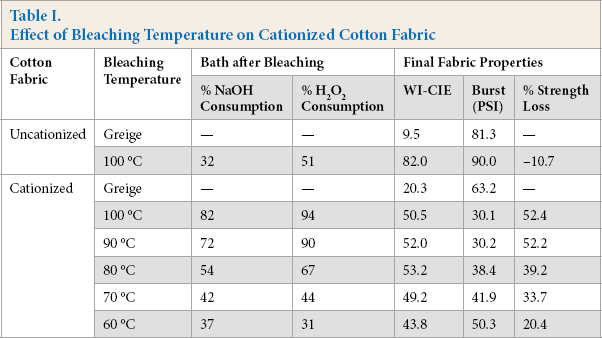
To estimate the loss of cationic sites during bleaching, the fabrics were dyed with 6.25% owf of Reactive Red 238 using the method described in the experimental section. Both the K/Ssum values before and after dyeing were measured to calculate the depth of color obtained during dyeing (K/Sdyeing). For bleached cationized cotton fabrics, the percent K/Sdyeing value was calculated by comparing the K/Sdyeing value with that of the cationized cotton dyed without bleaching. All colorimetric data and K/S values for both the cationized and uncationized cotton fabrics bleached at various temperatures are presented in Table II.
Effect of Bleaching Temperature on Cotton Fabric Cationization Level
As expected, the K/Sdyeing values for the uncationized fabrics were very low since no salt or alkali was added in the dyeing process. The unbleached cationized cotton had the highest K/Sdyeing value, and with increased bleaching temperature, the K/Sdyeing value decreased, indicating that more cationic sites were cleaved during bleaching. When bleached at 100 °C, only around 80% of the cationic sites were reserved for dyeing. Based on the WI-CIE and bursting strength values, and dyeability of the fabrics, the bleaching system must be better stabilized to more efficiently decolorize the cationized cotton prior to dyeing without severely damaging the fabric and cleaving cationic sites.
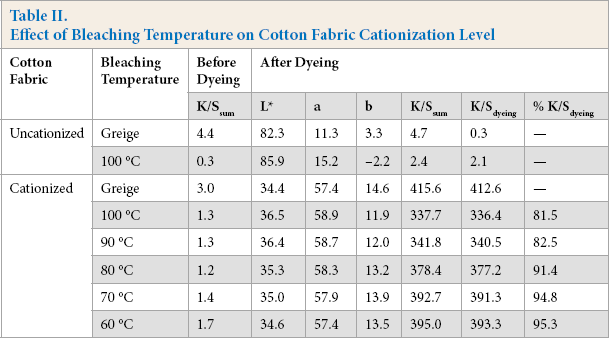
Effect of Magnesium Sulfate Stabilizer
To stabilize the bleaching system and more efficiently bleach the cationized cotton, magnesium sulfate (MgSO4) was added into the formula as a stabilizer, while other conditions were kept the same. Table III shows the chemical consumption, and WI-CIE, bursting strength, and percent K/Sdyeing values of the cationized cotton fabric bleached at 90 °C and 100 °C with and without 0.5 g/L of MgSO4. Adding MgSO4 into the bleaching bath significantly increased the WI-CIE, bursting strength, and percent K/Sdyeing values of the fabrics bleached at both 90 °C and 100 °C. As the percent NaOH and H2O2 values of the final bath indicate, MgSO4 successfully stabilized the bleaching bath by reducing the chemical consumption rate. With the use of MgSO4, the bleached cationized cotton fabrics had less than a 10% loss in bursting strength and K/Sdyeing values compared to the cationized fabric without bleaching. However, the WI-CIE value was still less than that for the bleached conventional cotton. Since cationized cotton is not supposed to be left undyed, a full-white is not required and a WI-CIE value of around 60 should be adequate for dyeing, even for a light to medium shade.
Effect of MgSO4 on Bleaching Cationized Cotton Fabric
Mechanism
Although the purpose of this work is to establish a viable bleaching process for cationic cotton, some discussion of the bleach mechanism of cationic cotton is warranted. From Table I, the presence of cationic cotton caused much higher consumption of not only H2O2, but of NaOH as well. Referencing Eq. 2, this undoubtedly led to the generation of the much more active bleaching species, HO2−.
In Table I, we also saw a corresponding decrease in the bursting strength. In Fig. 1, graphing H2O2 use with the decrease in bursting strength gave a very good linear ft, suggesting the formation of oxycellulose.
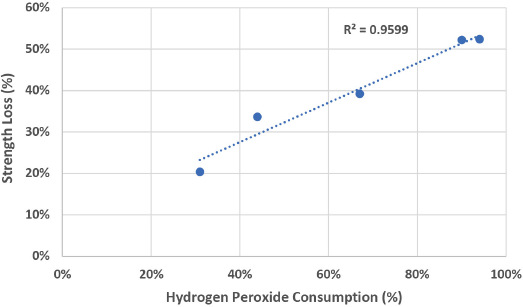
Relationship between H2 O2 consumption and fabric strength loss.
Again, although not the goal of this work, the formation of oxycellulose could be verified through simple analysis techniques such as Fehlings Test or Turnbulls Blue Test. 29 It would be expected that high levels of reducing and carboxyl groups would be found. The formation of oxycellulose and the accompanied loss in fiber strength would also suggest significant chain cleavage. A recent paper thoroughly investigates the mechanism of β-alkoxyl elimination of oxidized cellulose in alkaline media, concluding that this process is the key to cellulose degradation. 30 As Table II shows, the bleached cationic cotton showed a marked decrease in dyeability, further supporting chain cleavage and the cleavage of cationic sites. Fig. 2 shows the relationship of H2O2 consumption and the dyeability of the bleached fabrics. Similarly, a strong linear relationship was seen between H2O2 consumption and loss of cationic sites.
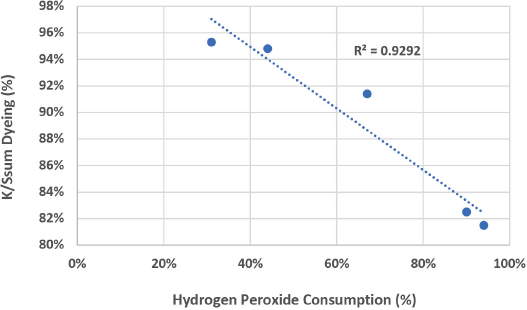
Relationship between H2O2 consumption and fabric dyeability.
It has been suggested that following Eq. 2, hydroxyl radicals are formed that can be terminated by the sequence of Eqs. 8 and 9, or a chain reaction as in Eqs. 10 and 11.18,19 The chain reaction component of the sequence increased as the temperature increased, analogous to the increased H2O2 consumption and increased fiber degradation and loss of cationic sites with increasing temperature found in this work.
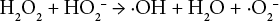
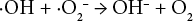
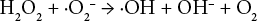
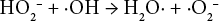
It was theorized that the inclusion of magnesium hydroxide effectively binds the superoxide radical (Eq. 12), as well as the formation of MgO in quenching a hydroxyl radical (Eq. 13).18,19
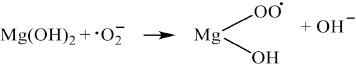
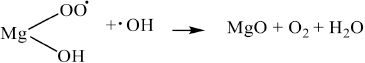
In our work using magnesium sulfate, the inclusion of magnesium appeared to corroborate the chelation of both radicals and sodium hydroxide as noted by the increase in fabric strength, increased dyeability, and lower consumption of sodium hydroxide and hydrogen peroxide. We do not believe that the cationic charge itself directly played a significant role in the rapid decomposition of hydrogen peroxide, as in the role of cationic transition metals, as the cationic charges are hard quats. Additionally, the cationized fiber was wet processed in high levels of alkali and much of the inherent metal content of the natural fiber was removed. However, it is well known that in the presence of alkali, cationic cotton generates free trimethylamine; 14 we suspect that this may serve as a catalyst for the rapid decomposition of hydrogen peroxide, especially at elevated temperatures. It is known that hydrogen peroxide reacts with tertiary amines to form amine oxides.31
Statistical Analysis and Modeling of Bleaching Parameters
To investigate the influence of different bleaching conditions on WI-CIE and bursting strength values, and dyeability of the cationized cotton fabric, a design of experiment with five factors, including bleaching temperature (60-100 °C), time (25-60 min), and NaOH (1-10 g/L), H2O2 (1-10 g/L), and MgSO4 (0.05-1 g/L) concentrations, was designed and 37 experimental runs were conducted. The WI-CIE, bursting strength, and color strength values from dyeing (percent K/Sdyeing) of bleached cotton fabrics were used as the response factors. The results of these experiments are presented in the Appendix and the data was analyzed using JMP 13 statistical software package (SAS Institute Inc.)
A standard least squares model was fitted to the data with the emphasis on effect screening. Fig. 3 presents the overall effect summary of the factors for all three responses and the half-normal probability plot for each response. Based on the p-values in the effect summary plot, the significant factors were identified, including temperature, H2O2 concentration, NaOH concentration, temperature*NaOH, time, MgSO4 concentration, temperature*H2O2, and H2O2*H2O2. The insignificant factors with higher p-values should be excluded from the model to improve the resolution of the remaining factors.
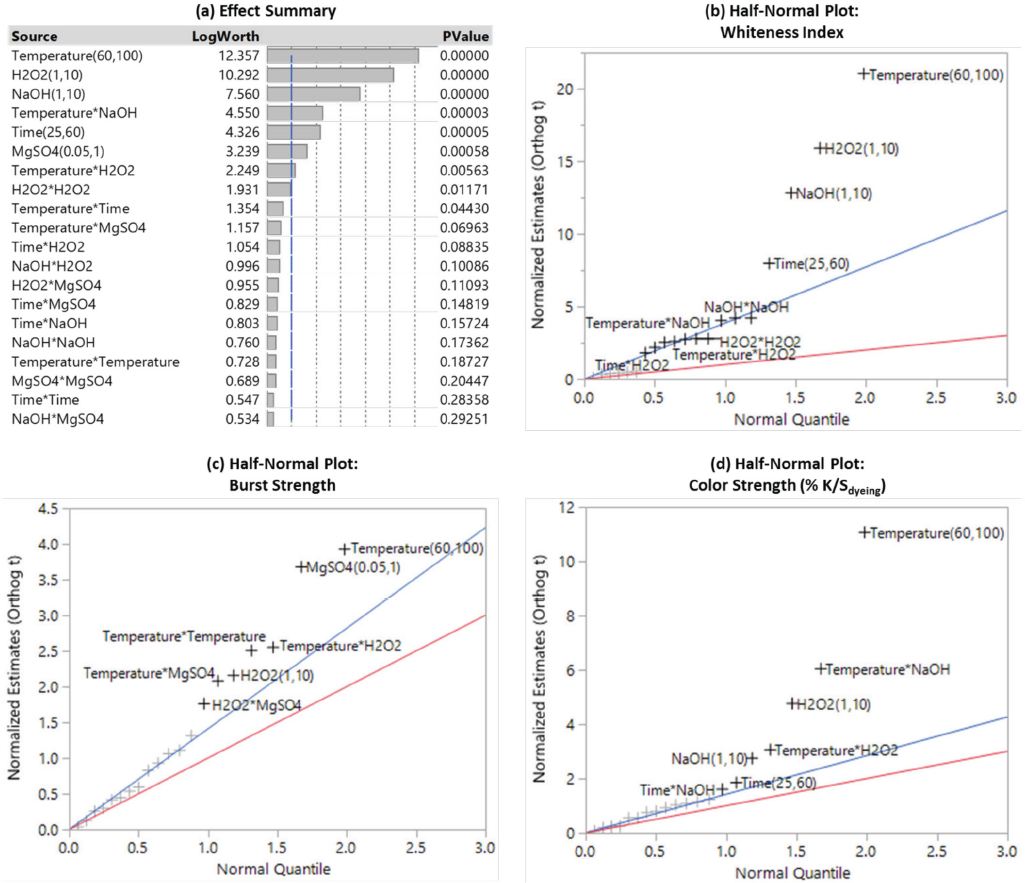
Effect summary (a) and half-normal plots (b to d) for screening out the significant main effects and two-factor interactions for bleaching cationic cotton.
The half-normal probability plot is also a graphical tool that can be used to separate and rank the important factors from the unimportant ones. In the half-normal plot, the significant main effects and interactions will typically be displaced well of the lines (the blue line has a slope equal to Length's PSE and the red line has a slope equal to 1 toward the upper right of the graph. It is clear that in consideration of the WI-CIE value, the bleaching temperature, and H2O2 and NaOH concentrations were the most important factors. Both bleaching temperature and MgSO4 concentration had a very significant influence on the bursting strength of the bleached fabrics. To maintain a high level of cationization and good dyeability of the fabrics, the bleaching temperature was critical.
After excluding the insignificant factors, a new standard least squares model was fitted to the data. Fig. 4 shows the renewed effect summary of the significant factors. Compared with the effect summary in Fig. 3a, the p-value of most of the significant factors were reduced by eliminating the insignificant factors from the model, indicating a better resolution of the remaining factors.
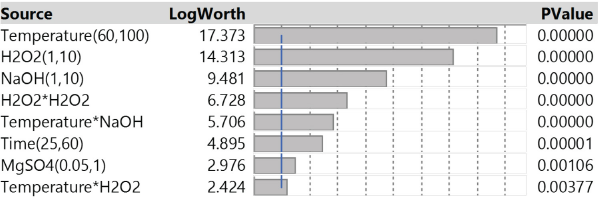
Effect summary of the significant factors for bleaching cationized cotton.
To illustrate how well the model ft the actual data, the actual by predicted plot and the R 2 value of the model for the three responses are presented in Fig. 5. For both WI-CIE and color strength values, the model had a very high R 2 value, indicating a good ft of the model to the actual data. For the bursting strength, the R 2 of the model was only 0.58. The variance in strength of the original fabric before bleaching and the limited accuracy of the test method likely influenced the final result and the R 2 value of the model. In general, with the significant factors included, the model for whiteness index and color strength ft the actual data nicely, while the model for bursting strength only partially explained the data.
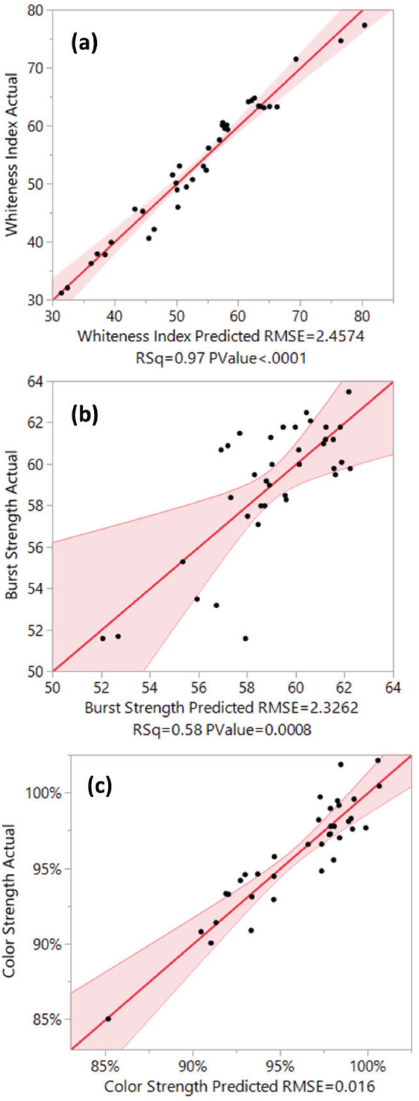
Actual by predicted plot of the standard least squares model for (a) WI-CIE, (b) bursting strength, (c) and color strength values.
Fig. 6 shows the prediction profiler of the bleaching model including only the significant factors. However, since the interactions of temperature*NaOH and temperature*H2O2 were also significant factors for bleaching, it was necessary to include the interaction profiles of temperature, NaOH, and H2O2 (Fig. 7) to better illustrate how the main factors affected the three responses under different conditions.
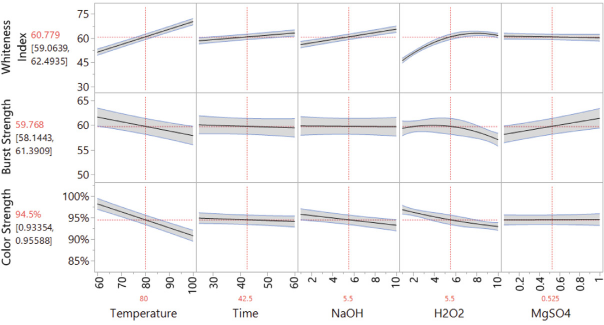
Prediction profiler of the bleaching model including only the significant factors.
Based on Figs. 6 and 7, increasing the bleaching temperature significantly increased the WI-CIE values of the bleached fabric, but lowered the bursting strength and color strength values for dyeing at the same time. A longer bleaching time slightly increased the WI-CIE value, but the influence was relatively small. Increased NaOH concentration increased the WI-CIE value, especially when bleached at high temperature. At a low bleaching temperature, the NaOH concentration had relatively small influence on color strength, but at high temperature (greater than 80 °C), a higher NaOH concentration resulted in a lower color strength of the dyed bleached fabric. H2O2 was the only factor that had a significant quadratic relationship with the responses. In consideration of all three factors, it was better to use 3-7 g/L of H2O2 since an extra amount of H2O2 had little influence on WI-CIE values, but significantly reduced bursting strength and color strength values of the fabric. A higher concentration of MgSO4 had a very small influence on WI-CIE and color strength values, but greatly increased bursting strength values of bleached fabrics.
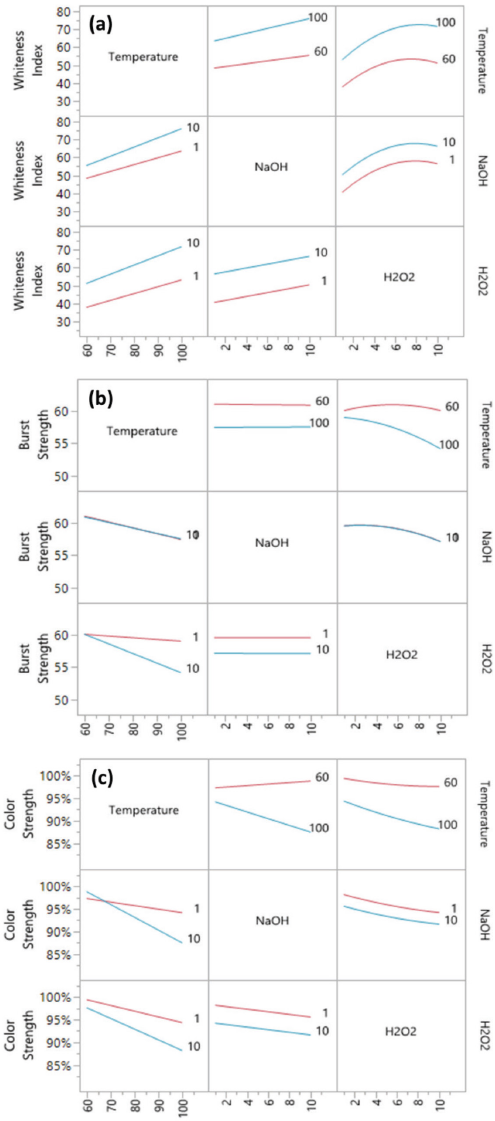
Interaction profiles of temperature, NaOH concentration, and H2O2 concentration for (a) WI-CIE, (b) bursting strength, and (c) color strength values.
Based on the model, 3-7 g/L of H2O2 and 0.5-1 g/L of MgSO4 are proposed for bleaching cationic cotton in consideration of all responses. The selection of the other conditions (temperature, time, and NaOH) should be based on the requirements of the fabric in production to reach a balance between WI-CIE, bursting strength, and dyeability. For example, if a light blue shade needs to be obtained on a fabric with relatively high strength, an acceptable WI-CIE value is probably the critical response to be considered. In real production, the model can be used as a good reference for choosing the conditions to bleach cationized cotton fabrics.
Conclusions
Using a conventional hydrogen peroxide (H2O2) bleaching formula for cationized cotton resulted in relatively low whiteness index (WI-CIE) values, and significant loss in bursting strength and cationic sites, due to cationized cotton over activating the H2O2 bleaching system. Adding an appropriate amount of MgSO4 efficiently stabilized the bleaching system and significantly improved the properties of bleached cationized cotton. A screening experiment with temperature, time, NaOH, H2O2, and MgSO4 as the factors was conducted and the models for WI-CIE, bursting strength, and color strength values of dyeing were generated. Since it was difficult to increase all three responses at the same time, selection of the bleaching conditions should be based on the requirements of the fabric in real production. By controlling the factors during bleaching and using the model as a reference, bleached cationized cotton with acceptable whiteness, bursting strength, and dyeability can be obtained.
Footnotes
Appendix
Matrix of Experiments
Factors
Responses
Temperature (°C)
Time (min)
50% NaOH (g/L)
35% H2O2 (g/L)
MgSO4 (g/L)
WI-CIE
Burst (PSI)
% K/Sdyeing
60
25
1
1
1
31.2
63.5
97.6
60
25
1
10
0.05
40.7
61.3
99.7
60
25
10
1
0.05
40.0
58.0
102.2
60
25
10
10
1
49.5
60.1
98.1
60
60
1
1
0.05
38.0
59.5
99.5
60
60
1
10
1
51.6
59.5
96.6
60
60
10
1
1
45.7
61.2
97.7
60
60
10
10
0.05
60.2
57.5
95.6
60
45
10
10
1
53.1
61.2
101.9
60
45
10
10
0.2
56.2
59.2
97.0
60
45
10
4
0.2
50.8
60.0
99.6
60
45
4
10
0.2
53.1
60.0
96.6
60
60
4
4
0.05
49.0
58.3
97.3
60
60
4
4
0.2
50.2
60.7
99.0
60
60
6
6
0.5
52.4
61.8
97.3
60
60
10
10
0.5
57.6
58.5
97.8
60
25
1
10
1
45.3
59.8
94.8
60
25
10
1
1
37.8
61.8
100.5
60
25
1
1
0.05
32.1
59.0
98.3
60
60
1
1
1
36.3
59.8
99.2
80
25
10
10
0.5
63.2
60.9
93.3
80
45
10
6
1
63.3
61.0
94.6
80
45
6
10
0.5
64.9
60.7
94.2
80
60
6
6
0.05
63.4
61.5
94.6
90
25
4
4
0.05
59.4
51.6
93.0
90
25
4
4
0.2
60.2
57.1
94.5
90
25
4
4
0.5
59.6
61.8
95.8
100
25
4
4
0.5
64.4
58.0
90.9
100
25
4
4
1
64.2
62.5
93.1
100
25
10
10
0.05
74.7
51.7
85.1
100
25
10
1
1
60.6
62.1
91.4
100
25
1
10
1
63.4
53.5
93.3
100
25
1
1
0.05
42.2
58.4
97.8
100
60
10
10
1
77.4
55.3
82.9
100
60
10
1
0.05
63.5
53.2
90.8
100
60
1
10
0.05
71.6
51.6
90.1
100
60
1
1
1
46.0
61.8
98.2